Affiliation:
1Department of Neurorehabilitation, Casa di Cura “Villa Verde”, 73100 Lecce, Italy
ORCID: https://orcid.org/0009-0009-8329-7295
Affiliation:
1Department of Neurorehabilitation, Casa di Cura “Villa Verde”, 73100 Lecce, Italy
2Department of Engineering for Innovation, University of Salento, 73100 Lecce, Italy
Email: gverrienti@villaverde.lecce.it
ORCID: https://orcid.org/0000-0003-3477-7195
Explor Neuroprot Ther. 2026;6:1004145 DOI: https://doi.org/10.37349/ent.2026.1004145
Received: November 25, 2025 Accepted: March 11, 2026 Published: April 10, 2026
Academic Editor: Maurizio Balestrino, University of Genoa and IRCCS Ospedale Policlinico San Martino, Italy
The article belongs to the special issue Neuropsychology of Handwriting
Background: Acquired brain injury (ABI) often causes long-lasting impairments in written language and handwriting that limit autonomy and daily functioning. Despite their relevance, these deficits have received limited research attention compared with spoken language disorders. The present work aims to systematically review interventions designed to improve reading, writing, and handwriting abilities in individuals with ABI.
Methods: Following PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines, PubMed, American Psychological Association (APA) PsycINFO, Cochrane Library, Web of Science, and Google Scholar were searched from inception to 1 November 2025. Eligible studies were randomized or non-randomized clinical trials (non-RCTs) involving adults or adolescents with ABI and documented written language impairments. Risk of bias was assessed using the Risk of Bias 2 tool (RCTs) and Risk of Bias in Non-randomized Studies—of Interventions tool (non-RCTs).
Results: Twelve studies met inclusion criteria (2 RCTs, 10 non-RCTs), all conducted in post-stroke populations, highlighting the absence of evidence from other ABI aetiologies. Three main intervention categories emerged: (1) Behavioral treatments, which consistently improved trained spelling and functional writing, with some advantages for errorless learning in maintaining gains. (2) Technology-assisted approaches, including assistive software, digital spelling aids, and handwriting-focused programs, which showed feasibility, high usability, and improvements in accuracy, legibility, and motor fluency. (3) Neuromodulation, with one RCT showing that dual-site transcranial direct current stimulation can modestly enhance behavioral writing therapy. Most non-RCTs showed serious or critical risk of bias, and sample sizes were small, limiting generalizability.
Discussion: Current evidence—although preliminary and restricted to post-stroke ABI—indicates that behavioral, technological, and neuromodulatory interventions can improve aspects of written language and handwriting after ABI. However, the available literature is characterized by small samples, substantial methodological variability, and a paucity of standardized and ecologically valid outcome measures. High-quality, adequately powered trials with standardized, functional outcomes are urgently needed, particularly in non-stroke ABI populations.
Acquired brain injuries (ABIs)—including traumatic brain injury (TBI), extensive stroke, anoxic-hypoxic damage, and infectious or inflammatory encephalopathies—represent one of the leading causes of long-term disability across adulthood [1–4]. Each year, millions of individuals worldwide experience an ABI. A substantial proportion of survivors present persistent cognitive, linguistic, and motor impairments that significantly affect independence and quality of life [5–7]. Among the wide spectrum of post-injury consequences, disturbances of written language and handwriting often receive less clinical attention than spoken language deficits, despite their crucial role in daily communication, education, and occupational participation [8]. For clarity, and in line with common usage in the literature, the terms used in this manuscript will be consistent as follows: written language refers to higher-level linguistic processes involved in writing (e.g., spelling, syntax, and text production), writing refers to the functional act of producing written output, and handwriting specifically denotes the grapho-motor component of writing.
Writing is a fundamental means of expressing ideas, managing personal affairs, interacting with services, and engaging in social and professional activities. Even in the digital era, handwriting remains essential for many everyday tasks, such as signing documents, completing forms, and taking notes. It also plays an important role in organizing information and communicating in settings where technology may not be easily accessible. For individuals recovering from ABI, the loss or reduction of handwriting abilities can severely hinder autonomy, limit participation in meaningful activities, and contribute to social withdrawal or dependency [9, 10].
Deficits in written language after ABI can manifest in multiple ways [8]. Some individuals develop alexia (impaired reading), agraphia (impaired writing), or both, reflecting disruptions at different stages of the cognitive-linguistic system. Others experience broader impairments that affect writing not only as a linguistic task but also as a complex motor, executive, and visuospatial function. In this context, handwriting impairments after ABI result from the interaction of linguistic, motor, attentional, and executive deficits, often worsened by fatigue and slowed processing.
Despite the clinical relevance of handwriting for functional independence and social reintegration, research on written language rehabilitation has historically been overshadowed by work on spoken language rehabilitation [11–13]. While several intervention approaches have been proposed—ranging from linguistic treatments targeting grapheme-phoneme conversion to motor-based training, computerized programs, and ecologically-oriented methods—evidence remains scattered and heterogeneous. As a result, findings are often difficult to compare across studies. Moreover, most existing reviews have focused broadly on agraphia or written language in aphasia rather than specifically addressing the unique population of individuals with ABI, who often present more complex profiles and require adapted therapeutic strategies [8, 14].
Given this gap, there is a pressing need to systematically examine the available literature on writing rehabilitation after ABI. To our knowledge, the studies conducted to date have been carried out predominantly in post-stroke populations. A systematic review (SR) would help clarify what approaches have been used, what outcomes they impact, and what evidence exists for their effectiveness. The introduction to the present SR is further developed through the following subsections, which provide an overview of the neurological basis of handwriting (The neurological basis of handwriting: a schematic overview) and a description of the handwriting deficits typically observed after ABI (Handwriting deficits in acquired brain injury).
Writing is a complex cognitive-motor activity that integrates linguistic, visuospatial, executive, and fine-motor processes [15–17]. Its successful execution relies on the coordinated function of a distributed neural network extending across frontal, parietal, temporal, and subcortical regions (Figure 1).
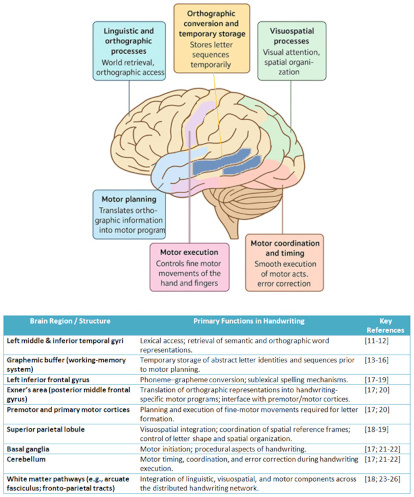
Neural network underlying handwriting. Linguistic, orthographic, visuospatial, motor-planning, and motor-execution processes are supported by a distributed system spanning temporal, frontal, parietal, cerebellar, and subcortical regions, with white matter pathways enabling integration across components; the brain icon was generated with ChatGPT (OpenAI, 2023; https://chatgpt.com/) and subsequently partially modified by the authors.
At the level of written language, the generation of written output begins with the activation of semantic and lexical representations within the left temporal lobe. In particular, the middle and inferior temporal gyri support word retrieval and orthographic access [18, 19]. These lexical-orthographic representations are then transformed into graphemic codes—abstract letter identities and sequences. This process is commonly attributed to the graphemic buffer, a working-memory system that temporarily stores serial letter information before motor planning [20–23].
The transition from graphemic representations to motor execution, corresponding to the handwriting component of writing, primarily involves frontal and parietal regions [24]. The left inferior frontal gyrus contributes to phoneme-grapheme conversion and sublexical spelling processes, whereas the superior parietal lobule is crucial for visuospatial integration, letter formation, and the coordination of spatial frames of reference [25, 26]. The Exner’s area, located in the posterior middle frontal gyrus, plays a key role in translating orthographic information into motor programs specific to handwriting [27, 28]. This region interfaces with premotor and primary motor cortices responsible for orchestrating fine-motor movements of the hand and fingers [24, 28].
Subcortical structures also participate in handwriting control. The basal ganglia regulate motor initiation and procedural aspects of writing, while the cerebellum contributes to motor timing, error correction, and the smooth execution of letter forms [24, 29, 30]. Additionally, white matter pathways—such as the arcuate fasciculus and fronto-parietal tracts—facilitate communication across this extended network. This connectivity enables the integration of written language and motor components [25, 31–34].
Given this distributed architecture, handwriting is particularly vulnerable to brain injury. Damage to different nodes of the network can lead to distinct impairments: lexical-semantic deficits from temporal lesions [35, 36], sublexical spelling deficits from left frontal involvement [37], visuoconstructive difficulties from parietal damage [38], and motor execution problems from cortical or subcortical motor system injury.
Handwriting impairments are a frequent consequence of ABI, particularly when the lesions involve networks supporting linguistic processing, visuospatial integration, and fine-motor control. These impairments may manifest as agraphia—the loss or impairment of previously acquired writing abilities—or as broader disturbances in written communication linked to executive, attentional, or motor deficits [28, 39]. The nature and severity of handwriting impairments vary widely based on the lesion’s etiology, location, and extent, as well as premorbid literacy skills.
At the linguistic level, ABI may disrupt access to lexical and orthographic representations, resulting in central agraphias. Individuals with central agraphia may show incorrect spelling of familiar words and phonologically implausible errors. They may also have difficulty with morphologically complex forms or failures in maintaining graphemic sequences due to impairments in the graphemic buffer [40–42]. Such deficits are especially common following left temporal or inferior frontal damage. Temporal lesions often produce lexical-semantic agraphia, characterized by semantic substitutions or difficulty retrieving irregular word spellings. In contrast, frontal lesions may lead to phonological agraphia, where nonwords and phonologically mediated spellings become disproportionately impaired [19, 37, 43]. Conversely, lesions affecting parietal, premotor, motor, or subcortical regions can produce peripheral agraphias, in which the linguistic plan for writing is preserved but the ability to execute written forms is compromised. These impairments can include distortions in letter shape, difficulty maintaining spatial alignment, and reduced legibility. Other issues may involve micrographia or problems with initiating or sustaining motor aspects of writing [44]. Injuries to the superior parietal lobule may impair visuospatial integration and letter construction, while cerebellar or basal ganglia damage can disrupt motor coordination, timing, and fluency. In patients with ABI, handwriting deficits are often multifactorial, reflecting the combined impact of linguistic, cognitive, and motor impairments. Co-occurring deficits in attention, working memory, executive functions, and visuospatial processing may further compromise written expression even when primary language mechanisms remain partially intact. Additionally, behavioral dysregulation or reduced awareness of deficits—frequent after traumatic or anoxic injuries—can interfere with the learning and application of writing strategies during rehabilitation.
Overall, handwriting impairments following ABI represent a significant obstacle to communication and functional independence. Understanding the diverse mechanisms underlying these deficits is essential for selecting and tailoring effective rehabilitation interventions, which is a central focus of the present SR.
Although research on rehabilitation approaches has increased, there is no comprehensive, updated synthesis focused specifically on written language rehabilitation in individuals with ABI.
The aim of this SR is therefore to critically evaluate current evidence on interventions designed to improve reading and written language abilities in this clinical population. Specifically, the objectives of the present work include:
The identification of rehabilitation approaches targeting written language disorders after ABI.
The evaluation of the effectiveness of these interventions using standardized linguistic, neuropsychological, and functional outcomes.
The identification of methodological limitations, gaps in the literature, and directions for future research.
A SR was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [45]. Although this review was not registered on PROSPERO (International Prospective Register of Systematic Reviews), the eligibility criteria, search strategies, and analytical procedures are presented systematically below to ensure methodological transparency and rigor. To mitigate potential risks of selection and reporting bias related to the lack of PROSPERO registration, the review protocol was defined a priori, including eligibility criteria, search strategy, outcomes of interest, and risk-of-bias assessment methods. The literature search was conducted using the following databases: PubMed, American Psychological Association (APA) PsycINFO, Cochrane Library, Web of Science (Clarivate Analytics), and Google Scholar, the latter included to increase search sensitivity and reduce the risk of missing relevant studies. All records retrieved were screened using the same predefined eligibility criteria and underwent formal risk-of-bias assessment, in line with studies identified from other databases. A comprehensive literature search was conducted, including all studies published up to 1 November 2025. Articles were identified using the following Medical Subject Headings (MeSH) and keywords: (brain injury OR traumatic brain injury OR stroke OR acquired brain injury) AND (alexia OR agraphia OR dyslexia OR handwriting OR dysgraphia) AND (rehabilitation OR therapy) AND (adult OR adolescent) AND (clinical trial). The eligibility criteria (PICOS framework) for inclusion are summarized in Table 1.
PICOS criteria.
| PICOS element | Description |
|---|---|
| Population (P) | Adults or adolescents with acquired brain injury (traumatic, anoxic, vascular, or infectious in origin) presenting documented written language impairments |
| Intervention (I) | Rehabilitation programs aimed at improving reading and/or writing abilities |
| Comparison (C) | No intervention, standard care, or alternative rehabilitation approaches |
| Outcomes (O) | Primary outcomes: performance on reading/writing tasks, spelling accuracy, lexical access in writing; secondary outcomes: functional communication, quality of life, independence in daily written communication |
| Study design (S) | Clinical trials (randomized and non-randomized) |
Citations resulting from the searches were screened for inclusion or exclusion by two authors independently. The full texts of all potentially relevant articles were subjected to additional screening by two independent authors in parallel. Conflicts were resolved by a third author. In addition, a forward and a backward search (checking the references of identified papers) were conducted to identify any studies that were not retrieved using the main search strategy.
The inclusion criteria were: (1) articles published in English; (2) randomized clinical trials (RCTs) and non-RCTs focused on the potential use of rehabilitation programs aimed at improving reading and/or writing abilities (e.g., phonological training, cognitive-linguistic therapy, computerized interventions, errorless learning, dual-route-based therapies, intensive therapeutic programs); and (3) studies published up to 1 November 2025. In the main search strategy, the following exclusion criteria were adopted: (1) studies for which the complete text could not be found; (2) studies with other designs such as case reports, expert opinions, reviews, and meta-analyses; and (3) articles not in English.
The data extraction form developed for this study aimed to determine the study characteristics (title, first author, study design, examined population, number of included subjects, and a brief description or main findings of the study).
We decided to use the Risk of Bias 2 tool (RoB 2) [46] to assess the quality of the RCTs, while the Risk of Bias in Non-randomized Studies—of Interventions tool (ROBINS-I) [47] was adopted for the quality assessment of non-RCTs. The RoB 2 tool assesses the risk of bias in RCTs across five domains: the randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result, providing an overall judgment of low risk, some concerns, or high risk. The ROBINS-I tool evaluates non-randomized studies across seven domains: bias due to confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes, and selection of the reported result, yielding overall ratings from low to critical risk.
A total of 536 articles were identified through the main search strategy, while no additional studies were identified through forward or backward citation tracking. After removing 35 duplicates, 501 studies were screened based on titles and abstracts. Of these, 435 were excluded, leaving 66 articles for full-text review. Fifty-four articles were not included for various reasons (e.g., not relevant, lacking rehabilitation content, published in languages other than English, etc.). Following the inclusion/exclusion process, the present SR includes 2 RCTs [48, 49] and 10 non-RCTs [13, 50–58]. The main characteristics of the included studies are reported in Table 2, and the entire inclusion/exclusion process is summarized in the PRISMA flow chart (Figure 2).
Main characteristics of the included studies.
| FA | YoP | SD | PT | NIP | Intervention | Comparator/Control | Outcome measures | Main findings | O-RoB |
|---|---|---|---|---|---|---|---|---|---|
| Moss [13] | 2024 | non-RCT | Aphasia with dysgraphia | 10 | Writing intervention using mainstream assistive technology | None | Writing accuracy, reading comprehension, functional communication | Improved writing and reading comprehension; benefits in functional communication | SR2 |
| Pisano [48] | 2021 | RCT | Chronic stroke | 14 | Dual-tDCS over the temporo-parietal cortex + writing tasks | Sham stimulation | Writing accuracy and speed | Active dual-tDCS enhanced writing performance; improvements stable post-treatment | SC1 |
| Marshall [49] | 2019 | RCT | Stroke with aphasia | 21 | Technology-enhanced writing therapy using assistive software | Usual care/waitlist | Functional writing, communication measures | Technology-based therapy improved functional writing performance | HR1 |
| Thiel [50] | 2016 | non-RCT | Stroke with writing impairment | 8 | Uni-modal vs multi-modal writing therapy | Between-condition comparison | Writing accuracy (trained/untrained), follow-up | Both therapies improved writing; no clear advantage of multi-modal; some gains not maintained at 6 weeks | SR2 |
| Johnson [51] | 2019 | non-RCT | Stroke with alexia and agraphia | 8 | Multi-step lexical + sublexical treatment | None | Reading and writing of trained/untrained words | Significant gains on trained items; generalization to related untrained items across modalities | SR2 |
| Thiel [52] | 2016 | non-RCT | Stroke with writing impairment | 45 | Learning-based writing therapy | None | Functional writing tasks | Improved functional writing; practice and error correction contributed to gains | SR2 |
| Mousavi [53] | 2025 | non-RCT | Stroke with chronic non-fluent aphasia | 6 | Persian writing treatment protocol | None | Writing accuracy for trained/untrained functional words | Significant improvement for trained words; stable results; no generalization to untrained words | SR2 |
| Simpson [54] | 2016 | non-RCT | Stroke with writing impairment | 7 | Home-based handwriting program | None | Handwriting legibility and speed | Feasible home-based program with preliminary handwriting improvements | CR2 |
| Thiel [55] | 2014 | non-RCT | Stroke with dysgraphia | 4 | Errorless vs errorful writing therapy | Errorless vs errorful comparison | Writing accuracy; follow-up | Both therapies effective; errorless slightly superior in one patient; gains maintained at 5 weeks | SR2 |
| Johansson-Malmeling [56] | 2022 | non-RCT | Stroke with spelling deficits | 6 | Digital spelling aid (tablet-based) | None | Spelling accuracy, generalisation tasks, usability | Significant improvements in spelling; some generalization; high usability and acceptance | SR2 |
| Curtis [57] | 2009 | non-RCT | Stroke with writing impairment | 5 | Interactive game-based handwriting rehab | None | Handwriting kinematics and legibility | Feasible and engaging; preliminary improvements in handwriting performance | CR2 |
| Raymer [58] | 2010 | non-RCT | Post-stroke acquired dysgraphia | 4 | Errorless spelling treatment | Errorful spelling treatment | Spelling accuracy (trained/untrained words) | Errorless training produced greater gains on trained words; some generalization across tasks | SR2 |
FA: first author; YoP: year of publication; SD: study design; PT: population type; NIP: number of included patients; O-RoB: overall Risk of Bias; tDCS: transcranial direct current stimulation; RCT: randomized clinical trial; SC: some concerns; HR: high risk of bias; SR: serious risk of bias; CR: critical risk of bias; 1: evaluated through the Risk of Bias 2 tool (RoB 2); 2: evaluated through the Risk Of Bias In Non-randomized Studies—of Interventions tool (ROBINS-I).
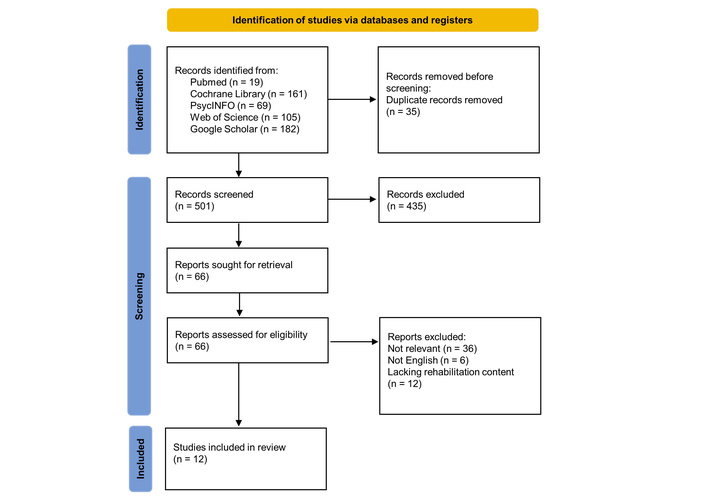
PRISMA flowchart. A total of 536 articles were identified, and after removing duplicates and screening titles and abstracts, 66 full-text articles were assessed. Following exclusions, 2 RCTs and 10 non-RCTs were included in the review. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; RCTs: randomized clinical trials. Adapted from [45]. © 2021 The Author(s). Distributed under a Creative Commons Attribution license (CC BY 4.0).
Across the 12 included studies, a wide variety of interventions were used to improve written language abilities and related language skills in the considered population. Although study designs, treatment components, and outcome measures varied substantially, the overall evidence supports the efficacy of targeted behavioral training, handwriting-focused interventions, technology-assisted programs, and neuromodulation in enhancing writing-related performance. Notably, several studies provided detailed insights into handwriting rehabilitation, referring to the grapho-motor component of writing, an area that remains comparatively underexplored but clinically relevant for functional communication.
To provide a structured overview, the following subsections summarize the interventions according to their main approaches: behavioral writing and spelling therapies (Behavioral writing and spelling interventions), technology-assisted programs with a focus on both functional writing and handwriting (Technology-assisted interventions), and neuromodulation approaches (Neuromodulation approaches). It should be noted that some studies could fit into more than one category, as interventions often combined behavioral, technology-assisted, and neuromodulation components. The current subdivision is intended for organizational clarity, but overlaps may exist.
A substantial portion of the reviewed studies investigated behavioral therapies grounded in lexical, sublexical, or learning-based principles. These interventions consistently improved accuracy on trained items and, in some cases, yielded generalisation to untrained stimuli. Learning-based written language treatments [50, 52] demonstrated gains in functional writing. These interventions emphasized the role of structured practice, feedback, and preserved learning mechanisms. Multi-step treatments integrating lexical and sublexical components [51] produced robust improvements in spelling. They also showed partial transfer to related words, supporting the value of hierarchical intervention protocols. Direct comparisons of therapeutic principles revealed that both errorless and errorful approaches can be effective. Errorless training, however, sometimes led to slightly superior outcomes and more stable maintenance over time [55, 58]. A structured, intensive behavioral intervention, recently released in the form of a protocol [53], further confirmed that intensive, structured practice can substantially improve trained functional words in chronic aphasia. Generalisation remains limited.
Several studies have explored the use of technology to support written language recovery. Technology-enhanced writing therapy using mainstream assistive software improved functional communication and writing accuracy [49]. Digital spelling aids facilitated significant gains in spelling accuracy, accompanied by positive user experiences [56]. Two studies specifically targeted handwriting and provided the clearest insights into motor aspects of written language recovery. Simpson et al. [54] evaluated a home-based handwriting program. They demonstrated that repeated practice can be delivered feasibly in domestic environments and may lead to improvements in legibility and motor fluency. Participant engagement was high, highlighting the acceptability of motor-focused writing rehabilitation. Curtis et al. [57] used interactive digital games to train handwriting movements in stroke survivors. Preliminary findings showed improvements in handwriting speed and control. These results suggest that motor learning principles—particularly repetition, feedback, and gamification—may enhance fine motor recovery for writing. Taken together, these results underscore that handwriting rehabilitation, though less often studied than lexical-orthographic interventions, represents a promising and feasible avenue for improving written communication. However, evidence remains preliminary, and sample sizes are small, indicating the need for more systematic research. Finally, Moss et al. [13] showed that practicing writing through commonly available assistive technology not only improved writing accuracy but also supported reading comprehension, suggesting productive cross-modal interactions.
In an RCT, Pisano et al. [48] investigated the effectiveness of transcranial direct current stimulation (tDCS) as an adjunct to writing or spelling interventions. For instance, the authors revealed that dual-site tDCS over temporo-parietal regions enhanced writing accuracy and supported maintenance of improvements after treatment. The arising data demonstrated modest and variable benefits of tDCS when paired with language training, indicating that neuromodulation may function as a supportive method.
In our review, 2 RCTs [48, 49] and 10 non-RCTs [13, 50–58] were included. Across the included studies, methodological quality varied substantially between randomized and non-randomized designs. Tables S1 and S2, provided in the appendix, present the RoB 2 assessment for the two RCTs and the ROBINS-I evaluation for the ten non-RCTs, respectively. With regard to RCTs, Marshall et al. [49] showed a high overall risk of bias, primarily due to concerns regarding the randomization process and deviations from intended interventions. Outcome measurement and selective reporting also raised concerns. In contrast, Pisano et al. [48] presented a robust methodological profile, with an overall low risk of bias, supported by appropriate randomization procedures, double blinding, and standardized outcome measures. Regarding the ten non-randomized studies, the majority of investigations fell within the “serious risk of bias” category, largely due to confounding, moderate concerns in participant selection, and issues in outcome measurement and reporting. Two studies [54, 57] were judged to be at critical risk of bias, reflecting substantial limitations in study design, particularly uncontrolled confounding and incomplete handling of missing data. No non-RCT achieved a low overall risk rating.
Overall, the risk-of-bias appraisal indicates that while the evidence base provides valuable insights into writing and handwriting rehabilitation after ABI, methodological limitations—especially in non-randomized designs—necessitate cautious interpretation of treatment effects.
The present SR sought to synthesize all existing evidence on behavioral, technological, and neuromodulatory approaches for enhancing written language abilities in individuals with ABI. Particular attention was given to interventions targeting handwriting, defined as the grapho-motor component of writing. Although the literature search was intentionally broad, encompassing all causes of ABI—including TBI, anoxic-hypoxic damage, and infectious or inflammatory encephalopathies—only studies conducted in post-stroke populations were identified. Therefore, it should be emphasized that the findings cannot be generalized to individuals with non-stroke ABI, and caution is warranted when extrapolating these results to other ABI aetiologies, as intervention effectiveness may differ. In line with previous studies [59, 60], this highlights a notable gap in the current evidence base, suggesting that the rehabilitation of handwriting and written language in non-stroke ABI remains largely unexplored.
Although the number of studies was modest and methodological heterogeneity considerable, the collective findings indicate that targeted rehabilitation can meaningfully improve several components of written communication—including spelling accuracy, functional writing, and, in some cases, handwriting quality. These results are particularly relevant given the central role of written language in daily life and the complex, multifactorial nature of post-stroke writing impairments, encompassing both linguistic and grapho-motor components [9, 10, 28, 59]. Behavioral interventions emerged as the most consistently investigated approach. The studies included in this SR demonstrate that structured training grounded in lexical, sublexical, or learning-based principles can yield significant improvements. Treatments emphasizing repeated practice, error management, and hierarchical learning—such as the multi-step protocols described by Johnson et al. [51] and the learning-based writing programs examined by Thiel and colleagues [50, 52]—were associated with improved performance on trained items and, in some cases, partial generalization to untrained stimuli. The balance between errorful and errorless practice continues to be debated, with some studies [55, 58] suggesting potential advantages for errorful training in long-term maintenance, while others report benefits of errorless learning for initial acquisition. Recent structured protocols, such as the Persian writing treatment protocol [53], further demonstrate that intensive behavioral practice can produce robust gains in individuals with chronic aphasia, though generalization remains a challenge. These findings align with theoretical models of relearning that emphasize preserved learning mechanisms and neural plasticity, even long after injury.
The growing use of technology in rehabilitation adds an important dimension to written language recovery. Several studies highlight the potential of assistive software and digital tools to complement or extend traditional therapy. For example, Marshall et al. [49] demonstrated that mainstream assistive technology can improve functional communication and spelling accuracy, while Johansson-Malmeling et al. [56] showed that digital spelling aids not only enhance performance but are also perceived positively by users, supporting their acceptability and feasibility in rehabilitation contexts.
A particularly noteworthy contribution of the reviewed literature concerns handwriting, a domain often neglected despite its importance for functional autonomy. Two studies focusing on targeted training—Simpson et al. [54] and Curtis et al. [57]—offered valuable insight into the motor aspects of written communication, providing evidence of feasibility, acceptability, and user engagement. Simpson and colleagues [54] reported that a structured, home-based handwriting program is feasible, engaging, and capable of improving legibility and motor fluency. In parallel, Curtis and colleagues [57] demonstrated that interactive, game-based motor training may enhance handwriting speed and fine-motor control by leveraging core principles of motor learning, including repetition, progressive difficulty, and real-time feedback. However, given the pilot nature of these studies, small sample sizes, and lack of controlled designs, conclusions regarding clinical efficacy remain preliminary. Despite these limitations, these findings underscore the clinical potential of explicitly targeting grapho-motor components within writing rehabilitation and highlight the need for adequately powered trials to establish efficacy.
While these studies demonstrate feasibility and functional benefits, the specific patient profiles that might benefit most from different technological interventions remain unclear. Importantly, the clinical benefits of technological interventions may vary depending on the patient’s specific profile, with different impacts for those with primarily linguistic versus grapho-motor impairments. It is plausible that assistive software may be particularly advantageous for individuals with central agraphia or spelling deficits, whereas handwriting-focused digital programs or gamified training could better support patients with pronounced grapho-motor impairments. Future research should aim to match technological approaches to patients’ clinical characteristics to optimize rehabilitation outcomes.
Neuromodulatory approaches, while less extensively studied, contribute an emerging and potentially valuable line of evidence [61, 62]. The RCT conducted by Pisano et al. [48] provided initial support for the use of dual-site tDCS over temporo-parietal regions as an adjunct to behavioral writing therapy. Their results indicate that tDCS may enhance spelling accuracy and help maintain treatment gains over time. However, the effects were modest and varied across participants, consistent with broader findings on neuromodulation in post-stroke language rehabilitation. The current evidence therefore suggests that tDCS is best conceptualized as a supplementary technique rather than a standalone treatment, with further research required to optimize stimulation parameters and clarify underlying mechanisms. Finally, the considerable inter-individual variability observed highlights the need for future studies to also identify clinical, cognitive, or neurofunctional markers that predict responsiveness to stimulation. Such markers would allow more targeted application of tDCS and could clarify which patients are most likely to benefit from adjunctive neuromodulatory interventions.
The interpretation of these findings must take into account the substantial methodological variability across studies. Only two RCTs [48, 49] were identified, one of which exhibited a high overall risk of bias. The ten non-randomized studies [13, 50–58] generally showed serious risks of bias, particularly with respect to confounding, participant selection, and outcome assessment, as indicated by the ROBINS-I evaluations. In addition, the conduct of a meta-analysis was not feasible due to multiple methodological limitations. Interventions differed markedly in therapeutic approach, treatment dosage, and delivery settings. Outcome measures were highly variable, ranging from spelling accuracy to functional writing and handwriting kinematics, and were often assessed using non-standardized tools. Furthermore, most studies involved small samples and non-randomized or single-case designs, limiting the availability of comparable effect size estimates. Collectively, these sources of clinical and methodological heterogeneity precluded meaningful quantitative synthesis and justified the adoption of a narrative SR approach. Another important methodological limitation concerns long-term maintenance, which was assessed in only a minority of studies, leaving questions about the durability of treatment effects unresolved.
Finally, an additional and important source of clinical heterogeneity concerns the presence and characterization of aphasia. In the majority of studies included in our review, aphasia was not considered as an a priori exclusion criterion. Moreover, it is reasonable to hypothesize that different aphasia profiles may differentially influence the rehabilitation trajectory of acquired dysgraphia, in a manner analogous to the recovery of motor functions. However, many studies failed to clearly specify the type or severity of aphasia in the included participants, thereby further limiting the interpretability and generalizability of the findings.
Despite these limitations, the collective findings offer meaningful clinical implications. Behavioral writing therapies remain the most evidence-supported approach, providing structured, theoretically grounded methods that can be adapted to different profiles of impairment. Technology-assisted interventions, including digital spelling aids and mainstream software, appear particularly promising for extending treatment intensity beyond clinical settings and enhancing patient engagement. The limited research on handwriting rehabilitation demonstrates that grapho-motor training is both feasible and potentially effective, suggesting that future rehabilitation models should more consistently address handwriting. Neuromodulation, although preliminary, represents an intriguing adjunctive technique that may enhance the effects of behavioral therapy, particularly as stimulation protocols become more refined.
In conclusion, from a clinical perspective, these findings suggest that handwriting interventions should be systematically integrated into interdisciplinary ABI rehabilitation programs, combining speech-language therapy, occupational therapy, and—when appropriate—technology-assisted and neuromodulatory approaches, in order to address both the written language and handwriting components and to promote functional, real-world communication outcomes.
From a broader perspective, the rehabilitation activities described in this review may also be interpreted within emerging frameworks that emphasize the role of health-promoting behaviors in maintaining and restoring brain function. The recently proposed concept of vitactions—defined as essential behaviors that support brain health and whose absence may contribute to physiological or neurological dysfunction—has been introduced as a non-pharmacological perspective on neuroprotection [63, 64]. Within this perspective, structured rehabilitation activities such as handwriting and writing practice could be viewed as targeted behavioral stimulations that promote neural plasticity and functional recovery following ABI.
Looking ahead, future research should prioritize more rigorous study designs, including adequately powered RCTs with standardized outcome measures and long-term follow-up. There is a need for greater ecological validity, with outcomes that reflect meaningful, functional writing tasks rather than isolated written language measures. Moreover, identifying the mechanisms through which specific interventions exert their effects—and understanding how linguistic, cognitive, and motor processes interact during writing—will be essential for developing individualized and comprehensive rehabilitation programs for individuals with ABI.
Written language rehabilitation after ABI remains poorly investigated and restricted exclusively to the stroke population. However, the literature available on this population type suggests potential benefits of intervention, but the overall evidence base remains weak due to small, heterogeneous studies.
In particular, behavioral therapies currently represent the most investigated intervention approach and show the most consistent, albeit preliminary, evidence—particularly for improving spelling accuracy and functional writing on trained items. However, the overall evidence base remains weak due to small, heterogeneous samples and a high risk of bias. Technology-assisted programs and neuromodulatory interventions should therefore be considered experimental or complementary, given the limited and low-quality evidence currently available.
Continued research is crucial to refine these methods, improve methodological quality, and ultimately enhance functional written communication and autonomy in individuals living with the long-term consequences of ABI. Importantly, future research should not be limited to post-stroke populations but should also include individuals with non-stroke ABI, such as traumatic or hypoxic-ischemic injuries. High-quality randomized trials with standardized protocols and meaningful functional outcomes related to written language, writing, and handwriting are urgently needed.
ABI: acquired brain injury
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
PROSPERO: International Prospective Register of Systematic Reviews
RCTs: randomized clinical trials
RoB 2: Risk of Bias 2 tool
ROBINS-I: Risk of Bias in Non-randomized Studies—of Interventions tool
SR: systematic review
TBI: traumatic brain injury
tDCS: transcranial direct current stimulation
The supplementary tables for this article are available at: https://www.explorationpub.com/uploads/Article/file/1004145_sup_1.pdf.
During the preparation of this work, the authors used ChatGPT (OpenAI, 2023; https://chatgpt.com/) to generate the brain icon in Figure 1. After utilizing the tool, the authors reviewed and edited the content as necessary and take full responsibility for the final content of the publication.
FL: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. ET: Data curation, Investigation, Formal analysis. GV: Validation, Supervision, Methodology, Visualization, Writing—review & editing, Project administration. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The datasets that support the findings of this study are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1065
Download: 24
Times Cited: 0
