Affiliation:
2Department of Pharmacology, Central University of Punjab, Bathinda 151401, Punjab, India
Email: shivani.pharmacology11@gmail.com
ORCID: https://orcid.org/0000-0002-1755-8241
Explor Endocr Metab Dis. 2026;3:101471 DOI: https://doi.org/10.37349/eemd.2026.101471
Received: March 06, 2026 Accepted: April 28, 2026 Published: May 12, 2026
Academic Editor: Seung-Soon Im, Keimyung University School of Medicine, Republic of Korea
The article belongs to the special issue Role of Dysregulated Cytokine Signaling Pathways in Metabolic Disease
Metabolic diseases, including obesity, insulin resistance, and type 2 diabetes mellitus, are increasingly recognized as conditions characterized by chronic low-grade inflammation driven by dysregulated cytokine signaling. Persistent elevation of pro-inflammatory mediators disrupts insulin signaling and contributes to metabolic dysfunction. Nutritional interventions capable of restoring cytokine balance are emerging as promising adjunct strategies for metabolic disease management. Chickpea (Cicer arietinum L.), a widely consumed legume, is rich in polyphenols, saponins, dietary fiber, and bioactive peptides with potential immunometabolic benefits. This review critically summarizes the role of chickpea bioactives in modulating cytokine signaling networks, with particular emphasis on the IL-6-JAK-STAT3-SOCS3 axis and NF-κB pathway. Chickpea components exert antioxidant and anti-inflammatory effects, suppress pro-inflammatory cytokine production, and may improve insulin sensitivity. Additionally, their potential chemoprotective effects in obesity-associated metabolic disorders are discussed. Furthermore, this review highlights key gaps in mechanistic and translational research and proposes future directions for pathway-oriented investigations. Although current evidence supports the therapeutic potential of chickpea, further mechanistic and clinical validation is required.
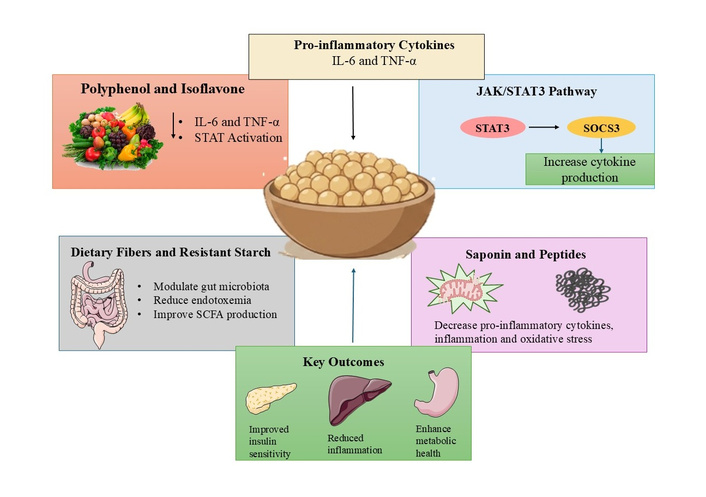
Metabolic diseases represent one of the most pressing global health challenges of the twenty-first century. The prevalence of obesity, insulin resistance, and type 2 diabetes mellitus (T2DM) has increased dramatically over recent decades, affecting both developed and developing nations. Obesity is now recognized not merely as a disorder of excess adiposity but as a complex metabolic condition characterized by endocrine and immune dysfunction. Persistent caloric excess and sedentary lifestyles promote adipose tissue expansion, ectopic lipid accumulation, and systemic metabolic disturbances that culminate in insulin resistance and hyperglycemia. Importantly, a growing body of evidence indicates that these disorders share a common inflammatory basis, leading to the conceptualization of “metabolic inflammation” or “meta-inflammation,” defined as chronic, low-grade, sterile inflammation driven by nutrient excess and metabolic stress rather than overt infection [1, 2]. This paradigm shift has transformed the understanding of metabolic disease pathogenesis from a purely metabolic imbalance to a state of sustained immune activation.
Central to meta-inflammation is the dysregulated production and signaling of pro-inflammatory cytokines within metabolically active tissues, particularly adipose tissue. In obesity, adipocyte hypertrophy and hypoxia promote immune cell infiltration, especially macrophages, which adopt a pro-inflammatory M1 phenotype. These cells secrete elevated levels of tumor necrosis factor-α (TNF-α), interleukin (IL)-6, and IL-1β, creating a self-perpetuating inflammatory milieu [3, 4]. TNF-α impairs insulin signaling by inducing serine phosphorylation of insulin receptor substrate-1 (IRS-1), thereby disrupting downstream phosphatidylinositol 3-kinase (PI3K)/Akt activation [5]. IL-6, although pleiotropic in function, contributes to hepatic glucose overproduction and systemic insulin resistance when chronically elevated, largely through activation of the Janus kinase (JAK)/signal transducer and activator of transcription (STAT) pathway [6]. IL-1β further exacerbates metabolic dysfunction by impairing pancreatic β-cell function and promoting apoptosis [7]. Sustained activation of these cytokines results in persistent intracellular signaling cascades, including nuclear factor-κB (NF-κB) and JAK/STAT pathways, which amplify inflammatory gene expression and interfere with metabolic homeostasis.
The JAK/STAT signaling axis occupies a pivotal position in cytokine-mediated metabolic dysregulation. Binding of cytokines such as IL-6 to their cognate receptors activates JAK kinases, leading to phosphorylation and nuclear translocation of STAT transcription factors, particularly STAT3. Activated STAT3 induces transcription of suppressor of cytokine signaling (SOCS) proteins, notably SOCS3, which serve as negative feedback regulators but paradoxically contribute to insulin resistance by directly inhibiting insulin receptor signaling [8]. This intricate immune-metabolic crosstalk underscores how inflammatory pathways intersect with classical metabolic signaling networks. Consequently, targeting cytokine signaling pathways has emerged as a promising therapeutic approach in metabolic disorders.
Pharmacological inhibitors of JAK kinases and other inflammatory mediators have demonstrated efficacy in autoimmune and inflammatory diseases; however, their application in metabolic disorders remains limited by safety concerns, immunosuppression, high cost, and long-term tolerability issues [9]. Moreover, metabolic diseases often require chronic management strategies, making sustained pharmacological inhibition of immune pathways less desirable. These limitations have stimulated interest in complementary and preventive approaches, particularly nutritional strategies capable of modulating inflammatory signaling without profound immunosuppression. Dietary bioactive compounds, including polyphenols, saponins, peptides, and fermentable fibers, have been shown to influence cytokine production, NF-κB activation, oxidative stress, and intracellular kinase signaling pathways [10]. Such compounds may act either directly on immune cells or indirectly through modulation of gut microbiota and short-chain fatty acid (SCFA) production, thereby influencing systemic cytokine profiles and metabolic outcomes.
Among functional foods, chickpea (Cicer arietinum L.) has garnered considerable attention due to its rich nutritional and phytochemical composition. As one of the most widely consumed legumes globally, chickpea provides high-quality protein, complex carbohydrates, dietary fiber, vitamins, and minerals. In addition to its nutritional value, it contains diverse bioactive constituents, including isoflavones, phenolic acids, saponins, bioactive peptides, and resistant starch, which have been associated with antioxidant, hypolipidemic, and anti-inflammatory effects [11, 12]. Emerging experimental and clinical evidence suggests that chickpea consumption can reduce circulating inflammatory markers, modulate adipokine secretion, and improve insulin sensitivity.
Despite these promising findings, the specific molecular mechanisms underlying these effects particularly in relation to cytokine signaling pathways, such as the JAK/STAT/SOCS axis, remain insufficiently characterized. Given the central role of cytokine dysregulation in metabolic disease and the growing interest in dietary immunomodulation as a complementary strategy, a pathway-oriented evaluation is warranted. Therefore, this review aims to critically synthesize current evidence on the role of chickpea bioactives in modulating dysregulated cytokine signaling pathways in metabolic disease, with a focus on mechanistic insights and translational relevance.
Obesity-associated metabolic dysfunction is characterized by sustained elevation of pro-inflammatory cytokines within adipose tissue and systemic circulation. Hypertrophic adipocytes and infiltrating macrophages form an inflammatory microenvironment that promotes chronic activation of immune signaling cascades. Among the key mediators, TNF-α plays a pivotal role in linking inflammation to insulin resistance. Increased TNF-α expression in adipose tissue correlates with body mass index and systemic insulin resistance [13]. Mechanistically, TNF-α impairs insulin signaling by inducing serine phosphorylation of IRS-1, thereby inhibiting its normal tyrosine phosphorylation by the insulin receptor. This modification attenuates downstream PI3K/Akt activation and reduces glucose transporter type 4 (GLUT4) translocation, ultimately decreasing cellular glucose uptake [14]. TNF-α also enhances lipolysis and increases circulating free fatty acids, further aggravating hepatic insulin resistance and metabolic stress.
IL-6 exhibits a dual and context-dependent role in metabolic regulation. Acute, transient increases in IL-6, such as those observed during physical exercise, may enhance glucose uptake and lipid oxidation. In contrast, chronic elevation of IL-6 in obesity contributes to hepatic gluconeogenesis, adipose tissue inflammation, and systemic insulin resistance [15]. Persistent IL-6 signaling sustains activation of intracellular pathways that interfere with insulin receptor signaling and amplify inflammatory gene expression. Thus, while IL-6 may exert protective effects in physiological contexts, its prolonged overproduction in obesity promotes metabolic dysregulation. IL-1β is another critical mediator of metabolic inflammation, particularly in pancreatic islets. Elevated IL-1β levels impair β-cell function by inducing nitric oxide production, oxidative stress, and apoptosis [16]. In obesity and type 2 diabetes, activation of the NLRP3 inflammasome enhances cleavage of pro-IL-1β into its mature form, intensifying local islet inflammation and progressive β-cell failure [17]. Collectively, TNF-α, IL-6, and IL-1β establish a chronic inflammatory milieu that disrupts insulin signaling across multiple metabolic tissues.
The JAK/STAT pathway serves as a central conduit for cytokine-mediated metabolic effects. The binding of IL-6 to its receptor complex, composed of IL-6 receptor α (IL-6Rα) and glycoprotein 130 (gp130), induces conformational changes that activate associated JAK kinases, primarily JAK1 and JAK2. Activated JAKs phosphorylate specific tyrosine residues on the receptor cytoplasmic domain, creating docking sites for STAT3. Subsequent phosphorylation of STAT3 promotes its dimerization and nuclear translocation, where it regulates transcription of genes involved in inflammation, cell survival, and metabolic control [18].
One of the principal STAT3 target genes is SOCS3, a classical negative feedback regulator. SOCS3 binds to phosphorylated JAKs and receptor complexes to attenuate further STAT activation, thereby limiting cytokine signaling. However, in chronic inflammatory states such as obesity, sustained IL-6-JAK-STAT3 activation leads to persistent SOCS3 expression, which paradoxically contributes to insulin resistance [19]. SOCS3 can interact directly with the insulin receptor and IRS proteins, inhibiting their tyrosine phosphorylation and promoting ubiquitin-mediated degradation. Elevated SOCS3 expression in liver and adipose tissue has been associated with impaired insulin signaling and glucose intolerance in experimental models [20].
Thus, while the JAK/STAT pathway is physiologically essential for immune regulation, its chronic activation in metabolic disease promotes maladaptive signaling loops that exacerbate insulin resistance. Persistent STAT3 activity and SOCS3 overexpression represent critical molecular links between cytokine dysregulation and metabolic impairment.
In parallel with JAK/STAT activation, the NF-κB pathway functions as a master regulator of inflammatory gene transcription in obesity. Elevated levels of circulating saturated fatty acids and lipopolysaccharide (LPS) in obese individuals stimulate toll-like receptor (TLR4) signaling on adipocytes and macrophages [21]. Activation of TLR4 recruits adaptor proteins such as MyD88, leading to stimulation of the IκB kinase (IKK) complex. Phosphorylation and subsequent degradation of IκB release NF-κB dimers, allowing their nuclear translocation and transcriptional activation of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β.
This NF-κB-mediated cytokine production establishes an amplification loop in which newly synthesized cytokines further activate JAK/STAT signaling and reinforce inflammatory responses. Crosstalk between NF-κB and STAT3 intensifies this effect, as STAT3 can cooperate with NF-κB at promoter regions to enhance transcription of inflammatory genes [22]. Such coordinated signaling sustains chronic low-grade inflammation and perpetuates insulin resistance in metabolic tissues.
The convergence of TLR4/NF-κB and IL-6/JAK/STAT pathways therefore constitutes a mechanistic nexus in obesity-associated metabolic dysfunction. Targeting these interconnected signaling networks offers a rational strategy for restoring immune-metabolic balance in metabolic disease.
Chickpea is one of the most widely cultivated and consumed grain legumes worldwide, serving as a major source of plant-based protein in Asia, the Middle East, the Mediterranean region, and parts of Africa. It is broadly classified into two main types, Desi and Kabuli, which differ in seed morphology and phytochemical composition. Beyond its macronutrient profile, chickpea contains a diverse array of bioactive constituents with potential metabolic and immunomodulatory properties. Its nutritional matrix includes high-quality proteins (18–24%), complex carbohydrates, dietary fiber, essential amino acids, unsaturated fatty acids, vitamins, and minerals [23, 24]. Importantly, chickpea also provides a rich spectrum of phytochemicals that may influence inflammatory signaling pathways implicated in metabolic disease.
Chickpea seeds contain substantial quantities of polyphenolic compounds, including phenolic acids (such as ferulic, caffeic, and p-coumaric acids) and flavonoids. These compounds exhibit antioxidant and anti-inflammatory activities that can modulate redox-sensitive signaling cascades. Polyphenols are known to inhibit NF-κB activation, reduce reactive oxygen species (ROS) production, and attenuate pro-inflammatory cytokine release in experimental models [25].
Isoflavones, structurally similar to estrogenic compounds, are also present in chickpea, although in lower concentrations compared to soybeans. Isoflavones such as biochanin A and formononetin have demonstrated anti-inflammatory effects through modulation of cytokine production and inhibition of pro-inflammatory transcription factors [26]. By influencing intracellular kinase activity and transcriptional regulators, chickpea-derived polyphenols may indirectly affect JAK/STAT signaling dynamics under inflammatory conditions.
Saponins constitute another important class of bioactive phytochemicals in chickpea. These amphipathic glycosides possess cholesterol-lowering, antioxidant, and anti-inflammatory properties. Chickpea saponins, particularly soyasaponins, have been reported to modulate immune cell activity and suppress production of TNF-α and IL-6 in inflammatory models [27]. Mechanistically, saponins may alter membrane microdomain organization, interfere with receptor-mediated signaling, and inhibit activation of NF-κB and mitogen-activated protein kinase (MAPK) pathways. Through attenuation of upstream inflammatory stimuli, saponins may indirectly reduce activation of cytokine-driven JAK/STAT signaling in metabolic tissues.
Enzymatic hydrolysis and gastrointestinal digestion of chickpea proteins generate bioactive peptides with functional properties extending beyond basic nutrition. These peptides have been shown to exert angiotensin-converting enzyme (ACE) inhibitory, antioxidant, and anti-inflammatory activities [28]. Certain legume-derived peptides can suppress pro-inflammatory cytokine expression and modulate macrophage polarization, thereby influencing systemic inflammatory tone. Although direct evidence linking chickpea peptides to JAK/STAT pathway modulation remains limited, their capacity to reduce TNF-α and IL-6 production suggests a plausible role in attenuating cytokine-mediated metabolic disturbances.
Chickpea is particularly rich in dietary fiber, including soluble fiber and resistant starch, which escape digestion in the small intestine and undergo fermentation by colonic microbiota. This fermentation process produces SCFAs such as acetate, propionate, and butyrate. SCFAs exert anti-inflammatory effects by enhancing gut barrier integrity, reducing endotoxemia, and modulating immune cell function [29]. Butyrate, in particular, has been shown to inhibit NF-κB activation and promote regulatory T-cell differentiation, thereby contributing to systemic immune homeostasis [30].
By altering gut microbiota composition and decreasing LPS-mediated TLR4 activation, chickpea fiber may reduce downstream cytokine production and inflammatory signaling cascades. This gut-immune-metabolic axis represents an indirect yet potentially powerful mechanism through which chickpea consumption may influence cytokine dysregulation in metabolic disease. Collectively, the major chickpea bioactives and their proposed molecular targets within cytokine signaling pathways are summarized in Table 1.
Chickpea bioactives and their modulatory effects on cytokine signaling pathways in metabolic disease.
| Chickpea bioactive class | Representative compounds | Targeted cytokine/Pathway | Specific molecular event affected | Metabolic outcome | Ref |
|---|---|---|---|---|---|
| Polyphenols | Quercetin, kaempferol | TNF-α/NF-κB | Inhibition of IκB degradation and NF-κB nuclear translocation | Reduced pro-inflammatory cytokine transcription; improved insulin sensitivity | [31, 32] |
| Polyphenols | Flavonoids | IL-6/JAK/STAT3 | Suppression of JAK-mediated STAT3 phosphorylation | Decreased SOCS3 induction; improved insulin signaling | [33, 34] |
| Isoflavones | Biochanin A | IL-6/STAT3 | Inhibition of STAT3 activation and DNA binding | Attenuation of inflammatory gene expression | [35–38] |
| Saponins | Soyasaponins | TLR4/NF-κB | Reduced TLR4-mediated cytokine amplification | Lower systemic inflammation | [39, 40] |
| Dietary fiber | Soluble fiber | TLR4/Endotoxin pathway | Improved gut barrier; reduced LPS translocation | Decreased metabolic endotoxemia | [41, 42] |
| Resistant starch | RS type I & II | IL-6/Gut microbiota-Inflammatory axis | Reduced IL-6 levels | Improved glycemic control | [43, 44] |
| Bioactive peptides | Seed-derived peptides | Oxidative stress-NF-κB axis | Reduction of ROS-mediated inflammatory signaling | Protection of the IRS-1 function | [45] |
| Indirect pathway effect | Whole chickpea consumption | STAT3-SOCS3 axis | Reduced circulating IL-6 → lower SOCS3 expression | Restoration of insulin receptor signaling | [46, 47] |
IL: interleukin; IRS-1: insulin receptor substrate-1; JAK: Janus kinase; LPS: lipopolysaccharide; NF-κB: nuclear factor kappa B; ROS: reactive oxygen species; SOCS3: suppressor of cytokine signaling 3; STAT3: signal transducer and activator of transcription 3; TLR4: toll-like receptor 4; TNF-α: tumor necrosis factor-α.
Accumulating experimental evidence suggests that chickpea-derived bioactive compounds attenuate the production of key pro-inflammatory cytokines implicated in metabolic disease, particularly TNF-α and IL-1β. In vitro studies using legume extracts have demonstrated suppression of LPS-induced TNF-α release in activated macrophages, largely through inhibition of upstream inflammatory signaling pathways [48]. Phenolic-rich fractions isolated from chickpea have been shown to reduce oxidative stress and downregulate inflammatory mediators in cellular models, indicating a potential role in dampening cytokine overproduction [49]. In animal models of diet-induced obesity, incorporation of chickpea into high-fat diets has been associated with reduced circulating TNF-α levels and decreased adipose tissue inflammation [50]. These effects are likely mediated by combined actions of polyphenols, saponins, and fiber components that limit macrophage infiltration and shift macrophage polarization away from the pro-inflammatory M1 phenotype. By lowering TNF-α availability, chickpea bioactives may indirectly preserve IRS-1 function and improve downstream insulin signaling.
IL-1β, a cytokine closely linked to inflammasome activation and β-cell dysfunction, may also be influenced by legume-derived compounds. Although direct mechanistic studies specifically evaluating chickpea and IL-1β maturation remain limited, plant-derived polyphenolic compounds have demonstrated inhibitory effects on NLRP3 inflammasome activation and IL-1β secretion in experimental systems [51]. Given the structural and functional similarities among legume phytochemicals, it is plausible that chickpea bioactives exert comparable modulatory effects on inflammasome-mediated cytokine release, thereby contributing to protection against metabolic inflammation.
Moreover, chronic elevation of IL-6 is a hallmark of obesity-related metabolic dysfunction. Dietary intervention studies incorporating chickpea or chickpea-based products have reported reductions in systemic inflammatory markers, including C-reactive protein (CRP), a downstream surrogate of IL-6 signaling [52]. While direct measurements of IL-6 in human chickpea intervention trials remain relatively sparse, improvements in inflammatory status and insulin sensitivity suggest modulation of upstream cytokine networks. In preclinical models, legume-rich diets have been shown to attenuate IL-6 expression in adipose tissue and liver [53]. These effects may be attributed to decreased activation of NF-κB and reduced oxidative stress.
In addition to suppressing pro-inflammatory cytokines, emerging evidence indicates that legume-derived bioactives may influence immunoregulatory cytokines such as IL-10, which plays a critical role in limiting excessive immune activation. Polyphenols and dietary fiber have been reported to enhance IL-10 production and promote anti-inflammatory immune responses in experimental models [54, 55]. Although direct evidence specific to chickpea remains limited, such effects may contribute to the restoration of cytokine balance and immune regulation in metabolic disease.
Additionally, chickpea consumption has been associated with improvements in metabolic and inflammatory profiles, which may indirectly influence adipokine regulation through modulation of gut microbiota and systemic inflammation [56]. Because adipokines interact closely with cytokine signaling pathways, normalization of adiponectin-leptin balance may further suppress IL-6 production and downstream JAK/STAT activation. The fiber fraction of chickpea also contributes indirectly to IL-6 regulation through modulation of gut microbiota composition. Fermentation of resistant starch generates SCFAs, which have been shown to reduce systemic inflammatory cytokines and enhance metabolic homeostasis [57]. By decreasing endotoxemia and TLR4 stimulation, chickpea fiber may attenuate IL-6 synthesis at its source.
Beyond their anti-inflammatory and antioxidant properties, chickpea bioactives may exert broader immunomodulatory effects by influencing immune cell function and cytokine balance. Emerging evidence suggests that dietary polyphenols and saponins can modulate macrophage polarization, promoting a shift from pro-inflammatory M1 phenotype toward an anti-inflammatory M2 phenotype. This transition is associated with restoration of immune homeostasis rather than generalized immune suppression. In addition, chickpea-derived components may contribute to balancing pro- and anti-inflammatory cytokines, thereby maintaining appropriate immune responsiveness while limiting chronic low-grade inflammation. Such immunoregulatory effects are particularly relevant in metabolic disease, where persistent immune activation rather than acute immune response drives pathology [58].
Moreover, chickpea bioactives may also exert context-dependent immunomodulatory effects that differ between metabolically compromised and healthy individuals. In individuals with normal immune function, dietary polyphenols and fiber-rich foods are not associated with immunosuppression but rather contribute to the maintenance of immune homeostasis. These compounds primarily attenuate chronic low-grade inflammation without impairing acute immune responses required for host defense against pathogens. Evidence from nutritional immunology suggests that plant-based diets rich in legumes support balanced cytokine responses and immune resilience, partly through modulation of gut microbiota and systemic inflammatory tone [59, 60]. Molecular mechanisms linking chickpea bioactives to cytokine signaling modulation in metabolic disease is illustrated in Figure 1.
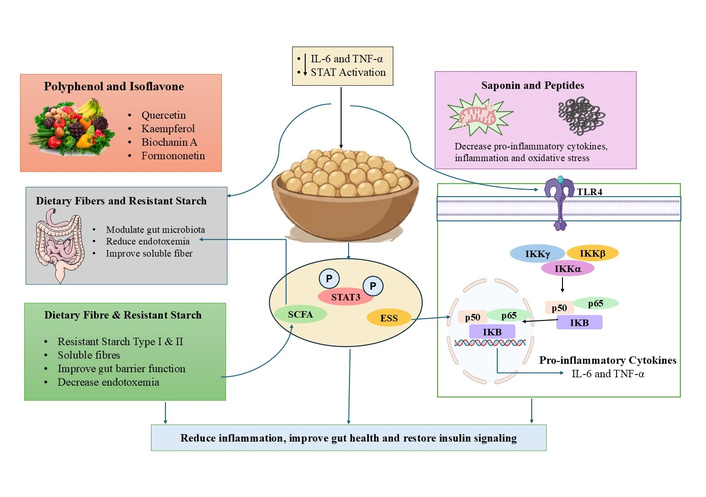
Molecular mechanisms linking chickpea bioactives to cytokine signaling modulation in metabolic disease. Figure created using PowerPoint Presentation. Reduced STAT3 activation limits transcription of SOCS3, thereby restoring IRS-1 signaling. Saponins and bioactive peptides attenuate TLR4-mediated NF-κB activation and oxidative stress, reducing inflammatory cytokine amplification. Dietary fiber and resistant starch enhance gut barrier integrity and promote SCFA production, decreasing metabolic endotoxemia and downstream cytokine activation. Collectively, these coordinated effects reduce chronic low-grade inflammation, improve insulin sensitivity, and support metabolic homeostasis. ESS: endoplasmic stress signalling; IKK: IκB kinase; IRS-1: insulin receptor substrate-1; SCFA: short-chain fatty acid; SOCS 3: suppressor of cytokine signaling 3; STAT: signal transducer and activator of transcription; TLR4: toll-like receptor 4.
NF-κB acts as a central transcriptional regulator controlling the expression of TNF-α, IL-6, and IL-1β, thereby sustaining the inflammatory state observed in metabolic disorders. Chickpea-derived phytochemicals, particularly polyphenols and saponins, have demonstrated the capacity to suppress NF-κB activation through multiple mechanisms. These compounds can stabilize IκB, preventing nuclear translocation of NF-κB subunits and reducing transcription of pro-inflammatory genes [61].
Saponins have also been shown to inhibit MAPK pathways, which further contribute to inflammatory gene expression [62]. In addition, oxidative stress is a key activator of NF-κB signaling, and the antioxidant properties of chickpea bioactives may reduce ROS-mediated activation of this pathway [63]. Through these combined actions, chickpea bioactives may disrupt the cytokine amplification loop and attenuate chronic inflammation associated with metabolic disease. Importantly, suppression of NF-κB signaling also reduces upstream cytokine production, thereby indirectly influencing downstream pathways such as JAK/STAT, highlighting the interconnected nature of inflammatory signaling networks.
In contrast to NF-κB-mediated transcriptional regulation, the JAK-STAT pathway primarily governs cytokine signal transduction and downstream metabolic responses. Persistent elevation of IL-6 in obesity promotes activation of JAK, leading to phosphorylation and nuclear translocation of STAT3, which drives the expression of inflammatory and metabolic regulatory genes [64]. Chronic activation of STAT3 induces SOCS3, which acts as a negative feedback regulator but paradoxically contributes to insulin resistance. SOCS3 interferes with insulin receptor signaling by inhibiting tyrosine phosphorylation of IRSs and promoting their degradation, thereby impairing PI3K/Akt signaling [65, 66].
Although direct studies evaluating chickpea-specific effects on STAT3 activation remain limited, polyphenolic compounds present in chickpea have demonstrated inhibitory effects on STAT3 phosphorylation by reducing upstream kinase activity and oxidative stress-mediated signaling [67, 68]. Additionally, reductions in circulating IL-6 levels following chickpea consumption may indirectly limit activation of this pathway [69]. Furthermore, dietary fiber from chickpea contributes to the modulation of this axis through gut microbiota interactions. Fermentation of resistant starch produces SCFAs, which improve gut barrier integrity and reduce endotoxin-mediated TLR4 activation, thereby decreasing cytokine production and indirectly attenuating JAK-STAT signaling [47]. These findings suggest that chickpea bioactives regulate the IL-6-STAT3-SOCS3 axis through both direct molecular interactions and indirect anti-inflammatory effects, ultimately contributing to improved metabolic homeostasis.
However, it is important to note that chickpea represents a complex dietary matrix composed of multiple bioactive constituents, including polyphenols, saponins, dietary fiber, peptides, and micronutrients. The immunomodulatory effects described above are therefore unlikely to be attributed to a single compound but rather to the combinatorial and potentially synergistic interactions among these components. Such interactions may enhance the overall efficacy of cytokine modulation but also introduce variability in biological responses. This compositional heterogeneity presents challenges in isolating specific mechanisms and may limit direct translation into controlled clinical settings, where standardization of bioactive exposure is required.
Chronic low-grade inflammation associated with metabolic disorders is increasingly recognized as a critical link between obesity and cancer development. Persistent activation of inflammatory pathways promotes a microenvironment that supports tumor initiation, progression, and metastasis. In particular, NF-κB and STAT3 signaling pathways play central roles in regulating genes involved in cell proliferation, angiogenesis, and resistance to apoptosis [70]. Chickpea bioactives may exert chemoprotective effects by targeting these interconnected inflammatory pathways. Polyphenols and saponins present in chickpea have been shown to suppress NF-κB activation, thereby reducing transcription of pro-inflammatory and pro-tumorigenic genes [71]. Similarly, inhibition of STAT3 activation can limit cytokine-driven oncogenic signaling and disrupt tumor-promoting feedback loops associated with chronic inflammation [37]. In addition to modulation of cytokine signaling, chickpea-derived compounds possess antioxidant properties that reduce oxidative stress and DNA damage, which are key contributors to carcinogenesis. Experimental studies have demonstrated that chickpea protein hydrolysates and phytochemicals can attenuate inflammation and improve metabolic parameters in obesity models, further supporting their protective role [46, 47]. Moreover, flavonoids and saponins from chickpea have shown antiproliferative potential in various experimental systems [72]. The combined regulation of inflammatory signaling, oxidative stress, and metabolic dysfunction suggests that chickpea bioactives may help mitigate obesity-associated cancer risk. However, direct clinical evidence remains limited, and further studies are required to validate these chemoprotective effects and determine their translational applicability.
While mechanistic studies provide important insights into the immunometabolic effects of chickpea bioactives, validation in human populations is essential to establish their clinical relevance. Current evidence from dietary intervention studies suggests that chickpea consumption, either alone or as part of legume-rich diets, is associated with improvements in metabolic and inflammatory parameters. Randomized controlled trials and dietary intervention studies have demonstrated that regular legume intake improves glycemic control and reduces circulating inflammatory markers in individuals with obesity and T2DM [47]. In particular, chickpea-based dietary interventions have been associated with reductions in CRP and improvements in insulin sensitivity indices [52]. Although most clinical studies focus on systemic biomarkers rather than intracellular signaling pathways, decreases in circulating IL-6 and TNF-α indicate attenuation of upstream inflammatory processes.
In addition to cytokine modulation, chickpea consumption has been shown to improve lipid profiles, reduce postprandial glucose excursions, and enhance satiety in controlled feeding trials [52, 73]. These metabolic improvements may indirectly contribute to reduced inflammatory burden by limiting adipose tissue dysfunction and associated cytokine production. Thus, the clinical benefits of chickpea are likely mediated through both direct immunomodulatory effects and secondary improvements in metabolic homeostasis. Furthermore, chickpea-derived dietary fiber plays a critical role in modulating gut-mediated inflammation. Fermentation of resistant starch promotes the production of SCFAs, which enhance gut barrier integrity and reduce endotoxemia-associated inflammation [42]. This reduction in endotoxin-driven TLR4 activation may contribute to decreased systemic cytokine levels and improved inflammatory status.
Despite these promising findings, several limitations should be considered. Many clinical studies are short-term, involve mixed legume interventions rather than isolated chickpea supplementation, and do not assess molecular signaling endpoints such as STAT3 activation or SOCS3 expression. Additionally, variability in study design, dosage, and participant characteristics limits direct comparison across studies. Overall, current translational evidence supports the role of chickpea intake in reducing systemic inflammation and improving metabolic outcomes. However, well-designed, long-term randomized controlled trials incorporating pathway-specific biomarkers are required to confirm the direct modulation of cytokine signaling pathways in human populations. Clinical and translational evidence linking chickpea intake to cytokine modulation in metabolic disease is illustrated in Figure 2.
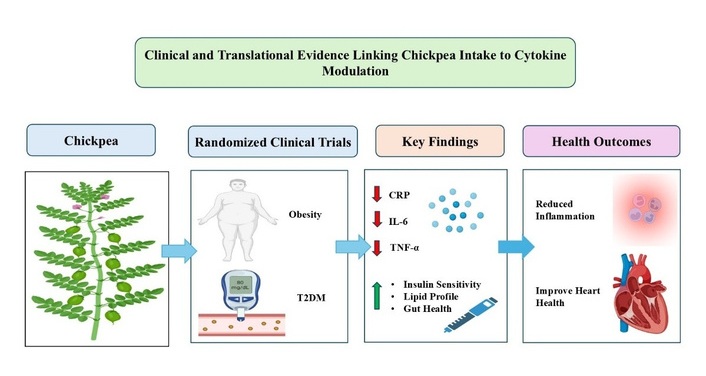
Clinical and translational evidence linking chickpea intake to cytokine modulation in metabolic disease. Figure created using PowerPoint Presentation. This figure summarizes evidence from clinical and dietary intervention studies demonstrating the impact of chickpea intake on inflammatory and metabolic parameters. Randomized clinical trials in individuals with obesity and type 2 diabetes mellitus (T2DM) indicate that chickpea consumption is associated with reductions in key inflammatory markers, including C-reactive protein (CRP), IL-6, and TNF-α. These changes are accompanied by improvements in insulin sensitivity, lipid profiles, and gut health. The beneficial effects are likely mediated through modulation of systemic inflammation, attenuation of cytokine signaling pathways, and improvement of gut-derived metabolic regulation. Collectively, these findings support the role of chickpea as a functional dietary component contributing to reduced metabolic inflammation and improved overall metabolic health.
Despite growing evidence supporting the anti-inflammatory potential of chickpea bioactives, several critical gaps remain in understanding their precise role in cytokine signaling and metabolic regulation. Addressing these limitations will be essential for advancing both mechanistic insight and clinical translation. Most available studies focus on circulating biomarkers such as CRP, IL-6, and TNF-α, whereas direct interrogation of intracellular signaling pathways, particularly the JAK-STAT-SOCS axis, remains limited. Future investigations should incorporate pathway-specific molecular endpoints, including phospho-STAT3 quantification, STAT3 nuclear translocation assays, SOCS3 gene expression, and IRS-1 phosphorylation status in relevant metabolic tissues. Such approaches will provide mechanistic validation of cytokine modulation beyond systemic markers.
Another major limitation is the scarcity of chickpea-specific mechanistic studies, with many conclusions extrapolated from broader legume or polyphenol research. Future studies should employ purified chickpea bioactive fractions, including phenolic extracts, saponin-enriched fractions, and resistant starch isolates, to delineate compound-specific effects on cytokine signaling pathways. In addition, dose-response analyses and pathway-targeted interventions using JAK or STAT3 inhibitors would help establish causal relationships and strengthen mechanistic interpretation. The inherent compositional heterogeneity of chickpea presents a significant challenge for mechanistic interpretation and clinical standardization. Future research should focus on developing standardized chickpea-derived formulations and well-characterized bioactive fractions to minimize variability in biological responses. Approaches such as fractionation, bioactivity-guided isolation, and controlled reconstitution studies may help delineate synergistic versus individual effects of specific components. Such strategies will be critical for improving reproducibility and facilitating translation into evidence-based dietary or nutraceutical interventions.
Integration of multi-omics approaches represents a promising strategy to achieve a systems-level understanding. Transcriptomic profiling of adipose and hepatic tissues following chickpea supplementation may reveal modulation of cytokine-regulated gene networks, while proteomic analyses could clarify changes in phosphorylation-dependent signaling cascades. Metabolomic profiling may further identify bioactive metabolites responsible for immunometabolic effects. The integration of these datasets through pathway enrichment and network analysis will provide comprehensive insights into chickpea-mediated regulation of inflammatory pathways. The gut-immune-metabolic axis also warrants deeper investigation. Although indirect evidence suggests that chickpea-derived fiber reduces endotoxin-mediated inflammation, mechanistic links between microbial metabolites and cytokine signaling pathways remain poorly defined. Future studies should incorporate gut microbiome sequencing alongside inflammatory and signaling biomarkers to clarify the role of microbiota-derived metabolites in modulating cytokine networks.
From a translational perspective, well-designed, long-term randomized controlled trials are required to validate preclinical findings. These studies should include standardized chickpea dosages, controlled dietary backgrounds, and stratification of participants based on metabolic phenotype. Importantly, incorporation of pathway-specific biomarkers will enable direct assessment of cytokine signaling modulation in human populations and support the development of evidence-based dietary interventions.
From a dietary perspective, most clinical studies evaluating chickpea or legume-rich interventions employ whole-food approaches rather than isolated bioactives. Typical intake levels of approximately 140 g/day of cooked chickpea have been used in human intervention studies and are feasible within habitual dietary patterns, with demonstrated improvements in glycemic responses [73]. However, precise dose-response relationships for cytokine modulation, particularly at the level of intracellular signaling pathways, remain inadequately defined. Standardization of intake and controlled dose-escalation studies are required to establish clinically effective and reproducible intervention strategies. Addressing these gaps through integrative, multi-level approaches will not only clarify the role of chickpea bioactives in metabolic disease but also contribute to the advancement of nutritional immunology and precision dietary strategies.
Metabolic diseases are characterized by chronic low-grade inflammation driven by cytokines such as IL-6, TNF-α, and IL-1β, which disrupt insulin signaling through JAK-STAT and NF-κB pathways. Chickpea contains polyphenols, saponins, and dietary fiber that may attenuate inflammatory signaling and improve metabolic homeostasis. Emerging evidence suggests its potential to modulate IL-6-STAT3 activation and reduce SOCS3-mediated insulin resistance, although direct mechanistic validation remains limited. Overall, chickpea represents a promising, accessible dietary immunomodulator, warranting further pathway-specific and clinical investigations to confirm its role in cytokine-driven metabolic dysfunction.
CRP: C-reactive protein
IL: interleukin
IRS-1: insulin receptor substrate-1
JAK: Janus kinase
LPS: lipopolysaccharide
MAPK: mitogen-activated protein kinase
NF-κB: nuclear factor kappa B
PI3K: phosphatidylinositol 3-kinase
ROS: reactive oxygen species
SCFA: short-chain fatty acid
SOCS: suppressor of cytokine signaling
STAT: signal transducer and activator of transcription
T2DM: type 2 diabetes mellitus
TLR4: Toll-like receptor 4
TNF-α: tumor necrosis factor-α
The authors acknowledge Central University of Punjab for providing resources to carry out the literature survey.
SAK: Writing—original draft. SC: Conceptualization, Supervision, Writing—original draft, Writing—review & editing. Both authors read and approved the submitted version.
The authors declare that they have no conflicts of interest, financial or non-financial, related to this work.
Not applicable.
Not applicable.
Not applicable.
Not available.
Not available.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 328
Download: 23
Times Cited: 0
Arbab Alam ... Vikrant Rai
