Affiliation:
1Lifecare Hospital Lahore, Nespak Housing Society, Lahore 54770, Pakistan
ORCID: https://orcid.org/0009-0007-0318-2443
Affiliation:
2Nursing Department, Al-Zaytoonah University, Amman 11733, Jordan
Email: omaralqaisi119@gmail.com
ORCID: https://orcid.org/0009-0006-9760-651X
Affiliation:
2Nursing Department, Al-Zaytoonah University, Amman 11733, Jordan
ORCID: https://orcid.org/0000-0002-8345-0140
Affiliation:
3Faculty of Medicine, University of Prishtina, 10000 Prishtina, Kosovo
ORCID: https://orcid.org/0009-0002-4880-1838
Affiliation:
3Faculty of Medicine, University of Prishtina, 10000 Prishtina, Kosovo
ORCID: https://orcid.org/0009-0000-9309-9959
Affiliation:
4Division of Oncology, University of Saskatchewan, Saskatoon, SK S7N 5A2, Canada
ORCID: https://orcid.org/0000-0001-5730-0088
Affiliation:
5Department of Oncology, University of Alberta, Edmonton, AB T6G 2R3, Canada
ORCID: https://orcid.org/0000-0003-3514-2183
Affiliation:
4Division of Oncology, University of Saskatchewan, Saskatoon, SK S7N 5A2, Canada
ORCID: https://orcid.org/0000-0001-7700-776X
Explor Med. 2026;7:1001388 DOI: https://doi.org/10.37349/emed.2026.1001388
Received: October 24, 2025 Accepted: January 30, 2026 Published: March 15, 2026
Academic Editor: Marco Cascella, University of Salerno, Italy
The article belongs to the special issue Practical Tips for Cancer Care: Guidance for Patients, Caregivers, and Healthcare Professionals
Opioid use disorder (OUD) is an emerging clinical and public health concern in cancer care. Although opioids remain essential for cancer pain management, a substantial subset of patients develops OUD, including many iatrogenic cases. This scoping review (ScR) synthesized evidence on the epidemiology, risk factors, screening approaches, and multidisciplinary management of OUD in adults with cancer. The review followed Joanna Briggs Institute (JBI) and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-ScR guidelines. Four databases (PubMed, Medline, Scopus, ScienceDirect) were searched for English language randomized trials, quasi-experimental studies, and observational studies published from 2019–2025. Eligible studies included adults aged ≥ 18 years with cancer. Two independent reviewers conducted the study selection (OA and YI). The search identified 1,044 records; 403 abstracts were screened; 164 full-texts were assessed; and 46 studies met the inclusion criteria. OUD prevalence averaged 8% (range 6–50%), with 23.5% of patients classified as at risk. New persistent opioid use occurred in 19% within 8 weeks. Postoperative persistent opioid use was 33.3% overall, higher among patients with prior opioid exposure (48.3%) than opioid-naive individuals (18.5%). Reported risk factors included prior opioid use, younger age, male sex, substance use history, anxiety, and financial distress. Screening tools identified approximately 20% of patients as high risk. Multidisciplinary interventions—including buprenorphine/naloxone treatment and structured monitoring—were effective in managing concurrent cancer pain and OUD. OUD in cancer populations is preventable and manageable through systematic risk assessment and integrated multidisciplinary care. Early screening with validated tools, structured monitoring, and compassionate communication support safe opioid use while maintaining adequate analgesia. Evidence supports expanding access to medication-assisted therapies, psychosocial support, and harm-reduction strategies. These findings provide a foundation for improving clinical outcomes and guiding future research.
Cancer-related pain is among the most prevalent and debilitating symptoms experienced by patients with cancer and remains a major factor in quality of life, functional status, and clinical outcomes [1, 2]. Despite substantial advances in anticancer therapies and supportive care, pain continues to affect a large proportion of patients throughout the disease course. Meta-analytic data indicate that approximately 55% of patients receiving active cancer treatment experience pain, and the prevalence rises to approximately 66% among those with advanced or terminal disease [3]. Consequently, effective pain control is widely recognized as a cornerstone of comprehensive cancer care and a core ethical obligation of oncology practice. However, access to adequate analgesia remains uneven worldwide, with most patients with moderate-to-severe pain in low- and middle-income countries still receiving inadequate treatment, underscoring the global relevance of optimizing pain management strategies [4, 5].
Opioid analgesics have long been the keystone for managing moderate to severe cancer pain. Clinical evidence and practice guidelines support the use of opioids as the gold standard for cancer-related pain relief [6]. These medications provide potent analgesia with flexible dosing, multiple routes of administration, and no absolute ceiling dose, making them indispensable for controlling severe cancer pain [7]. The introduction of the World Health Organization’s “analgesic ladder” in 1986 cemented opioids’ central role in cancer pain therapy, and they are listed as essential medication for palliative care [8]. However, opioid benefits come with well-known side effects (such as sedation, constipation, and respiratory depression) and the potential for tolerance and dependence.
Over the past two decades, an opioid epidemic has unfolded in the context of chronic non-cancer pain, marked by indiscriminate opioid prescribing and a surge in misuse and overdose deaths in the general population [9, 10]. This public health crisis has prompted the medical community to re-examine opioid prescribing practices even in oncology, raising awareness that patients with cancer are not immune to opioid use disorder (OUD) [2].
Increasing cancer survival rates and longer durations of cancer pain management have brought OUD in cancer patients into sharper focus [11]. Traditionally, it was thought that the risk of addiction was negligible when opioid was used for bona fide cancer pain. Recent data have challenged this assumption, showing that a meaningful subset of patients with cancer can and do develop opioid misuse or addiction. A 2024 systematic review found reported rates of opioid misuse in the cancer population ranging from approximately 6% to as high as 50%, depending on definitions, with a pooled estimate suggesting about 12% of patients may be at risk of aberrant opioid use [12]. Another review focusing on OUD in cancer pain patients reported an OUD prevalence of around 8%, with some studies documenting up to 20% of patients meeting criteria for opioid addiction or problematic use [13]. These findings make clear that aberrant opioid behaviors-including non-medical opioid use (NMOU) and “chemical coping”-occur in a notable minority of cancer patients receiving long-term opioid [14]. The issue is twofold: some individuals enter their cancer journey already struggling with substance use (given the high background prevalence of opioid misuse in society), while others develop iatrogenic OUD during or after cancer treatment [15]. In fact, epidemiologic data from the United States showed that opioid-related overdose deaths among patients with cancer increased significantly between 2006 and 2016, mirroring trends in the general population [16]. Such evidence dispels the misconception that cancer patients are categorically protected from addiction and underlines the emerging concerns about OUD in oncology settings. At the individual level, co-existing OUD can compromise cancer care by impairing patients’ ability to adhere to treatment, increasing hospitalization, and worsening overall prognosis [2, 17]. Opioid misuse carries the well-documented risk of overdose and death, which is a catastrophic outcome for patients already fighting cancer [18, 19]. Excessive or prolonged opioid use also leads to adverse health effects: studies indicate that chronic opioid therapy may suppress immune function (potentially affecting infection risk and even tumor progression), while causing endocrinopathies and cognitive impairment [20]. Patients on high-dose opioids often suffer side effects like sedation, mental clouding, respiratory depression, and gastrointestinal dysfunction, all of which can significantly diminish functional status and quality of life [21, 22]. Furthermore, opioid dependence may engender profound psychosocial consequences. Patients with cancer and OUD frequently experience stigma and psychological distress, which can deter them from reporting pain or taking adequate analgesia. Family members and healthcare providers might inadvertently stigmatize these patients, perceiving their pain compliant with suspicion, thereby straining the therapeutic alliance. On the societal level, the intersection of cancer and OUD places additional burdens on health systems [23]. Rising rates of OUD related emergency visits and complications in oncology add to healthcare costs and resource utilization [13]. Thus, OUD in the context of cancer is a serious comorbidity that not only endangers patients’ safety but also complicates the management of cancer itself and strains public health resources. These realities highlight the clinical dilemma of balancing adequate pain relief with the minimization of misuse, a central challenge in modern cancer care. This review will offer expert recommendations to bridge the gap between research and practice.
This scoping review (ScR) introduces and summarizes the updated evidence about the prevalence, screening methods, risk factors, and management strategies for OUD among adult patients with cancer. Additionally, we describe the rapid timeline of misuse emergence and assess the multidisciplinary approach that aimed at balancing effective pain control with opioid safety.
This ScR followed the JBI methodology for ScRs and PRISMA-ScR guidelines. The eligibility criteria were defined using the population, concept, context framework:
Population: adult patients (aged ≥ 18 years) diagnosed with cancer, regardless of cancer type or stage.
Concept: OUD, including opioid misuse, dependence, risk factors, screening tools, and management interventions.
Context: all clinical settings, including hospitals, primary care units, and oncology centers, without geographical restrictions.
A comprehensive literature review was conducted on four major databases: PubMed, Medline, Scopus, and ScienceDirect. Inclusion criteria were defined as articles: (1) only published English language, (2) published between 2019 and 2025, (3) full-text randomized controlled trials, quasi-experimental studies, and observational studies focusing on adult cancer patients receiving OUD interventions in primary care or hospital settings. This review included randomized controlled trials, quasi-experimental studies, observational studies (cohort, case control, cross-sectional), case series, and case reports. Systematic reviews were excluded to avoid data duplication, but were screened for primary references.
Search terms included “opioid use disorder”, “opioid dependence”, “opioid misuse”, “cancer”, “malignancy”, “oncology”, and “pain management”, combined using Boolean operators (AND/OR). The complete search strategy, including exact search terms for each database and filters applied, is provided in Table S1.
Two independent reviewers (OA and YI) screened all identified citations using predefined inclusion criteria. Potentially relevant studies were retrieved for full-text assessment. Any disagreements were resolved through discussion or consultation with a third reviewer (SAG). All citations were collated and managed using EndNote software, with duplicates removed prior to screening.
The study selection process is illustrated in Figure 1, following PRISMA-ScR guidelines. Database searches yielded 1,044 initial records. After removing 641 duplicates, 403 unique records were screened by title and abstract. Full-text assessment was performed for 164 articles, of which 118 were excluded for not meeting the inclusion criteria. Ultimately, 46 studies were included in the final ScR.
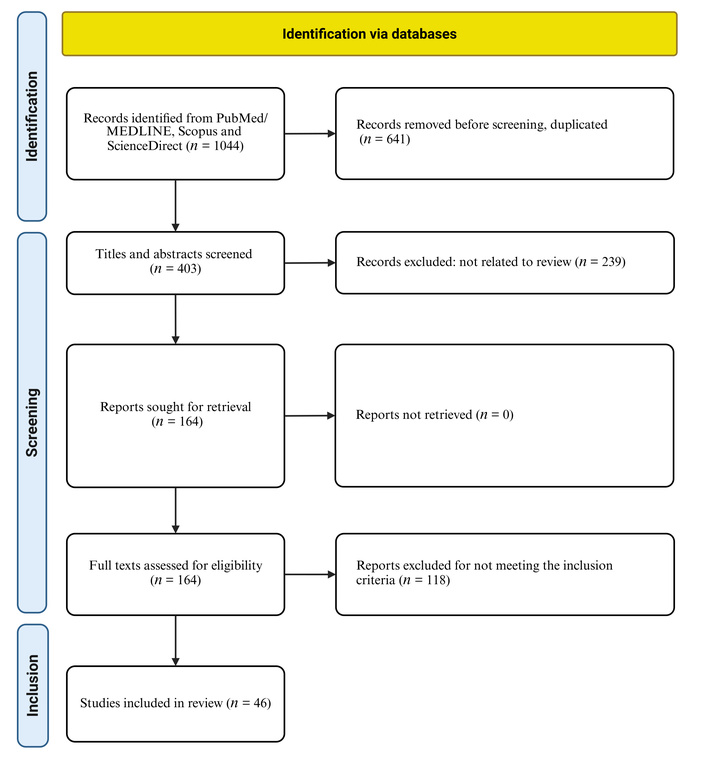
PRISMA flow diagram for ScR of opioid use disorder in cancer patients. The diagram illustrates the systematic study identification, screening, and selection process: initial records identified (n = 1,044) → records after deduplication (n = 403) → records screened → full-text articles assessed (n = 164) → studies included (n = 46), following PRISMA-ScR guidelines. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; ScR: scoping review. Adapted from “PRISMA” (http://www.prisma-statement.org/). Accessed May 30, 2025. © 2024–2026 PRISMA Executive. Distributed under a Creative Commons Attribution (CC BY 4.0) license.
Consistent with the JBI methodology for ScRs, the critical appraisal of individual studies (risk of bias) was not conducted. The primary aim of this review was to map the available evidence and identify key characteristics and gaps in the literature regarding OUD in cancer patients, rather than to synthesize effectiveness data. However, data regarding study design and sample characteristics were extracted to provide context on the strength of evidence.
From those 46 articles that were included in the final literature review, the following data were abstracted and inserted into Table S2: author and year of publication, purpose of study, country, sample size, design, and main findings. The process of selecting the final 46 studies is outlined in the figure, and the studies are listed in Table S2.
ScRs do not formally assess study quality; however, source credibility and risk of bias were considered during data extraction and reporting. Results were synthesized narratively and through thematic mapping, focusing on gaps and trends in the prevalence and risk factors of OUD among cancer patients, screening and assessment tools for identifying at-risk patients, treatment approaches including medication-assisted therapy and multimodal pain management strategies, barriers to adequate pain control in patients with co-occurring substance use disorders, interdisciplinary care models and opioid stewardship programs in oncology settings, expert consensus recommendations for managing cancer pain in patients with OUD, and patient outcomes related to pain control, quality of life, and addiction recovery in the cancer population. This analytical approach enabled comprehensive identification of evidence gaps and informed the development of expert recommendations to improve current clinical practice, policy frameworks, and research priorities for addressing OUD in cancer patients.
Following the PRISMA guidelines, our comprehensive search strategy across four major databases (PubMed, Medline, Scopus, and ScienceDirect) initially identified 1,044 potentially relevant studies. After removing 641 duplicates, 403 records underwent title and abstract screening. Subsequently, 164 studies were assessed for full-text eligibility, with 118 studies excluded based on predetermined criteria (non-cancer population: n = 42, non-opioid focus: n = 35, study design/editorial commentaries: n = 25, and full-text unavailable or language barriers = 16). The final ScR included 46 studies that met all inclusion criteria, spanning publication years from 2019 to 2025.
The included studies represented diverse geographical regions, with the majority conducted in the United States (n = 38), followed by Canada (n = 2), and other countries, including South Korea, France, Britain and Norway, etc (n = 6). Study designs varied considerably, including retrospective cohort studies (n = 18), reviews and meta-analyses (n = 12), case reports and series (n = 5), cross-sectional studies (n = 3), prospective interventional studies (n = 2), qualitative studies (n = 1) and miscellaneous (n = 5) (Table 1).
Characteristics of included studies on opioid use disorder (OUD) in cancer patients (refer to Table S2 for details).
| Factor/characteristics | Prevalence/rate or associated | Studies |
|---|---|---|
| Overall OUD prevalence | 6–50% (pooled mean: 8%) | Multiple systematic reviews & meta-analyses [24, 25] |
| Patients at high risk for OUD | 23.5% | Meta-analysis [25]; screening tools (screener and opioid assessment for patients with pain identified 20% as high-risk [11, 26]) |
| Development of non-medical opioid use | 19% within 8 weeks of opioid prescription | Early follow-up data [11] |
| Chronic opioid use after diagnosis | 14% (majority were opioid-naive) | Population-based studies [27] |
| Persistent postoperative opioid use | 33.3% overall (48.3% with prior exposure, 18.5% opioid-naive) | Head and neck surgery cohort data [28] |
| Key risk factors | Prior opioid exposure, younger age, male gender, substance use history, anxiety, and financial distress | Multi-study consensus [29, 30] |
| Intervention effectiveness | Multidisciplinary team-based care and buprenorphine/naloxone showed efficacy | Multi-case series and intervention studies [31] |
A critical finding emerged regarding the rapid development of aberrant opioid behaviors following treatment initiation. Multiple studies demonstrated that approximately 19% of cancer patients develop NMOU within just eight weeks of opioid initiation [11]. This rapid timeline underscores the importance of early identification and intervention strategies in cancer care settings.
Saraswathula et al. [28] specifically examined persistent postoperative opioid use in older head and neck cancer patients, finding that 33.3% of patients overall developed persistent use, with significantly higher rates among those with prior opioid exposure (48.3%) compared to opioid-naive patients (18.5%) (in Table S2). The study identified postoperative radiotherapy (odds ratio ≈ 1.99) and higher comorbidity burden as additional risk factors for persistent use in Table S2.
This review identified consistent risk factors across multiple studies that predispose cancer patients to developing OUD or aberrant opioid behaviors. Yennurajalingam et al. [11] conducted a comprehensive assessment of 745 cancer patients using the Screener and Opioid Assessment for Patients with Pain, finding that approximately 20% were identified as high-risk. Primary risk factors included male gender, higher pain severity scores, anxiety, financial distress, and positive screening for alcohol/drug use using the CAGE adapted to include drugs instrument.
Carmichael et al. [26] performed an integrative literature review analyzing 34 publications focused on opioid misuse risk assessment in cancer patients in Table S2. Their findings confirmed that screening tools and urine drug screens (UDSs) could effectively identify approximately 20% of cancer patients as being at risk for OUD. Importantly, the review demonstrated that high-risk patients showed significantly greater misuse behaviors and worse clinical outcomes compared to low-risk patients.
A study identified specific demographic and psychological predictors of aberrant opioid behavior in their case-control study of 100 cancer patients on chronic opioid therapy (see Table S2). The analysis revealed that younger age and higher baseline anxiety levels were significant predictors of aberrant use, with the most common aberrant behavior being early opioid refill requests.
Several studies examined the clinical consequences of OUD in cancer populations. Yoo et al. [29] conducted a multicenter retrospective cohort study of 2,066 advanced cancer patients in South Korean palliative care units, demonstrating that opioid use significantly increased delirium risk in a dose-dependent manner (see Table S2). The study identified specific high-risk subgroups, including older patients, males, those not receiving chemotherapy, and non-obese patients, who showed elevated delirium risk with opioid exposure.
Singh et al. [32] performed a matched case-control analysis examining pain management disparities in hospitalized cancer patients with documented OUD history. The study revealed that cancer patients with OUD received significantly lower opioid doses than matched non-OUD patients [median –3 morphine milligram equivalent (MME)/day vs. +37 MME/day], with 90% lower odds of dose escalation. These disparities only equalized after palliative care consultation, suggesting systematic undertreatment of pain in this vulnerable population.
Multiple studies evaluated intervention strategies for managing cancer patients with concurrent OUD. Hamlish et al. [33] presented a successful case report of multidisciplinary team (MDT) management for a 69-year-old head and neck cancer survivor with chronic pain who developed OUD (see Table S2). The coordinated approach, led by oncology teams and utilizing buprenorphine/naloxone, effectively managed both OUD and pain control while providing essential psychosocial support.
Kale et al. [34] described the development and implementation outcomes of a novel Palliative Harm Reduction and Resiliency clinic specifically designed for cancer patients with substance use disorders. The quality improvement study included 101 patients and demonstrated successful treatment outcomes with steady patient volume growth, 70% retention for follow-up visits, and 55% of OUD patients receiving buprenorphine as part of their comprehensive management.
Weiss et al. [35] presented a comparative case analysis of a 75-year-old cancer patient maintained on buprenorphine for OUD, demonstrating that continuing buprenorphine alongside other opioids proved more effective for cancer pain control than tapering protocols. When buprenorphine was discontinued, morphine dosing required doubling to achieve equivalent analgesia.
Yusufov et al. [31] developed and pilot-tested a behavioral intervention based on Acceptance and Commitment Therapy specifically designed to reduce OUD risk in cancer patients. The six-session program demonstrated feasibility, acceptability, and positive patient satisfaction among all ten adult participants, with patients finding mindfulness and coping strategies particularly beneficial.
Bulls et al. [23] conducted qualitative interviews with 31 participants (20 patients with advanced cancer and 11 support persons) to investigate attitudes about OUD and opioid risks. The study revealed conflicting perspectives, with many participants believing OUD risk was minimal in cancer pain settings and relying primarily on personal control as protection. These findings highlighted the need for improved communication strategies addressing realistic opioid risks and benefits.
Dobson and Blackhall [36] evaluated outcomes of universal substance use disorder screening in an outpatient palliative care clinic managing 204 cancer patients on opioid therapy. Universal screening using UDSs and opioid risk assessment tools identified fewer than 3% of patients with aberrant opioid use. High-risk patients were successfully managed through structured protocols including frequent clinic visits, limited opioid dispensing quantities, and opioid rotation or tapering when indicated. Notably, no patients in the study cohort experienced overdose events or required emergency care for substance use-related complications.
Several studies addressed OUD risks in specific cancer populations. Ji et al. [37] examined opioid prescribing patterns among 8,969 pediatric cancer survivors compared to 44,845 matched non-cancer controls during the first-year post-therapy. The study revealed significantly higher rates of potential opioid misuse among adolescent cancer survivors (22.9%) compared to age-matched peers without cancer (7.7%), with risk increasing progressively with age.
Beauchemin et al. [38] conducted a systematic review focusing on opioid use and misuse in children, adolescents, and young adults with cancer. Across 11 included studies, opioid usage prevalence in pediatric cancer populations ranged widely from approximately 12% to 90%. The review identified prior mental health disorders, substance use history, and previous opioid exposure as primary risk factors for misuse in these younger populations.
Jones et al. [30] conducted a modified Delphi consensus study involving 120 experts in palliative care and addiction medicine to develop evidence-based guidance for managing advanced cancer-related pain in patients with opioid misuse behaviors or confirmed OUD. The expert panel achieved consensus on several key recommendations, including the appropriateness of initiating buprenorphine/naloxone instead of full opioid agonists for patients with documented OUD, while noting that referral to methadone clinics was not typically recommended. For patients with short prognoses, split-dose methadone for pain management was considered appropriate, though recommendations were less certain for patients with longer life expectancies.
The consensus emphasized the importance of managing these patients within oncology/palliative care teams rather than specialized opioid treatment programs, while acknowledging the need for policy reform to expand access to buprenorphine and methadone for cancer patients. The expert panel also recommended enhanced monitoring protocols and discouraged opioid tapering or discontinuation for patients with aberrant use behaviors but without a confirmed OUD diagnosis.
The findings of this ScR indicate that OUD among cancer patients represents a significant clinical challenge that exceeds previous estimates and assumptions. The pooled prevalence of OUD ranging from 6% to 50% with a mean of 8%, alongside the identification that 23.5% of cancer patients are at high risk for developing problematic opioid use, challenges the traditional belief that cancer patients are inherently protected from addiction [27]. The 46 studies spanning 2016–2025 revealed that OUD in cancer populations is more prevalent than historically recognized. The pooled estimate of 8% OUD prevalence, with some studies reporting rates as high as 50%, underscores the need for heightened awareness among oncology providers [27]. This finding is particularly concerning given that 79% of patients who developed chronic opioid use were previously opioid-naive, indicating that cancer treatment itself may serve as a gateway to problematic opioid use patterns [39].
The identification that nearly one-quarter (23.5%) of cancer patients are at elevated risk for OUD development necessitates the implementation of systematic risk stratification protocols [26]. These findings contradict earlier assumptions that cancer-related pain inherently justifies unlimited opioid access without consideration of addiction potential.
Consistent risk factors emerged across multiple studies, including prior opioid exposure, younger age, male gender, substance use history, anxiety disorders, and financial distress [22, 26]. These demographics and clinical characteristics provide actionable targets for preventive interventions and enhanced monitoring protocols.
The finding that 19% of patients developed NMOU within eight weeks of initial prescription highlights the rapid timeline within which problematic patterns can emerge [11]. Similarly, the persistence of postoperative opioid use in 33.3% of surgical patients, with rates varying significantly based on prior opioid exposure (48.3% versus 18.5% in opioid-naive patients), demonstrates the critical importance of perioperative opioid stewardship [26].
While validated screening instruments may help identify patients at high risk, evidence suggests they should not rely on them as stand-alone tools [40]. Screening instruments must be incorporated with careful attention to opioid dose, duration of therapy, and ongoing clinical assessment, as reliance on screening alone may underestimate true risk. Close monitoring should focus on dose escalation, prolonged duration, early refill requests, and functional outcomes rather than sole dependence on screening scores. These tools successfully flagged approximately 20% of patients as high-risk, providing clinicians with objective criteria for enhanced monitoring and alternative pain management strategies. Evidence from non-cancer populations demonstrates that each additional day of opioid therapy beyond the third day substantially increases the likelihood of long-term use, with persistent risk observed years later [40]. Although cancer pain presents unique considerations, these findings underscore the importance of minimizing initial opioid dose and duration whenever clinically feasible.
The long-term implications of opioid exposure differ substantially between cancer survivors and patients with a limited prognosis. Among survivors, persistent opioid use and OUD may adversely affect long-term quality of life, functional recovery, and psychosocial well-being, underscoring the importance of conservative prescribing and early intervention.
Evidence supporting MDT-based interventions and the use of buprenorphine/naloxone for managing both cancer pain and OUD represents a significant advancement in clinical care [13, 29, 33]. The expert consensus supporting buprenorphine/naloxone as an appropriate first-line treatment for cancer patients with established OUD challenges traditional approaches that relied exclusively on full opioid agonists [35].
The successful implementation of specialized interdisciplinary clinics designed specifically for cancer patients with OUD demonstrates the feasibility and effectiveness of targeted interventions [24]. These programs integrate oncology, pain management, addiction medicine, and psychosocial support to address the complex needs of this vulnerable population.
The evidence presented necessitates fundamental changes in oncology pain management practices. Universal screening for OUD risk factors should become standard practice, particularly for patients requiring long-term opioid therapy [36]. Early identification of high-risk patients enables the implementation of enhanced monitoring protocols, alternative analgesic strategies, and prophylactic interventions.
Healthcare systems must develop infrastructure to support cancer patients with concurrent OUD, including access to medication-assisted treatment and specialized clinical expertise [36]. The integration of addiction medicine specialists into oncology teams represents a critical step toward comprehensive care delivery.
Despite the substantial evidence base, several limitations warrant consideration. Definitional inconsistencies across studies regarding OUD criteria may contribute to the wide prevalence range observed [30]. Standardization of outcome measures and diagnostic criteria would enhance the comparability of future research.
The underrepresentation of certain cancer populations and geographic regions limits the generalizability of findings. Future research should prioritize diverse patient populations and international perspectives to ensure global applicability of evidence-based interventions.
Universal risk assessment: implementation of validated screening tools for all cancer patients prescribed opioids, with particular attention to identified risk factors [22, 40].
Multidisciplinary care models: development of integrated teams including oncology, pain management, addiction medicine, and psychosocial support specialists [11, 24, 32].
Evidence-based pharmacotherapy: utilization of buprenorphine/naloxone as first-line treatment for cancer patients with established OUD, with management by oncology teams rather than external addiction programs [33].
Enhanced monitoring protocols: implementation of structured monitoring systems for high-risk patients, including regular reassessment and dose optimization strategies [37, 40].
Provider education: comprehensive training programs for oncology providers regarding OUD recognition, prevention, and management strategies [33].
OUD among cancer patients is no longer a theoretical concern but a real and measurable issue that directly affects patient safety, quality of life, and cancer outcomes. The pooled prevalence ranging from 6% to 50%, with nearly one in four patients at risk, illustrates the scale of this growing issue and underscores the need for systematic prevention efforts. This review highlights that a significant proportion of individuals receiving opioid therapy develop risky use behaviors within a short period, emphasizing the urgent need for vigilance, structured monitoring, and early preventive action. The growing recognition of OUD within oncology challenges the traditional assumption that cancer patients are somehow protected from addiction and calls for a more proactive, evidence-based response.
Effective cancer pain control and opioid safety are not mutually exclusive goals—they can and should coexist through thoughtful, patient-centered care. Routine risk assessment, open communication, and early involvement of MDTs can dramatically reduce misuse while preserving dignity and comfort for those in pain. Integrating addiction medicine expertise into oncology practice, expanding access to buprenorphine-based therapies, and fostering a culture of empathy rather than stigma are essential steps toward safer and more compassionate care. Implementing universal risk screening and standardized opioid stewardship protocols across oncology and palliative settings could significantly improve both safety and quality of life for patients.
Future research must focus on refining predictive tools, exploring personalized and gene-targeted pain management strategies, and understanding the long-term impact of opioid exposure on cancer progression and survivorship. Collaboration between oncologists, palliative care specialists, and addiction experts will be crucial in shaping new standards of care. Ultimately, addressing OUD in cancer care requires the same compassion, rigor, and innovation that define modern oncology itself, ensuring that every patient receives both effective pain relief and protection from harm. By uniting compassion with scientific discipline, oncologic healthcare workers can lead the way in redefining safe and humane pain care for patients worldwide.
JBI: Joanna Briggs Institute
MDT: multidisciplinary team
MME: morphine milligram equivalent
NMOU: non-medical opioid use
OUD: opioid use disorder
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
ScR: scoping review
UDSs: urine drug screens
The supplementary tables for this article are available at: https://www.explorationpub.com/uploads/Article/file/1001388_sup_1.pdf.
YI and OA: Conceptualization, Methodology, Writing—original draft, Writing—review & editing, Data curation, Formal analysis. SAG, OS, and KJ: Writing—review & editing. MS and LS: Conceptualization, Methodology, Writing—review & editing. PT: Conceptualization, Methodology, Writing—review & editing, Data curation, Formal analysis, Supervision. All authors read and approved the submitted version.
Patricia Tai, who is the Editorial Board Member and Guest Editor of Exploration of Medicine, and Omar Alqaisi, who is the Guest Editor of Exploration of Medicine, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
All relevant data are contained within the manuscript, as no new data are generated, and all selected studies are in the references.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1057
Download: 27
Times Cited: 0
Sara Diogo Gonçalves
Omar Alqaisi ... Aliaa Ezz Eldin Abd Elmoaty
