Affiliation:
1Faculty of Medicine, Tbilisi State Medical University, Tbilisi 0186, Georgia
ORCID: https://orcid.org/0009-0006-6693-8212
Affiliation:
1Faculty of Medicine, Tbilisi State Medical University, Tbilisi 0186, Georgia
Email: atifkhatib58@gmail.com
ORCID: https://orcid.org/0009-0009-7821-9292
Affiliation:
1Faculty of Medicine, Tbilisi State Medical University, Tbilisi 0186, Georgia
ORCID: https://orcid.org/0009-0000-6763-9595
Affiliation:
1Faculty of Medicine, Tbilisi State Medical University, Tbilisi 0186, Georgia
ORCID: https://orcid.org/0009-0005-2904-4626
Affiliation:
1Faculty of Medicine, Tbilisi State Medical University, Tbilisi 0186, Georgia
ORCID: https://orcid.org/0009-0000-0814-7937
Affiliation:
2Faculty of Medicine, Ivane Javakhishvili Tbilisi State University, Tbilisi 0179, Georgia
ORCID: https://orcid.org/0009-0001-6460-6111
Affiliation:
1Faculty of Medicine, Tbilisi State Medical University, Tbilisi 0186, Georgia
ORCID: https://orcid.org/0009-0009-7058-6565
Explor Endocr Metab Dis. 2026;3:101468 DOI: https://doi.org/10.37349/eemd.2026.101468
Received: January 24, 2026 Accepted: April 03, 2026 Published: April 23, 2026
Academic Editor: Osborne Almeida, University of Minho, Portugal
The global rise in consumption of added sugars and confectionery parallels the increasing prevalence of obesity, metabolic dysfunction, and male hypogonadism. Testosterone is essential for male reproductive, metabolic, and cardiovascular health, and growing evidence indicates that excessive sugar intake may disrupt hormonal regulation. This narrative review synthesizes data from human observational and interventional studies and experimental animal models to evaluate the effects of sugar consumption on testosterone homeostasis and male reproductive outcomes. Acute glucose ingestion is associated with hyperinsulinemia and inflammatory cytokine release, which may contribute to transient reductions in circulating testosterone of approximately 20–30%, although findings vary across populations and study designs. Chronic high-sugar intake, particularly from sugar-sweetened beverages, promotes oxidative stress, adiposity, insulin resistance, and leptin dysregulation, which may collectively impair Leydig cell steroidogenesis and contribute to testosterone suppression. Animal studies consistently demonstrate testicular structural damage, downregulation of steroidogenic enzymes, and impaired spermatogenesis under high-sugar dietary conditions. Epidemiological studies associate frequent intake of sugar-sweetened beverages and confectionery with lower testosterone levels and poorer semen parameters. Emerging evidence suggests partial reversibility with dietary interventions emphasizing reduced sugar intake and increased antioxidant consumption. Overall, current data support a biologically plausible link between excessive sugar intake and impaired testosterone regulation, underscoring the need for longitudinal studies and public health strategies targeting added sugar reduction.
Global sugar and confectionery consumption has risen substantially over recent decades due to the increased availability of processed food products and sweetened beverages. This trend coincides with growing concern over male reproductive health, including declining semen quality, reduced testosterone levels, and increasing rates of functional hypogonadism. Evidence suggests that diets high in simple sugars may adversely influence hormonal balance and reproductive outcomes in men [1].
Testosterone is a key hormonal regulator of male physiology, essential for spermatogenesis, libido, fertility, muscle mass, metabolic homeostasis, and overall well-being. Even mild reductions in circulating testosterone may contribute to metabolic dysfunction, impaired reproductive outcomes, and increased cardio-metabolic risk [2]. This is particularly relevant given the strong interplay between metabolic health, nutrition, and endocrine function. Excessive sugar consumption can impair insulin sensitivity, increase visceral adiposity, and promote systemic inflammation, all of which are known to disrupt testosterone homeostasis [3].
In industrialized or developed regions, dietary patterns have shifted from traditional high-fat diets to highly refined, sugar-dense diets. This transition parallels rising global rates of obesity, metabolic syndrome, and type 2 diabetes, which are significantly related to lower testosterone secretion and impaired testicular function [4]. Additional epidemiologic data confirm that frequent consumption of soft drinks, a major source of added sugars, is linked to impaired testicular function [5].
Short-term physiological studies also demonstrate that dietary macronutrient composition influences hormonal responses. Men with overweight and obesity exhibit altered postprandial testosterone dynamics following carbohydrate-rich meals, highlighting the acute hormonal impact of sugar intake [6]. Dietary pattern studies further support that unhealthy, sugar-dense diets adversely affect testicular function and reproductive hormone profiles [2].
Given the global burden of obesity and increasing prevalence of male hypogonadism, understanding how sugar and confectionery consumption affect hormonal and reproductive health has significant public health implications.
Accordingly, this narrative review aims to synthesize current human and experimental evidence on the effects of sugar and confectionery consumption on serum testosterone levels and related reproductive pathways.
A comprehensive literature search was conducted across PubMed/Medline, Scopus, and Web of Science using terms including “testosterone”, “sugar”, “fructose”, “sugar-sweetened beverages”, “Leydig cell”, “steroidogenesis”, “insulin resistance”, and “hypothalamic-pituitary-gonadal axis”. No date restriction was applied. Studies were considered if they examined sugar or sweetener intake in relation to male reproductive hormones or testicular function, and were available in English. Both human studies (observational and interventional) and experimental animal models were included. Review articles were incorporated to provide a mechanistic context. Editorials and non-peer-reviewed sources were excluded. Full search strings included combinations such as: (“testosterone” OR “androgens”) AND (“sugar” OR “fructose” OR “sucrose” OR “sugar-sweetened beverages” OR “confectionery”) AND (“male” OR “men” OR “spermatogenesis”). The search was conducted from database inception through December 2024. Records were screened by title and abstract against pre-specified inclusion criteria; full texts were retrieved for all potentially relevant records. Articles were included if they reported primary data or synthesized evidence on sugar or sweetener intake in relation to testosterone levels, steroidogenesis, or male reproductive function. Two authors independently screened records, and disagreements were resolved by consensus. Although the current review follows the principles of transparent reporting for narrative reviews, full PRISMA compliance was not required, given the narrative design; efforts to minimize selection bias included pre-specifying inclusion criteria and consulting multiple databases.
The hypothalamic-pituitary-gonadal (HPG) axis, commonly referred to as the hypothalamic-pituitary-testicular axis in males, is a crucial endocrine system that consists of the anterior hypothalamus, the pituitary gland, and the gonads. This axis is vital for regulating reproductive functions, including key processes such as spermatogenesis [7]. Any dysfunction in this system can lead to low testosterone levels, a condition known as male hypogonadism [8]. At the most essential level, the gonadotropin-releasing hormone (GnRH) neurons act as the primary regulators of the HPG axis, coordinating the release of gonadotropins from the pituitary gland by sending signals to the median eminence (ME). The synthesis and release of GnRH are mainly regulated by Kisspeptin, a significant central regulatory factor that functions as a gatekeeper for the HPG axis, promoting GnRH secretion through its receptor, KISS1R (Figure 1) [7].
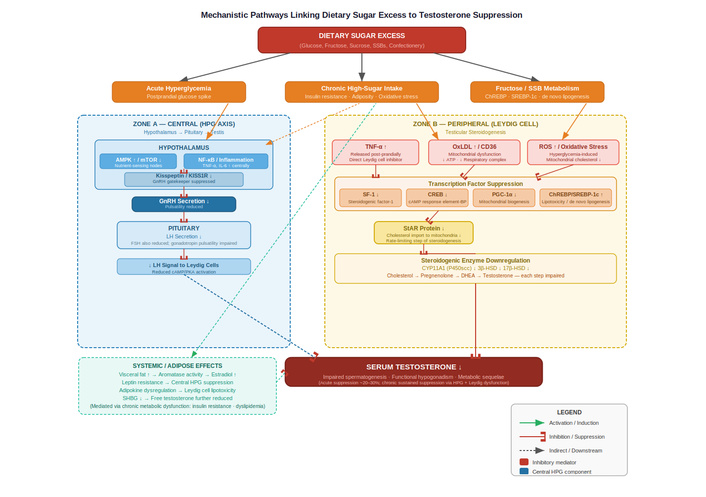
Mechanistic pathways linking dietary sugar excess to testosterone suppression. AMPK: AMP-activated protein kinase; ChREBP: carbohydrate-responsive element-binding protein; CREB: cAMP response element-binding; CYP11A1: cholesterol side chain cleavage enzyme; FSH: follicle-stimulating hormone; GnRH: gonadotropin-releasing hormone; HPG: hypothalamic-pituitary-gonadal; HSD: hydroxysteroid dehydrogenase; LH: luteinizing hormone; mTOR: mechanistic target of rapamycin; oxLDL: oxidized low-density lipoprotein; PGC-1α: peroxisome proliferator-activated receptor gamma coactivator 1-alpha; ROS: reactive oxygen species.SF-1: steroidogenic factor-1; SHBG: sex hormone binding globulin; SREBP-1c: sterol regulatory element-binding protein 1c; SSBs: sugar-sweetened beverages; StAR: steroidogenic acute regulatory; TNF- α: tumor necrosis factor-α.
It is proposed that a rapid rise in serum glucose levels may temporarily disrupt the synthesis and/or release of GnRH. This central disturbance is supported by in vitro findings indicating that elevated glucose levels impair GnRH secretion, reduce cell viability, and trigger apoptosis in GnRH-secreting neuronal cell lines [9]. However, no significant changes were observed in serum luteinizing hormone (LH) levels after either a glucose load or a mixed meal, even when serum LH was measured at various time points in both groups. The differences in findings could be attributed to the pulsatile nature of LH secretion, meaning that measuring it at limited intervals may not be sufficient to draw meaningful conclusions [10].
The decline in testosterone, which reduces Leydig cell stimulation and steroid hormone production, is influenced by both immediate metabolic factors and long-term consequences of obesity and insulin resistance [8, 9]. In the short term, the consumption of an oral glucose load or mixed meal prompts the release of the inflammatory cytokine tumor necrosis factor-α (TNF-α), which is known to increase in healthy males following glucose intake and may directly inhibit testosterone production by Leydig cells. TNF-α may accomplish this inhibition by reducing steroidogenesis in Leydig cells, particularly by decreasing the expression of the steroidogenic acute regulatory (StAR) protein [11]. Over the long term, high sugar intake from sugar-sweetened beverages (SSBs) has been associated with obesity and insulin resistance, which may lead to elevated leptin levels [8].
After multivariate adjustment, men in the highest quartile of SSB consumption (≥ 442 kcal/day) had significantly higher odds of low serum testosterone than those in the lowest quartile (≤ 137 kcal/day) (adjusted odds ratio [aOR] = 2.29, p = 0.041), according to Chen et al. [12] findings in its results section (“Associations between sugar-sweetened beverages and low testosterone level”). According to multivariate logistic regression analysis, the likelihood of having a low testosterone level rose considerably as SSB consumption increased (Q4 [≥ 442 kcal/day] vs. Q1 [≤ 137 kcal/day], aOR = 2.29, p = 0.041). Body mass index (BMI) was an independent risk factor for a low testosterone level after controlling for potential confounding variables. Subjects with a BMI ≥ 25 kg/m2 were more likely to have a low testosterone level than those with a BMI < 25 kg/m2 (aOR = 3.68, p = 0.044) [12]. The buildup of excess adipose tissue is viscerally linked to insulin resistance, dyslipidaemia, inflammation, hypothalamic leptin resistance, and gliosis, which underlie the functional hypogonadism of obesity, which has a diverse phenotypic spectrum. Nearly 20,000 obese men in a meta-analysis of 68 studies, 42.8% had serum total testosterone (TT) levels below 10.4 nmol/L (below 300 ng/dL) [8]. A 13–15% drop in serum testosterone levels has been associated with a 5 kg/m2 increase in BMI from 25 to 30 kg/m2, according to a Mendelian randomization analysis [8].
Other contributing inhibitory elements linked to obesity involve the buildup of oxidized low-density lipoprotein (oxLDL) within Leydig cells, impairing mitochondrial functionality crucial for testosterone synthesis [13]. Separately, obesity-associated metabolic disruption has been linked to impaired steroidogenesis in Leydig cells through multiple pathways, including oxidative stress and altered gene expression of steroidogenic enzymes [8, 14].
Steroidogenesis at the molecular level relies on the synchronized function of essential enzymes, such as cholesterol side chain cleavage enzyme (CYP11A1), 3β-hydroxysteroid dehydrogenase (HSD), and 17β-HSD. As indicated by experimental studies, diets high in fructose or sugar inhibit the expression of these enzymes in Leydig cells, thereby hindering testosterone production at several stages of the steroidogenic pathway. Oxidative stress induced by hyperglycemia further impairs mitochondrial cholesterol transport. It diminishes the activity of the StAR protein, thereby reducing the amount of substrate available for testosterone synthesis and exacerbating endocrine dysfunction [15, 16].
Several intracellular signaling pathways mediate the effects of excess glucose and fructose on testosterone biosynthesis. At the central (HPG axis) level, hyperglycemia-associated metabolic stress has been proposed to suppress GnRH and gonadotropin secretion through activation of AMP-activated protein kinase (AMPK) and inhibition of the mechanistic target of rapamycin (mTOR) pathway in hypothalamic neurons, both of which act as nutrient-sensing nodes [9]. Chronic nutrient excess also promotes low-grade inflammation via NF-κB-mediated inflammatory signaling, which may suppress kisspeptin-KISS1R signaling and thereby reduce GnRH pulsatility [15]. At the peripheral (Leydig cell) level, transcriptional regulation of steroidogenesis involves steroidogenic factor-1 (SF-1), cAMP response element-binding (CREB) protein, and peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), which coordinate the expression of StAR and steroidogenic enzymes [13, 14]. High glucose and fructose intake may suppress these transcription factors by promoting oxidative stress and mitochondrial dysfunction, thereby impairing substrate delivery and enzyme activity [14–16]. At the metabolic signaling level, carbohydrate-responsive element-binding protein (ChREBP) and sterol regulatory element-binding protein 1c (SREBP-1c) are induced by excess dietary sugar, promoting de novo lipogenesis and potentially contributing to lipotoxic disruption of Leydig cell function [8]. Together, these converging pathways provide a mechanistic framework linking dietary sugar excess to impaired steroidogenesis at multiple regulatory levels.
It is important to distinguish between direct and indirect mechanisms through which sugar may influence testosterone homeostasis. Direct effects refer to acute, rapid responses that occur independently of weight gain or metabolic disease: these include post-prandial hyperglycemia, which triggers transient inflammatory cytokine release (notably TNF-α), and hyperinsulinemia, both of which are proposed to act directly on Leydig cells to suppress steroidogenesis within hours of ingestion [10, 11]. These acute suppressive effects have been observed even in metabolically healthy, non-obese men, suggesting a mechanism that does not require chronic metabolic disruption.
Indirect effects, by contrast, are mediated through the chronic metabolic consequences of habitual high-sugar intake, primarily visceral adiposity, insulin resistance, and leptin dysregulation. These secondary pathways operate over longer time scales and may produce more sustained impairment of the HPG axis, Leydig cell function, and spermatogenesis [8, 12]. The distinction is clinically relevant: addressing acute glucose spikes (e.g., through glycemic index reduction) and chronic metabolic disease (e.g., through weight loss and insulin sensitization) may require different therapeutic approaches. Future research should separately quantify these direct and indirect pathways using controlled study designs that distinguish acute from chronic exposure.
Previously, it was believed that chronic sugar consumption, leading to visceral fat accumulation, suppressed the hypothalamic-pituitary-testicular axis by converting testosterone to estradiol, thereby inducing central negative feedback. However, this prevailing notion is currently under scrutiny, as contemporary mass spectrometry findings reveal lower circulating estradiol levels in obese men, correlating with reduced testosterone levels, alongside observations of diminished adipose tissue aromatase expression in obese men with low testosterone levels [8].
Diets high in fructose, studied in male rats, cause major reproductive damage, leading to primary hypogonadism, as evidenced by a notable decline in serum testosterone levels (about a 28% reduction) and a two-fold rise in LH and follicle-stimulating hormone (FSH) levels. This metabolic dysfunction results in significantly reduced testicular and epididymal weights, decreased sperm count, and disrupted spermatogenesis. The damage is primarily driven by oxidative stress, in which fructose-related dyslipidaemia elevates reactive oxygen species (ROS) levels, which harm sensitive sperm DNA and mitochondrial function, leading to germ cell death and DNA fragmentation [17].
These hormonal and functional issues indicate potential reversibility in humans: men experiencing subfertility who adhered to a three-month dietary program, particularly an antioxidant-rich, low-carbohydrate Mediterranean diet emphasizing 80% organic foods, demonstrated a significant rise in testosterone levels. Additionally, this method resulted in a considerable reduction in the sperm DNA fragmentation index (DFI) within the low-carbohydrate group, validating the reparative effect of antioxidant-rich diets on metabolic harm [18].
Low testosterone levels are associated with a higher cardiovascular risk, non-alcoholic fatty liver disease, mood disorders, and decreased muscle mass, conditions commonly observed in men with high sugar intake and central obesity. Chronic low-grade inflammation caused by a poor diet affects not only metabolic pathways but also spermatogenesis, leading to reduced fertility and overall reproductive potential [19].
Lifestyle changes during and after COVID-19, such as increased snacking, stress-induced sugar consumption, reduced physical activity, and disrupted sleep, have worsened the metabolic profiles of men. Although SARS-CoV-2 infection has been linked to testicular dysfunction, the more significant factor in ongoing hormonal imbalance seems to be the post-pandemic shift in dietary and lifestyle habits that increase metabolic stress [20].
Accumulating evidence from human and experimental studies indicates that excessive intake of sugar and confectionery products adversely affects testosterone regulation, suggesting a dose-dependent impairment of male reproductive endocrine function. Findings from short-term feeding experiments, population-based cohort studies, and dietary interventions are summarized in Table 1.
Methodological characteristics and hormonal outcomes of human studies evaluating the impact of dietary sugar intake on serum testosterone levels in males.
| Study | Study type | Population | Sugar exposure | Fasting status/Sampling context | Testosterone assay method | Confounder adjustment | Main hormonal finding |
|---|---|---|---|---|---|---|---|
| Acute studies: Single glucose load or short-term postprandial feeding | |||||||
| Iranmanesh et al., 2012 [10] | Acute physiological interventional study | 57 healthy adult men (19–78 years) | 75 g oral glucose load vs. water | Overnight fast; intensive 10-min interval sampling for 6.5 h | Chemiluminescent immunoassay | Multivariate regression for age, BMI, visceral fat, and metabolic hormones | Basal T secretion: 5,256 → 4,608 ng·dL−1·6.5 h−1 (↓12.3%, p < 0.0001). Total T secretion: 6,065 → 5,474 ng·dL−1·6.5 h−1 (↓9.7%, p < 0.001). T concentration slope 5.5× steeper after glucose vs. water (p < 0.001). Pulsatile LH secretion: 20 → 18 IU·L−1·6.5 h−1 (p = 0.043). |
| Gagliano-Jucá et al., 2019 [11] | Acute metabolic intervention | Healthy eugonadal men (aging OGTT cohort + young MMTT cohort) | OGTT and carbohydrate-rich mixed meal | 10–12 h overnight fast; serial early-morning sampling every 20 min | LC-MS/MS | Within-subject paired design (no covariate adjustment required) | ~20–30% transient decline in serum total testosterone within 60–90 min of glucose ingestion. |
| Van de Velde et al., 2020 [6] | Observational postprandial physiology study | 43 adult men across the age/BMI spectrum | Standardized mixed meal tolerance test | ≥ 8 h fast; sampling at baseline, 30, 60, 120 min | LC-MS/MS | Linear mixed-effects modelling including metabolic variables | Mean total T decrease: 13 ± 12% at 30 min; 15 ± 15% at 60 min (p < 0.001 at all timepoints). Free T decrease: 15 ± 13% at 30 min; 17 ± 16% at 60 min. Younger men (≤ 40 years) showed an additional 2.7 nmol/L steeper decline at 60 min vs. older men (p < 0.001). SHBG unchanged. |
| Pearce and Tremellen, 2019 [3] | Dietary metabolic pilot intervention | Overweight/obese men (18–50 years) | Macronutrient feeding protocols, including the refined carbohydrate arm (orange juice) | Overnight fast; baseline and hourly sampling over 5 h | Not explicitly specified in methods | Adjustments for age, BMI, habitual diet, sleep, and activity | Carbohydrates alone (orange juice): no significant change vs. fasting (p = NS). Carbohydrates + PUFA: significantly reduced T vs. fasting (p = 0.040). Mean baseline T: 11.7 ± 3.0 nmol/L. PUFA alone: ↓3.2 nmol/L at 1 h (p = 0.023), persisting to 5 h (p = 0.012). |
| Chronic/Epidemiological studies: habitual dietary patterns or long-term dietary intervention | |||||||
| Chen et al., 2018 [12] | Cross-sectional epidemiological study (NHANES) | 991 US men aged 20–39 years | SSB intake (24-h recall) | Single blood sampling; timing accounted for circadian variation | Electrochemiluminescence immunoassay | Multivariable adjustment (age, BMI, race, lifestyle, socioeconomic factors) | Highest SSB quartile (≥ 442 kcal/day) vs. lowest (≤ 137 kcal/day): aOR for low testosterone = 2.29 (p = 0.041). BMI ≥ 25 kg/m2: aOR = 3.68 (p = 0.044) as an independent risk factor. |
| Nassan et al., 2021 [5] | Cross-sectional fertility cohort study | ~2,935 Danish young men (~18 years) | Soft drink intake via validated FFQ | Morning venous sampling; time-of-day adjustment in models | Time-resolved fluoroimmunoassay/ELISA (platform changed during study) | Extensive adjustment (age, BMI, lifestyle, diet patterns, SES, substance use) | Total testosterone: p-trend = 0.15 (not statistically significant across SSB quartiles). Primary significant effects: sperm concentration –13.0 million/mL in the highest SSB quartile vs. non-consumers (95% CI: –21.0, –5.5; p-trend = 0.001); inhibin-B –12 pg/mL (95% CI: –21, –4). |
| Corsetti et al., 2023 [18] | Prospective dietary lifestyle intervention | 50 subfertile men (35–45 years) | Low-carbohydrate organic Mediterranean diet (3 months) | Post-intervention hormonal assessment; fasting status not clearly specified | Not specified | Minimal reported adjustment | Testosterone: 3.2 ± 0.3 ng/mL → 6.92 ± 1.16 ng/mL (↑116%; p = 0.011; n = 30). Sperm DFI: 44.2 ± 3.02% → 23.2 ± 3.57% (↓47.5%; p = 0.001; n = 20 in low-carb subgroup). |
aOR: adjusted odds ratio; BMI: body mass index; DFI: DNA fragmentation index; ELISA: enzyme-linked immunosorbent assay; FFQ: food frequency questionnaire; LC-MS/MS: liquid chromatography-tandem mass spectrometry; LH: luteinizing hormone; MMTT: mixed meal tolerance test; NHANES: National Health and Nutrition Examination Survey; OGTT: oral glucose tolerance test; PUFA: polyunsaturated fats; SES: socioeconomic status; SHBG: sex hormone binding globulin; SSB: sugar-sweetened beverages.
Short-term clinical studies indicate that glucose ingestion may be associated with a rapid 20–30% decline in circulating testosterone in healthy eugonadal men, although findings may vary across populations; the effect is potentially driven by hyperinsulinemia and transient suppression of gonadotropin activity [11]. Long-term population data further reinforce this relationship. Epidemiological analyses have reported that higher intake of SSBs is associated with reduced testosterone and poorer reproductive hormone profiles among men in large observational cohorts, including those evaluating dietary quality and endocrine function in healthy and subfertile males [2, 5]. Dietary pattern studies similarly demonstrate that diets rich in confectionery, refined carbohydrates, and ultra-processed foods correspond with poorer semen parameters, increased oxidative stress, and reduced steroid hormone levels, suggesting a metabolic and inflammatory contribution to gonadal dysfunction [1, 12].
Although limited, interventional evidence supports these associations. A controlled dietary trial demonstrated that transitioning men from a high-glycemic, sugar-rich diet to a low-glycemic index pattern improved testosterone and sex hormone binding globulin (SHBG) levels, suggesting that metabolic suppression is reversible when sugar intake is reduced. Mechanistic clinical investigations also reveal that obesity, insulin resistance, and low-grade inflammation, conditions associated with excessive sugar intake, represent proposed routes linking diet to testosterone insufficiency [3]. Methodological variability, including differences in hormone assays (immunoassay vs. liquid chromatography-tandem mass spectrometry [LC-MS/MS]), fasting status, and confounding factors such as BMI and lifestyle, contributes to heterogeneity in reported magnitudes but does not diminish the consistent direction of effect (Table 1).
Experimental animal models provide strong mechanistic support for the associations observed in human studies. Key findings from representative rodent investigations are summarized in Table 2.
Experimental animal studies investigating the effects of high-sugar and metabolically adverse diets on testosterone regulation and male reproductive outcomes.
| Study | Animal model | Diet composition | Dose/Exposure mode | Duration | Testosterone outcome | Testicular/Reproductive outcome |
|---|---|---|---|---|---|---|
| Tkachenko et al., 2020 [17] | Juvenile male Wistar rats | 10% fructose drinking solution (~100 g/L) with standard pellet chow | Ad libitum fructose solution replacing drinking water | 60 days (juvenile → pubertal transition) | ↓Serum testosterone with compensatory ↑LH and FSH (ELISA-based measurement) | Seminiferous epithelial degeneration, reduced spermatogenesis index, ↓epididymal sperm count, impaired fertilising capacity |
| Hsia et al., 2022 [21] | Adolescent male Sprague-Dawley rats | High-fructose diet (65% fructose chow) vs. standard diet | Ad libitum high-fructose feeding | 21 weeks (chronic exposure) | ↓Plasma and Leydig cell testosterone production (RIA-based measurement) | Impaired LH/cAMP-PKA signaling responsiveness, reduced steroidogenic capacity in isolated Leydig cells, ↓sperm vitality with metabolic dysregulation (hyperinsulinemia, hyperglycemia) |
| Sertorio et al., 2022 [22] | Male Wistar rat offspring | HFHS diet (32% fat; 50% carbohydrates with ~25% from sugar; sweetened condensed milk source) vs. control diet | Ad libitum parental dietary exposure (paternal preconception + maternal gestation/lactation) followed by offspring standard diet | 10 weeks paternal exposure + gestation/lactation programming; offspring evaluated at 90 days | ↓Serum testosterone (chemiluminescence assay) with reduced expression of steroidogenic genes (StAR, Hsd17b3) | Testicular inflammation (↑cytokines), oxidative stress, impaired spermatid number and sperm morphology, and altered epigenetic regulatory markers |
| Jing et al., 2020 [13] | Adult male C57BL/6J mice + primary rat Leydig cells/TM3 Leydig cell line | HFD (34.9% fat; 26.3% carbohydrate) vs. standard diet | Ad libitum high-fat feeding; mechanistic in-vitro oxLDL exposure (25–100 µg/mL) | 16 weeks (chronic metabolic exposure) | ↓Serum testosterone and reduced Leydig cell steroidogenesis (ELISA-based hormone measurement) | oxLDL accumulation via CD36 signaling, mitochondrial dysfunction (↓respiratory complex activity, ↓ATP), ↑ROS production and MAPK activation, reduced expression of steroidogenic proteins (StAR, P450scc, 3β-HSD, 17β-HSD), impaired sperm motility and count |
| Suleiman et al., 2020 [14] | Adult male Sprague-Dawley rats (obesity model) | HFD: 68 g standard chow + 32 g ghee + 12% cholesterol (energy-dense obesogenic diet) vs. standard diet | Ad libitum high-fat feeding | 12 weeks (chronic exposure) | ↓Serum and intra-testicular testosterone; altered LH and FSH levels | ↓Expression of steroidogenic genes (StAR, CYP11A1, 3β-HSD, 17β-HSD), ↑oxidative stress in epididymal tissue, impaired sperm motility, viability, and morphology, reduced Leydig cell number, and spermatogenic disruption |
ATP: adenosine triphosphate; CD36: cluster of differentiation 36; CYP11A1: cholesterol side chain cleavage enzyme; ELISA: enzyme-linked immunosorbent assay; FSH: follicle-stimulating hormone; HFD: high-fat diet; HFHS: high-fat high-sugar; HSD: hydroxysteroid dehydrogenase; LH: luteinizing hormone; MAPK: mitogen-activated protein kinase; oxLDL: oxidized low-density lipoprotein; RIA: radioimmunoassay; ROS: reactive oxygen species; StAR: steroidogenic acute regulator.
Rodent studies complement human findings by demonstrating that high-fructose or high-sucrose diets lead to significant reductions in serum testosterone, testicular degeneration, and decreased expression of steroidogenic enzymes such as StAR and 17β-HSD. These changes are frequently accompanied by increased oxidative stress, mitochondrial impairment, and inflammatory signaling within the testes, mechanisms similarly identified in human metabolic and fertility disorders [15, 16]. Prolonged exposure amplifies these effects, and liquid sugar appears to produce metabolic disruption more rapidly than solid confectionery forms, mirroring human post-prandial metabolic differences [21]. These models offer important mechanistic insight into how long-term sugar may affect steroidogenesis and testicular function. However, species-specific metabolic variations restrict direct translation of these models. Notably, rodents used in these studies were often exposed to extreme direct conditions (e.g., 60–65% fructose by weight) that far exceed typical human intake, limiting direct dose extrapolation [21]. Human-equivalent dose considerations and comparative metabolic pharmacokinetics should be incorporated in future experimental designs to strengthen translational relevance [17, 21].
Together, human and animal data converge on a coherent model wherein excessive sugar intake contributes to testosterone suppression through metabolic dysregulation, oxidative stress, and impaired steroidogenesis. While species-specific differences limit direct extrapolation from rodent models, the biological plausibility and consistency across mechanistic and clinical observations underscore a likely causal pathway. Current evidence highlights the urgent need for well-controlled, long-term human trials using standardized hormonal assessments to determine dose thresholds and reversibility [8] (Table 2).
Males without diabetes exhibit higher testosterone levels than those with the disease, while more active men have higher testosterone levels than sedentary men. Consuming SSBs has been linked to several medical conditions, including weight gain, type 2 diabetes, cardiovascular disease, hyperuricemia, and gout, according to several studies that used the National Health and Nutrition Examination Survey (NHANES) database [12]. Available evidence on SSB intake and sperm motility remains inconclusive; one study reported no significant association between SSB intake and sperm motility, while observing a positive association with normal sperm morphology and estradiol level [23]. Changes in the HPG axis, elevated scrotal temperature due to scrotal and abdominal fat deposition, or the build-up of liposoluble endocrine disruptors in adipose tissue have all been proposed as explanations for the effects of excess body weight on sperm production. SSBs have been linked to weight gain and obesity in numerous studies, including randomised studies [24, 25]
Non-caloric sweeteners (NCS), a popular sugar alternative that delivers strong sweetness with little to no energy, have seen increased consumption in recent years. Our food supply is rich in NCS, which can be found in consumables such as chewing gum, yoghurt, and diet drinks. NCS appears to affect male fertility in rodents; some studies indicate that animals consuming aspartame, saccharin, and stevia have significantly lower sperm quality and quantity. The effects of NCS may be dose-dependent, with higher intakes associated with worse outcomes than lower intakes and controls. However, there has been little research on how NCS affects human reproduction, especially in relation to male reproductive health [26]. Sweeteners can be broadly categorized into nutritive sweeteners (e.g., sucrose, fructose) that provide caloric energy and NCS (e.g., aspartame, sucralose, saccharin) that provide minimal or no energy [27].
Although the literature is conflicting, sweeteners have a variety of effects on glycemia, hormone levels, and cellular pathways [27]. The impact of sweeteners on hormone levels has been the subject of conflicting research [28]. Significantly, there is limited evidence linking NCS to human testosterone regulation [26]. The majority of the available information comes from rodent models, in which high-dose exposure to sweeteners, including aspartame and saccharin, has been linked to decreased androgen levels, oxidative stress, and diminished spermatogenesis [26, 28]. Despite these findings in rodents, only a few human clinical investigations are assessing endocrine outcomes, including testosterone, LH, or SHBG. As a result, it remains unclear whether NCS has independent endocrine effects or whether the reported changes are due to broader metabolic changes [26]. Without altering insulin concentration, sucralose increases body weight, reduces food intake, and lowers glycemia and glucose insulinotropic peptide (GIP) release. Non-nutritive sweeteners in beverages (either by themselves or within blends) do not, however, considerably affect the responses to glucose, insulin, GLP-1, GIP, PYY, ghrelin, and glucagon, in contrast to water controls [28].
The link between SARS-CoV-2 infection and primary testicular damage is evident, characterized by disrupted steroidogenesis and deteriorating spermatogenesis. The lack of the virus in seminal fluid suggests a minimal risk of sexual transmission of the infection to partners and offspring. Nevertheless, most findings are based on short-term follow-ups, and only a limited number of studies have explored the long-term effects of the viral infection. Therefore, additional research is necessary to assess the long-term implications for male reproductive health [29].
The pro-inflammatory diet was associated with decreased TT and estradiol E2 levels in male adolescents, but no statistically significant association was detected in male children. Managing diet may be crucial for the developmental and reproductive health benefits in male adolescents. However, additional studies are necessary to establish the causal link between DII and sex hormones [30]. The levels of testosterone and estradiol found in younger adults lower the production of pro-inflammatory cytokines by human macrophages when triggered by COVID-19-related stimuli. Observational data suggest that older age and obesity-related hypogonadism are associated with higher COVID-19 severity and mortality, though the precise role of declining sex hormone levels in this relationship requires further investigation [31].
Mass spectrometry is the gold-standard method for measuring steroid hormones [32]. And these methods are preferable for measuring sex steroid hormones [33]. Given its high sensitivity and specificity, LC-MS/MS has emerged as the preferred method for quantifying steroid hormones in human serum [32]. Although technical advancements in the previous decade have dramatically improved the accuracy, sensitivity, and throughput of steroid analysis, these analytical tools are not without flaws. However, adhering to standard parameters for the accurate assessment of steroid concentrations, including method and assay standardization, provides researchers and clinicians with the superior analytical power of validated GC- and LC-MS/MS methods, which surpass that of immunoassays. The simultaneous detection and quantification of multiple end-point steroids as well as their intermediates in a single analysis is one of the greatest advantages of using LC-MS/MS—a snapshot of an entire metabolic pathway as well as single end-point steroids [34].
The real-time data collection capabilities of continuous glucose monitoring (CGM), combined with advanced analytical tools, have enhanced the precision and timeliness of glucose assessment. CGM allows for continuous, individualized monitoring, providing clinicians with detailed insights into glucose fluctuations that can inform diagnostic decisions and guide early intervention strategies. Future research should prioritize the development of higher-precision CGM devices, improved data interpretation algorithms, and integrated monitoring systems. Through interdisciplinary collaboration and a supportive policy framework, CGM technology has the potential to become a standard component of metabolic health assessment [35].
CGM also demonstrates potential for better dietary practices by providing immediate feedback on glucose responses to food intake. While its impact on physical activity remains unclear, CGM enables personalized interventions that account for individual physiological variability, allowing more precise and targeted recommendations [36]
Regarding sugar intake, the updated WHO guidelines reflect stronger evidence linking high sugar consumption to adverse health outcomes. The guidelines highlight the personal, social, and economic consequences of excessive sugar consumption, underscoring the need for systematic public health action. Public health nutritionists and policymakers should leverage these guidelines to implement interventions that encourage healthier diet patterns and reduce population-level sugar intake [37].
There is now increasing evidence that sugar and SSBs are associated with lower serum testosterone levels in some male populations. However, significant gaps remain that prevent causal inference and therapeutic translation in real-world settings. There are only a few large-scale, long-term randomized or well-controlled interventional trials that measure testosterone longitudinally, and the majority of human data are cross-sectional (e.g., NHANES analyses) [12]. There also exist measurement and comparability issues between studies, mainly caused by assay heterogeneity (immunoassays vs. LC-MS/MS), differing sampling settings (fasting vs. non-fasting), and inconsistent confounding correction (BMI, insulin resistance, SHBG) [33, 38]. There are limited studies that differentiate liquid SSBs from solid forms of confectionery, even though their metabolic fates and postprandial endocrine reactions are most likely different. Literature and evidence are rare in adolescents and older/aging males, despite the developmental and age-related changes of the HPG axis [39].
Future research should focus on multi-center longitudinal cohorts and randomized feeding/intervention trials employing the gold standard technique of LC-MS/MS testosterone quantification and standardized sample methodology [32, 40]. Mapping of the diet, metabolite, and hormonal pathways, identifying mediators and biomarkers, can be helped by integration of multi-omics (metabolomics, proteomics, gut microbiome) [41, 42]. Controlled trials are also required for non-nutritive sweeteners and endocrine effects, as they have given mixed signals in humans and animal models [43, 44]. From a public-health standpoint, it’s important to evaluate sugar taxes and fiscal policies for their endocrine and metabolic benefits for high-risk men [45, 46], alongside product information, labeling, and primary care screening protocols [45].
Growing evidence from human observational studies and animal models links excessive consumption of added sugars and confectionery to adverse effects on serum testosterone regulation and male reproductive health. Acute excessive sugar intake can briefly suppress testosterone secretion. Meanwhile, chronic intake, especially of sweetened beverages, has been linked to metabolic dysfunction, insulin resistance, inflammation, and oxidative stress, which may impair Leydig cell steroidogenesis and disrupt HPG axis signaling. These pathways are supported by experimental findings demonstrating testicular injury and suppression of steroidogenic enzymes under chronic high-sugar diets.
Despite methodological variability and sparse interventional data limiting causal inference, the consistency of findings across mechanistic, clinical, and epidemiological domains supports a biologically plausible association between excessive sugar intake and testosterone suppression. It is important to emphasize, however, that the majority of human evidence derives from cross-sectional and observational studies, which are susceptible to residual confounding and do not permit definitive causal conclusions. The distinction between association and causation must therefore be maintained when interpreting these findings. Recent studies suggest that dietary changes, weight loss, and improved metabolic health may partially reverse testosterone-suppressing effects, though controlled trial specifically designed to test this reversibility are lacking.
These findings have significant implications for public health because sugar intake, obesity, and functional hypogonadism are on the rise globally, especially among younger men. Future research featuring longitudinal designs, integrative metabolic methodologies, and standardized hormone measuring techniques is crucial to identify dose thresholds, susceptible populations, and optimal therapies. Currently, improved public health policies and nutritional advice targeted at lowering added sugar consumption might be an effective strategy to promote better male reproductive, endocrine health, and broader metabolic benefits.
aOR: adjusted odds ratio
BMI: body mass index
CGM: continuous glucose monitoring
GIP: glucose insulinotropic peptide
GnRH: gonadotropin-releasing hormone
HPG: hypothalamic-pituitary-gonadal
HSD: hydroxysteroid dehydrogenase
LC-MS/MS: liquid chromatography-tandem mass spectrometry
LH: luteinizing hormone
NCS: non-caloric sweeteners
NHANES: National Health and Nutrition Examination Survey
SHBG: sex hormone binding globulin
SSBs: sugar-sweetened beverages
StAR: steroidogenic acute regulatory
TNF- α: tumor necrosis factor-α
TT: total testosterone
Artificial intelligence—based tools were used to assist with language editing and formatting of tables and figures. All scientific content, data interpretation, and final editorial decisions were made solely by the authors, who take full responsibility for the accuracy and integrity of the work.
RHAR: Conceptualization, Writing—original draft, Writing—review & editing, Supervision. ASK: Conceptualization, Writing—original draft, Writing—review & editing, Supervision. NT: Conceptualization, Writing—original draft, Writing—review & editing. SIA: Conceptualization, Writing—original draft, Writing—review & editing. SSA: Conceptualization, Writing—original draft, Writing—review & editing. TL: Conceptualization, Writing—original draft, Writing—review & editing. RG: Conceptualization, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2575
Download: 49
Times Cited: 0
