Affiliation:
1Division of Rheumatology, Centro de Diagnóstico de Osteoporosis y Enfermedades Reumáticas (CEDOR), Lima 15036, Perú
Email: maritzavw@gmail.com
ORCID: https://orcid.org/0000-0001-8494-5260
Affiliation:
2Department of Internal Medicine and Rheumatology, School of Medicine, University of California at Davis, Sacramento, CA 95817, USA
ORCID: https://orcid.org/0000-0001-7578-8603
Explor Endocr Metab Dis. 2026;3:101466 DOI: https://doi.org/10.37349/eemd.2026.101466
Received: January 24, 2026 Accepted: March 25, 2026 Published: April 09, 2026
Academic Editor: Harold Lebovitz, State University of New York Downstate Medical Center, USA.
Osteoarthritis (OA) is the most prevalent form of arthritis and a leading cause of disability worldwide. Epidemiological studies indicate that its global prevalence is rising, particularly among women, who often experience more severe disease than men. This sex disparity is most pronounced around menopause and is influenced by female-specific biological and hormonal factors, joint morphology, reproductive history, and sex-specific comorbidities that contribute to disease onset and progression. A mechanistic understanding of these factors is essential to identify individuals at risk, implement preventive strategies, enable early diagnosis, and mitigate complications, ultimately preserving function and quality of life. This review focuses on the determinants underlying the higher incidence and severity of OA in women, highlighting the interplay between hormonal changes, anatomy, and systemic factors.
Osteoarthritis (OA) imposes a considerable and growing burden on women’s health worldwide. Women not only exhibit higher prevalence but also experience more pronounced symptoms, faster radiographic progression, and greater functional limitations than men [1–4]. These differences become particularly evident after menopause, when hormonal shifts may influence cartilage integrity and inflammatory pathways [1, 5]. The effects on mobility, daily activities, and quality of life drive substantial healthcare utilization across populations. With global aging trends, the number of women affected by OA is expected to rise further, emphasizing the need for earlier recognition and more effective long-term management strategies [3, 6–8]. Despite increasing attention to sex-specific aspects of OA, much of the existing evidence comes from studies that do not stratify results by sex. Consequently, conclusions regarding female-specific mechanisms are often extrapolated from general OA research. Identifying these gaps is crucial to guide future studies and improve understanding of sex-related differences in OA.
This narrative review was conducted using a comprehensive literature search of PubMed/MEDLINE, Scopus, and Web of Science for articles published up to March 2026. Search terms included combinations of “osteoarthritis”, “sex differences”, “women”, “hormones”, “menopause”, “metabolic syndrome”, and “polycystic ovary syndrome (PCOS)”. Additional relevant studies were identified through manual review of the reference lists of selected articles. Original research articles, cohort studies, case-control studies, cross-sectional studies, and meta-analyses published in English were considered. Priority was given to studies with sex-stratified analyses and those specifically addressing female populations. Given the narrative nature of this review, no formal quality assessment or meta-analysis was performed; however, emphasis was placed on high-quality and widely cited studies.
Global OA prevalence has risen sharply, influenced by epigenetic factors, diet, sedentary lifestyles, and improved diagnostic imaging, allowing earlier identification [2, 6, 9]. Prevalence estimates vary depending on whether clinical or radiographic criteria are used and the joints assessed [10]. The Global Burden of Disease Study reports a 132.2% increase in OA since 1990, with 7.6% of the global population affected in 2020 [6]. Women consistently exhibit higher OA prevalence, accounting for approximately 60% of cases, with around 18% of women over 60 affected, likely reflecting genetic, hormonal, and anatomical factors [11, 12]. In the United States, OA affects 32.5 million people, with symptomatic knee OA (KOA) occurring in about 10% of men and 13% of women aged ≥ 60 years [13, 14]. This disparity emerges around age 40 [11, 15], with higher risks for incident hand and KOA in women documented [16]. Meta-analyses confirm a greater KOA prevalence in women than men (21.7% vs. 11.9%, respectively) [15]. Before age 50, women may have higher OA prevalence at most joint sites; after 50, hand, knee, and foot OA predominate in women, whereas hip OA (HOA) is more common in men [17]. Given its substantial disability burden, OA underscores the need for strengthened prevention and early intervention strategies at younger ages [18].
OA exhibits important sex-specific differences that vary by joint. Hand and KOA are consistently more prevalent in women, particularly after menopause, suggesting a role for hormonal factors such as estrogen deficiency [11, 16, 17]. In contrast, HOA is more evenly distributed between sexes or may be slightly more frequent in men in some populations, with structural and morphological factors contributing more prominently [16, 17]. Spine OA shows a heterogeneous pattern, with sex-related differences influenced by both hormonal and mechanical factors across the lifespan [19–21]. These patterns indicate that OA in women is not uniform across joints and likely reflects a complex interplay between biological and biomechanical determinants (see Table 1). Evolving lifestyle and demographic trends in women, including increased participation in high-impact physical activities, greater occupational demands, rising prevalence of obesity and metabolic risk factors, and hormonal transitions across the lifespan, may further influence OA distribution and progression. However, longitudinal data specifically addressing these sex-related changes remain limited, with much of the available evidence derived from epidemiological and mechanistic studies rather than longitudinal analyses [13–16, 19–21].
Sex-specific differences in OA by joint: epidemiology and contributing factors in women.
| Joint | Prevalence in women vs. men | Female-specific factors | Key references |
|---|---|---|---|
| Hand OA | Higher in women, especially after age 50, incidence increases markedly after menopause | Menopause-related estrogen decline, higher frequency of inflammatory/erosive phenotypes, polyarticular involvement | [4, 10–13, 16, 17] |
| Knee OA | Higher in women, particularly after menopause | Obesity, altered biomechanics (alignment, muscle strength), greater symptom severity, and progression | [4, 10, 12, 13, 15, 16] |
| Hip OA | Similar or slightly higher in men (population-dependent), more strongly associated with structural and morphological factors | Differences in hip morphology and a less clear hormonal influence | [13, 16, 17] |
| Spine OA | Mixed distribution | Postmenopausal degenerative changes, interaction of hormonal factors with mechanical load, and sex-specific degeneration patterns | [10, 13, 19–21] |
OA: osteoarthritis.
Articular cartilage is a specialized connective tissue, primarily composed of an extracellular matrix (ECM) rich in type II collagen and proteoglycans (glycosaminoglycans), along with smaller amounts of glycoproteins and other non-collagenous proteins [18, 22]. Its only resident cells are chondrocytes, which rely entirely on nutrient diffusion from synovial fluid because the tissue lacks vascular, neural, and lymphatic supply [18, 20, 22–25]. Structurally, cartilage is organized into distinct zones: a superficial layer with flattened chondrocytes and collagen fibrils aligned parallel to the surface; an intermediate zone containing rounder cells and obliquely oriented fibers; and a deep zone characterized by columnar chondrocytes and perpendicular collagen bundles [18, 22, 24–27] (See Figure 1). The tidemark, which is rich in proteoglycans, separates these layers from the underlying calcified cartilage, where hypertrophic chondrocytes anchor the matrix to the subchondral bone [18]. This zonal architecture is critical for distributing load and minimizing friction during joint motion [26]. The ECM is composed predominantly of type II collagen, with type XI collagen contributing to fibrillar organization, while proteoglycans such as aggrecan, hyaluronic acid, and chondroitin sulfate fill the interfibrillar space [22, 28–30]. Together, these components provide the mechanical stability and resilience characteristic of healthy articular cartilage [22, 24, 25, 30].
Recent studies have demonstrated clear sex differences in cartilage structure and composition, which might explain why OA is more frequent and often more severe in women [11]. A recent systematic review involving nearly 2,000 participants reported that men generally have larger cartilage volumes and different morphological features, while women, particularly after menopause, tend to show more rapid degenerative changes [31]. This lower cartilage volume in females may predispose them to faster structural decline. In established OA, higher T1-rho values measured by magnetic resonance imaging (MRI) in women indicate reduced proteoglycan content and altered cartilage composition, aligning with the greater frequency of lateral compartment joint-space narrowing observed in women [11, 32]. Another study using ultrasound in 232 healthy adults observed consistently thicker cartilage in men across multiple knee regions. Age-related variation was mainly seen in men, while women showed no meaningful age-related differences, underscoring that sex and age should be considered when interpreting cartilage thickness as a potential early marker of KOA [33]. Some anatomical differences suggest greater structural vulnerability for OA in women, as demonstrated by Eckstein et al. [34]. Their study showed consistent sex-based disparities: women exhibited smaller joint space width, more frequent meniscal damage, and smaller ligaments and cartilage dimensions, even after adjusting for confounders, while cartilage composition remained largely similar between sexes [34]. Finally, other MRI-based work points to sex-specific differences in bone geometry, such as the posterior condylar offset of the femur and tibial slope, which may affect how cartilage is loaded and contribute to differing OA susceptibility [35].
OA in women arises from a complex interaction of biological, mechanical, metabolic, and reproductive factors that collectively influence joint integrity across the lifespan. Unlike men, women experience distinct hormonal transitions, sex-specific biomechanics, and metabolic profiles that modify tissue vulnerability and accelerate degenerative changes. These influences act alongside non-modifiable determinants such as age, genetics, and family history, creating a multifactorial risk landscape [36]. The categories below outline the main factors that have been linked to a higher risk of developing OA in women (See Table 2).
Factors associated with the development of osteoarthritis (OA) in women.
| Type | Factor |
|---|---|
| Non modifiable | GeneticsFemale sexAge |
| Modifiable | ObesityJoint injuryOccupational load |
| Female-specific | Anatomic and biomechanicalSmaller vertebral bodiesLower muscle supportGreater ligament flexibilityReduced stability Q angleGreater lumbar curvatureLimb alignmentQuadriceps strengthAcetabular and femoral morphology |
| Hormonal and metabolicOverweight and obesityVitamin D deficiencyMetabolic syndromePolycystic ovary syndromeLow estrogen and progesterone levelsLow testosterone levels | |
| Reproductive and menstrual factorsEarly menarcheEarly menopauseEarly age of parityLower age at first live birthPregnancy biomechanicsMultiparityDuration of breastfeedingEarlier age of oral contraceptive pills |
Q angle: quadriceps angle.
Genetics is a major contributor to OA risk, shaping height, weight, and body mass index (BMI) from childhood through adolescence [37], and this hereditary influence is further supported by family and inheritance studies [38]. A twin study involving 130 monozygotic and 120 dizygotic women aged 48–70 reported a heritability estimate of 0.54 (95% CI: 0.43–0.65), with site-specific estimates ranging from 0.39 to 0.65, and consistently higher intraclass correlations in identical compared with non-identical twins [39]. As a polygenic disease, more than 300 OA genomic loci have been identified by using genetic analyses and genome-wide association studies (GWAS) of affected individuals; results have shown that OA genes are associated with the process of synovial joint development, playing an important role in the onset of OA as well as during the late stages of the disease [38, 40–42]. While mutations in cartilage structural genes like COL2A1 and ACAN can contribute to early-onset OA, they seem less relevant to typical late-onset cases [40, 43]. Most genetic variants found through GWAS affect transcriptional regulation, suggesting that genetics may influence cartilage repair capacity rather than trigger disease onset. Epigenetic mechanisms are also increasingly recognized but require larger studies for clarification [43]. In women, gene expression analyses have identified a set of genes closely tied to molecular heterogeneity in OA, and there is increasing evidence that some genetic markers linked to inflammatory pathways seem to heighten susceptibility to OA. Some variants show clear sex-dependent effects; for example, the FRZB T/G haplotype has been implicated in OA pathogenesis only in women, and COMP haplotypes associated with KOA differ between sexes. Conversely, certain COL2A1 haplotypes may offer some protection in men but not in women [11]. Additionally, other genes involved are part of the Wnt pathway [HMG-Box transcription factor 1 (HBP1); B cell CLL/lymphoma 9 protein (BCL9)] and transforming growth factor (TGF) pathway [TGF beta 1 (TGFβ1)], latent transforming growth factor beta binding protein 1 and 3 (LTBP1, LTBP3), SMAD3 and ROCR long non-coding RNA (lncRNA) that regulates chondrogenesis [38, 44]. Although molecular pathways such as Wnt signaling play a central role in the pathogenesis of OA, current evidence supporting sex-specific differences in these mechanisms remains limited. Therefore, their contribution to the higher burden of disease in women is not yet fully understood. Recent discoveries suggest that OA risk is driven more by altered gene regulation than by structural mutations in cartilage proteins. Epigenetic mechanisms, such as DNA methylation, histone changes, and microRNAs, likely explain why individuals with similar genetic risk can have very different clinical outcomes [45]. Progress in the field requires larger, better-defined cohorts and a clearer separation of OA into biologically distinct endotypes.
A twin study of nearly 60,000 Swedish adults evaluated how strongly genetics contribute to OA compared with other musculoskeletal and rheumatic diseases. The results showed that OA, especially in the hip, has one of the highest heritability estimates among the conditions examined. The researchers also found that OA, at any joint site, commonly co-occurred with shoulder pain and back or neck pain, and a substantial portion of these overlaps could be attributed to shared genetic factors, suggesting underlying biological mechanisms with several pain-related conditions, influenced by heredity [46]. A nationwide Swedish cohort study assessed familial risk of several OA subtypes by linking the Multigeneration Register with national healthcare records. Across more than 6.5 million individuals, all OA forms showed elevated familial hazard ratios, increasing with closer genetic relatedness. The strongest hereditary signals were observed for first carpometacarpal and polyarticular OA, followed by HOA, while knee and other OA types showed weaker but still significant familial effects [47]. Supporting this pattern, Weldingh et al. [48] examined hereditary influences on hip, knee, and hand OA in two population-based Norwegian cohorts, and reported that having a mother with OA increased both risk and severity in daughters, whereas paternal OA showed a weaker association.
Clinical and epidemiological data consistently show that women have a higher prevalence of OA across adulthood, a disparity that becomes more pronounced after menopause as symptom onset and disease progression accelerate [31, 36, 49]. This increased susceptibility is supported by evidence that women exhibit lower cartilage volume and a faster rate of cartilage loss than men; although body size contributes to these differences, sex-specific structural and biochemical characteristics of cartilage, how they change with age, also play a significant role [31]. As a result, women demonstrate higher rates of hip, knee, and hand OA compared with men [36].
A systematic review reported sex-specific risk profiles; in women, high BMI, alcohol use, and atherosclerosis were linked to greater KOA risk, whereas in men, the main factors were high physical activity, soft-drink intake, and abdominal obesity [50]. OA incidence rises around the cessation of menses [16], likely reflecting hormonal changes, body composition, joint anatomy, and muscle strength. Segal et al. [11] noted that 60% of individuals with OA were women, who also showed higher pain perception, radiographic severity, and greater functional limitations, with possible contributions from differences in central pain processing and coping strategies. Other studies similarly reported higher pain scores among women using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) [51] and visual analogue scale (VAS), with socioeconomic factors further influencing pain perception in older women [52]. These findings might be explained by the influence of psychological aspects related to chronic pain. Catastrophizing, characterized by heightened threat appraisal and persistent pain-related thoughts, has been proposed as a partial explanation for these sex differences and appears more common in women [53–55]. Pain and disability are related to the magnification and rumination of the pain-related information shown in catastrophizing, which tends to happen more often in women [55]. In a cohort of 168 OA patients, women showed greater pain, pain behaviors, and disability [53]. Experimental studies also support increased pain sensitivity and reduced pain inhibition in women [55]. Failla et al. [56] found that women perceived heat-evoked pain at lower temperatures and displayed different age-related patterns in descending pain modulatory system activity compared to men, suggesting neurobiological differences in pain processing across sexes.
The Q angle, the angle formed by the anterior superior iliac spine (ASIS)-patella and patella-tibial tubercle lines, represents the combined vector of quadriceps and patellar tendon forces and serves as a clinical indicator of patellofemoral alignment [57, 58] (See Figure 2). In women, a relatively shorter femur, wider pelvis, and larger Q angle alter lower-limb alignment and increase knee valgus forces, shifting load toward the lateral compartment and elevating the risk of lateral tibiofemoral OA. When combined with greater hip internal rotation, higher knee torque, and increased varus-valgus laxity, these structural and biomechanical features reduce joint stability during landing or rapid directional changes and help explain the markedly higher rate of ligament injuries, particularly anterior cruciate ligament tears [59].
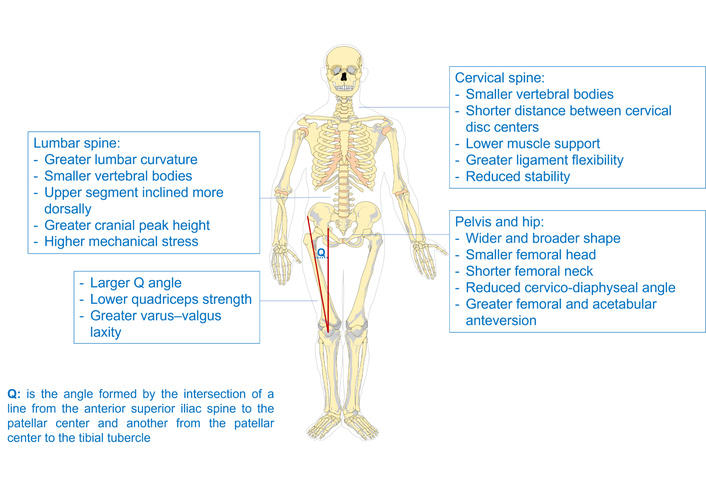
Female anatomical features of the spine, pelvis, quadriceps, and the quadriceps angle (Q angle). The original skeletal image was adapted with permission from https://pixabay.com/vectors/bones-color-anatomy-skeleton-307297/. Accessed January 12, 2026. Licensed under a Pixabay License.
Hormonal fluctuations and pregnancy may further affect ligament laxity and joint mechanics, adding to this vulnerability. Sex-specific differences in bone morphology, such as femoral rotation, condylar curvature, and tibial dimensions, have been documented across Asian populations and appear more pronounced in women. As OA progresses, women also show characteristic changes in joint alignment and morphology that differ from men, potentially contributing to less favorable loading patterns. Consistent with these structural distinctions, women exhibit a larger normalized medial tibiofemoral contact area and reduced joint congruity across OA grades [11, 59].
Women exhibit lower quadriceps strength compared to men, placing them closer to the threshold for insufficient knee stabilization. In women only, quadriceps weakness consistently predicts symptomatic and radiographic KOA, structural progression, functional decline, and future knee arthroplasty, likely reflecting reduced specific strength rather than muscle mass. Strength gains correlate with improved performance in women but not men. Hip abductor weakness similarly increases the risk of worsening knee pain in women alone. Although women demonstrate greater hamstring coactivation during quadriceps activity, this pattern is more strongly associated with cartilage deterioration in men [11]. Studies have shown that higher quadriceps strength was linked to reduced worsening of lateral patellofemoral cartilage damage, bone marrow lesions, and effusion-synovitis in women, and knee pain, suggesting a protective effect on specific joint structures in women with or at risk for KOA [60, 61]. Another study compared quadriceps-only strengthening with a combined exercise program in women aged 50–60 with KOA. After four weeks of training, both interventions significantly improved pain, balance, and the quadriceps-to-hamstring strength ratio (P ≤ 0.05), whereas the control group showed no changes. Both exercise approaches were effective for improving symptoms and function [62]. Overall, resistance training and quadriceps-focused strengthening seem to be an effective strategy for improving pain and quality of life in older women with KOA [63].
Women show a distinct pattern of cervical spine degeneration compared with men, driven in part by anatomical and biomechanical differences, which may contribute to a higher burden of cervical OA in women [64]. More broadly, structural and functional characteristics of joint tissues have been well described [18]. Studies report that women have shorter distances between cervical disc centers, a feature that declines with age and reflects sex-specific degeneration of the vertebrae and discs [65]. Clinically, they are more likely to develop persistent neck pain after trauma, a vulnerability linked to smaller vertebrae, lower muscle support, and more compliant ligaments, all of which reduce stability and increase tissue strain [49, 66]. Computed tomography (CT)-based work further shows that, even when body size is matched, men still have larger vertebral dimensions, offering greater mechanical resistance [64, 67]. These anatomical differences may have important clinical implications, as they influence load distribution and spinal biomechanics, potentially contributing to sex-specific patterns of degeneration. In clinical practice, this may be reflected in differences in pain presentation, functional limitation, and susceptibility to facet joint OA, spinal instability, and chronic low back pain in women.
Variations in collagen, cartilage, and bone properties may predispose women to degenerative conditions and specific joint pathologies across the spine [18, 29, 31, 34, 68]. Some studies report that thoracic degeneration increases with age in both sexes, with a different pattern among sexes. Older women tended to show more eburnation, while older men showed more lipping and porosity, and these contrasts became more pronounced in the elderly group [69]. Estrogen helps maintain intervertebral disc structure by supporting vascular supply, matrix turnover, hydration, and anti-inflammatory signaling while limiting apoptosis [18, 70]. Moreover, large imaging studies also demonstrate more severe disc degeneration after menopause, with the most rapid decline occurring in the first decade following the menopausal transition [71]. When men and women are matched for age and body size, women still exhibit greater disc degeneration, underscoring estrogen’s protective effect [18–20]. Dual-energy X-ray absorptiometry (DEXA)- based analyses similarly show that disc space remains stable until midlife and then decreases substantially, especially in early post menopause [72].
Sex-related differences in lumbar anatomy and degeneration are evident across imaging, biomechanical, and epidemiological studies. CT-based modeling shows that women have a greater lumbar curvature with the upper segment inclined more dorsally and a greater cranial peak height. These features create the appearance of a deeper curve even though the total degree of lordosis is similar to that of men, and may represent adaptations that reduce spinal loading during pregnancy [73]. Biomechanically, smaller vertebral dimensions further disadvantage women. Quantitative CT shows that although cancellous and cortical bone densities are comparable between sexes, women’s vertebral bodies are about 25% smaller in cross-sectional area and volume (P < 0.001). As a result, equivalent loads generate 30–40% higher mechanical stress in female vertebrae, helping explain the greater fracture susceptibility seen later in life [74]. Although this difference has been linked to anatomical and biomechanical factors, the specific contribution of body weight, muscle mass, and fat distribution remains incompletely understood, as sex-stratified data are limited.
Longitudinal evidence from the Framingham Study reinforces these patterns. Disc height narrowing and facet joint OA were already common by midlife and progressed more rapidly in women, particularly in the lumbar spine [20]. Epidemiologic data also point to a higher burden of lumbar OA in women. In a large population-based cohort, symptomatic lumbar OA affected roughly 9% of adults, with higher prevalence in women (P = 0.021) and strong associations with age, obesity, and physical labor among others [75]. Sex-specific bone geometry and joint mechanics likely contribute to the higher prevalence and severity of HOA in women compared to men [76]. Overall, HOA in women tends to occur within a broader polyarticular pattern and is characterized by more pronounced symptomatic and structural burden [77]. Females generally have a wider pelvis, a shorter femoral neck, and a smaller femoral head, along with lower femoral offset and a reduced cervico-diaphyseal (CCD) angle, as well as greater femoral and acetabular anteversion. These features can alter hip congruence and joint mechanics, increasing load on the articular surface [78]. As a result, the effective contact area may be reduced, and with routine daily loading, this configuration can contribute to accelerated cartilage wear and subchondral bone changes [76]. These meaningful differences between sexes might determine how the disease presents and progresses. In a five-year multicenter cohort of 508 adults with painful HOA, women entered the study with more widespread joint involvement, more frequent superomedial femoral head migration, and more severe symptoms than men. Although average joint-space narrowing over the first year was similar across sexes, women were more likely to experience rapid structural deterioration (OR = 2.34). Over the full follow-up period, total hip arthroplasty (THA) was performed more often in women; however, multivariate analyses indicated that surgery was primarily driven by disease severity rather than sex itself [77].
Understanding the morphological differences between sexes is essential for diagnosing and managing disorders linked to anatomical variation, as they influence hip motion before impingement occurs. In a CT-based analysis of 106 healthy hips from older adults, researchers compared acetabular and femoral features and assessed the range of motion (ROM) until bony impingement in four directions. Clear sex differences emerged in both joint orientations, such as acetabular inclination and anteversion, and femoral neck anteversion, and in the contours of the acetabular rim and femoral neck. These anatomical distinctions were accompanied by sex-specific limits in ROM across all tested movements [79]. Muscle mass distribution and lower-limb biomechanics also shape how forces are transmitted through the hip. Gait studies show clear sex differences; men generate higher mass-specific mediolateral ground reaction forces (GRF), whereas women exhibit greater hip abductor moments, a more medially positioned trochanter, and a larger GRF moment arm, indicating that female hip morphology places higher functional demands on the abductors and modifies their mechanical advantage during single-leg stance [80]. As previously mentioned, anatomical differences between men and women play a significant role in the development of biomechanical alterations; however, it is important to note that these factors alone cannot fully explain the development of OA. Other systemic factors associated with the development of the disease must be taken into account, as discussed in the following section.
Endogenous sex hormones influence the pathogenesis, pain perception, and progression of OA [81]. Higher estrogen levels have been linked to reduced pain sensitivity, while hormonal fluctuations during the menstrual cycle and menopause modulate pain responses in women [11, 82]. Although hormonal and reproductive factors appear relevant to OA development, their independent contribution relative to obesity and inflammation remains unclear [81]. Menopause represents a period of significant estrogen decline, associated with loss of bone mineral density and musculoskeletal pain, and women with more intense menopausal symptoms may have increased vulnerability to chronic back pain, arthralgia, and OA [83–87]. A recent systematic analysis showed a 1.3-fold increase in OA incidence, prevalence, and disability-adjusted life-years (DALYs) in postmenopausal women, with higher hand OA incidence at ages 55–64 and HOA at 60–64 [86]. This rise during menopause suggests a link between ovarian function and OA [88].
Sex hormones and their receptors are widely expressed in the central and peripheral nervous system and contribute to pain transmission [55]. During menopausal transition, declining estrogen increases arthralgia and pain sensitivity, whereas testosterone is associated with higher pain thresholds in men [87, 89]. In the Women’s Health Initiative, estrogen therapy reduced total joint replacement by 50% in highly adherent users [90]. Estrogen also promotes cartilage homeostasis by preventing structural damage, enhancing proteoglycan synthesis, and regulating subchondral bone growth and mineralization [91]. Low endogenous estrogen metabolites have also been associated with KOA development. In a cohort of 842 middle-aged women, those with estradiol levels in the lowest tertile (< 47 pg/mL) had increased odds of radiographic KOA (OR = 1.88; 95% CI: 1.07–3.51), as did those with low urinary 2-hydroxyestrone (OR = 2.9; 95% CI: 1.49–5.68) [92]. Hormone replacement therapy (HRT) shows protective effects on cartilage and bone. In a 2-year study of 171 postmenopausal women, both oral and transdermal estradiol, administered as continuous combined therapy (with progestins), reduced the cartilage degradation marker, urinary C-telopeptides of type II collagen (uCTX-II), by 19–30% after one year (P = 0.02 and P = 0.003 vs. placebo), and lowered the bone resorption marker urinary C-telopeptides of type I collagen (uCTX-I) by 70% compared with placebo (P < 0.0001). Oral regimens included 1 mg 17β-estradiol combined with 1–3 mg drospirenone, while transdermal therapy consisted of 45 μg 17β-estradiol combined with 30–40 μg levonorgestrel. These findings support estrogen’s protective effects on cartilage and bone [93].
Progesterone also contributes to cartilage integrity by suppressing matrix-degrading enzymes such as matrix metalloproteinases (MMPs) [87, 94]. In a study of 200 adults, low estradiol, progesterone, and testosterone levels were associated with greater knee effusion-synovitis and other OA-structural abnormalities; among women, progesterone correlated positively with cartilage volume [95]. Despite these findings, the role of HRT in OA remains controversial. Evidence regarding its clinical benefits is not entirely consistent, and most studies have focused on surrogate biomarkers or observational outcomes rather than long-term structural progression. Moreover, HRT is not without risks, including increased incidence of thromboembolic events, breast cancer, and cardiovascular complications in certain populations [90]. Current clinical guidelines do not recommend HRT specifically for the prevention or treatment of OA, and its use should be individualized based on overall risk-benefit assessment. Therefore, while estrogen may exert protective effects on joint tissues, caution is warranted when extrapolating these findings to routine clinical practice.
Age-related testosterone decline contributes to reduced muscle function, bone mass, and increased joint pain [96]. In 272 older adults undergoing unilateral total knee replacement (TKR), higher testosterone levels were associated with lower pain in both sexes and lower disability in women [97]. Similarly, NHANES data (2011–2016) show that low testosterone increases OA risk (OR = 1.22; 95% CI: 1.02–1.46), particularly among obese women with hyperlipidemia [98]. Other studies also link low testosterone to greater arthritis risk [99] and reduced muscle mass and strength, with evidence from interventional studies demonstrating the role of testosterone in muscle adaptation and strength responses [100]. In summary, maintaining adequate levels of estrogen, progesterone, and testosterone may support cartilage integrity, reduce pain, and help limit OA progression.
Obesity is a major modifiable risk factor for OA and appears to have a particularly significant impact in women [13–16, 101]. In many populations, women exhibit higher rates of obesity, influenced by a combination of behavioral, nutritional, and socioeconomic factors, which may contribute to increased disease burden and associated comorbidities. In addition to mechanical loading, systemic and metabolic factors associated with obesity, including low-grade inflammation, may further exacerbate joint degeneration [18–20]. However, it is important to note that several studies evaluating the relationship between obesity and OA include both men and women, and sex-specific analyses remain limited in some cases.
Beyond mechanical overload on weight-bearing joints, adipose deposition, immune dysregulation, dyslipidemia, and insulin resistance contribute to OA in non-weight-bearing joints [102, 103]. Higher BMI consistently increases OA risk. A meta-analysis reported a positive association between BMI and KOA (RR = 1.25; 95% CI: 1.17–1.35), with each 5-unit BMI increment raising risk by 35% and showing stronger effects in women [104]. Obesity markedly increases the likelihood of KOA up to 6.8-fold [101], and this risk increases progressively from 0.1 in subjects with BMI < 20 kg/m2 to 13.6 in those with BMI ≥ 36 kg/m2 [105]. Associations with knee and hand OA have been confirmed in multiple cohort studies [106, 107] and in a recent meta-analysis identified higher BMI as a major risk factor for incident KOA [108]. Similar trends were observed in an Australian population study [109]. This study demonstrated that the odds of OA, as well as arthritis, were up to 7 times higher for obese individuals compared with those underweight or of normal weight [109].
Weight loss should be the primary goal for overweight or obese patients, as excess body weight strongly increases the risk of knee and hand OA and, to a moderate extent, HOA. Clinical symptoms should always be considered alongside radiographic findings [101–111]. Losing more than 7.5% of body weight reduces the risk of TKR [112], and even a ≥ 5% reduction can improve disability in patients with KOA [113]. Obesity also significantly elevates perioperative risk; in a meta-analysis of over 2 million THA cases, obese individuals had higher rates of complications, infection, dislocation, reoperation, revision, and readmission, with risks further increasing in morbid obesity [114]. Notably, up to 24% of surgeries could be prevented by achieving a normal BMI or losing just 5 kg [105].
The progression of OA in obese patients is multifactorial; mechanical forces, increased joint load, inflammatory cytokines, and the inability to completely repair the tissue are implicated in the degeneration of cartilage [115]. OA in non-bearing joints, such as the hand and wrist, is associated with obesity, suggesting that adipose tissue has a systemic inflammatory effect [103], and some authors even consider that pain intensity might be a reflection of adipose tissue dysfunction [116]. Additionally, overload is associated with matrix degradation and apoptosis of the cartilage, mainly because of activation of inflammatory pathways, as we have described previously [103, 115]. OA-related joint inflammation might be explained by the cartilage fragments of the degraded tissue acting as a foreign body when contacting the synovium, an inflammatory cascade starts when synovial cells react, activating chondrocytes and MMPs synthesis [117]. Patients with OA show differences in the cytokine and chemokine levels in synovial fluid, even in the mild or moderate stages [118]. Furthermore, it has been demonstrated that pain severity is associated with higher high-sensitivity C-reactive protein (CRP) levels in patients with advanced OA [119]. Similar findings showing an association between systemic inflammation and OA have been reported, and interestingly, female sex was associated with greater painful joint count and higher CRP [120].
While several of the mechanisms described in this section are involved in OA pathophysiology in general, evidence specifically addressing sex-related differences remains limited. Where available, female-specific findings are highlighted; however, many studies include both sexes without stratified analyses. Metabolic syndrome is a complex condition characterized by hypertension, dyslipidemia, hyperglycemia, and central obesity, and is closely linked to a chronic low-grade inflammatory state that accelerates OA progression [121–123]. Oxidative stress, advanced glycation end-products, and lipid and glucose dysregulation disrupt cartilage homeostasis, while adipose-derived cytokines such as TNF-α, IL-6, IL-17, and IL-18 promote catabolic enzyme activity, subchondral bone remodeling, and matrix breakdown [122–124]. Epidemiologic data from the UK Biobank show that metabolic syndrome increases the risk of OA by 15%, with an even significantly higher risk among individuals with elevated CRP (HR = 1.35), central obesity (HR = 1.58), hyperglycemia (HR = 1.13), and dyslipidemia in triglycerides (HR = 1.07) [125]. These findings align with other studies reporting metabolic syndrome in 80% of patients with OA vs. 26% in controls, along with higher total cholesterol and LDL levels in the OA group [122]. Vitamin D deficiency, common in metabolic syndrome due to sequestration in adipose tissue, further amplifies inflammatory and degradative pathways, partly through upregulation of MMP-2 and MMP-9 [126–128]. Notably, correcting deficiency may improve symptoms; in a population-based cohort, 25-hydroxyvitamin D (25OHD) levels < 10 ng/mL predicted greater worsening of hip and knee pain, and supplementation attenuated these changes over time [129]. Given its impact on MMP expression, inflammation, and chondrocyte integrity, vitamin D optimization represents a plausible therapeutic target in OA management [130].
PCOS shares several pathophysiological features with metabolic syndrome, including insulin resistance and chronic inflammation, but also presents distinct endocrine characteristics relevant to female-specific OA risk. Characterized by ovarian dysfunction and hyperandrogenism, PCOS is frequently accompanied by obesity and broader metabolic disturbances [131–133]. These patients demonstrate increased cardiometabolic risk, including higher rates of cardiovascular disease, early-onset type 2 diabetes, and systemic inflammation [132–134]. Notably, several rheumatic conditions appear more common in women with PCOS. A retrospective study reported a significantly higher prevalence of OA in PCOS, 5.44% vs. 2.92% in controls (P = 0.0030), with an OR of 1.913 (95% CI: 1.239–2.955) [135]. Similar findings were observed in a large Danish registry, where the PCOS cohort showed increased risks of knee, hip, and hand OA over two decades, along with an earlier onset in both weight-bearing and non-weight-bearing joints [136, 137]. Prospective cohort data further support this association, suggesting that PCOS may act as an independent risk factor for OA; women with KOA and PCOS had more than twice the odds of developing the condition (OR = 2.734) [137]. The biological mechanisms underlying the association between PCOS and OA are likely multifactorial. Hyperandrogenism, a hallmark of PCOS, may influence cartilage metabolism by altering chondrocyte activity and ECM turnover, although its direct effects remain incompletely understood. Insulin resistance, another central feature of PCOS, contributes to systemic metabolic dysfunction [132–134] and has also been implicated in cartilage degradation through increased production of advanced glycation end products and oxidative stress [134, 138]. In parallel, the chronic low-grade inflammatory state observed in PCOS, characterized by elevated cytokines such as TNF-α and interleukins, may promote matrix degradation and synovial inflammation [134, 139, 140]. These metabolic and inflammatory pathways may act synergistically to accelerate joint degeneration, providing a plausible link between PCOS and increased OA risk.
Symptom severity also appears greater, as demonstrated by Kabakchieva et al. [138], who found worse functional limitations and increased femoral cartilage thickness in PCOS patients. Beyond peripheral joints, PCOS has been associated with altered lumbopelvic alignment, including increased pelvic inclination and lumbar lordosis in nulliparous females without obstetric history compared to controls (P < 0.05). Additionally, a strong correlation was found between the luteinizing hormone (LH)/follicle-stimulating hormone (FSH) ratio and lumbar angle and pelvic inclination [134]. Temporomandibular joint disorders (TJD) are also more prevalent, likely reflecting hormonal and inflammatory influences; studies report markedly higher TJD rates in PCOS alongside elevated TNF-α, MMP-1, and MMP-8 levels [139, 140]. Given the presence of estrogen receptors (ERα and ERβ) in cartilage, hormonal dysregulation in PCOS may contribute to OA susceptibility, underscoring the need for more research into joint degeneration in this population [140, 141].
Female reproductive factors, particularly pregnancy and childbirth-related biomechanical adaptations, appear to influence musculoskeletal structure and loading patterns, potentially contributing to the risk of OA [142]. Pregnancy-related biomechanical load has been associated with degradation of the femoral articular cartilage [143, 144], and parity has been linked to changes in size, volume, and cartilage defects in the tibial and patellar compartments [145]. These reproductive-related alterations may contribute to OA development in the lower limbs and help explain the gait impairments observed in parous women [143]. A population-based cohort of 815 middle-aged women reported a clear, incremental association between the number of pregnancies, parity, and knee breadth. Each pregnancy was linked to a 0.11–0.14% increase in mediolateral knee width (P < 0.05), and each delivery to a further 0.20% increase (P < 0.01). Overall, multiparous women showed 0.68–1.01% larger knee breadth compared with nulliparous and primiparous women (P < 0.05), implying that repeated pregnancy-related loading may contribute to subtle structural adaptations of the knee [143].
Another study evaluated gait mechanics in healthy nulliparous and parous women across preferred, fast, and standardized walking speeds (1.4 m/s) and observed distinct kinematic differences between groups. Parous women showed a greater knee flexion angle at toe-off (P = 0.0002), along with smaller knee extension moments at heel strike (P = 0.0006), reduced first-peak knee flexion moments (P = 0.040), and lower peak hip adduction moments (P = 0.003). These alterations in joint loading patterns have been proposed to contribute to a higher long-term risk of OA, particularly in multiparous women [146]. In imaging studies of older women, parity has been consistently linked to structural cartilage changes. In a cohort of 489 middle-aged women, higher parity was associated with lower total knee cartilage volume and reduced volume in the tibial compartment, along with a greater likelihood of patellar cartilage defects [145]. Although joint space narrowing and osteophytes were more common in parous women, these findings were not statistically significant [145]. Additional data from 100 postmenopausal women showed that multiparity correlated with thinner femoral cartilage across lateral, central, and medial regions, reinforcing the connection between childbearing history and reduced cartilage thickness [144].
Earlier menarche has been linked to a higher likelihood of KOA in several studies. In a case-control study of 204 women over 50, those with KOA reported a significantly younger age at menarche (P = 0.031), along with a higher number of pregnancies and longer breastfeeding duration (P = 0.017 and P = 0.039, respectively). Higher parity markedly increased the odds of KOA (OR = 8.726), whereas an older age at menarche appeared to have a protective effect (OR = 0.851) [147]. In a large prospective cohort of 1.3 million middle-aged women followed for 6.1 person-years, higher parity was associated with an increased likelihood of knee and hip joint replacement, rising by 8% and 2% per birth, respectively. Early menarche also conferred a modest increase in the risk of both knee and hip replacement, particularly among women with menarche at ≤ 11 years (RR = 1.09) or at 12 years (RR = 1.15) [148]. Tan et al. [149] examined additional reproductive factors and found that earlier age at first live birth was associated with higher odds of overall OA (OR = 0.97) and KOA (OR = 0.60). Early menopause was linked to increased HOA risk (OR = 1.12), and younger age at initiation of oral contraceptives also showed modest associations with overall OA (OR = 0.97) and KOA (OR = 0.58) [149], underscoring how different aspects of reproductive history may contribute to OA susceptibility and the importance of conducting an evaluation that includes patients' gynecological history.
Prolonged breastfeeding has also been proposed as a potential factor influencing OA risk, although the underlying mechanisms are not fully understood. Lactation is characterized by a hypoestrogenic state, with suppressed ovarian function and reduced circulating estrogen levels, which may negatively affect cartilage homeostasis and joint health. In addition, breastfeeding is associated with increased calcium mobilization and transient bone loss, which could influence subchondral bone remodeling. From a biomechanical perspective, repetitive physical demands related to childcare, including lifting and carrying, may further contribute to joint loading. However, current evidence remains limited, and the relationship between breastfeeding duration and OA risk requires further investigation, particularly in studies with sex-specific and longitudinal designs [150–152].
Epidemiological evidence also suggests a potential association between prolonged breastfeeding and OA risk. In a large cross-sectional study including 3,454 women aged ≥ 50 years from the Korea National Health and Nutrition Examination Survey, longer breastfeeding duration was associated with a higher prevalence of both joint pain and radiographic KOA. Compared with women who had never breastfed, those with extended breastfeeding histories showed increased odds of joint pain, with a significant dose-response relationship observed as duration increased. Notably, breastfeeding for more than 25 months was associated with approximately a twofold higher likelihood of KOA, and this association was more pronounced in women aged ≥ 60 years. Although causality cannot be established due to the cross-sectional design, these findings support a potential link between cumulative reproductive factors and later joint health [151]. These findings are supported by a larger population-based analysis using data from the Korea National Health and Nutrition Examination Survey (2010–2018), which included 10,102 women aged ≥ 50 years. In this study, a history of breastfeeding was associated with a higher likelihood of physician-diagnosed OA compared with women who had never breastfed (OR = 1.55; 95% CI: 1.18–2.03). A duration-dependent effect was also observed, as women who breastfed for more than 25 months had a similarly increased risk (OR = 1.56; 95% CI: 1.19–2.06). These associations remained significant after adjusting for multiple sociodemographic, behavioral, and metabolic factors. However, as with other cross-sectional analyses, causal relationships cannot be definitively established [152].
Joint replacement surgery is increasingly performed for OA, with rates rising markedly over recent decades, particularly among women. Total joint arthroplasty (TJA) has increased substantially; data from the National Center for Health Statistics indicate that between 2000 and 2010, TKR rates increased by 99% in women, exceeding the corresponding 86% increase observed in men during the same period [153]. A regression analysis of US Medicare data projected annual growth rates of 5.2% for THA and 4.44% for TKA. Overall, TJA increased by 177% between 2000 and 2019, with further projected increases of 176% for THA and 139% for TKA by 2040. This significant increase in joint replacement surgeries represents an important health problem that needs to be addressed [154]. Orthopedic procedures, including joint arthroplasty, are associated with inherent perioperative risks. Evidence indicates that sex differences influence these outcomes, with female sex linked to greater healthcare resource utilization and the need for tailored perioperative management. A systematic review of 42 studies reported that women have a higher prevalence of OA, experience greater postoperative pain and inflammation, and show poorer long-term functional outcomes and slower recovery after TKR compared with men [155].
A retrospective analysis showed marked increases in TKA rates over 10 years and more advanced disease at surgery in women, who had a higher prevalence of obesity (9.0% vs. 6.2%), morbid obesity (4.1% vs. 2.4%), postoperative transfusion requirements (18.9% vs. 12.6%) and rate and length of stay compared to men (all P < 0.001). At discharge, men were more frequently discharged home, whereas women were more often transferred to rehabilitation facilities [156]. Consistently, female sex is reported to be associated with a greater need for preoperative functional assistance, higher risks of readmission, reoperation, and wound infection after THA, and longer hospitalization [157]. Large cohort analyses further demonstrated lower overall adverse event rates in men after THA and TKA (OR = 0.8, P < 0.001), while female sex emerged as an independent risk factor for urinary tract infection, perioperative blood transfusion, and non-home discharge [158, 159].
Patient-reported outcomes (PROs) by sex have also been evaluated. In a study of 475 patients undergoing total ankle replacement, women reported significantly worse preoperative PROs across multiple measures, including the VAS, 36-Item Short Form Health Survey (SF-36), the American Orthopaedic Foot & Ankle Society (AOFAS) hindfoot scale, and the Short Musculoskeletal Function Assessment (SMFA), with all comparisons yielding P < 0.05, and had higher rates of non-revision reoperations (32.2% vs. 22.6%; P = 0.0191), although overall failure rates did not differ by sex [160]. Similarly, an analysis of 133 patients with moderate-to-severe OA prior to knee arthroplasty found that women, particularly African Americans, reported worse preoperative Knee Injury and Osteoarthritis Outcome Scores (KOOS) scores for symptoms, pain, daily activities, and quality of life prior to knee arthroplasty, while postoperative outcomes were comparable across groups despite baseline differences [161]. A study assessing physicians’ perceptions of the impact of sex on KOA and TKA outcomes found that female sex was associated with more severe symptoms, higher pain intensity, and lower pain tolerance. Physicians also reported that women often require different pharmacological management strategies than men, highlighting the importance of personalized, sex-specific treatment approaches to optimize clinical outcomes [155–162].
Potential sex-related disparities in access to surgical care should be considered. Women may experience delays in referral and undergo surgery at more advanced disease stages despite greater symptom severity, suggesting possible referral or decision-making biases. However, the relative contributions of physician behaviour, patient preferences, and systemic factors remain unclear, and although disparities may be narrowing, contemporary data are limited. [156, 161, 163, 164]. From a clinical perspective, these differences support targeted perioperative strategies in women, including optimization of anemia and comorbidities, individualized pain management, and measures to reduce complications and enhance recovery. Timely referral and improved preoperative counselling may be important considerations to mitigate more advanced disease at presentation [155–159, 161, 163, 164].
Taken together, the factors discussed in this review can be conceptualized within a framework linking underlying biological and biomechanical mechanisms to distinct clinical phenotypes and outcomes in women with OA. Hormonal changes, metabolic dysfunction, and chronic inflammation interact with joint-specific biomechanics to influence cartilage degradation and pain processing. These mechanisms may manifest as different phenotypic patterns, including earlier disease onset, greater symptom severity, and increased functional limitation. From a clinical perspective, this framework supports a more individualized approach to risk stratification and management, incorporating sex-specific factors such as menopausal status, metabolic profile, and comorbid conditions. Recognizing these interconnections may help guide earlier diagnosis, targeted prevention strategies, and more personalized therapeutic interventions in women with OA.
Overall, the available evidence indicates that OA in women follows a distinct clinical course shaped by sex-specific biological and mechanical factors. Hormonal changes, particularly the decline in estrogen during the menopausal transition, appear to influence cartilage integrity, inflammatory pathways, and pain perception. Concurrently, differences in joint morphology, limb alignment, muscle function, and gait biomechanics contribute to the increased susceptibility of women to symptomatic and progressive OA. Reproductive history and female-predominant conditions, including metabolic syndrome and autoimmune diseases, further intersect with OA risk and may partly explain the steeper age-related increase in prevalence observed in women. Recognition of these interacting factors is critical for advancing targeted prevention strategies and improving early diagnosis. As global aging accelerates, the number of women affected by OA is expected to rise substantially, with important consequences for mobility, independence, and quality of life. Although male sex has been associated with higher postoperative risks, women face a distinct set of challenges, including a greater preoperative burden of pain and symptoms, higher rates of certain postoperative complications such as urinary tract infections and transfusion requirements, and slower functional recovery. Consequently, women are more frequently discharged to rehabilitation facilities, which may further influence long-term outcomes and overall well-being. Delayed diagnosis may compound these issues, contributing to poorer mental health and increased healthcare utilization.
Despite growing recognition of these differences, important knowledge gaps remain. There is a lack of validated sex-specific biomarkers for early OA detection and risk stratification, limiting the ability to implement targeted preventive strategies in women. In addition, mechanistic studies exploring sex differences in pain perception and central sensitization in OA are still limited, despite consistent evidence that women report greater pain severity and functional impairment. A major limitation in the current body of literature is the lack of consistent sex-stratified analyses. Many mechanisms implicated in OA pathogenesis are derived from mixed or predominantly male populations, which may not fully capture female-specific biological and biomechanical factors. Future studies should systematically incorporate sex-disaggregated data to better define the unique OA phenotype in women. Future research should also prioritize the identification of female-specific molecular and imaging biomarkers, as well as a deeper understanding of neurobiological pain mechanisms. Greater inclusion of women in clinical and translational research, along with more refined imaging, biomechanical assessment, and longitudinal study designs, will be essential. Addressing these gaps will be critical to advancing precision medicine approaches and moving toward more effective, equitable, and sex-informed management of OA in women.
BMI: body mass index
CRP: C-reactive protein
CT: computed tomography
ECM: extracellular matrix
GRF: ground reaction forces
GWAS: genome-wide association studies
HOA: hip osteoarthritis
HRT: hormone replacement therapy
KOA: knee osteoarthritis
MMPs: matrix metalloproteinases
MRI: magnetic resonance imaging
OA: osteoarthritis
PCOS: polycystic ovary syndrome
PROs: patient-reported outcomes
Q angle: quadriceps angle
ROM: range of motion
THA: total hip arthroplasty
TJA: total joint arthroplasty
TJD: temporomandibular joint disorders
TKR: total knee replacement
VAS: visual analogue scale
MV: Conceptualization, Writing—original draft, Writing—review & editing. NEL: Conceptualization, Writing—original draft, Writing—review & editing. Both authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1240
Download: 37
Times Cited: 0
