Affiliation:
1Shifa College of Pharmaceutical Sciences, Shifa Tameer-e-Millat University, Islamabad 44000, Pakistan
Email: mhhammad497@gmail.com
ORCID: https://orcid.org/0000-0001-8894-3692
Explor Endocr Metab Dis. 2026;3:101467 DOI: https://doi.org/10.37349/eemd.2026.101467
Received: February 27, 2026 Accepted: April 03, 2026 Published: April 15, 2026
Academic Editor: Myron R. Szewczuk, Queen’s University, Canada
Circadian rhythms are intrinsic 24-hour cycles that coordinate key metabolic processes, including glucose homeostasis, lipid metabolism, and energy expenditure. Disruption of these rhythms, due to sleep disturbances, shift work, or irregular feeding schedules, contributes to the development of obesity, insulin resistance, and dyslipidemia. Core clock genes, including circadian locomotor output cycles kaput (CLOCK), brain and muscle ARNT-like 1 (BMAL1), period gene (PER), and cryptochrome (CRY), play a central role in orchestrating these metabolic pathways. Chronotherapy, aligning lifestyle, behavioral, and pharmacologic interventions with circadian timing, represents a promising, yet underexplored, strategy for metabolic disease management. Evidence suggests that interventions such as time-restricted feeding, light therapy, pharmacologic modulators of clock gene expression, and wearable technology can improve glucose control, lipid profiles, and body weight. This review synthesizes current knowledge on the molecular regulation of metabolism by circadian clocks, elucidates mechanistic links between rhythm disruption and metabolic dysfunction, and explores translational strategies to restore circadian homeostasis. By targeting circadian rhythms, personalized and cost-effective interventions can be developed to mitigate the global burden of metabolic disorders. Future research should focus on large-scale clinical trials, precision chronotherapy, and integration of wearable devices to optimize the timing of interventions, ultimately enhancing treatment efficacy and long-term metabolic health.
Metabolic diseases, including obesity, type 2 diabetes mellitus (T2DM), and dyslipidemia, have reached epidemic proportions globally, creating a substantial burden on healthcare systems [1]. Recent estimates indicate that over 529 million adults are currently living with diabetes, a figure inextricably linked to the rising tide of obesity. Projections now suggest this prevalence will escalate to 1.31 billion cases by 2050, highlighting an urgent need for systemic interventions to address these escalating metabolic trends [2]. Conventional management strategies such as pharmacotherapy, dietary modification, and physical activity often provide only partial success, with high rates of relapse and limited long-term efficacy. This highlights the urgent need for novel, integrative approaches that go beyond calorie restriction and medication [3, 4].
Circadian rhythms are endogenous 24-hour cycles that play a fundamental role in metabolic regulation by coordinating the timing of physiological processes such as hormone secretion, glucose metabolism, lipid homeostasis, insulin release, and energy expenditure in response to environmental cues like light-dark cycles and feeding patterns [5–8]. The central circadian pacemaker, located in the suprachiasmatic nucleus (SCN) of the hypothalamus, synchronizes peripheral clocks in metabolically active tissues, including the liver, pancreas, adipose tissue, and skeletal muscle, through neural and hormonal signaling pathways. Disruption of these rhythms disturbs metabolic homeostasis and is strongly associated with disorders such as obesity and diabetes [9]. Consequently, aligning lifestyle and therapeutic interventions such as meal timing and physical activity with circadian biology may enhance the effectiveness of strategies aimed at preventing and managing metabolic diseases [10, 11].
Disruption of circadian rhythms resulting from shift work, irregular sleep-wake patterns, late-night food intake, or chronic jet lag has been consistently associated with metabolic dysregulation. Circadian misalignment adversely affects key metabolic pathways, including insulin secretion and sensitivity, adipokine oscillations, hepatic glucose production, and overall energy expenditure, thereby increasing susceptibility to obesity, insulin resistance, dyslipidemia, and cardiometabolic disorders [12–14]. Recent advances in chronotherapy, which involves synchronizing therapeutic interventions with endogenous circadian timing, have highlighted promising translational approaches [15]. These strategies encompass time-restricted dietary regimens, pharmacological modulation of clock-related genes, light-based interventions, and wearable technology-guided lifestyle optimization, all aimed at restoring circadian homeostasis [16–18]. This review provides a comprehensive overview of the molecular and physiological mechanisms underlying circadian regulation of metabolism, the metabolic consequences of circadian disruption, and emerging chronotherapeutic strategies for the prevention and management of metabolic diseases.
Circadian rhythms are endogenous, approximately 24-hour cycles that regulate numerous physiological and metabolic processes in humans. At the molecular level, these rhythms are orchestrated by interconnected transcriptional-translational feedback loops (TTFLs) of core clock genes, including CLOCK, brain and muscle ARNT-like 1 (BMAL1), period gene (PER)1–3, and cryptochrome (CRY)1/2 [19–21]. The CLOCK and BMAL1 proteins form a heterodimer in the nucleus, which binds to E-box elements on DNA to activate the transcription of target genes, most notably the PER and CRY family [22]. Once transcribed and translated in the cytoplasm, PER and CRY proteins accumulate over several hours, form complexes, and translocate back into the nucleus, where they inhibit CLOCK-BMAL1 activity, effectively closing the negative feedback loop. This cycle of transcription, translation, and inhibition takes approximately 24 hours, creating a self-sustained oscillation that forms the molecular basis of circadian rhythmicity [23, 24].
Rhythmic expression of core clock genes extends beyond the autoregulation of their own transcription and plays a central role in orchestrating the temporal organization of downstream metabolic pathways. These genes regulate the circadian timing of key physiological processes, including hepatic gluconeogenesis, pancreatic insulin secretion, adipocyte lipolysis, and mitochondrial energy metabolism [25]. In hepatic tissue, the transcriptional activity of the CLOCK-BMAL1 complex modulates the expression of enzymes essential for glucose production and lipid metabolism, whereas in adipose tissue it governs adipokine secretion and fatty acid mobilization. In the pancreas, circadian oscillations in clock gene expression influence both the timing and amplitude of insulin release, thereby establishing a direct mechanistic link between circadian rhythmicity and glucose homeostasis [26, 27].
Peripheral circadian clocks are present in metabolically active tissues, including the liver, skeletal muscle, adipose tissue, and pancreas, where they regulate tissue-specific gene expression and metabolic timing. These peripheral oscillators are entrained by the central pacemaker located in the SCN via hormonal signals (such as cortisol and melatonin) and autonomic neural pathways. Disruption of either central or peripheral circadian clocks arising from genetic alterations, shift work, irregular sleep-wake patterns, or environmental stressors results in circadian misalignment, metabolic dysfunction, and an increased risk of obesity, insulin resistance, and metabolic syndrome [28, 29].
To further elucidate these processes, circadian regulation can be conceptualized as an integrated regulatory network in which core clock genes interact dynamically with downstream metabolic pathways. The CLOCK-BMAL1 complex initiates transcriptional activity, while PER and CRY proteins provide inhibitory feedback, forming the central oscillatory loop. This core molecular clock regulates a wide array of clock-controlled genes involved in glucose metabolism, lipid homeostasis, mitochondrial function, and hormonal signaling [30, 31]. Importantly, these interactions are bidirectional, as metabolic states and nutrient availability can also give feedback to influence clock gene expression. A schematic representation of these regulatory flow relationships is provided in Figure 1, illustrating how disruption at any level of this network can propagate through metabolic systems to promote insulin resistance, dyslipidemia, and obesity. The principal clock genes and their metabolic functions, along with the consequences of their dysregulation, are summarized in Table 1.
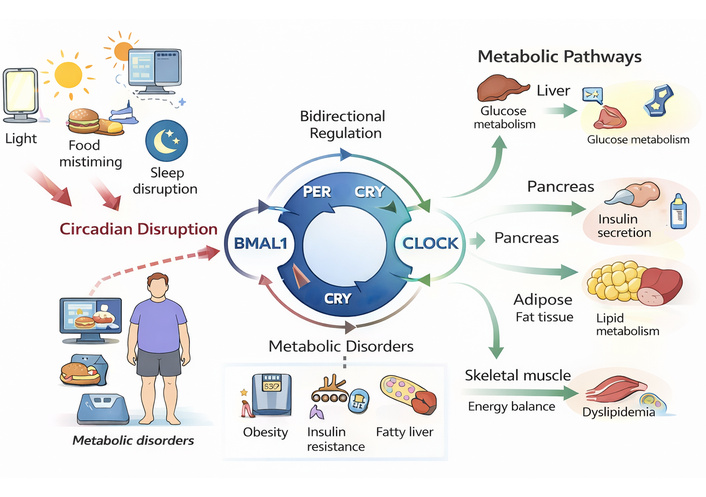
Regulatory network linking circadian clock genes to metabolic pathways. BMAL1: brain and muscle ARNT-like 1; CLOCK: circadian locomotor output cycles kaput; CRY: cryptochrome; PER: period gene.
Key clock genes and their metabolic roles.
| Gene | Primary function | Metabolic role | Dysregulation effect |
|---|---|---|---|
| CLOCK | Transcription factor | Glucose homeostasis, lipid metabolism | Obesity, insulin resistance |
| BMAL1 | Transcription factor | Circadian regulation of insulin | Hyperglycemia, fatty liver |
| PER1/2 | Feedback repressors | Appetite, energy expenditure | Altered feeding rhythm, metabolic syndrome |
| CRY1/2 | Feedback repressors | Hepatic gluconeogenesis | Hyperglycemia, insulin resistance |
BMAL1: brain and muscle ARNT-like 1; CLOCK: circadian locomotor output cycles kaput; CRY1/2: cryptochrome 1 and 2; PER1/2: period gene 1 and 2.
While Table 1 summarizes the key clock genes and their metabolic roles, it is important to note that these mechanisms are largely derived from mammalian studies, including both animal models and human investigations. The core circadian machinery is highly conserved across mammalian species, allowing translational insights from preclinical models. However, species-specific differences in metabolic regulation, feeding behavior, and circadian organization should be considered when applying these findings to human physiology and clinical practice.
Through these tightly regulated molecular mechanisms (Table 1), circadian clocks integrate environmental cues with metabolic processes, providing temporal organization that optimizes energy utilization, hormonal balance, and overall metabolic homeostasis. As illustrated in Figure 1, these processes operate through interconnected regulatory pathways linking central and peripheral clocks with metabolic effectors, reinforcing the systems-level nature of circadian control. Disruption of these pathways contributes to the pathophysiology of modern metabolic disorders, highlighting their potential as therapeutic targets for chronotherapy and precision medicine [32, 33].
The circadian clock plays a pivotal role in regulating hormonal and metabolic processes by synchronizing energy intake, storage, and expenditure with environmental and behavioral cycles. This temporal coordination is essential for preserving metabolic homeostasis and mitigating the risk of metabolic disturbances, including obesity, insulin resistance, and dyslipidemia [5].
Insulin and glucagon: Insulin sensitivity exhibits a pronounced circadian rhythm, reaching its peak during the active phase of the day, when nutrient intake is greatest, and declining during the biological night. This temporal pattern facilitates efficient glucose utilization and limits excessive postprandial hyperglycemia [34]. Circadian misalignment arising from shift work, sleep deprivation, or irregular eating schedules disrupts insulin responsiveness, leading to persistent hyperglycemia, compensatory hyperinsulinemia, and the gradual development of insulin resistance. In parallel, glucagon secretion is subject to circadian regulation, enabling the coordination of hepatic glucose production with fasting and activity cycles. Disruption of these tightly regulated hormonal rhythms impairs glucose homeostasis and increases susceptibility to T2DM [35].
Cortisol and melatonin: Cortisol secretion by the adrenal cortex follows a distinct diurnal rhythm, characterized by peak concentrations in the early morning hours. This morning surge facilitates the mobilization of energy substrates through the stimulation of gluconeogenesis, lipolysis, and glycogenolysis, thereby preparing the organism for daytime metabolic demands [36]. In contrast, melatonin is predominantly secreted during the nocturnal period and plays a central role in regulating the sleep-wake cycle while also modulating insulin secretion and peripheral glucose metabolism. Disruptions in melatonin rhythmicity, including delayed onset or reduced nocturnal secretion, have been associated with impaired glucose tolerance, dysregulation of appetite control, and increased adiposity [37].
Adipokines: Circadian rhythms also regulate the secretion of adipose-derived hormones, notably leptin and adiponectin. Leptin plays a central role in the control of satiety and energy expenditure, whereas adiponectin enhances insulin sensitivity and promotes lipid metabolism. Disruption of circadian rhythmicity attenuates the normal oscillatory patterns of these adipokines, thereby favoring increased food intake, adipose tissue accumulation, and the development of insulin resistance [38].
Collectively, these hormonal signals interact with core clock gene networks to establish a coordinated temporal framework for metabolic regulation. Consequently, the timing of nutritional intake, physical activity, and pharmacological interventions is of critical importance, as synchronizing these behaviors with endogenous circadian rhythms can optimize metabolic efficiency and improve clinical outcomes in obesity, diabetes, and related metabolic disorders. This temporal perspective on metabolic control highlights the translational significance of chronotherapy as an emerging strategy in metabolic disease management.
Epidemiologic studies provide strong evidence linking circadian misalignment to an increased risk of metabolic diseases. However, the metabolic impact of shift work is not uniform and depends on the type and duration of shifts. Long-term night shifts, particularly permanent night schedules, are consistently associated with obesity, T2DM, dyslipidemia, and hypertension, whereas rotating shifts may cause more variable effects on circadian rhythms and metabolic outcomes due to repeated clock gene desynchronization and partial adaptation. The study suggests that night shift workers can have a higher risk of developing metabolic syndrome compared to day workers, underscoring that both the nature of the shift system and chronicity of exposure influence the degree of metabolic disruption [39].
Sleep deprivation: It constitutes a significant contributor to circadian misalignment. Both partial and total sleep restriction increase circulating ghrelin levels while decreasing leptin concentrations, thereby enhancing appetite, caloric intake, and weight gain. Experimental studies in humans indicate that even short-term sleep restriction impairs glucose tolerance, diminishes insulin sensitivity, and elevates inflammatory markers, collectively increasing the risk of cardiometabolic disorders [40].
Irregular feeding patterns: Irregular feeding patterns further exacerbate metabolic dysregulation. Intake of meals during the biological night disrupts the alignment of peripheral clocks in the liver, adipose tissue, and pancreas, impairing glucose homeostasis and lipid metabolism. Such circadian misalignment is associated with elevated fasting glucose, dyslipidemia, and increased adiposity. Collectively, epidemiological evidence indicates that behavioral and environmental factors that perturb circadian rhythms, such as shift work, sleep deprivation, and mistimed eating, play a central role in the development of obesity, insulin resistance, and related metabolic disorders [41].
Mechanistic links between circadian disruption and metabolic disease have been elucidated through both preclinical and clinical studies, demonstrating that misalignment at molecular and systemic levels contributes to obesity, insulin resistance, and dyslipidemia, as shown in Figure 2. Evidence from animal models indicates that mutations in core clock genes have direct metabolic consequences: mice with disrupted CLOCK or BMAL1 expression develop obesity, hyperglycemia, and dyslipidemia even under standard dietary conditions. Similarly, alterations in PER and CRY genes affect feeding behavior, hepatic glucose production, and adipose tissue metabolism, underscoring the essential role of circadian clock components in maintaining metabolic homeostasis [42].
Mitochondrial dysfunction: It represents an additional critical mechanism linking circadian disruption to metabolic disease. Misalignment of circadian rhythms impairs mitochondrial bioenergetics, diminishing oxidative capacity and energy expenditure while promoting lipid accumulation in skeletal muscle and liver. This energetic impairment contributes to insulin resistance and ectopic fat deposition, thereby connecting molecular clock dysregulation with systemic metabolic disturbances [43].
Adipose tissue and hepatic metabolism: They are tightly regulated by circadian rhythms. Disruption of adipose tissue clocks impairs lipolysis and dysregulates the secretion of key adipokines, including leptin and adiponectin, which are critical for the regulation of appetite and insulin sensitivity. Similarly, perturbation of hepatic circadian clocks alters gluconeogenesis and triglyceride synthesis, thereby exacerbating hyperglycemia and dyslipidemia [26].
Inflammatory pathways: Inflammatory pathways are closely integrated with circadian regulation. Circadian misalignment elevates pro-inflammatory cytokines, including interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), thereby promoting systemic inflammation, insulin resistance, and early atherogenic processes [44].
Gut microbiota: Gut microbiota displays pronounced diurnal oscillations in both composition and metabolic activity, largely driven by host feeding-fasting cycles and intrinsic circadian clock signaling. Circadian disruption, particularly due to mistimed feeding, perturbs microbial rhythmicity, impairing nutrient absorption, bile acid metabolism, and energy uptake. These microbial alterations, in turn, influence host metabolism, further amplifying metabolic risk [45].
Emerging evidence suggests that circadian misalignment leads to a loss of microbial diversity and a shift toward obesogenic microbial profiles, characterized by an increased Firmicutes-to-Bacteroidetes ratio and altered short-chain fatty acid (SCFA) production. SCFAs, particularly butyrate, play a critical role in maintaining gut barrier integrity, modulating inflammation, and regulating glucose and lipid metabolism. Disruption of SCFA rhythmicity has been linked to increased intestinal permeability (“leaky gut”), facilitating translocation of endotoxins such as lipopolysaccharides (LPS), which trigger systemic low-grade inflammation and insulin resistance [46, 47].
In addition, the gut microbiota interacts closely with host circadian genes through bidirectional signaling pathways. Microbial metabolites can influence the expression of core clock genes (e.g., BMAL1 and PER), while host circadian regulators shape microbial composition via rhythmic secretion of bile acids, hormones, and antimicrobial peptides. This bidirectional crosstalk underscores the integrative role of the gut microbiome in circadian-metabolic homeostasis [45].
Importantly, preclinical and emerging clinical studies suggest that interventions such as time-restricted feeding (TRF), probiotic/prebiotic supplementation, and dietary modulation can partially restore microbial rhythmicity and improve metabolic outcomes. However, inter-individual variability in microbiome composition and the lack of standardized therapeutic approaches remain significant challenges for clinical translation [48].
Collectively, these mechanistic insights demonstrate that circadian disruption impacts multiple, interconnected pathways, including genetic, hormonal, metabolic, inflammatory, and microbial networks, thereby creating a permissive environment for the development of obesity, insulin resistance, and metabolic syndrome. Elucidation of these mechanisms provides a critical foundation for the design and implementation of targeted chronotherapeutic interventions.
TRF is a chrono-nutrition strategy that limits daily caloric intake to a fixed window, usually spanning 8–10 h, with the goal of aligning food consumption with endogenous circadian rhythms. This approach leverages the temporal dimension of metabolism, optimizing the interaction between central and peripheral clocks to enhance metabolic health. By restricting feeding to the active phase of the circadian cycle, TRF helps synchronize hepatic, pancreatic, adipose, and skeletal muscle clocks, which collectively regulate glucose homeostasis, lipid metabolism, and energy expenditure [49]. Preclinical studies have consistently demonstrated the metabolic benefits of TRF. In rodent models, mice subjected to TRF are protected from diet-induced obesity even when consuming high-fat or high-calorie diets. TRF restores oscillatory expression of core clock genes (CLOCK, BMAL1, PER, and CRY) in the liver and adipose tissue, improving rhythmic hormonal secretion and enhancing insulin sensitivity. TRF also reduces hepatic triglyceride accumulation, improves mitochondrial function, and promotes adipose tissue lipolysis, collectively mitigating risk factors for metabolic syndrome [50]. Clinical evidence supports these findings. Early TRF in overweight or prediabetic adults improves postprandial insulin sensitivity, reduces fasting glucose, and enhances lipid profiles. Participants often experience modest reductions in body weight and waist circumference, accompanied by decreases in blood pressure and systemic inflammation. Importantly, TRF may improve adherence to dietary interventions by providing a simple temporal framework rather than imposing strict caloric restriction [51].
Beyond the duration of the feeding window, emerging evidence indicates that the specific timing, frequency, and distribution of meals throughout the day significantly influence metabolic outcomes. Early-day caloric intake, particularly a larger breakfast and reduced evening consumption, has been associated with improved insulin sensitivity, better glycemic control, and enhanced diet-induced thermogenesis. In contrast, late-night eating and irregular meal timing are linked to impaired glucose tolerance, increased adiposity, and a higher risk of metabolic disorders [52].
Meal frequency also plays a role, as fewer, well-timed meals within the active circadian phase may optimize metabolic efficiency by reducing prolonged postprandial hyperglycemia and aligning nutrient intake with peak metabolic capacity. Conversely, frequent snacking or erratic eating patterns can disrupt peripheral clock synchronization and promote metabolic dysregulation. These findings highlight that both “when” and “how often” individuals eat are critical determinants of metabolic health within TRF paradigms [53, 54].
Mechanistically, TRF promotes metabolic flexibility by enhancing nutrient partitioning and optimizing the temporal coordination of energy intake and expenditure. By aligning feeding with periods of maximal insulin sensitivity and hepatic metabolic capacity, TRF reduces circadian misalignment, improves glucose tolerance, and supports long-term metabolic health. As a non-pharmacologic, low-cost intervention, TRF represents a promising lifestyle approach for the prevention and management of obesity, insulin resistance, and related metabolic disorders.
Sleep quality, duration, and timing are fundamental determinants of circadian rhythm entrainment and metabolic regulation. The SCN, the central circadian pacemaker, relies on consistent sleep-wake cues and environmental light signals to synchronize peripheral clocks in metabolically active tissues such as the liver, adipose tissue, pancreas, and skeletal muscle. Disruption of sleep timing or exposure to inappropriate light at night can desynchronize these clocks, leading to impaired glucose metabolism, altered lipid homeostasis, and increased risk of obesity and insulin resistance [55].
Consistent sleep schedules play a critical role in reinforcing SCN signaling. Maintaining regular bedtimes and wake times stabilizes the oscillatory expression of clock genes and harmonizes peripheral metabolic rhythms. Experimental studies have shown that irregular sleep patterns reduce insulin sensitivity, elevate evening cortisol, and impair nocturnal leptin secretion, collectively predisposing individuals to weight gain and metabolic dysfunction [56]. Light exposure serves as a powerful zeitgeber, or time cue, for circadian entrainment. Morning exposure to natural light reinforces SCN signaling, advancing the circadian phase and improving alignment of metabolic processes with behavioral cycles. Conversely, excessive exposure to blue-enriched light during the evening delays circadian phase, suppresses nocturnal melatonin secretion, and disrupts glucose regulation. Minimizing artificial light at night, especially from screens, has been associated with improved sleep quality, better glycemic control, and reduced appetite in both preclinical and clinical studies [57]. Moreover, the metabolic impact of optimized sleep and light hygiene is significant. Adequate, well-timed sleep restores leptin and ghrelin rhythms, reduces hyperphagia, and enhances insulin sensitivity. Improved sleep quality lowers evening cortisol, attenuates inflammatory signaling, and promotes lipid homeostasis. Integrating sleep hygiene strategies with light management can synergistically reinforce circadian entrainment, supporting weight regulation, glucose metabolism, and overall cardiometabolic health [40].
Collectively, sleep and light interventions represent non-pharmacologic, cost-effective approaches to mitigate circadian misalignment, offering a practical avenue for the prevention and management of obesity, insulin resistance, and related metabolic disorders.
Physical activity is a potent non-photic zeitgeber that plays a critical role in modulating circadian rhythms and metabolic homeostasis. Beyond its well-established metabolic benefits, exercise influences the temporal organization of physiological processes by interacting with peripheral clocks in skeletal muscle, adipose tissue, and liver. These interactions contribute to the regulation of glucose metabolism, lipid utilization, and energy expenditure [58]. The timing of exercise has emerged as an important determinant of its metabolic effects. Morning or early-day exercise has been shown to enhance circadian alignment, improve insulin sensitivity, and promote better glycemic control, particularly in individuals with metabolic disorders. In contrast, late-night high-intensity exercise may delay circadian phase, disrupt sleep architecture, and attenuate some metabolic benefits [59]. These findings suggest that aligning physical activity with an individual’s circadian phase can optimize therapeutic outcomes.
Mechanistically, exercise modulates the expression of core clock genes, including BMAL1 and PER, in peripheral tissues. It also activates key metabolic regulators such as AMP-activated protein kinase (AMPK) and enhances mitochondrial biogenesis, thereby improving metabolic flexibility and substrate utilization [60]. Moreover, exercise-induced improvements in skeletal muscle glucose uptake and insulin signaling further reinforce its role in chronometabolic regulation.
Emerging evidence also indicates that time-of-day-dependent variations in exercise performance and metabolic response may be influenced by chronotype, highlighting the need for personalized exercise timing strategies [59]. Integrating exercise timing into chronotherapy frameworks, alongside dietary and sleep interventions, may provide synergistic benefits for the prevention and management of obesity, insulin resistance, and related metabolic disorders.
Pharmacologic targeting of circadian regulators represents a promising translational strategy to manage metabolic diseases, particularly obesity, insulin resistance, and dyslipidemia. While lifestyle interventions such as TRF and sleep hygiene are effective, pharmacologic approaches offer the potential to directly modulate clock gene activity and restore circadian homeostasis, complementing behavioral strategies [61, 62] as shown in Table 2. Although much of the evidence originates from preclinical mammalian studies, particularly rodents, several interventions, such as melatonin and reverse erythroblastosis virus alpha/beta (nuclear receptors) (REV-ERB) agonists, have demonstrated translational relevance in humans. Clinical translation of these therapies, however, requires careful consideration of long-term safety, tolerability, and real-world applicability.
Pharmacologic interventions targeting circadian pathways.
| Drug/Class | Target | Effect on metabolism |
|---|---|---|
| REV-ERB agonists | CLOCK/BMAL1 | Reduces hepatic steatosis |
| Melatonin | SCN/Peripheral clocks | Improves insulin sensitivity |
| SIRT1 activators | CLOCK regulation | Enhances energy expenditure |
| Nobiletin | PER/CRY modulation | Reduces adiposity |
BMAL1: brain and muscle ARNT-like 1; CLOCK: circadian locomotor output cycles kaput; CRY: cryptochrome; PER: period gene; REV-ERB: reverse erythroblastosis virus alpha/beta (nuclear receptors); SCN: suprachiasmatic nucleus; SIRT1: sirtuin 1.
REV-ERB agonists are synthetic ligands that activate REV-ERB nuclear receptors, key transcriptional repressors in the circadian clock machinery. By modulating CLOCK-BMAL1 activity, REV-ERB agonists regulate hepatic lipid metabolism, suppress triglyceride synthesis, and improve mitochondrial function. Preclinical studies in mouse models demonstrate that REV-ERB activation reduces hepatic steatosis, enhances fatty acid oxidation, and improves glucose tolerance, even in the context of high-fat diets. These findings highlight the potential of REV-ERB agonists to target both central and peripheral circadian mechanisms for metabolic benefit [63]. Nevertheless, their clinical applicability remains limited due to a lack of human trials, uncertain pharmacokinetics, and potential off-target effects, particularly given the widespread role of nuclear receptors in multiple physiological systems.
Melatonin and its analogs are widely recognized for their role in regulating sleep-wake cycles and circadian phase. Beyond sleep modulation, melatonin also influences peripheral glucose metabolism and insulin secretion. Although clinical studies in patients with T2DM suggest potential benefits of exogenous melatonin such as modest improvements in nocturnal glycemic control, fasting glucose, and insulin sensitivity, these findings are preliminary and derived from small, short-term studies [64]. Melatonin therapy may help mitigate circadian misalignment in shift workers or individuals with delayed sleep phase disorders, providing a potential adjunctive approach to metabolic management [62]. However, the evidence remains limited, with significant uncertainties regarding optimal dosing, timing of administration, and long-term metabolic effects. Furthermore, the safety profile of chronic melatonin supplementation in T2DM patients has not been fully established, and larger, well-controlled trials are needed before routine clinical application.
Sirtuin1 (SIRT1) activators modulate circadian rhythms through interaction with CLOCK-BMAL1 and other transcriptional regulators. SIRT1 enhances energy expenditure, promotes lipid catabolism, and supports mitochondrial biogenesis. Preclinical studies indicate that pharmacologic activation of SIRT1 improves glucose homeostasis and reduces adiposity, emphasizing its potential as a circadian-modulating metabolic therapy [65]. Despite these promising findings, most SIRT1 activators (e.g., resveratrol and synthetic compounds) face limitations related to bioavailability, inconsistent clinical efficacy, and insufficient long-term safety data in humans.
Nobiletin, a natural flavonoid, acts on PER and CRY proteins to modulate circadian feedback loops. Animal studies demonstrate that nobiletin supplementation reduces adiposity, enhances glucose tolerance, and mitigates diet-induced metabolic dysfunction. Its dual role in circadian regulation and metabolic improvement makes it a compelling candidate for translational research [66]. However, its clinical use is still experimental, with limited human data and unclear optimal dosing strategies, necessitating further investigation before routine therapeutic application.
Collectively, these pharmacological interventions highlight the therapeutic potential of targeting circadian pathways in the management of metabolic disorders. While early-phase clinical evidence supports feasibility, widespread clinical adoption is currently constrained by gaps in safety profiling, limited large-scale randomized trials, and challenges in chrono-pharmacological optimization (i.e., timing of drug administration). Although much of the evidence to date is preclinical, emerging clinical studies demonstrate both feasibility and preliminary efficacy, suggesting a future in which circadian-based pharmacotherapy can be integrated with lifestyle interventions to enable personalized metabolic management. Figure 3 illustrates the mechanistic linkages between circadian regulators and key metabolic outcomes, as well as the pharmacologic interventions that can modulate these pathways.
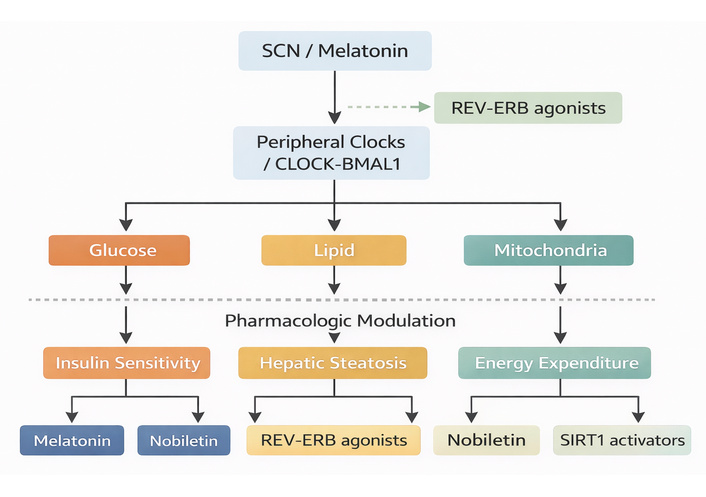
Mechanistic linkages between circadian regulators and metabolic outcomes. BMAL1: brain and muscle ARNT-like 1; CLOCK: circadian locomotor output cycles kaput; REV-ERB: reverse erythroblastosis virus alpha/beta (nuclear receptors); SCN: suprachiasmatic nucleus; SIRT1: sirtuin 1.
While circadian-based strategies such as TRF, sleep hygiene, and timed exercise have demonstrated clear metabolic benefits, their real-world effectiveness is often limited by behavioral, psychological, and social factors [67]. Adhering to strict temporal schedules can be particularly challenging due to work demands, social obligations, cultural practices, and individual lifestyle habits. For example, TRF requires that all daily calories be consumed within a defined window, which may conflict with evening social meals, late work shifts, or family routines. Similarly, maintaining consistent sleep-wake cycles or aligning exercise with one’s circadian phase may be difficult in the context of shift work, academic responsibilities, or irregular daily routines [68, 69].
Psychological factors also significantly influence adherence. Stress, anxiety, mood fluctuations, and low motivation can reduce an individual’s capacity to maintain circadian-based interventions, often leading to missed feeding windows, irregular sleep, or skipped exercise sessions [70]. The effectiveness of chronotherapy is often undermined by specific behavioral tendencies, including emotional eating, impulsive snacking, and irregular activity patterns. These habits can disrupt the biological synchrony that the therapy aims to establish. Beyond these behaviors, individual variations in chronotype, the natural predisposition toward “morningness” or “eveningness”, heavily influence how feasible these interventions actually are in a clinical or real-world setting. A person with a late-shifted or evening chronotype, for example, typically struggles to adhere to early-day TRF or rigid morning exercise routines. This misalignment not only limits the potential metabolic benefits but also heightens the risk of patient frustration and, ultimately, study dropout or treatment non-compliance. These individual differences suggest that a one-size-fits-all approach to circadian timing may be less effective than personalized protocols that account for a patient's baseline rhythm [71–73].
Social and environmental contexts further modulate adherence. Household routines, workplace culture, peer influences, and access to food and exercise resources can either facilitate or impede consistent implementation of circadian strategies. Strong social support is associated with better compliance and long-term success, whereas conflicting obligations, cultural practices such as late-night communal meals, or lack of supportive infrastructure may reduce adherence [74, 75]. These factors highlight that metabolic efficacy alone is insufficient; the human context in which interventions are applied plays a critical role in outcomes.
Addressing these challenges requires integrating behavioral and psychological support with circadian interventions. Patient education, counseling, and personalized strategies that account for lifestyle constraints, chronotype, and social environment can improve adherence and engagement. Self-monitoring using diaries, mobile applications, or wearable devices can reinforce behavior change by providing real-time feedback and accountability. Motivational strategies, including goal setting, reinforcement, and social support, further enhance long-term adherence. By combining these psychosocial approaches with biological and pharmacologic circadian interventions, it is possible to maximize translational potential, improve metabolic outcomes, and ensure that interventions are sustainable in real-world settings [76]. While circadian-based interventions hold promise for preventing and managing metabolic disorders, their success depends not only on physiological efficacy but also on addressing behavioral, psychological, and social determinants. Future research and clinical programs should focus on holistic strategies that integrate lifestyle, pharmacologic, and behavioral components to optimize adherence, feasibility, and long-term health benefits.
Recent advances in wearable technology offer unprecedented opportunities to implement precision chronotherapy in metabolic disease management. By continuously monitoring physiological and behavioral parameters, these devices enable real-time assessment of an individual’s circadian patterns, facilitating interventions tailored to their unique biological rhythms. Continuous glucose monitors (CGMs) provide detailed temporal profiles of glucose fluctuations, capturing responses to meals, physical activity, and sleep. This high-resolution data allows for precise identification of periods of peak insulin sensitivity and glucose tolerance, which can inform the timing of meals and pharmacotherapy. For example, adjusting carbohydrate intake or insulin administration based on circadian glucose patterns may improve postprandial glycemia and reduce overall metabolic risk [77].
Actigraphy devices and smartwatches track sleep-wake cycles, physical activity, heart rate variability, and exposure to environmental light. By analyzing these patterns, clinicians can identify circadian misalignment, sleep deprivation, or irregular activity that contribute to metabolic dysregulation. For instance, optimizing morning light exposure and maintaining consistent sleep schedules can reinforce central and peripheral clock entrainment, thereby improving insulin sensitivity and energy balance [78].
Importantly, wearable-derived data can also guide the timing of physical activity, which acts as a key non-photic regulator of circadian rhythms. Strategically timed exercise may enhance glucose uptake, mitochondrial efficiency, and metabolic flexibility, thereby complementing dietary and pharmacologic interventions. Moreover, integration with machine learning algorithms enhances the utility of wearable data by enabling predictive and adaptive chronotherapy. These models integrate multidimensional inputs, including glucose levels, activity patterns, sleep cycles, and light exposure to forecast optimal timing for meals, exercise, and medication. While such approaches hold promise for improving adherence and metabolic outcomes, their effectiveness depends on robust validation, data quality, and applicability across diverse populations [79, 80].
As illustrated in Figure 4, wearable technology serves as a central integrative hub within a multi-modal chronotherapy framework. Specifically, the figure depicts four key intervention domains: TRF, light therapy, pharmacologic modulation, and physical activity (captured via wearable devices), all converging toward circadian alignment at the individual level. The directional arrows emphasize how these inputs interact dynamically with the central and peripheral clocks, rather than acting in isolation. Wearable devices are uniquely positioned within this model as both monitoring and feedback tools, enabling continuous assessment of circadian biomarkers (e.g., glucose rhythms, activity cycles, and sleep patterns) and facilitating real-time adjustment of these interventions.
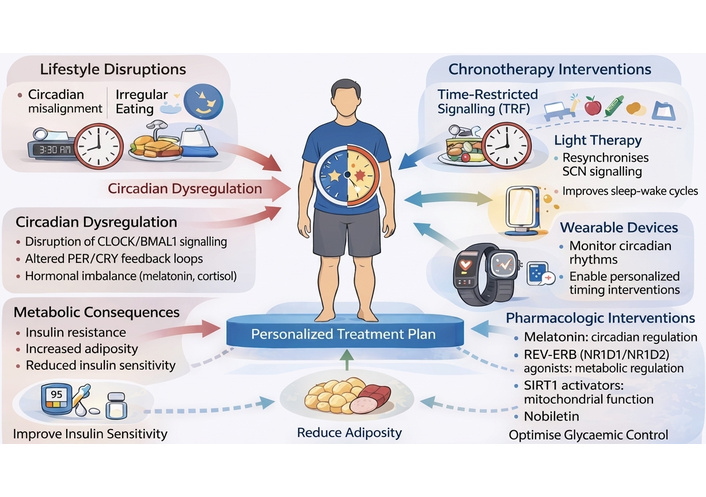
Translational applications of chronotherapy in metabolic disease. BMAL1: brain and muscle ARNT-like 1; CLOCK: circadian locomotor output cycles kaput; CRY: cryptochrome; PER: period gene; REV-ERB: reverse erythroblastosis virus alpha/beta (nuclear receptors); SCN: suprachiasmatic nucleus; SIRT1: sirtuin 1.
Furthermore, it also highlights the downstream metabolic outcomes of this integrated approach, including improved insulin sensitivity, reduced adiposity, and optimized glycemic control. This visual representation reinforces the concept that synchronized timing of lifestyle and pharmacologic interventions guided by wearable-derived data can produce synergistic metabolic benefits. By explicitly linking intervention timing to physiological outcomes, the figure strengthens the mechanistic basis of precision chronotherapy.
Despite these advantages, important limitations must be acknowledged. The high cost and limited accessibility of wearable devices and CGMs may restrict their implementation in low- and middle-income settings, potentially exacerbating healthcare disparities. Moreover, sustained user adherence, data interpretation challenges, and integration into routine clinical workflows remain significant barriers to widespread adoption. Combined with advanced analytics, these technologies offer a transformative, data-driven approach to precision chronotherapy, bridging circadian biology with real-world management of obesity, insulin resistance, and dyslipidemia, and establishing a scalable framework for personalized metabolic care [81–83].
Translation of circadian rhythm modulation into the clinical management of metabolic diseases presents both significant opportunities and notable challenges. By leveraging the temporal regulation of metabolism, interventions can be tailored to an individual’s intrinsic biological rhythms, representing a paradigm shift in personalized medicine [79]. From a translational perspective, the primary opportunity lies in the ability to align therapeutic interventions with circadian biology to enhance efficacy and reduce treatment burden. Personalized treatment strategies can be informed by integrating data on chronotype, sleep-wake patterns, feeding schedules, physical activity, and hormonal profiles. This approach allows precise timing of dietary, pharmacologic, or lifestyle interventions, thereby maximizing efficacy by synchronizing therapeutic actions with periods of peak metabolic responsiveness [84, 85].
In pharmacologic management, circadian-informed scheduling of medications can enhance drug effectiveness and potentially reduce required doses, decreasing side effects while maintaining or improving clinical outcomes. For example, administering insulin sensitizers or lipid-lowering agents at times of day when peripheral tissues are most responsive may optimize glucose and lipid control [86]. However, real-world implementation of chrono-pharmacology remains limited due to insufficient guideline-based recommendations and variability in patient response. Similarly, lifestyle interventions, such as TRF, sleep hygiene, and structured exercise, are more effective when timed to reinforce endogenous circadian oscillations, improving adherence, metabolic flexibility, and overall cardiometabolic health. The integration of wearable technologies and continuous monitoring devices further strengthens these approaches by providing real-time feedback, enabling dynamic adjustments tailored to individual circadian profiles [87, 88]. These technologies also offer scalability, making precision chronotherapy increasingly feasible in routine clinical settings.
Despite these promising opportunities, several challenges must be addressed to achieve successful translation of circadian-based interventions. Inter-individual variability shaped by genetic polymorphisms, age, sex, and environmental factors, complicates the determination of optimal timing for therapy. Importantly, individual chronotypes, such as “night owls” versus “early birds”, influence sleep-wake patterns, metabolic responses, and the efficacy of behavioral or pharmacologic interventions, underscoring the need for chronotype-tailored strategies [89]. Socio-cultural and occupational constraints (e.g., shift work, variable work schedules) further limit adherence to circadian-aligned interventions, particularly in low- and middle-income settings. Sustained compliance with TRF, sleep, and light exposure protocols may therefore require personalized scheduling, adaptive behavioral counseling, and integration of wearable-guided monitoring to align interventions with each individual’s intrinsic circadian preferences [90].
Moreover, clinical validation remains limited, necessitating robust, large-scale randomized controlled trials to establish evidence-based guidelines, evaluate long-term efficacy, and ensure safety across diverse populations. Current evidence is predominantly derived from preclinical studies or small-scale clinical trials, which restricts generalizability and delays integration into standard care pathways. Effective implementation into routine clinical practice will require education of healthcare professionals, adaptation of clinical workflows, and patient engagement strategies to ensure interventions are both practical and sustainable.
Future directions emphasize an integrated, multi-modal approach, combining pharmacologic agents that target clock genes with lifestyle modifications and precision-guided interventions via wearable devices to optimize circadian alignment. Importantly, future research should also focus on chronotype-based stratification, the development of standardized timing protocols, and the incorporation of circadian biomarkers into clinical decision-making. Novel small molecules and gene-targeted therapies that modulate clock gene expression represent an emerging frontier in translational chronotherapy, and incorporation of circadian principles into clinical guidelines for obesity, insulin resistance, and dyslipidemia will be essential to fully realize the therapeutic potential of this strategy.
Circadian rhythm modulation represents a multifaceted and promising approach for the prevention and management of metabolic diseases, including obesity, T2DM, and dyslipidemia. Evidence increasingly demonstrates that aligning behavioral, dietary, and pharmacologic interventions with endogenous circadian rhythms can improve insulin sensitivity, optimize lipid metabolism, and enhance energy homeostasis. Lifestyle strategies such as TRF, sleep and light optimization, and circadian-aligned exercise can restore peripheral and central clock synchronization, while pharmacologic agents, including melatonin, REV-ERB agonists, SIRT1 activators, and nobiletin, offer targeted modulation of clock gene activity. Wearable-guided precision chronotherapy further enables individualized, data-driven interventions that incorporate real-time monitoring of glucose, activity, and sleep patterns to improve adherence and efficacy.
However, despite compelling preclinical and early clinical evidence, translational challenges remain. Conflicting findings regarding optimal timing, feeding windows, pharmacologic targets, and chronotype-specific responses highlight the complexity of circadian interventions in heterogeneous human populations. Behavioral, psychological, and social determinants, such as stress, chronotype, occupational demands, and cultural practices, further modulate intervention feasibility and metabolic outcomes, underscoring that physiological efficacy alone is insufficient. Standardized protocols, rigorous large-scale clinical trials, and mechanistic studies that integrate genetic, hormonal, metabolic, and microbial perspectives are critically needed to establish evidence-based circadian guidelines.
While circadian-based interventions hold substantial potential to transform metabolic disease management, their real-world success will depend on holistic strategies that combine biological, pharmacologic, and psychosocial components. Integrating circadian principles into patient-centered approaches may offer a more precise, personalized, and sustainable framework for mitigating the global burden of metabolic disorders. Importantly, future research should not only refine therapeutic strategies but also critically examine conflicting evidence, identify population-specific limitations, and address implementation barriers to ensure that circadian modulation moves from a promising concept to clinical reality.
AMPK: AMP-activated protein kinase
BMAL1: brain and muscle ARNT-like 1
CGMs: continuous glucose monitors
CLOCK: circadian locomotor output cycles kaput
CRY: cryptochrome
IL-6: interleukin-6
LPS: lipopolysaccharide
PER: period gene
REV-ERB: reverse erythroblastosis virus alpha/beta (nuclear receptors)
SCFA: short-chain fatty acid
SCN: suprachiasmatic nucleus
SIRT1: sirtuin 1
T2DM: type 2 diabetes mellitus
TNF-α: tumor necrosis factor-alpha
TRF: time-restricted feeding
TTFLs: transcriptional-translational feedback loops
The authors acknowledge the use of a generative artificial intelligence-based image creation software (ChatGPT and/or DALL·E, Adobe Illustrator, OpenAI, 2026) for developing the figures in this manuscript. After utilizing the tool, the authors reviewed and edited the content as necessary and took full responsibility for the final content of the publication.
MH: Conceptualization, Investigation, Validation, Supervision, Writing—original draft, Writing—review & editing. KS: Writing—original draft, Writing—review & editing. Both authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1917
Download: 44
Times Cited: 0
