Affiliation:
Gastroenterology and Gastrointestinal Endoscopy Unit, University Department of Pediatrics, Children’s Hospital, ASST Spedali Civili, 25123 Brescia, Italy
Email: alberto.ravelli@ymail.com
ORCID: https://orcid.org/0000-0001-7362-8040
Explor Asthma Allergy. 2026;4:1009112 DOI: https://doi.org/10.37349/eaa.2026.1009112
Received: December 22, 2025 Accepted: February 06, 2026 Published: March 02, 2026
Academic Editor: Umit Murat Sahiner, Hacettepe University Faculty of Medicine, Turkey
The article belongs to the special issue Beyond Eosinophilic Gastrointestinal Diseases: Pathogenetic Mechanisms and Therapeutic Strategies
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated inflammatory disease of the esophagus that has emerged as a major cause of esophageal dysfunction in all ages. Over the past two decades, its frequency has increased worldwide, reflecting both heightened recognition and a rise in occurrence. EoE predominantly affects males and frequently coexists with atopic conditions, underscoring its relationship with allergy. The pathogenesis involves genetic susceptibility, epithelial barrier dysfunction, and dysregulated type 2 immune responses. Variants in genes related to epithelial integrity and immune signaling, such as TSLP and CAPN14, predispose susceptible individuals to aberrant immune responses to food antigens, leading to eosinophil recruitment, mast cell activation, and chronic inflammation, which in turn promotes tissue remodeling and progression toward fibrostenotic disease. Clinical presentation varies with age. Infants and younger children often exhibit feeding difficulties, vomiting, and abdominal pain, whereas older children and adolescents usually present with dysphagia and food impaction. Diagnosis requires integration of clinical symptoms with histologic confirmation of esophageal eosinophilia (≥ 15 eosinophils per high-power field) and exclusion of alternative causes. Management of pediatric EoE aims to achieve and maintain clinical and histologic remission while preventing long-term complications and preserving quality of life. First-line therapeutic options include proton pump inhibitors, swallowed topical corticosteroids, and dietary elimination strategies. Biologic therapy has expanded treatment options for severe or refractory disease. Because symptom improvement alone does not reliably reflect disease control, objective reassessment with endoscopy and biopsies is recommended after treatment and during follow-up. Long-term outcomes of EoE are strongly influenced by diagnostic timing and adequacy of treatment. Early diagnosis, sustained anti-inflammatory therapy, and transition from pediatric to adult care are critical components of an appropriate management. Future directions include the development of precision medicine, identification of biomarkers to guide therapy selection, non-invasive tools for disease monitoring, and strategies aimed at disease modification.
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated inflammatory disease of the esophagus, defined by symptoms of esophageal dysfunction and eosinophil-predominant mucosal inflammation [1]. Although now widely recognized, EoE is a relatively recent clinical entity, particularly in pediatric practice. Isolated descriptions of esophageal eosinophilia appeared in the late 1970s and 1980s, and were largely interpreted within the context of gastroesophageal reflux disease (GERD) or systemic eosinophilic disorders [2]. The concept of EoE as a distinct disease emerged in the early 1990s, when adult patients with dysphagia and dense esophageal eosinophilia plus an increased number of mast cells were shown to have normal esophageal acid exposure and to respond to dietary modification or corticosteroids rather than acid suppression [3, 4]. Shortly thereafter, pediatric case series demonstrated similar histologic findings in infants and children presenting with feeding difficulties, vomiting, and failure to thrive, establishing EoE as a disease affecting all age groups [5].
Subsequent population-based studies demonstrated a marked increase in the incidence and prevalence of EoE worldwide [6]. The parallel rise in atopic diseases suggests a true increase in disease burden, consistent with the strong association between EoE and allergic comorbidities [7, 8]. Advances in translational research have clarified that EoE is primarily a food antigen-driven, type 2 inflammatory disorder, characterized by epithelial barrier dysfunction and cytokine-mediated immune activation, as well as eosinophil and mast cell recruitment [9]. Persistent inflammation may lead to tissue remodeling and fibrostenotic complications, highlighting the importance of early diagnosis and effective long-term disease control, particularly during childhood. This review summarizes the current understanding of EoE in infancy and childhood, highlighting key aspects of historical evolution, age-specific clinical presentation, diagnostic principles, and current therapeutic strategies.
Over the past three decades, EoE has transitioned from a rare and poorly recognized condition to a common cause of chronic esophageal symptoms in children and adolescents. This change reflects improved disease recognition and diagnostic practices, as well as a true rise in disease occurrence, as demonstrated by population-based studies from multiple regions worldwide [6, 7, 10].
In North America and Europe, the incidence of pediatric EoE is estimated at approximately 5–10 cases per 100,000 children per year, while prevalence ranges from 30 to 90 cases per 100,000 in children [6, 7, 10, 11]. Population-based studies report a 2- to 4-fold increase in incidence since the early 2000s [11, 12]. Similar trends have been reported in Australia and parts of Asia, although data from low- and middle-income countries remain limited [6, 7, 13].
Pediatric EoE can present across all age groups. However, the incidence peaks during school age and early adolescence, with a median age at diagnosis ranging from 7 to 10 years. A consistent male predominance is observed, with male-to-female ratios of approximately 2:1 to 4:1 across pediatric cohorts [6, 7, 10–13].
EoE has been reported worldwide, with the highest prevalence documented in North America and Western Europe in predominantly Caucasian populations. Within these regions, urban populations appear to have higher rates than rural populations, suggesting a potential influence of environmental exposures [14, 15]. Lower rates have been described in Asian and South American cohorts; however, underdiagnosis and limited access to endoscopy likely contribute to these differences [10–13].
Atopic comorbidities are highly prevalent among children with EoE. Between 60% and 80% of pediatric patients have at least one concomitant atopic disorder, including asthma, allergic rhinitis, atopic dermatitis, or IgE-mediated food allergy [8, 11–13]. Family history of atopy is common, further supporting a shared genetic and immunologic background. Seasonal variation in diagnosis has been reported in some studies, with higher diagnosis rates during spring and summer months. This observation suggests a potential contributory role of aeroallergen exposure, particularly in sensitized individuals, although food antigens remain the primary disease triggers [8, 11–14].
Environmental and early-life exposures have been implicated in the rising incidence of EoE. Factors such as early antibiotic use, acid suppression, cesarean delivery, and altered microbial colonization have been associated with an increased disease risk, although effect sizes vary across studies [14]. Furthermore, epidemiologic studies indicate that EoE is a chronic disease with low rates of spontaneous remission [10–12]. Pediatric patients frequently transition into adulthood with persistent disease, underscoring the importance of early recognition and long-term management. Diagnostic delay has been associated with an increased risk of fibrostenotic complications, highlighting the clinical relevance of epidemiologic insights for patient outcomes [16, 17].
EoE is an antigen-driven, chronic inflammatory disease characterized by a complex interplay between genetic susceptibility, environmental exposures, and immune dysregulation. Current evidence supports EoE as a type 2 immune-mediated disorder in which epithelial barrier dysfunction and antigen-driven immune responses lead to eosinophilic inflammation and tissue remodeling.
Family aggregation studies demonstrate a significant heritable component. First-degree relatives of affected individuals have an approximately 10–40-fold increased risk of developing EoE compared with the general population. Twin studies further support genetic susceptibility, with concordance rates exceeding 40% in monozygotic twins compared with less than 10% in dizygotic twins [18–20]. Genome-wide association studies have identified susceptibility loci involving calpain-like protease 14 (CAPN14, esophagus-restricted calpain-like protease), thymic stromal lymphopoietin (TSLP), filaggrin (FLG), and signal transducer and activator of transcription 6 (STAT6)/IL4R pathways, and chromosomal regions such as 5q22 [18–20]. TSLP plays a key role in initiating type 2 immune responses at epithelial surfaces, while CAPN14 encodes a calpain protease expressed predominantly in the esophageal epithelium and induced by IL-13. Variants affecting these pathways are thought to predispose the esophagus to aberrant immune activation in response to luminal antigens. Collectively, however, known genetic variants explain approximately only 15–20% of disease heritability, indicating an important contribution of environmental factors.
IL-13-driven epithelial remodeling is central to EoE pathogenesis. Transcriptomic analyses reveal an IL-13 signature in more than 80% of pediatric patients with active disease. IL-13 induces CAPN14 expression, thereby reducing the expression of junctional proteins desmoglein-1 and FLG. Barrier dysfunction facilitates antigen penetration and promotes the release of epithelial-derived cytokines, including TSLP, IL-25, and IL-33. These cytokines activate dendritic cells and innate lymphoid cells, amplifying downstream type 2 immune responses. Epithelial abnormalities may persist even in histologic remission, suggesting a role in disease recurrence [21, 22].
Type 2 inflammation is central to EoE pathogenesis [23, 24]. Antigen presentation in genetically predisposed individuals leads to the activation of CD4+ T helper 2 (Th2) cells, which secrete cytokines such as IL-4, IL-5, and IL-13. These mediators orchestrate eosinophil recruitment, activation, and survival within the esophageal mucosa. IL-5 plays a critical role in eosinophil maturation and trafficking, while IL-13 induces epithelial remodeling and promotes expression of eotaxin-3 (CCL26), a potent eosinophil chemoattractant. Elevated levels ofeotaxin-3 are a hallmark of active EoE and correlate with tissue eosinophilia. Eotaxin-3 expression is increased by more than 20-fold in the affected esophageal epithelium. In addition to adaptive immune responses, innate immune pathways contribute significantly to disease activity. Group 2 innate lymphoid cells (ILC2s) respond rapidly to epithelial cytokines and produce large amounts of IL-5 and IL-13, linking epithelial injury to downstream inflammation [21–24].
The inflammatory infiltrate in EoE is dominated by eos, mast cells, and type 2 lymphocytes. Eos are the defining histologic feature of EoE, as their density frequently exceeds 50–100 eos/high power field (HPF) in active disease. Eos, however, play an active pathogenic role rather than serving solely as disease markers [21, 25]. Upon activation, they release cytotoxic granule proteins such as major basic protein, eosinophil peroxidase, and eosinophil-derived neurotoxin, which directly injure the esophageal epithelium. In addition, eos secrete cytokines and chemokines that sustain type 2 inflammation and recruit additional immune cells to the tissue. Beyond epithelial injury, eos contribute to tissue remodeling through the release of profibrotic mediators, including transforming growth factor-β (TGF-β). These signals promote fibroblast activation, collagen deposition, and smooth muscle hypertrophy, leading over time to esophageal stiffening and luminal narrowing. In addition to eosinophilic infiltration, the esophageal mucosa of children with EoE frequently exhibits increased mast cell density, often with evidence of degranulation. In fact, mast cells are increasingly recognized as important contributors to disease manifestations [21, 22, 26]. Mast cell-derived mediators, such as histamine, tryptase, and leukotrienes, influence esophageal smooth muscle function, sensory nerve activation, and pain perception, and also contribute to the development of fibrosis. Pediatric studies have shown that mast cell quantification, particularly when combined with eosinophil counts, improves histologic discrimination between EoE and GERD [27, 28]. Importantly, persistent mast cell infiltration has been associated with ongoing symptoms and endoscopic abnormalities despite histologic remission defined by eosinophil thresholds alone, suggesting an eosinophil-independent contribution to disease activity [28].
Mechanistic human and experimental data indicate that eosinophil-IL-9-mast cell interactions sustain mucosal mastocytosis and contribute primarily to epithelial dysfunction, esophageal remodeling, and dysmotility rather than initial eosinophil recruitment [29, 30]. Overall, mast cells are increasingly recognized as key effector and amplifier cells in pediatric EoE, with potential implications for disease monitoring and future therapeutic targeting [31].
Chronic type 2 inflammation promotes fibroblast activation, collagen deposition, and smooth muscle dysfunction, leading to progressive subepithelial fibrosis and reduced esophageal distensibility, which in turn results in esophageal stiffness, rings, and strictures. Biomarkers of tissue remodeling, such as periostin and TGF-β signaling pathways, are upregulated in patients with long-standing disease and correlate with disease duration [26, 32].
EoE presents with a broad spectrum of clinical manifestations in children, varying by age and disease duration. Because symptoms are often non-specific—particularly in younger patients—diagnosis is frequently delayed. A high index of suspicion, combined with a structured diagnostic approach, is essential to prevent diagnostic delay, disease progression, and long-term complications [1, 10, 33].
Infants and toddlers typically present with non-specific symptoms. Feeding difficulties, food refusal, prolonged mealtimes, vomiting, and poor weight gain are reported in approximately 40–60% of children younger than 6 years, while vomiting or regurgitation occurs in 30–50%. School-aged children frequently report abdominal pain, nausea, early satiety, or intermittent dysphagia. Abdominal pain is present in approximately 30–50% of patients in this age group. Adolescents usually develop classic, adult-type esophageal symptoms. Dysphagia is reported in 60–80%, and food impaction in 20–40%, particularly in those with long-standing disease [1, 10]. Clinical features such as progressive dysphagia, a history of recurrent food impaction, avoidance of solid foods, and prolonged eating times suggest fibrostenotic disease and warrant prompt endoscopic assessment [1, 10, 16, 33]. Between 60% and 80% of children with EoE have coexisting atopic conditions, including asthma, allergic rhinitis, atopic dermatitis, or IgE-mediated food allergy. A personal or family history of atopy should heighten clinical suspicion but is not required for the diagnosis of EoE [1, 10, 13, 34].
Upper gastrointestinal (GI) endoscopy with biopsies is mandatory for the diagnosis [1, 10, 33, 34]. Typical endoscopic features include: edema with decreased vascular markings, fixed or transient rings (so-called “trachealization”), linear furrows, white exudates or plaques, and esophageal narrowing or strictures [10, 13, 34] (Figure 1).
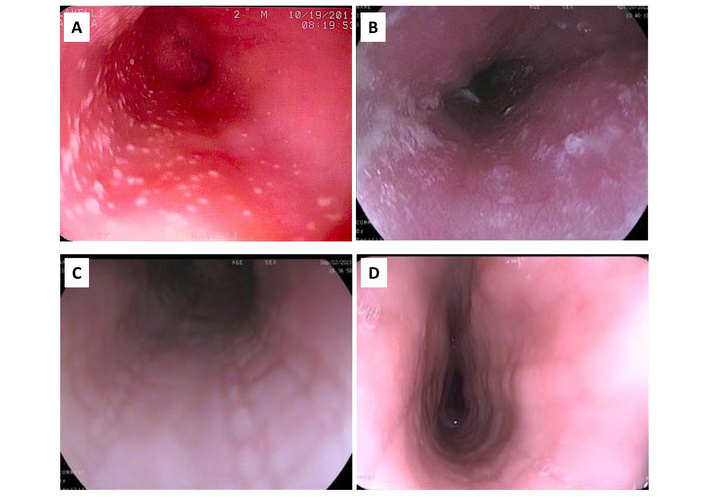
Endoscopic features of pediatric eosinophilic esophagitis (EoE). In pediatric EoE, the most typical and common endoscopic features are exudates, appearing as small (A) or large (B) white plaques on the esophageal mucosa. Longitudinal linear furrows (C) are also common, whereas circular rings and narrowing of the esophagus (D) are seen less frequently. Edema of the esophageal mucosa (A–D) is almost always present.
At least one abnormal endoscopic feature (edema, rings, exudates, furrows, strictures) is present in 70–90% of pediatric patients. Nevertheless, 10–30% of pediatric patients with histologically-proven and clinically active EoE have a macroscopically normal esophagus [10, 35]. Therefore, biopsies are mandatory regardless of macroscopic appearance [10, 34, 35].
To standardize assessment and improve reproducibility, an objective endoscopic scoring system—the Eosinophilic Esophagitis Endoscopic Reference Score (EREFS)—was developed [36, 37]. The acronym EREFS also serves as a reminder of the abnormal features—edema, rings, exudates, furrows, stricture—that can be found during endoscopy (Table 1). The use of EREFS is recommended, as it improves diagnostic consistency and disease monitoring [36–38].
The Eosinophilic Esophagitis Endoscopic Reference Score (EREFS).
| Endoscopic feature | Grading criteria |
|---|---|
| Edema | Grade 0: distinct vascularityGrade 1: absent or decreased vascularity |
| Rings | Grade 0: noneGrade 1: mild (ridges)Grade 2: moderate (distinct rings)Grade 3: severe (scope will not pass) |
| Exudates | Grade 0: noneGrade 1: mild (≤ 10% surface area)Grade 2: severe (> 10% surface area) |
| Furrows | Grade 0: noneGrade 1: mildGrade 2: severe |
| Stricture | Grade 0: absentGrade 1: present |
Although practiced only in a small number of centers, transnasal endoscopy (TNE) may not require deep sedation or general anesthesia and provides adequate biopsy specimens for the evaluation of EoE. Therefore, TNE may significantly reduce the diagnostic burden in conditions such as EoE, which often require repeated endoscopy [39, 40].
Five endoscopic features (edema, rings, exudates, furrows, and stricture) are graded according to their absence, presence, and severity. An EREFS of ≤ 2 is considered normal, whereas the most severe cases of EoE usually have an EREFS of 8 or 9, mostly depending upon the presence of esophageal stricture.
Current guidelines recommend obtaining at least 6 biopsies from multiple esophageal levels, including proximal and distal segments, in order to maximize the diagnostic yield of endoscopy and address patchy disease distribution [10, 34, 41]. Diagnosis of EoE requires ≥ 15 eos/HPF [1, 10, 34, 41, 42] (Figure 2). Additional features supporting the diagnosis include basal zone hyperplasia, dilated intercellular spaces, eosinophil microabscesses, and lamina propria fibrosis, when evaluable [10, 34, 41]. Gastric and duodenal biopsies are recommended to exclude alternative causes of eosinophilia when clinically indicated [10, 34].
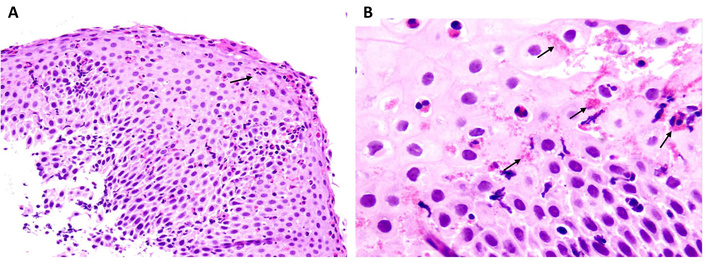
Histology from esophageal biopsies taken from a child with eosinophilic esophagitis (EoE). (A) Eosinophil granulocytes in the esophageal epithelium are in number exceeding 15/HPF, and some of them are clustered onto the epithelial surface (arrow), a feature that accounts for the whitish exudates seen at endoscopy (H&E, original magnification 40×). (B) Most eosinophils are degranulating (arrows), which is a sign of functional activation (H&E, original magnification 100×). Adapted with permission from [42]. © Copyright-2014 European Society for Pediatric Gastroenterology, Hepatology, and Nutrition and North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition. HPF: high power field.
Conditions to consider in the differential diagnosis of pediatric EoE include GERD, achalasia, functional dysphagia, infectious (especially Candida) esophagitis, drug-induced esophagitis, eosinophilic GI disorders, Crohn’s disease with esophageal involvement, and connective tissue disorders [10, 34, 41].
The correct diagnosis of EoE requires the presence of a significant eosinophilic infiltrate (≥ 15 eos/HPF) in the esophageal epithelium and the presence of symptoms of esophageal dysfunction [10, 34]. Common pitfalls in the diagnostic assessment of pediatric EoE include reliance on symptoms alone, dissociation between symptoms and histologic activity, normal endoscopic appearance, and response to empirical therapy with proton pump inhibitors (PPIs). Histologic and clinical response to PPIs occurs in approximately 30–50% of pediatric patients with EoE. Therefore, response to an empirical course of PPIs does not exclude EoE and should not delay diagnosis [43, 44]. The median diagnostic delay of pediatric EoE ranges from 2 to 6 years and is associated with increased risk of fibrostenotic complications. Each additional year of untreated disease increases the likelihood of esophageal narrowing, particularly in adolescents transitioning to adult care [16]. These findings emphasize the importance of early endoscopic evaluation in children with persistent esophageal symptoms [10, 33].
Symptom improvement alone is insufficient to assess disease control. Objective reassessment with endoscopy and biopsies is recommended after induction therapy, typically after 8–12 weeks, to document histologic remission [10, 34, 44]. Endoscopic scoring systems such as EREFS enhance consistency in follow-up evaluations [37, 38].
The management of pediatric EoE aims to achieve sustained symptom control, histologic remission, prevention of esophageal remodeling and fibrostenosis, and preservation of growth and quality of life [10, 34, 44]. EoE is a chronic disease, and long-term maintenance therapy is often required. Current management strategies include pharmacologic therapy, restricted diet, and, in selected cases, biologic agents [1, 10, 34, 44]. First-line therapy may include PPIs, swallowed topical corticosteroids, or dietary elimination, selected through shared decision-making. Esophageal dilation is reserved for patients with fixed fibrostenotic disease. Treatment selection should be individualized based on age, disease phenotype, severity, comorbid atopic disease, family preferences, and resource availability (Figure 3) [10, 44].
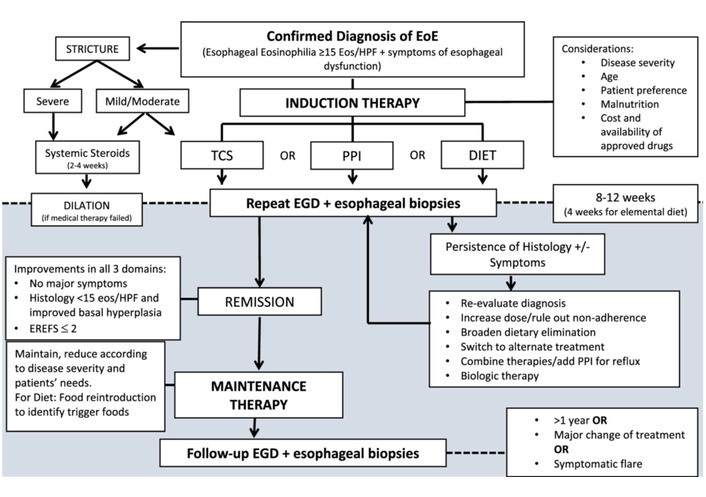
European Society of Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) therapeutic algorithm for pediatric eosinophilic esophagitis (EoE). Eos/HPF: eosinophils per high power field; TCS: topical corticosteroids; PPI: proton pump inhibitor; EGD: esophago-gastro-duodenoscopy; EREFS: Eosinophilic Esophagitis Endoscopic Reference Score. See reference in text. Reprinted with permission from [10]. © Copyright-2024 ESPGHAN and North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition.
PPIs (omeprazole, esomeprazole, lansoprazole, or pantoprazole) are widely used as first-line therapy due to their favorable safety profiles and ease of administration. The therapeutic effect of PPIs extends beyond acid suppression and includes anti-inflammatory properties, such as downregulation of CCL26 expression and improvement of epithelial barrier function [43–45]. Histologic remission is achieved in approximately 40–60% of pediatric patients, with symptom improvement in 50–70%. In the induction phase, PPIs are usually given at a daily dose of 1–2 mg/kg body weight for 8–12 weeks. The current pediatric guidelines report that there is a trend towards a higher response rate when PPIs are administered twice daily compared with once daily [10]. Current EoE guidelines also recommend that in responder patients, PPIs should be given at the lowest effective dose [10, 44]. In this respect, it might be worth investigating whether, in maintenance therapy, a once-daily administration could improve compliance without affecting efficacy. PPIs are generally well tolerated; clinically significant adverse events occur in fewer than 5% of children [10, 44].
Topical steroids are highly effective for both induction and maintenance therapy of pediatric EoE [46]. Unlike systemic steroids, swallowed formulations provide targeted anti-inflammatory effects with minimal systemic exposure. Histologic remission occurs in approximately 60–80% of pediatric patients, with symptom improvement in 50–70%. Swallowed topical corticosteroids include budesonide viscous slurries 0.5–1 mg twice daily, and fluticasone 440–880 μg/day [10, 44, 46]. Both products improve mucosal contact and treatment efficacy, should be taken after meals, rinsing the mouth to reduce candidiasis risk, and avoiding food or drinks for at least 30 minutes afterwards. Esophageal candidiasis occurs in 5–15% of cases and is usually asymptomatic, while clinically relevant adrenal suppression is reported in fewer than 5%, mostly younger children or those receiving concurrent inhaled steroids [10, 46]. Since prolonged and/or repeated courses of therapy are needed in order to maintain remission, we should remain cautious about the systemic effects of topical steroids in the pediatric population.
Nutritional management is a cornerstone of pediatric EoE care and requires close collaboration between gastroenterologists, allergists, and dietitians [1, 10, 34, 44]. Dietary therapy targets food antigens driving inflammation, and includes elemental diets, empiric elimination diets, step-up or step-down strategies tailored to disease severity [47, 48]. Each approach must balance efficacy with nutritional adequacy, palatability, and family burden. Elemental diets induce histologic remission in more than 90% of children but are limited by palatability and psychosocial burden. Empiric elimination diets are commonly used. Six-food elimination diets achieve remission in approximately 65–75% of pediatric patients, while four-food elimination diets achieve remission in 50–65% [47–49]. Single food (cow’s milk) elimination diet is much simpler and tolerable, and has shown comparable histological remission rates (50–65%) and improved quality of life (QoL) compared with four- and six-food elimination diets [50–52]. Targeted elimination diets are usually based on allergy testing and have variable efficacy [47], mostly because currently available allergy tests, like skin prick/patch or blood tests, are unreliable to determine trigger food(s) in the setting of EoE [53, 54].
Dietary therapy requires collaboration with a pediatric dietitian to ensure nutritional adequacy and adherence. Children with EoE who are prescribed dietary treatment should undergo regular growth assessment, monitoring for micronutrient deficiencies, and dietitian counseling to ensure a balanced intake [10, 44]. In EoE, the unpleasant experience of food-related symptoms and the poor palatability of elemental formula and elimination diets are risk factors for the development of feeding dysfunction, such as avoidant restrictive food intake disorder (ARFID), especially in young children. Feeding dysfunction indeed occurs in 30–60% of young children, and micronutrient deficiencies are reported in 10–25% without dietetic supervision [10, 17]. Feeding dysfunction may thus persist even after histologic remission, and behavioral feeding therapy and psychological support are often necessary components of comprehensive care [10, 17, 49].
Biologic therapies represent a major therapeutic advance in pediatric EoE, reflecting growing understanding of the molecular drivers of type 2 inflammation. These agents target key cytokines and signaling pathways involved in eosinophil recruitment, epithelial remodeling, and immune dysregulation. Biologic therapy is generally considered for children and adolescents with moderate-to-severe EoE, particularly those with inadequate response or intolerance to dietary therapy and topical corticosteroids, or with coexisting severe atopic disease.
Dupilumab is a fully human monoclonal antibody directed against the IL-4 receptor α subunit, thereby blocking signaling of both IL-4 and IL-13. Clinical trials and pediatric extension studies have demonstrated that dupilumab 300 mg weekly or every two weeks produces marked reductions in esophageal eosinophilia, improves dysphagia and feeding-related symptoms, reverses disease-associated transcriptional signatures, and provides benefit across pediatric age groups, including adolescents and younger children [55, 56]. Dupilumab achieves histologic remission in approximately 55–65% of treated adolescents compared with 5–15% in placebo groups and reduces eosinophil counts by more than 80%. Injection-site reactions occur in approximately 10–15% of patients. Dupilumab is particularly attractive in pediatric patients with multiple atopic comorbidities, as it simultaneously improves asthma, atopic dermatitis, and allergic rhinitis [57].
Therapies targeting IL-5 or the IL-5 receptor (e.g., mepolizumab, reslizumab, benralizumab) reduce eosinophil survival and circulating eosinophil counts. While these agents consistently reduce eosinophilia, their effects on symptoms and histologic remission in EoE have been more variable than those observed with IL-4/IL-13 blockade. At present, anti-IL-5 therapies are considered investigational or adjunctive in pediatric EoE, though they may be appropriate in selected patients with severe eosinophilic disease phenotypes [29, 58].
Novel biologic agents targeting upstream epithelial alarmins, including TSLP and IL-33, are under active investigation [22, 23, 58]. These therapies aim to intervene earlier in the inflammatory cascade and may offer disease-modifying potential, particularly in pediatric patients. The selective sphingosine 1-phosphate (S1P) receptor modulator etrasimod showed promising results in adults with EoE, leading to sustained symptomatic, endoscopic, and histologic improvement. As additional biologics become available, molecular endotyping may guide therapy selection and enable precision medicine approaches in pediatric EoE [22, 23, 26, 29, 32, 58].
Esophageal dilation is reserved for patients with fixed strictures or significant luminal narrowing causing dysphagia [59]. Although esophageal dilation is effective for symptom relief in fibrostenotic disease, it does not alter the underlying inflammatory process and should be combined with anti-inflammatory medical therapy [59, 60].
Initial therapy should be reassessed after 8–12 weeks with repeat endoscopy and biopsy [10, 34, 44]. In fact, symptom improvement does not reliably predict histologic remission. Relapse is common after treatment discontinuation, and prolonged maintenance therapy is often required. Patients with relapsing or refractory disease may require therapy escalation, combination strategies, or referral to specialized centers (Figure 3) [10, 34, 44].
Pediatric EoE is a chronic, progressive disease with important implications for long-term outcomes and quality of life extending into adulthood. Longitudinal studies demonstrate that approximately 60–80% of children with EoE have persistent disease continuing into adulthood. Rates of sustained, treatment-free remission are low, generally reported in < 10% of patients [10, 16, 17, 60]. Early, pediatric-onset disease is predominantly inflammatory; however, with increasing disease duration, a transition toward fibrostenotic phenotypes may occur. Persistent inflammation may lead to impaired esophageal distensibility, recurrent food impactions, and the need for endoscopic dilation [16, 17]. The median diagnostic delay in pediatric EoE ranges from approximately 2 to 6 years, and each year of untreated disease is associated with an estimated 5–7% increase in the risk of developing fibrostenotic complications, including strictures [16, 17, 60, 61]. Strictures are uncommon in young children, occurring in fewer than 5%, but are observed in approximately 30–40% of adolescents and young adults with long-standing disease. Male sex confers an approximately 2- to 3-fold increased risk of fibrostenotic disease. Additional risk factors for fibrostenotic disease and a more complicated course include early disease onset, high peak eosinophil density, prolonged diagnostic delay, and poor compliance with treatment [60, 61]. With appropriate therapy, most pediatric patients achieve clinical and histologic disease control; however, relapse is common following treatment discontinuation. Current pediatric guidelines, therefore, emphasize the need for maintenance strategies after induction of remission and caution against relying solely on symptom resolution as a marker of disease inactivity [10]. Available evidence indicates that relapse after treatment discontinuation is common and may occur within a relatively short time frame. In prospective pediatric studies evaluating swallowed topical corticosteroids, discontinuation of therapy has been associated with high relapse rates. In one cohort treated with viscous budesonide, only 45% of children maintained remission 12 weeks after stopping therapy, highlighting the tendency toward early disease recurrence [62]. Consistent findings have been reported in larger studies, including adolescents and adults: following successful induction with topical steroids, recurrence of symptoms occurred in more than half of patients within one year, and histologic relapse was documented in the majority of those with recurrent symptoms [63]. Randomized withdrawal data further demonstrate significantly higher relapse rates in patients switched to placebo compared with those continuing maintenance therapy (approximately 61% vs. 28% over 36 weeks) [64]. Despite this generally relapsing course, a minority of pediatric patients appear capable of sustaining clinical and histologic remission after treatment withdrawal. Small pediatric cohorts have described prolonged remission even after reintroduction of previously eliminated foods, suggesting biological heterogeneity and the existence of more favorable disease phenotypes in selected children [65]. In clinical practice, these data support a cautious approach to treatment de-escalation, with close follow-up and, when appropriate, repeat endoscopic assessment to confirm sustained remission, particularly in patients with prior severe inflammation or fibrostenotic features.
Recognition of negative prognostic factors may help identify patients who would benefit from closer monitoring and/or more aggressive early therapy [10, 58, 60, 61].
Concerns regarding growth impairment are particularly relevant in pediatric patients with EoE [10, 44]. While most children maintain normal growth trajectories with appropriate management, those with early-onset disease, delayed diagnosis, severe disease, and restrictive dietary therapy have an increased risk for feeding dysfunction, nutritional deficiencies, and impaired growth [66]. Multidisciplinary care involving pediatric gastroenterologists and dietitians is therefore essential to prevent feeding difficulties and nutritional deficiencies, thus ensuring normal growth and development [67]. Furthermore, growth should also be carefully monitored in infants and children who are being treated with recurrent or prolonged courses of topical corticosteroids, in view of their possible repercussions on adrenal function [66, 67].
EoE has a substantial impact on health-related quality of life (HRQoL) for both patients and families [66, 68]. A clinically meaningful impairment in HRQoL is reported in more than 50% of affected children and families. Mealtime anxiety related to eating and choking, fear of food impaction, social limitations around meals and school activities, emotional distress associated with procedures and medications, and parental stress are commonly reported [69]. Adolescents may report impaired autonomy and social participation, particularly in the context of dietary restrictions and recurrent symptoms. Caregivers often experience significant psychosocial stress related to dietary planning, fear of food impaction, and the need for repeated endoscopic procedures [68, 69]. Validated pediatric EoE-specific QoL instruments have been developed and are increasingly being incorporated into routine care and clinical trials [10, 68]. Studies demonstrate that caregiver burden and anxiety may parallel disease activity and improve with effective disease control. Effective treatment of EoE improves not only endoscopic and histologic outcomes but also quality of life [10, 67–69].
As EoE is a lifelong condition for most patients, a structured transition from pediatric to adult care is crucial to prevent loss to follow-up and disease progression. Transition programs should address disease education and self-management skills, adherence to maintenance therapy, monitoring strategies, and psychosocial support [10, 70, 71].
Despite major advances in the understanding and management of pediatric EoE, important unmet needs remain. Ongoing research aims to improve disease modification, reduce reliance on invasive monitoring, and personalize therapy. Next-generation biologics targeting IL-13, IL-5, IL-5 receptor, S1P receptor, and upstream epithelial alarmins are under investigation, with early-phase trials demonstrating significant reductions in esophageal eosinophilia [23, 26, 58]. Molecular endotyping and transcriptomic profiling may allow prediction of response to PPIs, dietary therapy, or biologics, facilitating individualized treatment strategies [32, 58]. Minimally invasive tools such as the esophageal string test and Cytosponge are being evaluated as alternatives to repeated endoscopy in children [10, 58]. Treat-to-target strategies and earlier initiation of effective therapy may reduce progression to fibrostenotic disease, particularly during childhood. Long-term safety of biologics, optimal maintenance strategies, role of environmental factors, and the impact of EoE on growth and neurodevelopment remain priorities for future research.
EoE is now recognized as a leading cause of chronic esophageal symptoms in children and adolescents, with a disease course that is typically persistent and prone to progression if left untreated [1, 10, 13, 16, 17]. The clinical heterogeneity observed across pediatric age groups, particularly the non-specific presentation in infants and young children, continues to pose diagnostic challenges and contributes to delays in diagnosis. Timely recognition of EoE and confirmation through appropriate endoscopic and histologic evaluation are essential to prevent long-term complications, including esophageal remodeling and stricture formation [16, 17]. Current therapeutic options—including dietary elimination strategies, swallowed topical corticosteroids, and PPIs—are effective in inducing disease remission in many pediatric patients when appropriately selected and monitored [10, 44]. The recent introduction of targeted biologic therapy, particularly dupilumab, represents a major advance for children with moderate to severe or treatment-refractory disease and marks an important step toward precision medicine in pediatric EoE [55, 58]. Ongoing research aimed at identifying noninvasive biomarkers, optimizing dietary approaches, and defining the long-term safety of emerging therapies will further refine disease management.
In conclusion, pediatric EoE is a chronic but increasingly manageable condition. Early recognition, individualized therapy, and multidisciplinary care—combined with continued advances in research and therapeutics—are essential to improving long-term outcomes and quality of life for affected children and their families.
CAPN14: calpain-like protease 14
CCL26: eotaxin-3
EoE: eosinophilic esophagitis
eos: eosinophils
EREFS: Eosinophilic Esophagitis Endoscopic Reference Score
FLG: filaggrin
GERD: gastroesophageal reflux disease
GI: gastrointestinal
HPF: high power field
HRQoL: health-related quality of life
PPIs: proton pump inhibitors
QoL: quality of life
S1P: sphingosine 1-phosphate
TGF-β: transforming growth factor-β
TNE: transnasal endoscopy
TSLP: thymic stromal lymphopoietin
AR: Conceptualization, Investigation, Methodology, Supervision, Writing—original draft, Writing—review & editing. The author read and approved the submitted version.
The author declares that he has no conflicts of interest.
The study involved in Figure 1 complies with the Declaration of Helsinki. Ethical approval for that study was not required because the endoscopic procedures by which the images were obtained were carried out according to Good Clinical Practice.
Not applicable.
Not applicable.
The data involved in Figure 1 could be available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1865
Download: 38
Times Cited: 0
Nayantrishna Nath ... Prasenjit Das
Adrianna Wierzbicka, Andrew Ukleja
