Affiliation:
College of Pharmacy, Jinan University, Guangzhou 510006, Guangdong, China
ORCID: https://orcid.org/0009-0008-2506-5843
Affiliation:
College of Pharmacy, Jinan University, Guangzhou 510006, Guangdong, China
Email: huangzhengw@jnu.edu.cn
ORCID: https://orcid.org/0000-0003-2351-7347
Affiliation:
College of Pharmacy, Jinan University, Guangzhou 510006, Guangdong, China
Email: zhanghongdou0223@126.com
ORCID: https://orcid.org/0000-0003-3330-3537
Explor Drug Sci. 2026;4:1008154 DOI: https://doi.org/10.37349/eds.2026.1008154
Received: January 04, 2026 Accepted: February 23, 2026 Published: March 10, 2026
Academic Editor: Fernando Albericio, University of KwaZulu-Natal, South Africa, Universidad de Barcelona, Spain
The article belongs to the special issue Nanoformulations for Non-Intravenous Drug Delivery
Intranasal drug delivery exhibits therapeutic potential for the treatment of central nervous system (CNS) diseases, as it allows pharmaceuticals to bypass the blood-brain barrier (BBB) via the olfactory and trigeminal pathways. The primary advantage of this method lies in its non-invasiveness and low systemic toxicity. Nevertheless, this delivery method faces notable challenges, including limited nasal mucosal absorption and short residence time in the olfactory region. To address these limitations, intranasal nanomedicine has gained research attention. Nanomedicines can improve brain bioavailability by enhancing drug solubility, permeability, and stability. The following discussion summarizes recent advancements in nanotechnology-enabled nose-to-brain delivery systems, with the aim of synthesizing progress in the field and outlining future research directions.
Conventional administration routes for central nervous system (CNS) diseases include oral, intravenous, and transdermal delivery [1]. However, due to the restrictions imposed by the blood-brain barrier (BBB), these traditional methods often face challenges regarding low bioavailability [2]. Furthermore, the lack of site-specific targeting leads to non-specific systemic distribution and severe adverse effects [3]. To address these bottlenecks, intranasal drug delivery has emerged as a promising alternative for the treatment of CNS diseases.
Intranasal drug delivery exploits the unique anatomical and physiological connections between the nasal cavity and the brain, which enable drugs to bypass the BBB for direct brain targeting. This route enables non-invasive drug administration and circumvents first-pass metabolism to achieve rapid onset and high bioavailability [4]. Despite these advantages, drug residence in the nasal mucosa prior to brain entry remains a significant challenge. The application of nanocarriers offers valuable opportunities to enhance brain targeting and selective delivery [5] and further promotes the accumulation of drugs in the CNS.
Nanocarriers, such as vesicular nanocarriers, lipid-based nanocarriers [6], emulsion-based nanocarriers, polymeric nanocarriers [7], and other related formulations, provide substantial advantages in drug encapsulation, stability, and targeted delivery. Through physicochemical properties optimization or surface modification of the nanoparticles, these platforms can effectively protect therapeutic cargo from physiological degradation or denaturation. This strategic design ensures precise delivery to target cells and target sites, thus minimizing the systemic distribution of the active pharmaceutical ingredient (API) and reducing the risk of off-target adverse effects [8].
When nanomedicines are delivered to the nasal cavity, the dendrites of the olfactory sensory neurons (OSNs) are distributed within the mucus layer on the surface of the olfactory epithelium. The axons of OSNs pass through the lamina propria and the cribriform plate, enter the subarachnoid space, and end in the olfactory bulb. Consequently, nanomedicines deposited in this region can be directly transported to the brain via the olfactory neural pathway, allowing drugs to bypass the BBB [9]. Intranasal nanomedicine achieves effective brain targeting, enhances patient compliance, and mitigates systemic toxicity and associated side effects. Consistent with the aforementioned characteristics of nanocarriers, intranasal nanomedicines can also improve drug solubility and realize controlled drug release, thus significantly enhancing the efficacy of nose-to-brain delivery [10].
These inherent advantages render intranasal nanomedicine particularly suitable for the treatment of CNS diseases. As specified in the World Health Organization (WHO) International Classification of Diseases, 11th Revision (ICD-11) for nervous system diseases, CNS diseases include multiple sclerosis, Alzheimer’s disease, dementia, Parkinson’s disease [11], schizophrenia, amyotrophic lateral sclerosis [12], glioblastoma [13], and other severe conditions (Figure 1).
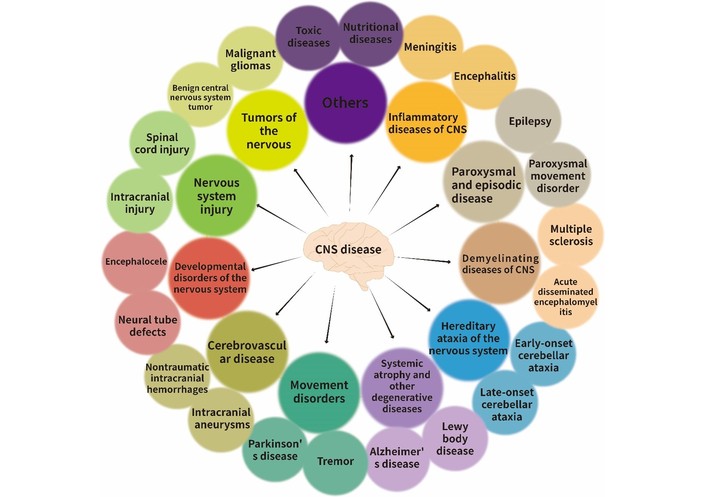
The classification of CNS diseases in the World Health Organization (WHO) International Classification of Diseases, 11th Revision (ICD-11). CNS: central nervous system.
As a result, this innovative drug delivery approach has garnered extensive attention within the academic community, catalyzing a surge of intensive research efforts across the academic landscape. To propel the frontier of intranasal nanomedicine into a new era of discovery and application, it is essential to conduct a thorough and systematic assessment of recent progress. This involves synthesizing the current corpus of research findings to clarify feasible development pathways and inspire transformative future investigations.
Nanocarriers are regarded as a promising strategy for nose-to-brain drug delivery; however, they encounter inherent challenges, including particle aggregation and rapid clearance due to reticuloendothelial system activation. To address these limitations, surface modification of nanocarriers with mucoadhesive polymers, permeation enhancers, or targeting ligands offers a promising strategy to improve their transport efficiency and facilitate more effective delivery of therapeutic agents to the brain.
Surface modification of nanomaterials using mucoadhesive substances can prolong the residence time of drugs within the nasal cavity, which in turn enhances drug absorption and elevates bioavailability. Lectins are an abundant and structurally diverse family of glycoproteins that bind to specific carbohydrate moieties in a selective and stereospecific manner, a property that has been validated to boost mucosal adhesion. Researchers demonstrated that nanoparticles functionalized with wheat germ agglutinin (WGA) indicated high binding capacity in the human nasal respiratory epithelium with a mucosal association concentration of 225.2 ± 36.26 nM at 4°C in fluorescence labeling experiments [14]. Additionally, Bilapatte et al. [15] highlighted that the mucoadhesive polymers of in situ gels, synergistic with their sustained release and targeting profiles, offers a viable strategy to enhance adherence and efficacy for epileptic patients. These studies prove the role of functionalized polymeric nanocarriers in maximizing nasal residence time and optimizing bioavailability [16].
The drugs delivered through the nasal brain delivery system can reach the brain only when they achieve sufficient adequate penetration through the nasal mucosa. Nevertheless, hydrophilic drugs face challenges in penetrating the nasal epithelium barrier. Therefore, the surface of the nanoparticles can be modified to enhance permeability and improve the delivery efficiency of hydrophilic drug molecules. Wang et al. [17] constructed an intranasal nanomedicine modified with borneol: borneol-modified lipid nanoparticle nasal spray (BLNP-NS). Borneol acts as a natural membrane penetration enhancer, which further increases the efficiency of drug delivery into the brain by promoting the opening of tight junction channels. Compared to the unmodified lipid nanoparticle nasal spray (LNP-NS), BLNP-NS increased the nasal mucosa absorption rate by a factor of 2.25. Meanwhile, the area under the curve (AUC) of BLNP-NS in the brain was 23.29% higher than that of LNP-NS, indicating a further improvement in total brain targeting accumulation [17].
Targeted surface modification of nanomedicine can effectively strengthen the targeting capability of therapeutic agents to improve their bioavailability. Usama Ashhar et al. [18] found that after nasal administration of a bromocriptine-loaded nano-emulsion for brain-targeted delivery, the concentration and residence time of the drug in brain tissue were significantly increased. Another relevant investigation centered on lactoferrin-mediated nano-surface modification. Polyethyleneglycol-poly ε-caprolactone (PEG-PCL) nanoparticles modified with lactoferrin showed significantly elevated brain accumulation in a rat model of Alzheimer’s disease after intranasal administration. Compared with the unmodified group, the AUC in brain pharmacokinetic of a short peptide composed of the sequence Asn-Ala-Pro-Val-Ser-Ile-Pro-Gln (NAPVSIPQ, abbreviated as NAP) in the glycoprotein lactoferrin-modified group increased significantly [19].
The physicochemical properties of intranasal nanomedicines, such as surface tension, viscosity, and osmotic pressure, exert a significant impact on the nose-to-brain drug delivery process [20]. Clementino et al. [21] found that decreasing the surface tension of the formulation can significantly enhance its wettability and mucus penetration ability on the nasal mucosa. A series of studies has demonstrated that adjusting the viscosity of formulations significantly influences the efficiency of nose-to-brain delivery. For instance, Yue et al. [22] reported that elevated viscosity improved the deposition rate within the olfactory region (ORF%). In addition, osmotic pressure must be strictly controlled within the range of 280–320 mOsm/kg. Excessively high osmotic pressure (e.g., 400 mOsm/kg) may lead to the dehydration of nasal epithelial cells, trigger local irritation, and induce mucosal injury. This not only significantly impairs drug absorption efficiency but also increases the risk of adverse reactions [23]. These findings underscore the need for the rational design and optimization of intranasal nanomedicines with respect to their macroscopic physicochemical properties.
The nasal spray device comprises a trigger, metering valve, and medication bottle [24]. The trigger and metering valve are critical components in spray formation, which can influence droplet size, spray pattern, and spatial distribution, as they regulate the compression force, spraying velocity, and frictional forces acting on the liquid formulation [25]. D’Angelo et al. [26] found that because of the relatively high initial velocity of the sprayed liquid (> 10 m/s), the generated droplets are relatively large, and the main droplets are deposited and accumulated on the surface of the nasal vestibule when using a standard swirl nozzle. In contrast, a nasal spray device with a Medspray mist nozzle produces smaller droplets with a lower initial velocity (< 1 m/s), enabling more uniform coverage of the nasal cavity and a higher deposition rate in the olfactory region [26]. Furthermore, Fang et al. [27] investigated drug delivery performance using three distinct nozzles. Their study revealed that nozzle design significantly impacts the efficiency of nasal spray devices. Specifically, nozzles with narrower plume angles showed improved drug delivery efficiency [27]. These findings indicate that effective nasal drug delivery depends on both the device design and the drug formulation.
Current research indicates that parameters such as particle size and surface charge of nanomedicines significantly influence their biodistribution. The trigeminal nerve and olfactory nerve serve as the primary pathways for intracellular substance transport; nanoparticles must interact with those nerves to access the CNS. However, the diameter of OSNs ranges from 100 to 700 nanometers, which limits the particle size of nanoparticles that can pass through. A multitude of studies have shown that using smaller-diameter particles can promote their transmission in the trigeminal nerve [28]. Intranasal levodopa nanoparticles (200–300 nm) showed efficient brain delivery for the treatment of Parkinson’s disease [16]. In addition, numerous studies have demonstrated that positively charged nanoparticles exhibit enhanced targeting efficiency and greater cellular uptake in nose-to-brain delivery systems [29]. Xiao et al. [30] observed that the electrostatic interaction between positively charged nanomedicines and cell membranes can effectively enhance the efficiency of cellular uptake. In summary, the physicochemical properties optimization of nanoformulations plays a critical role in nose-to-brain drug delivery and enhancing cellular uptake in the olfactory region. Therefore, this strategy should be prioritized in the design of intranasal nanomedicines.
Intranasal nanomedicine utilizes the non-invasive nasal-to-brain delivery pathway, which bypasses the BBB via the olfactory nerve while capitalizing on the enhanced mucoadhesion and targeted delivery capabilities of nanocarriers. This perspective aims to summarize the four main aspects of intranasal nanomedicine design: (1) surface modification of nanoparticles to improve mucosal adhesion, facilitate epithelial penetration, and enable precise targeting; (2) optimization of formulation parameters to enhance drug absorption and minimize mucosal irritation; (3) rational design of nasal spray devices to ensure effective and reproducible deposition in the olfactory region; and (4) systematic investigation of nanoparticle physicochemical properties to modulate biodistribution, promote cellular uptake, and increase bioavailability (Figure 2). These four intranasal nanomedicine design approaches offer a strategic framework for the design and clinical translation of next-generation intranasal nanomedicines.
Each research direction in intranasal nanomedicine constitutes a sophisticated and in-depth system. Intranasal nanomedicines should be engineered with surface modifications and tuned physicochemical properties, which are tailored to the therapeutic needs of different diseases. For instance, glioblastoma necessitates nanoparticles engineered with specific ligands for deep tumor targeting. Conversely, in contrast, therapeutic strategies for neuroinflammatory diseases (e.g., multiple sclerosis and meningitis) focus on improving permeability to facilitate deeper drug delivery. Here, mucoadhesive polymers (e.g., chitosan) or hydrogel vehicles are utilized to prolong nasal residence time and thus increase the penetration rate. Functional components of delivery devices, such as triggering mechanisms, metering valves, and nozzles, can be optimized to improve droplet size, pattern, and morphology, enhancing patient comfort. The key differences among various intranasal nanomedicines are summarized in Table 1.
The key differences among various intranasal nanomedicines.
| Optimization category | Specific strategy | Key differences |
|---|---|---|
| Surface engineering | Mucoadhesive modification | • Prolongs residence time.• Extends the absorption window along olfactory/trigeminal pathways.• Minimizes systemic exposure. |
| Permeation enhancement | • Enhances mucosal penetration (especially for hydrophilic drugs).• Significantly increases brain AUC and absorption rate (e.g., 2.25× increase). | |
| Active targeting | • Increases brain accumulation and bioavailability.• Improves specific uptake of therapeutic agents (e.g., NAP peptide) in disease models. | |
| Physicochemical properties | Surface tension | • Enhances wettability and mucus penetration ability. |
| Viscosity | • Improves deposition rate within the olfactory region (ORF%). | |
| Osmotic pressure | • Prevents nasal irritation, dehydration, and mucosal injury.• Avoids impaired drug absorption caused by hypertonicity (> 400 mOsm/kg). | |
| Delivery device | Medspray mist nozzle and other nozzles | • Ensures uniform coverage of the nasal cavity.• Higher deposition in the olfactory region (avoids vestibule accumulation).• Narrower plume angles demonstrate superior drug delivery efficiency. |
| Droplet size control | • Prevents inhalation into the lower respiratory tract (lung deposition).• Ensures anterior nasal deposition. | |
| In vivo fate factors | Particle size | • Smaller particles facilitate transport via the trigeminal nerve pathway. |
| Surface charge | • Enhances cellular uptake efficiency.• Improves targeting efficiency via mucosal interaction. |
AUC: area under the curve.
Intranasal nanomedicine offers significant advantages. However, several fundamental constraints continue to hinder its clinical application. A primary limitation is that the limited volume of the nasal cavity inherently restricts the maximum deliverable dose. Besides, the efficiency of nose-to-brain targeting is modulated by a complex interplay between drug physicochemical properties, formulation characteristics, and experimental methodologies. In addition, physiological barriers, including the intricate nasal anatomy, mucociliary clearance (MCC), enzymatic degradation, and pathological state, impede drug absorption as well as contribute to substantial individual variability. Another limitation lies in biocompatibility and long-term safety for clinical translation. Nanocarrier impacts on nasal mucosal health must be rigorously evaluated, specifically regarding ciliary beat frequency (CBF), epithelial integrity, and chronic toxicity.
Rodent models are commonly used in intranasal nanomedicine research due to their similar nose-to-brain pathways to humans, and this is significant in studying the specific mechanisms of intranasal drug delivery to the brain. Their olfactory structures are far more extensive, roughly 50% of the nasal surface is dedicated to olfaction. By contrast, the olfactory region in humans occupies a mere 10% of the total nasal area. These profound anatomical differences pose a significant challenge to the reliable extrapolation of pharmacodynamic and toxicological research results across species, this may lead to an overestimation of the nose-to-brain delivery efficiency in human applications.
Therefore, in future studies, in vivo investigations into brain delivery efficiency, regional distribution profiles, therapeutic efficacy, and mechanisms of action in disease models, as well as the translational efficiency of nose-to-brain delivery across species, are critical for supporting clinical translation and advancing effective intranasal nanomedicines for medical use. Only in this way can the clinical translation of nasal nanomedicines be advanced, enabling the development of a range of novel and highly effective formulations for clinical use.
AUC: area under the curve
BBB: blood-brain barrier
BLNP-NS: borneol-modified lipid nanoparticle nasal spray
CNS: central nervous system
LNP-NS: lipid nanoparticle nasal spray
OSNs: olfactory sensory neurons
RY: Conceptualization, Investigation, Writing—original draft. ZH: Validation, Writing—review & editing, Supervision. XZ: Validation, Writing—review & editing, Supervision. All authors read and approved the submitted version.
Zhengwei Huang, who is the Editorial Board Member and Guest Editor of Exploration of Drug Science, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2266
Download: 26
Times Cited: 0
