Affiliation:
1National Institute of Agronomic Research, Errachidia 52000, Morocco
2Research Team on Health and Natural Resources Development, Laboratory of Natural Resources, Health and Environment, Faculty of Sciences and Techniques, Moulay Ismail University, Errachidia 52000, Morocco
Email: bouchra.ouhejjou@inra.ma
ORCID: https://orcid.org/0009-0001-1894-5622
Affiliation:
3Chemistry and Environment Research Team, Faculty of Sciences and Techniques, Moulay Ismail University, Errachidia 52000, Morocco
ORCID: https://orcid.org/0009-0004-4971-2006
Affiliation:
2Research Team on Health and Natural Resources Development, Laboratory of Natural Resources, Health and Environment, Faculty of Sciences and Techniques, Moulay Ismail University, Errachidia 52000, Morocco
4Multidisciplinary Laboratory in Exact and Applied Sciences, Higher School of Technology of Fkih Ben-Salah, Sultan Moulay Slimane University, Beni Mellal 23200, Morocco
ORCID: https://orcid.org/0000-0003-3275-3093
Affiliation:
5Environmental Technology, Biotechnology, and Valorization of Bio-resources Team, Laboratory of Research and Development in Engineering Sciences, Faculty of Science and Techniques Al-Hoceima, Abdelmalek Essaadi University, Tetouan 32000, Morocco
ORCID: https://orcid.org/0000-0002-1571-5354
Affiliation:
6National Institute of Agronomic Research, Oujda 60000, Morocco
ORCID: https://orcid.org/0009-0001-4443-0366
Affiliation:
1National Institute of Agronomic Research, Errachidia 52000, Morocco
ORCID: https://orcid.org/0000-0002-8366-4739
Affiliation:
7Bioresources, Environment and Health Team, NRHE-UMI Laboratory, Faculty of Sciences and Technology Errachidia, Moulay Ismail University, Errachidia 52000, Morocco
ORCID: https://orcid.org/0000-0001-7486-2664
Affiliation:
2Research Team on Health and Natural Resources Development, Laboratory of Natural Resources, Health and Environment, Faculty of Sciences and Techniques, Moulay Ismail University, Errachidia 52000, Morocco
Email: n.elalamielhassani@umi.ac.ma
ORCID: https://orcid.org/0000-0002-5247-7236
Affiliation:
2Research Team on Health and Natural Resources Development, Laboratory of Natural Resources, Health and Environment, Faculty of Sciences and Techniques, Moulay Ismail University, Errachidia 52000, Morocco
ORCID: https://orcid.org/0000-0001-6004-7319
Explor Foods Foodomics. 2026;4:1010143 DOI: https://doi.org/10.37349/eff.2026.1010143
Received: January 20, 2026 Accepted: April 12, 2026 Published: May 10, 2026
Academic Editor: Celestino Santos-Buelga, University of Salamanca, Spain
Aim: This study aimed to evaluate the physicochemical, functional, and microbiological quality of Tahlaout, a traditional date-based product from the Draa-Tafilalet region of Morocco. The main objective is to compare these characteristics with international standards, including Gulf date syrup (dibs) and Egyptian date honey, to assess their potential for market valorization and formal recognition.
Methods: A cross-sectional analytical and comparative study was conducted on thirteen Tahlaout samples collected from different women-led cooperatives. Physicochemical parameters, including pH, degree Brix (°Bx), water activity, dry matter (DM), ash content, reducing sugars, viscosity, and color, were determined using standardized methods. Functional and nutritional properties were assessed by quantifying total polyphenols, flavonoids, antioxidant activity, and mineral composition. Microbiological quality was evaluated by enumerating aerobic mesophilic flora, yeasts, molds, and total coliforms.
Results: The majority of Tahlaout samples complied with international quality standards and exhibited high levels of bioactive and nutritional compounds: mean total polyphenol content (TPC) of 26.68 mg GAE/gS (range: 14.16–33.88), mean total flavonoid content (TFC) of 14.46 mg RE/gS (range: 5.93–23.19), mean ferric reducing antioxidant power (FRAP) of 7.80 mmol Fe2+/gS (range: 3.52–11.50), and mean DPPH inhibition of 56.93% (range: 50.81–79.32). Mineral analysis yielded a mean iron content of 5.20 mg/100 g DM (4.27–6.58), zinc of 0.43 mg/100 g DM (range: 0.25–1.45), copper of 0.96 mg/100 g DM (range: 0.77–1.35), and manganese of 1.9 mg/100 g DM (range: 1.34–2.49). Microbiological analyses indicated generally satisfactory quality. However, elevated coliform and yeast counts were detected in certain samples, suggesting possible contamination during or after processing.
Conclusions: Tahlaout demonstrates strong potential as a high-quality traditional date-based product. Nevertheless, improvements in hygiene practices and the implementation of standardized certification protocols are necessary to ensure product safety and consistency. These findings support the formalization and sustainable development of the Tahlaout sector and contribute to the valorization of the date industry in Moroccan oasis regions.
The date palm (Phoenix dactylifera L.) is one of the oldest cultivated fruit crops in the world, grown primarily for its nutritious fruits, which serve as a staple food in many regions [1]. Dates are rich in bioactive compounds, including polyphenols, carotenoids, vitamins, and minerals, providing a significant source of dietary energy while exhibiting potential health-promoting and therapeutic effects against a range of diseases [2]. Their remarkable preservation properties, especially in dried form, further enhance their value in international trade [3].
In Morocco, date production is projected to reach approximately 160,000 tonnes during the 2025–2026 season, representing a substantial increase relative to previous years [4]. However, nearly half of this output is derived from low-market-value varieties that are frequently directed toward animal feed, despite their considerable potential for processing and value addition [5].
In the Draa-Tafilalet region, local communities have developed traditional methods for transforming dates into various products, including jams, fruit pastes, date flour, and most notably Tahlaout, an emblematic traditional sweet preparation, deeply rooted in local food culture.
Tahlaout is broadly comparable to industrial date-based syrups such as dibs (produced in Gulf countries) and Egyptian date honey, as it follows analogous production principles [6, 7]. However, it is crafted using traditional techniques that confer distinct sensory characteristics and significant cultural value. Across various Moroccan regions, this product is known by several local names, including Roub, Tizefte, Tamante, and Takayen. Despite its regional importance, Tahlaout remains largely unrecognized at both national and international levels.
A central challenge facing Tahlaout is the absence of regulatory frameworks governing its production and marketing in Morocco. Unlike other Arab countries, where established standards exist for date syrups, Morocco currently lacks specific regulations for this product, thereby limiting the structuring and development of this traditional sector. This regulatory gap prevents Tahlaout from obtaining official recognition and constrains its potential for wider commercialization and cultural valorization.
The present study constitutes the first comprehensive scientific evaluation of Tahlaout in Morocco, with a focus on the Draa-Tafilalet region, which accounts for approximately 76% of the national date production [4]. The objectives were to characterize its physicochemical, microbiological, nutritional, and antioxidant properties, and to compare these with existing standards for Gulf date syrups and Egyptian date honey, thereby providing a scientific basis for the valorization, standardization, and potential commercialization of this traditional Moroccan product.
Tahlaout samples were collected in autumn 2024 from thirteen cooperatives (out of 21 active at that time) located in the Draa-Tafilalet region of Morocco. The dates used for production belonged primarily to the Bouslikhene variety, alongside the Khalt and Boustahmmi varieties. For each cooperative, approximately 3 liters of freshly prepared Tahlaout were collected in sterile containers, transported to the laboratory under appropriate conditions, and stored at 4°C until analysis. Each batch was thoroughly homogenized, and a 100 mL aliquot was subsequently withdrawn as a representative sample for further analyses (Figure 1). Analyses were conducted sequentially, beginning with microbiological examinations, followed by chemical, physicochemical, and functional characterizations.

Tahlaout samples were collected from thirteen cooperatives in the Draa-Tafilalet region of Morocco.
An internal survey confirmed that standardized artisanal practices were consistently followed across cooperatives, ensuring both the representativeness of the samples and the relative uniformity of the production process. These steps comprised date harvesting, sorting, cleaning, cooking, filtration, juice concentration at 50°C, cooling, and packaging, as illustrated in Figure 2. All samples were transported to the laboratory under appropriate conditions, homogenized upon arrival, and analyzed in triplicate for each parameter to ensure the precision and reliability of the results.
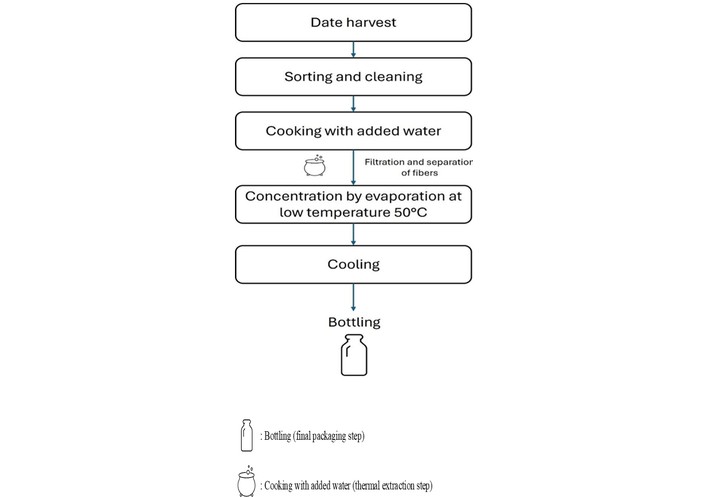
Schematic representation of the traditional Tahlaout production process as practiced by cooperatives of the Draa-Tafilalet region, from date harvesting to bottling.
All chemicals and reagents used in this study were of analytical grade. Formaldehyde, methanol, and hydrochloric acid were obtained from Reidel de Haen, HiPerSolv Chromanorm (Seelze, Germany). Sodium carbonate, 2,4,6-tripyridyl-s-triazine (TPTZ), and gallic acid were purchased from Fluka Co (Seelze, Germany). All other reagents were supplied by Sigma-Aldrich Chemical Co (St. Louis, Missouri, USA).
For microbiological analyses, specific culture media were selected based on the target microbial groups. Plate count agar was used to enumerate total mesophilic aerobic flora. Potato dextrose agar, a carbohydrate medium, was used to cultivate and enumerate yeasts and molds. Violet red bile lactose agar was utilized for total coliforms detection and quantification, as it supports the growth of Gram-negative enteric bacteria while inhibiting the growth of Gram-positive organisms. All culture media were supplied by Merck, Darmstadt, Germany.
The pH of the various Tahlaout samples was measured using a pH-meter (Thermo Scientific Orion A 211, Thermo Fischer Scientific, Waltham, MA, USA) according to AOAC Method No. 981.12 [8]. Titratable acidity was determined using the potentiometric titrimetric method [9]. Briefly, 5 g of date syrup was dissolved in 45 mL of distilled water. The solution was titrated with 0.05 M sodium hydroxide (NaOH) until a pH of 8.1 ± 0.2 was reached. Results are expressed as a percentage (%) of citric acid equivalent (g of citric acid per 100 g of syrup). Dry matter (DM) content was determined by drying 5 g of each sample at 70°C for 72 h in a WiseVen® oven (WiseVen®, Gyeonggi-do, South Korea), following AOAC Method No. 920.151 [9]. Total soluble content (°Bx) was measured using a portable digital refractometer (ATAGO Pocket Digital Refractometer, Tokyo, Japan). Water activity (aw) was determined using an aw-meter (LABSWIFT NOVASINA, Lachen, Switzerland), with 5 g of the sample placed in capsules, and equilibrium was reached after approximately 2 h [10]. Ash content was determined by incinerating 5 g of the sample at 500°C in an electric furnace (Ney® VULCAN D‑550, Oxnard, CA, USA), following AOAC Method No. 940.26 [9].
Reducing sugars were determined using the dinitrosalicylic acid (DNS) method, as described by Gail Lorenz Miller [11]. A reaction mixture containing 50 µL of Tahlaout extract (5 mg/mL), 250 µL of DNS reagent, and 700 µL of distilled water was heated at 100°C for 6 min. After cooling to room temperature, the absorbance was measured at 540 nm using a VIS-723G spectrophotometer (Beijing, China). A calibration curve was prepared using glucose standards (1–8 mg/mL). The reduced sugar content was expressed as milligrams of glucose equivalent per gram of syrup (mg GluE/gS) [12].
Total phenolic compounds, including polyphenols and flavonoids, were determined according to the method of Singleton and Rossi [13]. A volume of 100 µL of Tahlaout extract (5 mg mL–1) was mixed with 500 µL of 10% Folin-Ciocalteu reagent, then 400 µL of 7.5% sodium carbonate. After 30 min of incubation at room temperature, absorbance was measured at 765 nm using a UV-Vis spectrophotometer. Results were expressed as milligrams of gallic acid equivalent per gram of sample (mg GAE/gS).
Flavonoid content was determined by colorimetry based on the formation of flavonoid-aluminum complexes. A volume of 200 µL of extract was sequentially mixed with 100 µL of 5% sodium nitrite, 100 µL of 10% aluminum chloride, and 1 mL of 1 M sodium hydroxide. The mixture was incubated in the dark for 15 min, and absorbance was read at 510 nm. Flavonoid concentrations were expressed as milligrams of rutin equivalent per gram of sample (mg RE/gS).
The antioxidant activity of Tahlaout samples was assessed using two standard methods: the DPPH radical scavenging assay and the ferric reducing antioxidant power (FRAP) assay. For the DPPH test, 100 µL of each sample was mixed with 1 mL of a 0.1 mM DPPH solution in methanol and incubated in the dark for 20 min at room temperature. Absorbance was measured at 517 nm using a methanolic blank [14]. The percentage of inhibition was calculated as:
The FRAP assay evaluates the reduction of ferric (Fe3+) to ferrous (Fe2+) ions in an acidic medium. A volume of 25 µL of extract was added to 1 mL of freshly prepared FRAP reagent, composed of 30 mL of acetate buffer (300 mM, pH 3.6), 3 mL of 10 mM TPTZ in 40 mM HCl, and 3 mL of 20 mM FeCl₃. A standard curve was generated using FeSO₄ (0.1–1 mM). Results were expressed as mmol of Fe2+ equivalents per gram of extract (mmol Fe2+ E/gE), and reported as mean ± standard deviation from three replicates [15].
The mineral content was determined following the method described by Bouhlali et al. [9]. Two grams of each Tahlaout sample were ashed at 550°C, dissolved in HCl, evaporated, treated with H₂O₂ and deionized water, and diluted to 25 mL. The resulting solutions were analyzed in triplicate for copper, iron, manganese, and zinc using atomic absorption spectrometry (PerkinElmer Analyst 300, Waltham, United States), with results expressed in mg/100 g of DM.
Dynamic viscosity was measured with a Höppler viscometer (Königswinter, Germany) by timing a 14 mm nickel ball falling through the sample [16]. Viscosity, expressed in centipoise (cP), was then calculated using the equation:
Where:
K is an instrumental constant = 7;
ρ₁ is the density of the nickel ball = 8.1 g·cm–3;
ρ₂ is the density of the sample = 1.34 g·cm–3;
t represents the measured fall time.
The different samples’ color indices (L*, a*, b*) were measured using a Hunter MiniScan EZ colorimeter spectrophotometer (Model 4500L; Hunter Labs, Inc., Reston, USA). Measurements were performed according to the CIELab system, with three readings taken for each sample [17]. In this system, L* represents lightness, with values ranging from 0 (black) to 100 (white); a* indicates the position between green (negative values) and red (positive values); and b* reflects the position between blue (negative values) and yellow (positive values).
Microbiological analysis of Tahlaout samples was performed to determine microbial loads. Serial decimal dilutions were prepared by mixing 1 mL of sample with 9 mL of sterile Ringer’s solution to obtain a 10⁻1 dilution, followed by successive dilutions up to 10⁻2, each performed in triplicate. Appropriate aliquots were plated on selective media and incubated under aerobic conditions: mesophilic aerobic flora and total coliforms at 37 ± 2°C for 48 h, yeasts at 25°C for 72 h, and molds at 25°C for 120 h. Results were calculated as colony-forming units per gram (CFU/g) using the following equation:
Two serial dilutions (10⁻1 and 10⁻2) were prepared, each analyzed in triplicate for every sample. The mean values obtained were expressed as CFU/g. When required, data were subsequently converted to log₁₀ CFU/g for statistical analysis and presentation. The enumeration of yeasts, coliforms, and mesophilic aerobic flora (total aerobic mesophilic flora) was performed in accordance with ISO 21527-2:2008, ISO 4832:2006, and ISO 4833-1:2013, respectively [18, 19].
Statistical analyses were performed using IBM SPSS Statistics, version 26 (IBM Corp., Armonk, NY, USA). Initially, correlation analyses were conducted to assess the relationships between physicochemical parameters, including water activity (aw), soluble solids content (°Bx), pH, titratable acidity, dry matter (DM), ash content, reducing sugars, mineral composition, viscosity, and chromaticity, as well as microbiological analyses and bioactive compounds, such as total polyphenol content (TPC), total flavonoid content (TFC), and antioxidant activity. To further investigate the interrelationships among the measured variables, a principal component analysis (PCA) was performed. Prior to statistical testing, the normality of the data distribution was assessed. Variables exhibiting a normal distribution were analyzed using one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test for multiple comparisons, whereas non-normally distributed variables were analyzed using the Kruskal-Wallis non-parametric test. Differences with p-values less than 0.05 were considered statistically significant.
The physicochemical analysis of the thirteen Tahlaout samples collected across the Draa-Tafilalet region revealed significant variability in the measured parameters (Table 1). Soluble sugar content, expressed in °Bx, ranged from a minimum of 67.8 ± 0.2 °Bx (sample 4) to a maximum of 76.3 ± 0.1 °Bx (sample 10), with an overall average of 71.8 °Bx. The water activity (aw) remains low, oscillating between 0.62 ± 0.00 (sample 10) and 0.72 ± 0.00 (sample 2). Pearson correlation analysis demonstrated a strong and statistically significant negative correlation between aw and °Bx (r = −0.793; p < 0.01; n = 39).
Results of the physicochemical analyses of the thirteen Tahlaout samples developed in the Draa-Tafilalet region.
| Sample | Brix (°Bx) | aw | pH | Titrable acidity (%) | Dry matter (%) | Ash content (%) | Reducing sugars (mg GluE/gS) |
|---|---|---|---|---|---|---|---|
| 1 | 71.8 ± 0.2f | 0.69 ± 0.01cd | 3.83 ± 0.01ab | 0.561 ± 0.0097d | 81.2 ± 0.5h | 2.83 ± 0.4ab | 608.3 ± 22.1ab |
| 2 | 68.4 ± 0.5b | 0.72 ± 0.00e | 3.83 ± 0.00ab | 0.491 ± 0.0084c | 78.6 ± 0.9f | 2.64 ± 0.07ab | 577.2 ± 14.7ab |
| 3 | 70.3 ± 0.3d | 0.70 ± 0.00cd | 3.83 ± 0.03ab | 0.480 ± 0.0097c | 70.4 ± 0.5b | 3.07 ± 0.07abcd | 696.5 ± 32.6a |
| 4 | 67.8 ± 0.2a | 0.70 ± 0.00d | 3.95 ± 0.00b | 0.498 ± 0.00242c | 69.7 ± 0.8b | 2.50 ± 0.05a | 617.2 ± 3.7ab |
| 5 | 73.3 ± 0.2g | 0.69 ± 0.00cd | 3.89 ± 0.02b | 0.396 ± 0.04228b | 74.4 ± 0.2d | 2.99 ± 0.01abc | 650.7 ± 17.7ab |
| 6 | 74.7 ± 0.2h | 0.69 ± 0.01cd | 3.92 ± 0.02b | 0.442 ± 0.00242bc | 77.7 ± 0.5e | 3.86 ± 0.06e | 669.5 ± 42.3a |
| 7 | 70.9 ± 0.2e | 0.69 ± 0.01cd | 3.88 ± 0.05ab | 0.412 ± 0.02635c | 73.5 ± 0.4c | 3.4 ± 0.7acde | 698.1 ± 27.3ab |
| 8 | 70.0 ± 0.02cd | 0.68 ± 0.00c | 3.82 ± 0.03ab | 0.405 ± 0.00874c | 65.6 ± 0.2a | 3.65 ± 0.26de | 614.8 ± 51.4ab |
| 9 | 71.1 ± 0.05e | 0.69 ± 0.00cd | 3.88 ± 0.03ab | 0.462 ± 0.0042bc | 65.3 ± 0.2a | 3.54 ± 0.07cde | 618.1 ± 1.4ab |
| 10 | 76.3 ± 0.1i | 0.62 ± 0.00a | 4.12 ± 0.03c | 0.316 ± 0.02072a | 81.3 ± 0.2h | 2.64 ± 0.03ab | 658.9 ± 25.8ab |
| 11 | 69.8 ± 0.1c | 0.70 ± 0.00d | 4.38 ± 0.06d | 0.714 ± 0.042e | 73.3 ± 0.2c | 3.60 ± 0.05de | 560.1 ± 17.0bc |
| 12 | 74.5 ± 0.2h | 0.66 ± 0.00b | 3.68 ± 0.24a | 0.620 ± 0.08126d | 80.3 ± 0.5g | 3.44 ± 0.01cde | 658.1 ± 29.4ab |
| 13 | 74.7 ± 0.1h | 0.66 ± 0.00b | 4.10 ± 0.07c | 0.581 ± 0.00642d | 78.6 ± 0.3f | 3.17 ± 0.17bcd | 524.9 ± 7.5c |
| Average | 71.8 | 0.68 | 3.93 | 0.491 | 74.6 | 3.18 | 627.1 |
| Range | 67.8–76.3 | 0.62–0.72 | 3.68–4.38 | 0.316–0.714 | 65.3–81.3 | 2.50–3.86 | 524.9–698.1 |
* Values represent the mean ± standard deviation of three replicates (n = 3). Within the same column, means sharing the same letter are not significantly different according to Tukey’s post hoc test (p > 0.05). aw: water activity; °Bx: degree Brix.
The pH values of the samples ranged from 3.68 ± 0.24 (sample 12) to 4.38 ± 0.06 (sample 11). Titratable acidity varied from 0.316 ± 0.02% (sample 10) to 0.714 ± 0.04% citric acid (sample 11), with an average of 0.491%. A moderate but significant positive correlation was found between pH and titratable acidity (r = 0.317; p = 0.049; n = 39).
Dry matter (DM) content ranged from 65.3 ± 0.2% to 81.3 ± 0.2%, averaging 74.6%. The ash content, representing the mineral fraction, varied between 2.50 ± 0.05% and 3.86 ± 0.06% of DM. Regarding sugar composition, reducing sugars ranged from 524.9 ± 7.5 mg GluE/gS (52.5%) to 698.1 ± 27.3 mg GluE/gS (69.8%).
A comparison of the average physicochemical parameters (pH, DM, ash, and Brix) of Tahlaout with international standards for dibs and date honey is presented in Table 2.
Physicochemical properties of Tahlaout samples were compared to the established standards of dibs and date honey.
| Parameter | Tahlaout | Dibs | Date honey |
|---|---|---|---|
| pH | 3.93 | 4.00 | 4.60 |
| Dry matter (%) | 74.6 | 63.0 | 65.0 |
| Ash (%) | 3.18 | 2.00 | 1.80 |
| Brix (°Bx) | 71.8 | 70.0 | 75.0 |
°Bx: degree Brix
Colorimetric analysis shows that Tahlaout samples possess very low luminance (L*: 0.02 to 12.6; average: 1.9), low redness (a*: 0.07 to 16.5; average: 2.4), and low yellow-blue values (b*: 0.02 to 18.6; average: 2.1). Sample 10 stands out with significantly higher values (L* = 12.6, a* = 16.5, b* = 18.6) compared to others (Table 3).
Physical and rheological properties of the thirteen Tahlaout samples collected across the Draa-Tafilalet region, Morocco.
| Sample | Color parameters (L*, a*, b*) | Dynamic viscosity (cP) | ||
|---|---|---|---|---|
| L* | a* | b* | ||
| 1 | 0.15 ± 0.02a | 0.80 ± 0.07ab | 0.16 ± 0.03a | 1,275 ± 32a |
| 2 | 0.03 ± 0.01a | 0.17 ± 0.04a | 0.04 ± 0.01a | 2,554 ± 40b |
| 3 | 0.44 ± 0.28a | 1.4 ± 0.2b | 1.0 ± 0.2a | 1,056 ± 15af |
| 4 | 0.84 ± 0.05a | 2.9 ± 0.2c | 0.02 ± 0.02a | 9,520 ± 238c |
| 5 | 0.02 ± 0.01a | 0.07 ± 0.02a | 0.02 ± 0.00a | 2,572 ± 50b |
| 6 | 0.19 ± 0.06a | 0.61 ± 0.15ab | 0.42 ± 0.17a | 3,602 ± 513d |
| 7 | 10.2 ± 3.6b | 5.5 ± 1.3d | 5.0 ± 1.2b | 4,792 ± 215e |
| 8 | 0.16 ± 0.02a | 0.84 ± 0.04ab | 0.32 ± 0.03a | 784 ± 24f |
| 9 | 0.06 ± 0.03a | 0.32 ± 0.05ab | 0.09 ± 0.02a | 1,349 ± 73ah |
| 10 | 12.6 ± 0.3c | 16.5 ± 0.7e | 18.6 ± 0.9c | 1,063 ± 24af |
| 11 | 0.09 ± 0.02a | 0.51 ± 0.02ab | 0.17 ± 0.01a | 1,666 ± 48h |
| 12 | 0.4 ± 0.6a | 0.7 ± 0.4ab | 0.7 ± 0.9a | 2,475 ± 48b |
| 13 | 0.20 ± 0.03a | 0.90 ± 0.15ab | 0.28 ± 0.06a | 6,664 ± 95i |
| Average | 1.9 | 2.4 | 2.1 | 3,029 |
| Range | 0.02–12.6 | 0.07–16.5 | 0.02–18.6 | 784–9,520 |
Values represent the mean ± standard deviation of three replicates (n = 3). Within the same column, means sharing the same letter are not significantly different according to Tukey’s post hoc test (p > 0.05). cP: centipoise.
Dynamic viscosity shows extreme variability, ranging from 784 ± 24 cP (sample 8) to 9,520 ± 238 cP (sample 4), with a general average of 3,029 cP.
The samples exhibited TPC ranging from 14.16 ± 0.06 to 33.88 ± 0.3 mg GAE/gS (Table 4), while TFC ranged from 5.93 ± 0.17 to 23.19 ± 0.2 mg RE/gS. Antioxidant activity, as measured by DPPH assay, yielded inhibition percentages between 50.81 ± 2.3% and 79.32 ± 4.7%. FRAP values ranged from 3.52 ± 0.36 to 11.5 ± 0.35 mmol Fe2+ eq/100 g. Strong positive correlations were observed among TPC, TFC, and antioxidant activities (r = 0.837–0.973, p < 0.01).
Nutritional composition of the thirteen Tahlaout samples collected across the Draa-Tafilalet region, Morocco.
| Sample | Total polyphenol content (TPC) and total flavonoid content (TFC) | Antioxidant activity | Mineral elements | |||||
|---|---|---|---|---|---|---|---|---|
| TPC(mg GAE/gS) | TFC(mg RE/gS) | FRAP(mmol Fe2+/gS) | DPPH(% inhibition) | Iron(mg/100 g DM) | Zinc(mg/100 g DM) | Copper(mg/100 g DM) | Manganese(mg/100 g DM) | |
| 1 | 31.6 ± 0.9f | 18.8 ± 0.3f | 10.5 ± 0.4ef | 51.7 ± 0.8d | 4.95 ± 0.01bc | 0.25 ± 0.10ab | 1.35 ± 0.20b | 1.34 ± 0.01ab |
| 2 | 23.2 ± 0.2b | 10.5 ± 0.3c | 5.93 ± 0.11abc | 51.1 ± 0.8bc | 5.14 ± 0.50c | 0.28 ± 0.00a | 0.93 ± 0.10a | 1.51 ± 0.10ab |
| 3 | 31.0 ± 1.2f | 17.0 ± 0.1e | 9.01 ± 0.26def | 52.0 ± 5.1d | 5.53 ± 0.20abc | 0.30 ± 0.00a | 0.91 ± 0.10a | 1.75 ± 0.10abc |
| 4 | 29.3 ± 0.2e | 18.6 ± 0.8f | 8.14 ± 1.00cde | 51.5 ± 1.7d | 4.84 ± 0.10a | 0.43 ± 0.00ab | 0.93 ± 0.00a | 1.38 ± 0.00a |
| 5 | 27.2 ± 0.2d | 11.1 ± 0.3c | 4.80 ± 3.84ab | 50.81 ± 2.3bc | 5.31 ± 0.00abc | 0.36 ± 0.00a | 0.83 ± 0.10a | 1.86 ± 0.10abcd |
| 6 | 24.45 ± 0.06c | 9.19 ± 0.09b | 6.11 ± 0.27abc | 51.2 ± 0.2b | 6.08 ± 0.20d | 0.53 ± 0.00bc | 0.77 ± 0.10a | 1.97 ± 0.10cd |
| 7 | 15.0 ± 0.4a | 5.93 ± 0.17a | 3.52 ± 0.36a | 53.8 ± 0.2a | 4.76 ± 0.10abc | 0.42 ± 0.00abc | 0.90 ± 0.10a | 1.94 ± 0.00bcd |
| 8 | 33.5 ± 0.6g | 21.1 ± 0.1h | 10.4 ± 0.2ef | 58.7 ± 0.8e | 5.95 ± 0.10abc | 0.46 ± 0.10bc | 0.96 ± 0.00a | 2.12 ± 0.10abcd |
| 9 | 27.2 ± 0.4d | 15.5 ± 0.1d | 8.14 ± 0.06cde | 56.3 ± 2.4d | 6.58 ± 0.00c | 0.44 ± 0.00abc | 1.09 ± 0.00a | 2.31 ± 0.10d |
| 10 | 14.16 ± 0.06a | 6.06 ± 0.08a | 4.22 ± 0.98ab | 55.6 ± 4.8a | 4.68 ± 0.30ab | 0.49 ± 0.00c | 0.87 ± 0.00a | 1.95 ± 0.10cd |
| 11 | 31.0 ± 0.7f | 19.8 ± 0.4g | 10.3 ± 0.3ef | 65.6 ± 0.1e | 5.14 ± 0.20abc | 0.38 ± 0.00ab | 1.09 ± 0.10a | 2.40 ± 0.10d |
| 12 | 26.4 ± 0.4d | 11.3 ± 0.1c | 6.85 ± 0.7bcd | 61.5 ± 0.8c | 4.62 ± 0.10abc | 1.45 ± 0.10c | 0.98 ± 0.00 a | 2.47 ± 0.10d |
| 13 | 33.88 ± 0.3g | 23.19 ± 0.2i | 11.5 ± 0.35f | 79.32 ± 4.7f | 4.27 ± 0.10ab | 0.33 ± 0.00a | 1.02 ± 0.10 a | 2.49 ± 0.40d |
| Average | 26.68 | 14.46 | 7.80 | 56.93 | 5.20 | 0.43 | 0.96 | 1.9 |
| Range | 14.16–33.88 | 5.93–23.19 | 3.52–11.50 | 50.81–79.32 | 4.27–6.58 | 0.25–1.45 | 0.77–1.35 | 1.34–2.49 |
DM: dry matter; FRAP: ferric reducing antioxidant power. * Values are expressed as means ± standard deviation (n = 3). Means within the same column followed by the same letter are not significantly different according to Tukey’s post hoc test (p > 0.05).
Mineral analysis revealed notable levels of iron (4.27 to 6.58 mg/100 g DM), zinc (0.25 to 1.45 mg/100 g DM), copper (0.77 to 1.35 mg/100 g DM), and manganese (1.34 to 2.49 mg/100 g DM). To synthesize these findings, a PCA was performed on the correlation matrix, capturing 100% of the total variance across two components (PC1 = 59.14%; PC2 = 40.86%) (Figure 3). The variables were clustered into three distinct dimensions: (i) a physicochemical and mineral axis, predominantly oriented along PC1, associating aw, titratable acidity, iron, copper, and manganese. All exhibiting strong positive loadings on PC1; (ii) a bioactive axis, oriented along PC2, grouping TPC, TFC, and antioxidant activities (DPPH, FRAP), with DM and the a* color parameter also contributing to this dimension; and (iii) a color and pH axis, contrasting L*, zinc, and FRAP (negative PC1 loadings) to pH and b* (positive PC2 loadings). Statistical comparisons using Kruskal-Wallis and one-way ANOVA tests confirmed that inter-sample differences were statistically significant for all parameters (p < 0.001 to 0.003).
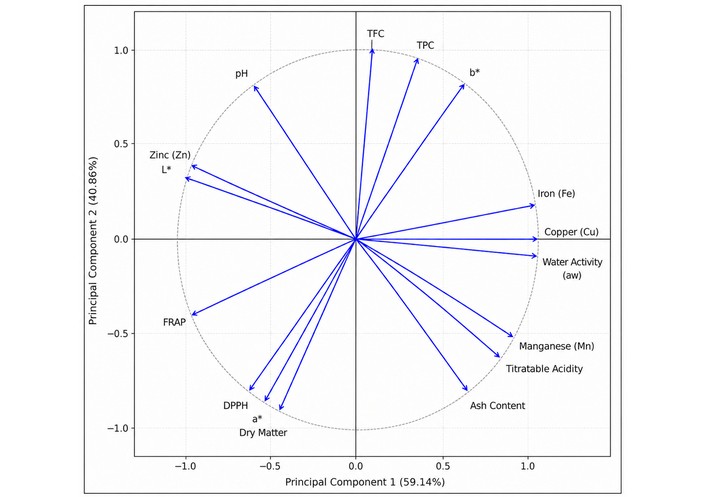
Principal component analysis loading plot of physicochemical, bioactive, and mineral properties of Tahlaout samples.
Microbiological analysis of the Tahlaout samples revealed that the total aerobic mesophilic flora was present in samples 4, 6, and 8, with levels of 3.39 ± 0.11 log CFU/g, 4.48 ± 0 log CFU/g, and 3.38 ± 0.08 log CFU/g, respectively (Table 5). Yeasts were detected in samples 4, 6, and 8, with concentrations between 2.22 ± 0.16 and 2.48 ± 0 log CFU/g, while molds were not detected in any samples. Total coliforms were identified in samples 2, 3, 4, 7, 9, and 12, with values ranging from 4.48 ± 0 to 5.21 ± 0 log CFU/g. Notably, samples 1, 5, and 11 showed no contamination.
Microbiological analysis of the thirteen Tahlaout samples collected across the Draa-Tafilalet region, Morocco.
| Sample | Total aerobic mesophilic flora (log CFU/g) | Yeasts (log CFU/g) | Molds (log CFU/g) | Total coliforms (log CFU/g) |
|---|---|---|---|---|
| 1 | ND | ND | ND | ND |
| 2 | ND | ND | ND | 5.16 ± 0b |
| 3 | ND | ND | ND | 5.06 ± 0a |
| 4 | 3.39 ± 0.11de | 2.48 ± 0de | ND | 5.21 ± 0d |
| 5 | ND | ND | ND | ND |
| 6 | 4.48 ± 0e | 2.22 ± 0.16e | ND | ND |
| 7 | ND | ND | ND | 4.48 ± 0a |
| 8 | 3.38 ± 0.08de | 2.48 ± 0de | ND | ND |
| 9 | ND | ND | ND | 4.84 ± 0b |
| 10 | ND | ND | ND | ND |
| 11 | ND | ND | ND | ND |
| 12 | ND | ND | ND | 4.66 ± 0a |
| 13 | ND | ND | ND | ND |
The values are expressed as log CFU/g (colony-forming units per gram). The superscript letters (a, b, d, e) indicate statistically significant differences between the sample means, analyzed by an ANOVA test followed by a Tukey test (p < 0.05). Samples marked with the same letter do not show a statistically significant difference (p > 0.05). “ND” indicates that the measurement was not detected in the sample. ANOVA: analysis of variance.
The physicochemical variability observed across the thirteen Tahlaout samples is a direct reflection of the diversity in date cultivars, artisanal extraction methods, and local storage conditions inherent to the Draa-Tafilalet region. The Brix values (average 71.8 °Bx) were high, yet notably lower than those reported for Tunisian date syrups [20]. This is a critical point for stability: while a minimum threshold of 74 °Bx is typically required to effectively inhibit microbial growth, previous research suggests that a range between 71 °Bx and 92 °Bx is favorable for storage stability [21]. Despite falling slightly below the 74 °Bx threshold in some samples, aw remained between 0.62 and 0.72. These levels are below the critical range (0.7 to 0.9) associated with microbial proliferation as cited by Bouhlali et al. [9], suggesting that Tahlaout is inherently bio-stable. This contrasts markedly with Algerian date syrups, where an aw of 0.92 has been reported, making them considerably more susceptible to microbiological deterioration [10].
The syrup acidity (pH 3.68–4.38) constitutes an additional hurdle against spoilage. These values are in close agreement with Bouhlali et al. [9] (pH 3.94–4.37) and Boussaid et al. [21] (pH 4.35–4.53). This natural acidity is likely attributed to the intrinsic organic acids of the date fruit combined with the specific storage conditions [22]. Generally, maintaining a low pH is a key favorable factor for the long-term preservation of the product [23]. Furthermore, the titratable acidity (0.316% to 0.714%) was consistent with Messaoudi and Fahloul (0.58%) [10], although lower than that documented in some traditional syrups (1.6% to 2.9%) by Bouhlali et al. [9]. This indicates that Tahlaout retains sufficient acidity for preservation without compromising its organoleptic qualities. The observed buffering effect, whereby pH increases slightly alongside titratable acidity, is a characteristic of the chemical behavior of organic acids in concentrated fruit matrices.
Regarding composition, the DM content (65.3% to 81.3%) exceeded values reported by Bouhlali et al. [9] (54.07% to 69.59%) and Boussaid et al. [21] (51.8% to 58.9%), likely owing to more intensive evaporation practices or the use of high-density cultivars by local cooperatives. Similarly, ash content (2.50% to 3.86%) was substantially higher than that of Algerian Mech-Deglet syrups (1.02% to 1.21%) [24], but perfectly aligned with the ranges documented by Djafri et al. [25] (1.73% to 3.70%). The total sugar content (average 71.8% sucrose) and reducing sugars (up to 69.8%) were comparable to syrups derived from Ghars and Deglet Nour varieties [26], confirming the high nutritional value of this product.
The physical attributes, particularly its dark, near-black color (low L*, a*, and b* values), are characteristic of traditional date concentrates and result from non-enzymatic browning and Maillard reactions occurring during processing, further intensified by the presence of bioactive polyphenols. Although these color parameters were lower than those reported by Bouhlali et al.’s [9], the dynamic viscosity (average 3,029 cP) exhibited considerable variation across samples. These values exceed those of Djafri et al. [25] (419 to 803.7 cP) but fall within the range reported by Al-Hilphy et al. [27]. The fact that some samples reached 9,520 cP while Barhi syrup has been shown to exceed 35,000 cP underscores the extent to which processing temperature and sugar polymerization degree govern the textural properties of Tahlaout [28].
From a nutritional standpoint, Tahlaout presents a complex profile. Total polyphenol concentrations were relatively low compared to those reported in a comparative study of date syrup and concentrate by Farahnaky et al. [29]. However, flavonoid concentrations (5.93 to 23.19 mg RE/gS) remained comparable to the values (11.93 mg RE/gS) measured by the same authors. Although polyphenol levels were lower than those found in the Saudi Khalas variety [30] or Algerian varieties such as Ghars [26], antioxidant activity (DPPH inhibition up to 79.32%) was significantly higher than that of Tunisian date syrups (14% to 23%) [20]. The FRAP values also exceeded those reported in studies comparing different production methods [31]. These findings suggest that Tahlaout may function as a potent functional food, particularly given its high mineral content. Iron and zinc levels were consistent with existing literature, while copper concentrations (up to 1.35 mg/100 g) were notably higher than those reported for Saudi (0.08 mg/100 g) and Tunisian (0.34 mg/100 g) varieties [32].
This nutritional complexity is further supported by the PCA results (Figure 3), which captured 100% of the total variance (PC1 = 59.14%; PC2 = 40.86%), revealing a clear multidimensional structure of Tahlaout syrup. Along PC2, the positive loadings of TFC, TPC, and b* contrasted with the negative loadings of DPPH, a*, and DM, indicating that color parameters and bioactive compounds are distributed across two opposing poles of this axis. Along PC1, the strong positive loadings of iron (Fe), copper (Cu), aw opposed the negative loadings of zinc (Zn), L*, pH, and FRAP, delineating a physicochemical and mineral axis that structurally separates hydration and trace mineral richness from color brightness and reducing antioxidant capacity. The intermediate positioning of manganese (Mn), titratable acidity, and ash content in the positive PC1/negative PC2 quadrant reflects the close association between mineral content and the acidic character of the syrup. Notably, FRAP projected distinctly toward the negative PC1 pole alongside zinc and pH, suggesting that reducing antioxidant capacity in Tahlaout may be linked to the alkaline mineral fraction rather than exclusively to phenolic content. This multidimensional coherence confirms that Tahlaout represents a significant source of essential minerals and constitutes a potent functional food with high antioxidant stability.
Comparison with international standards (Table 2) confirms that Tahlaout meets the benchmarks set by Gulf legislation [7] and Egyptian standards [6], attesting to its overall quality. However, the microbiological results presented in Table 5 reveal a significant concern. The presence of yeasts, coliforms, and mesophilic flora in some samples, despite low aw values, strongly suggests post-production contamination or insufficient standardization of hygienic practices (ONSSA Order No. 293-19/2019) [33]. Consequently, the formal recognition of Tahlaout within Moroccan oasis regulations is essential to guarantee sanitary quality and to support the transformation of this ancestral product into a competitive and sustainable industry for local communities.
The richness of Tahlaout in minerals, antioxidant compounds, and natural sugars demonstrates its considerable nutritional potential and underscores its value as a traditional product from Morocco’s oasis regions. Process optimization and standardization are essential to enhance traceability, hygiene management, and product consistency. Formalizing production and implementing quality standards would safeguard the sanitary integrity of Tahlaout and increase its competitiveness compared to dates, honey, and other fruit-based syrups on the market. Official recognition of Tahlaout as a certified regional product could support the sustainable development of oasis communities by integrating ancestral practices into modern agro-food value chains and promoting the valorization of underexploited date cultivars. While achieving such recognition represents a considerable challenge, it simultaneously offers a strategic opportunity for sector development and a promising economic pathway for local communities.
°Bx: degree Brix
ANOVA: analysis of variance
aw: water activity
CFU/g: colony-forming units per gram
cP: centipoise
DM: dry matter
DNS: dinitrosalicylic acid
FRAP: ferric reducing antioxidant power
PCA: principal component analysis
TFC: total flavonoid content
TPC: total polyphenol content
TPTZ: 2,4,6-tripyridyl-s-triazine
We extend our sincere gratitude to the thirteen cooperatives of the Draa‑Tafilalet region for generously providing samples and sharing their traditional know-how. We also warmly thank all organizations, administrations, and institutions whose support made this research possible.
BO: Conceptualization, Methodology, Formal analysis, Writing—original draft. ML: Methodology, Formal analysis, Writing—original draft. HE: Visualization, Formal analysis, Writing—original draft. HZ: Visualization, Validation. AM: Methodology, Formal analysis. EDTB: Formal analysis, Writing—review & editing. KS: Supervision, Validation, Writing—review & editing. NEAEH: Supervision, Validation, Writing—review & editing. CA: Supervision, Validation, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
All data generated and analyzed during this study are available from the corresponding author, Bouchra Ouhejjou at the following email address: bouchra.ouhejjou@inra.ma upon reasonable request.
The authors did not receive support from any organization for the submitted work.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 373
Download: 41
Times Cited: 0
