Affiliation:
1Department of Medicine, Trinity Medical Sciences University School of Medicine, Kingstown VC01000, Saint Vincent and the Grenadines
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
3Department of Medicine, Armed Forces Postgraduate Medical Institute, Islamabad 44040, Pakistan
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
2Department of Medicine, Ajman University, College of Medicine, Ajman 6263, United Arab Emirates
Affiliation:
6Department of Family Medicine, University of Illinois College of Medicine Peoria, Bloomington, IL 61701, USA
Email: ayoolaawosika@yahoo.com
ORCID: https://orcid.org/0000-0002-3506-6734
Explor Med. 2026;7:1001389 DOI: https://doi.org/10.37349/emed.2026.1001389
Received: November 07, 2025 Accepted: February 05, 2026 Published: March 16, 2026
Academic Editor: Attila Nemes, University of Szeged, Hungary
Background: Transcatheter mitral valve intervention, including repair (e.g., edge-to-edge repair) and replacement, is now a cornerstone of treatment for severe mitral regurgitation (MR) in high-risk patients, providing a less invasive alternative to surgical treatment. The purpose of this systematic review is to assess the efficacy and safety of different antithrombotic strategies to prevent thromboembolic events and bleeding complications after transcatheter mitral valve repair (TMVR).
Methods: According to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a systematic literature search on PubMed, Embase, Web of Science, Scopus, and Cochrane Library was conducted from January 2015 to January 2025. Eligible studies included adult patients who underwent transcatheter mitral valve intervention (including repair and replacement procedures) and compared different antithrombotic regimens, including direct oral anticoagulants (DOACs), vitamin K antagonists (VKAs), and antiplatelet therapies. Two reviewers independently extracted data and assessed quality. The studies were too heterogeneous, so a narrative synthesis was performed.
Results: Fifteen studies involving 20,956 patients were included. DOACs were associated with a lower risk of major bleeding compared to VKAs [hazard ratio (HR): 0.21, p = 0.02, in one large study], with similar rates of stroke. Mortality was lower with DOACs in several analyses (e.g., HR: 0.67). Triple therapy and dual antiplatelet therapy (DAPT) were associated with increased bleeding risk without providing additional thromboembolic protection.
Discussion: DOACs have a safer profile in post-TMVR patients, with reduced bleeding risk and lower mortality compared with VKAs. Triple therapy and DAPT should only be used in high-risk patients with specific indications due to their greater risk of bleeding. Optimizing outcomes requires a tailored approach to antithrombotic therapy, considering patient factors and procedural considerations. Definitive standards may still demand further investigation, such as multicenter randomized controlled trials evaluating antithrombotic treatments after TMVR.
Transcatheter mitral valve repair (TMVR) has emerged as a transformational therapy for severe mitral regurgitation (MR) in high-risk patients, giving a minimally invasive alternative to traditional surgical procedures [1, 2]. Nonetheless, the enduring efficacy of TMVR is jeopardized by the lack of defined antithrombotic procedures following treatment, posing a considerable barrier to post-interventional management.
The MitraClip device, an innovative TMVR technique, has shown favorable clinical results while reducing invasiveness [3, 4]. Despite its procedural benefits, TMVR has unresolved issues in post-interventional care, particularly thromboprophylaxis. The implantation procedure may injure arterial linings and modify blood flow dynamics, potentially resulting in thrombus development [5]. This risk is exacerbated by the significant occurrence of atrial fibrillation (AF) in patients with TMVR, which naturally elevates thromboembolic risk [6].
Antithrombotic therapy following TMVR seeks to avert stroke and systemic embolism while mitigating the considerable risk of hemorrhagic consequences. Therapy for thromboembolic hazards following procedures often includes direct oral anticoagulants (DOACs), vitamin K antagonists (VKAs), and antiplatelet agents [7, 8]. The ideal antithrombotic regimen is still debated, given the intricate risk profile of TMVR patients, who frequently exhibit advanced age, diminished bone mineral density, and comorbidities, such as AF [6, 9].
Contradictory results mark existing research on post-TMVR antithrombotic care. OAC effectively mitigates thromboembolism in AF patients [10]. However, its hemorrhagic hazards are alarming in geriatric populations [10]. Dual antiplatelet therapy (DAPT) may mitigate device thrombosis but presents an elevated risk of hemorrhage relative to single antiplatelet regimens [11]. These contradictory results highlight the need for rigorous research to provide definite clinical guidelines.
The long-term effectiveness of TMVR is dependent on successful stroke prevention and hemorrhage management efforts. These problems can markedly affect the quality-of-life enhancements that TMVR seeks to deliver to patients with severe MR [12]. Implementing suitable antithrombotic measures is essential to enhance the safety and effectiveness of TMVR.
This systematic review investigates the clinical effectiveness and safety of various anticoagulation and antiplatelet regimens for patients undergoing transcatheter mitral valve intervention. Given the evolving landscape, we include evidence related to both transcatheter edge-to-edge repair (TEER) and transcatheter mitral valve replacement to provide a comprehensive overview of antithrombotic management in this heterogeneous patient population. These findings will contribute to developing evidence-based guidelines, ultimately enhancing patient care and outcomes in this rapidly evolving field of interventional cardiology.
This systematic review followed the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [13]. The study protocol was registered in the International Prospective Register of Systematic Reviews (CRD420251000976). This systematic review addressed the following research question: In adult patients undergoing TMVR, what are the effectiveness and safety of various antithrombotic strategies in preventing thromboembolic events and bleeding complications? We expanded our inclusion to all study designs (randomized trials, registries, and cohort studies) reporting post-TMVR antithrombotic outcomes.
The PICO framework (P—population, I—intervention, C—comparison, and O-outcome) was employed to formulate a focused clinical question. The characteristics of the study are detailed below.
P. Adult patients (≥ 18 years) undergoing transcatheter mitral valve intervention, including edge-to-edge repair (TEER), valve replacement, and valve-in-valve or valve-in-ring procedures.
I. Any post-procedural antithrombotic regimen [e.g., single antiplatelet therapy (SAPT), DAPT, OAC with VKAs or DOACs, or combinations like triple therapy].
C. This review compared different antithrombotic strategies used post-TMVR. This involved comparisons between different types of anticoagulation (e.g., DOACs vs. VKAs), antiplatelet regimens (e.g., SAPT vs. DAPT), and antithrombotic therapy versus no antithrombotic therapy.
O. Thromboembolic events (stroke, transient Ischemic attack, systemic embolism), major bleeding complications [Bleeding Academic Research Consortium (BARC) ≥ 3 or equivalent], and all-cause mortality.
Due to significant heterogeneity in procedures (repair vs. replacement), devices, and patient populations, data synthesis was planned and performed narratively, with findings discussed separately for TEER and TMVR studies where applicable.
The articles selected for the analysis were written in English, and only human studies were included. Randomized controlled trials (RCTs), cohort studies, case-control studies, and case series of more than five cases were considered to capture a broad spectrum of evidence.
Publications were excluded from the analysis if they did not meet the PICO criteria, were not in English, were not conducted in humans, focused on surgical rather than TMVR techniques, or lacked sufficient detail for data extraction and quality assessment.
To identify relevant studies, the following international databases were searched: PubMed, Embase, Web of Science, Scopus, and Cochrane Library. The search encompassed scientific literature on antithrombotic therapy following TMVR from January 2015 to January 2025. The search strategy involved the use of Medical Subject Headings terms and relevant keywords, including: ‘transcatheter mitral valve repair’, ‘TMVR’, ‘MitraClip’, ‘edge-to-edge repair’, ‘anticoagulation’, ‘antiplatelet’, ‘direct oral anticoagulants’, ‘vitamin K antagonists’, ‘stroke’, ‘thromboembolism’, ‘bleeding complications’, ‘hemorrhage’, and ‘mortality’.
Two independent reviewers independently screened the titles and abstracts of all the identified studies. Studies that did not meet the inclusion criteria were excluded. The full texts of potentially relevant studies were retrieved and assessed according to the inclusion and exclusion criteria. Any disagreements between reviewers were resolved through discussion.
A standardized data extraction form was developed, and the following information was extracted from each study. The study’s characteristics included authors, year of publication, study design, sample size, and intervention summary. Information on the antithrombotic regimen was also gathered, including the type of therapy (such as SAPT, DAPT, or OAC), the specific agent used (e.g., aspirin, clopidogrel, warfarin, or DOACs), dosage, and duration. The outcomes were recorded, including thromboembolic events (such as stroke, transient ischemic attack, and systemic embolism), bleeding complications (both major and minor), mortality, cardiovascular events, and hospitalization for heart failure. Finally, the follow-up duration or length of time the patients were monitored after the TMVR procedure was noted. Two reviewers independently performed data extraction, and any discrepancies were resolved through discussion.
The methodological quality of the included studies was assessed using appropriate tools based on study design. The Cochrane risk of bias tool was used to assess the risk of bias in RCTs. This tool evaluates bias across several domains: selection, performance, detection, attrition, and reporting biases. The Newcastle-Ottawa Scale (NOS) was used to assess the quality of observational studies. NOS evaluates studies based on selection, comparability, and outcome assessment.
Owing to the anticipated heterogeneity of study designs, patient populations, TMVR procedures, and antithrombotic regimens, a quantitative synthesis (meta-analysis) may not be appropriate. Instead, narrative synthesis was performed to summarize and synthesize the findings of the included studies. This involved organizing the data into meaningful categories and identifying key themes and patterns.
A comprehensive literature search was conducted across five major electronic databases: PubMed/MEDLINE, Embase, Cochrane Library, Scopus, and Web of Science, and 1,677 records were identified. After removing duplicates, 1,252 records were screened. Reports assessed for eligibility (n = 26). Finally, 15 studies were included in the study. These include various study types, such as prospective cohort studies, retrospective studies, RCTs, and registry-based observational studies. The PRISMA flow diagram (Figure 1) illustrates the study selection process.
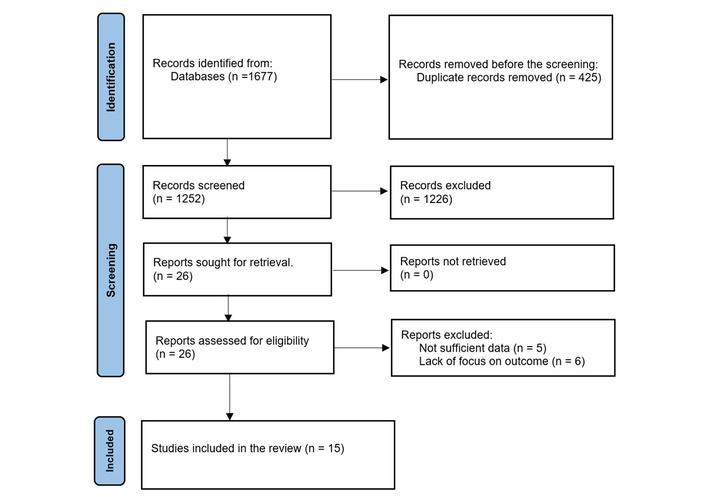
PRISMA-2020 flow diagram. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Adapted from [13]. © The Author(s) 2021. Licensed under a Creative Commons Attribution (CC BY 4.0).
The total number of patients across all studies was 20,956, with individual study sample sizes ranging from 11 to 11,267 participants in the most extensive study by Mentias et al. [14], which analyzed a Medicare database. The patient population primarily consisted of individuals with severe MR or specific valve conditions. The mean age of participants was generally in their 70s and 80s, with the sex distribution varying across studies. The included studies evaluated a range of transcatheter mitral interventions, including TEER devices (e.g., MitraClip) and transcatheter mitral valve replacement devices (e.g., Tendyne and Tiara systems). The follow-up period ranged from 30 days to 4 years, depending on the study. A detailed overview of the study’s characteristics is provided in Table 1.
Study characteristics with population and intervention summary.
| Study | Study design | Sample size | Mean age | Follow-up | Population summary | Procedure | Intervention summary |
|---|---|---|---|---|---|---|---|
| El Bèze et al. [15] | Prospective cohort study | 156 | 65 ± 18.5 years | Median 4.7 months (Q1–Q3) | Adult patients (66% female) undergoing TMVR with SAPIEN prosthesis. | TMVR with SAPIEN | DOACs vs. VKAs. 20.5% DOACs, 79.5% VKAs. |
| Guerrero et al. [16] | Registry-based observational study | 820 | 72.2 ± 10.4 years | 1 year | Adult patients (50.9% female) undergoing TMVR via MViR. | Valve-in-ring | MViR with SAPIEN 3 valve, transseptal access. |
| Giustino et al. [17] | Randomized controlled trial (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients with Functional Mitral Regurgitation) | 614 | Not available | 4 years | Patients with secondary MR due to heart failure. | TEER (MitraClip) | TEER (MitraClip) vs. guideline-directed medical therapy for severe secondary MR. |
| Körber et al. [18] | Single-center cohort study | 347 | 76 years | Not specified | Adult patients (41.5% female) undergoing percutaneous mitral valve repair with MitraClip. | TEER (MitraClip) | MitraClip implantation for MR, various antithrombotic regimens. |
| Paukovitsch et al. [19] | Single-center registry-based cohort | 586 | 76.8 years | Median 577 days | Adult patients (40.1% female) undergoing TMVR with MitraClip. | TEER (MitraClip) | TMVR with MitraClip, echocardiographic, and fluoroscopic guidance. |
| Nathan et al. [20] | Retrospective cohort study (claims-based) | 1,997 | 71 (valve replacement), 81 (valve replacement) | 90 days post-discharge | Patients with AF undergoing MVR (surgical or transcatheter). | Mixed (surgical & transcatheter repair) | Evaluated anticoagulation (VKAs, DOACs, or no therapy) post-MVR. |
| Li et al. [21] | Single-center observational study | 33 | 70.1 years | Median 28 months, max 47 months | Patients undergoing ViV-TMVR due to bioprosthetic valve failure. | ViV-TMVR | ViV-TMVR with J-Valve, Warfarin anticoagulation. |
| Geis et al. [22] | Retrospective cohort study | 86 | 66.5 years | Mean 424 ± 287 days | Patients with severe MR undergoing TEER with MitraClip. | TEER (MitraClip) | MitraClip implantation for severe MR-analyzed ventricular arrhythmias and implantable cardioverter defibrillator therapies. |
| Malaisrie et al. [23] | Prospective multicenter study (PARTNER 3 MViV) | 50 | 70.1 ± 9.7 years | 1 year | Patients with failing mitral bioprosthetic valves undergoing MViV with SAPIEN 3. | ViV-TMVR | MViV with SAPIEN 3 via transseptal approach. |
| Mentias et al. [14] | Retrospective cohort study (Medicare database) | 11,267 | 77.5 years (DOAC), 75.0 years (warfarin) | Median 468 days (MVR) | Patients with AF undergoing MVR (surgical or transcatheter). | Mixed (surgical & transcatheter repair) | DOACs vs. Warfarin in AF patients post-TMVR. |
| Ludwig et al. [24] | Single-center observational study | 11 | 77 years (73–84) | 6 months | Patients with severe symptomatic MR undergoing TMVR using Tendyne™ or Tiara™ devices. | TMVR (Tendyne or Tiara) | TMVR with Tendyne™ or Tiara™ via transapical access. |
| Hohmann et al. [25] | Retrospective observational study | 1,342 | 76 ± 9 years | 30 days to 6 months | Patients undergoing TMVR with MitraClip. 62% had AF, 91% hypertensive, 78% congestive heart failure. | TEER (MitraClip) | Post-TMVR anticoagulation patterns and thromboembolic risk. |
| Tichelbäcker et al. [26] | Single-center retrospective study | 453 | 79.1 years | Up to 6 months | Patients undergoing MitraClip for severe MR. 60.5% had AF, 71.7% hypertensive. | TEER (MitraClip) | Left ventricular thrombus formation post-TMVR and anticoagulation association. |
| Schipper et al. [27] | Single-center retrospective observational cohort | 206 | 78.6 years | Median 833 days | High-risk patients with severe MR and AF, undergoing mitral valve TEER. | TEER (MitraClip) | Post-procedure oral anticoagulation with either DOAC or VKA. |
| Alaour et al. [28] | Prospective multicenter registry analysis | 2,908 | 82 years | 30 days to 5 years | Consecutive high-risk patients with severe aortic stenosis and AF undergoing TAVR. | TAVR (comparative context) | Post-procedure oral anticoagulation with either DOAC or VKA. |
AF: atrial fibrillation; DOACs: direct oral anticoagulants; MR: mitral regurgitation; MViR: mitral valve-in-ring; MViV: mitral valve-in-valve; MVR: mitral valve repair; TAVR: transcatheter aortic valve replacement; TEER: transcatheter edge-to-edge repair; TMVR: transcatheter mitral valve repair; ViV: valve-in-valve; VKAs: vitamin K antagonists.
Across the studies, different antithrombotic therapy regimens were used, including DOACs, VKAs, and DAPT, etc. Several studies compared these therapies regarding bleeding risk, stroke, and mortality, providing insightful findings.
Four studies [14–17] compared DOACs with VKAs and found that DOACs were associated with a significantly lower bleeding risk [hazard ratio (HR): 0.21, p = 0.02] than VKAs, with similar stroke and mortality outcomes across both groups. Specifically, Mentias et al. [14] demonstrated that DOACs led to reduced mortality (HR: 0.67) and lower major bleeding rates compared to VKAs. Similarly, Guerrero et al. [16] reported a higher bleeding risk with VKAs but no significant differences in stroke or mortality outcomes, confirming the safety benefits of DOACs.
A recent single-center observational study by Schipper et al. [27] found no significant difference between DOAC and VKA for the composite of ischemic and bleeding events. Notably, overall mortality was higher in the VKA group (HR 2.56, p = 0.002) [27], suggesting a possible advantage of DOACs. Schipper et al. [27] conclude that DOACs may be a safe alternative but emphasize that randomized trials are needed.
In a similar analysis of transcatheter valve patients, Alaour et al. [28] examined DOAC vs. VKA after transcatheter aortic valve replacement (TAVR) in a large registry (n = 2,908) and found no difference in net clinical benefit or bleeding at 1–5 years. Consistent with Schipper et al. [27], they observed higher long-term mortality with VKAs (HR = 1.25 at 5 years). These findings across transcatheter valve cohorts suggest DOACs are at least comparable to VKAs, although all data remain observational.
In addition, Paukovitsch et al. [19] and Guerrero et al. [16] explored the efficacy of triple therapy (OAC + DAPT) in patients post-TMVR. Although triple therapy resulted in a higher risk of bleeding (24%), it did not provide additional protection against thromboembolic events or reduce mortality. This suggests that triple therapy carries substantial bleeding risks without clear benefits compared to DOAC/DAPT alone.
Several studies [19–22] examined DAPT and found it associated with a higher bleeding rate than SAPT. However, DAPT did not offer any additional benefit for stroke prevention, highlighting the need for careful consideration of bleeding risks when prescribing DAPT. For a detailed comparison of antithrombotic therapies, see Table 2.
Antithrombotic therapy comparison among the included studies.
| Study | Antithrombotic therapy | Antithrombotic regimen | Dosage | Duration of therapy | Comparator & impact on outcomes (bleeding, stroke, mortality) |
|---|---|---|---|---|---|
| El Bèze et al. [15] | DOACs vs. VKAs | DOACs (apixaban, rivaroxaban, dabigatran) | ≥ 3 months, stopped after imaging | Post-TMVR | DOACs are associated with a 79% reduction in bleeding risk (9% vs. 40% with VKAs) and similar stroke/mortality outcomes. |
| Guerrero et al. [16] | VKAs, DOACs, antiplatelet therapy | VKAs (INR 2–3), DOACs, antiplatelet | Long-term therapy | - | Higher bleeding risk with VKAs, no significant difference in stroke/mortality. DOACs are safer with lower bleeding risk. |
| Giustino et al. [17] | DAPT (aspirin + clopidogrel) | Aspirin: 75–100 mg/day, clopidogrel: 75 mg/day | 6 months (aspirin), 1 month (clopidogrel) | Post-TEER | Higher bleeding rate with DAPT vs. SAPT, no added thromboembolic benefit. |
| Körber et al. [18] | SAPT, DAPT, VKAs, DOACs, combined therapy | SAPT (aspirin), DAPT, VKAs, DOACs | SAPT: 75–100 mg/day | Long-term | DAPT is associated with a higher bleeding risk (HR: 3.52) and no additional stroke prevention benefit. |
| Paukovitsch et al. [19] | Triple therapy (OAC + DAPT) | OAC + DAPT | Not specified | Post-TEER | Triple therapy increased bleeding risk (24%), with no additional thromboembolic protection or mortality benefit. |
| Nathan et al. [20] | VKAs, DOACs, no therapy | VKAs (INR 2–3), DOACs | Long-term | - | DOACs are associated with lower bleeding risk, similar stroke prevention, and reduced mortality compared to VKAs. |
| Li et al. [21] | VKA (warfarin) | Warfarin (INR 2.5) | 3–6 months | Post-valve-in-valve-TMVR | Stroke: 6.1%, mortality: 6.1%. No major bleeding data was reported. |
| Geis et al. [22] | DAPT (aspirin + clopidogrel) | Aspirin + clopidogrel | 3–6 months | Post-TEER | Reduced ventricular arrhythmias, with no significant impact on stroke or bleeding. |
| Malaisrie et al. [23] | Warfarin + DAPT | Warfarin (INR ≥ 2) + aspirin/clopidogrel | 6 months | Post-mitral valve-in-valve | 6% valve thrombosis resolved with anticoagulation, and no stroke or mortality was reported. |
| Mentias et al. [14] | DOACs vs. warfarin | DOACs (apixaban, rivaroxaban, dabigatran) | Standard dosing | Continuous | DOACs are associated with lower mortality (HR: 0.67) and major bleeding (HR: 0.79) compared to warfarin. |
| Ludwig et al. [24] | VKA (warfarin) | Warfarin (INR 2.5–3.5) | Lifelong | Post-TMVR | Major bleeding: 18.2%, no strokes reported. |
| Hohmann et al. [25] | NOACs, VKAs, SAPT, DAPT, triple therapy | NOACs, VKAs, SAPT, DAPT | Variable | Continuous (OAC), ≤ 6 months (SAPT/DAPT) | Higher mortality in SAPT/DAPT patients (HR: 3.84) and NOACs are associated with lower bleeding risk. |
| Tichelbäcker et al. [26] | DOACs, VKAs, SAPT, DAPT | DOACs, VKAs, SAPT, DAPT | Variable | Continuous (OAC), 1–6 months (SAPT/DAPT) | Left ventricular thrombus: 1.1% overall, 4.4% in LVEF < 30%. Thrombus cases despite DOAC therapy. VKAs are associated with higher bleeding risk. |
| Schipper et al. [27] | DOAC vs. VKA | DOAC, VKA | Variable | Continued through follow-up | Bleeding, no significant difference in major bleeding rates between DOAC and VKA groups; stroke, similar rates in both groups; Mortality, higher long-term mortality in the VKA group. |
| Alaour et al. [28] | DOAC vs. VKA | DOAC, VKA | Variable | Assessed at 30 days, 1 year, and 5 years post-transcatheter aortic valve replacement | Bleeding rates of life-threatening or major bleeding were similar with DOACs and VKAs through 5 year follow-up. Stroke, long-term incidence of disabling stroke was lower with VKA (HR 0.64, 95% confidence interval 0.46–0.90 for VKA vs. DOAC). Mortality, all-cause mortality was higher with VKA (VKA vs. DOAC HR 1.28 at 1 year and 1.25 at 5 years). Net clinical composite did not differ between therapies at 30 days, 1 year, or 5 years. |
HRs and outcomes are as reported in the individual primary studies; no pooled meta-analysis was performed. -: no data. DAPT: dual antiplatelet therapy; DOACs: direct oral anticoagulants; HR: hazard ratio; INR: international normalized ratio; LVEF: left ventricular ejection fraction; NOAC: non-vitamin-K antagonist oral anticoagulants; OAC: oral anticoagulant; SAPT: single antiplatelet therapy; TEER: transcatheter edge-to-edge repair; TMVR: transcatheter mitral valve repair; VKAs: vitamin K antagonists.
Four studies [15, 18–20] reported bleeding complications, revealing significant differences based on the anticoagulation regimen. Patients receiving triple therapy or DAPT had the highest rates of bleeding, whereas those treated with DOACs had significantly lower rates of major bleeding.
Two studies [14, 15] confirmed that DOACs were associated with a lower incidence of major bleeding events than VKAs without increasing the risk of stroke or mortality. Specifically, El Bèze et al. [15] reported that DOACs reduced bleeding risk by 79% (9% vs. 40% with VKAs), while Mentias et al. [14] found that DOACs were associated with a lower risk of major bleeding (HR: 0.79) compared to VKAs. Additionally, Paukovitsch et al. [19] found that combining DOACs with DAPT reduced bleeding complications without compromising thromboembolic protection, making it a safer alternative to triple therapy (Table 2).
Four studies [14, 22, 23, 26] reported mortality outcomes, indicating that DOACs are associated with reduced mortality after TMVR. Mentias et al. [14] and Malaisrie et al. [23] found that patients treated with DOACs had significantly lower mortality rates than those treated with VKAs. Specifically, Mentias et al. [14] reported a HR of 0.67 for mortality with DOACs compared to VKAs, highlighting the superior survival benefits of DOACs in the post-TMVR population.
Interestingly, Tichelbäcker et al. [26] observed a 1.1% incidence of left ventricular thrombus formation even in patients treated with DOACs, underscoring the need for vigilant monitoring of thromboembolic events despite effective anticoagulation therapy. This finding suggests that while DOACs are effective in reducing bleeding and mortality, they may not eliminate the risk of thrombotic events in high-risk patients (Table 2).
Regarding quality of life (QoL), several studies reported significant improvements in patients’ New York Heart Association class and Kansas City Cardiomyopathy Questionnaire scores following TMVR. Two studies [22, 23] demonstrated improvements in functional capacity, particularly in heart failure symptoms, after undergoing TMVR. These findings suggest that TMVR improves mortality and complications and significantly enhances patients’ overall well-being.
Furthermore, Geis et al. [22] reported a reduction in ventricular arrhythmias and a decreased need for implantable cardioverter defibrillator therapies following the procedure, highlighting the potential of TMVR to improve cardiac rhythm control and reduce the arrhythmic burden. These improvements in functional status and QoL are critical for patients with severe MR, as they often experience significant limitations in daily activities due to heart failure symptoms. For more details, see Table 3.
Aggregated primary and secondary outcomes across included studies.
| Study | Primary outcome(s) | Secondary outcome(s) |
|---|---|---|
| El Bèze et al. [15] | Bleeding complications (9% DOAC vs. 40% VKA) | Stroke, mortality, major vascular complications, and length of hospital stay. |
| Guerrero et al. [16] | 1 year mortality (22.4%) | Stroke (1.5%), device thrombosis (1.7%), mitral valve reintervention (9.1%), and major vascular complications (2.3%). |
| Giustino et al. [17] | Incidence of CVE (7.8% at 4 years) | Impact of anticoagulation on CVE risk (HR: 0.24 with anticoagulation, HR: 2.27 without). |
| Körber et al. [18] | MVARC-defined bleeding complications (21.6%) | Major bleeding (7.4%), transfusion requirement (4.9%), and mortality. |
| Paukovitsch et al. [19] | MVARC-defined bleeding complications (13.3%) | Major bleeding (1.7%), transfusion requirement (4.9%), and mortality. |
| Nathan et al. [20] | Trends in anticoagulant use (DOAC use increased from 12.4% in 2014 to 37.2% in 2018) | Stroke prevention, thromboembolic risk, and anticoagulant adherence. |
| Li et al. [21] | Survival and procedural success (97%) | Stroke (6.1%), mortality (6.1%), NYHA class improvement, and quality of life (KCCQ-12). |
| Geis et al. [22] | Reduction in VA and implantable cardioverter defibrillator therapies (VA reduced from 2.24 to 1.26 events/patient/month) | Mortality (21.2% at 1 year), heart failure hospitalization, and NYHA class improvement. |
| Malaisrie et al. [23] | Composite outcome of 1 year mortality and stroke (0% mortality, 0% stroke) | Quality of life (NYHA, KCCQ), valve thrombosis (6%), and valve performance. |
| Mentias et al. [14] | All-cause mortality (DOACs: HR 0.67 vs. warfarin) | Stroke (DOACs: HR 0.72 vs. warfarin), major bleeding (DOACs: HR 0.79 vs. warfarin). |
| Ludwig et al. [24] | Procedural success and mortality (100% procedural success, 6 month mortality: 22.2%) | NYHA class improvement, mitral regurgitation elimination, and major bleeding (18.2%). |
| Hohmann et al. [25] | All-cause mortality (HR: 3.84 for no anticoagulation vs. single antiplatelet therapy) | Major bleeding, ischemic stroke, and thromboembolic events. |
| Tichelbäcker et al. [26] | Left ventricular thrombus formation (1.1% overall, 4.4% in LVEF < 30%) | Stroke, mortality, and the effectiveness of the anticoagulation strategy. |
| Schipper et al. [27] | Composite of ischemic CVEs and major bleeding | Overall survival, no difference in the composite primary endpoint between DOAC and VKA; mortality was significantly lower with DOACs.Composite of ischemic events and major bleeding*. |
| Alaour et al. [28] | Net clinical benefit composite and primary safety composite (life-threatening or major bleeding) | VKAs vs. DOACs showed higher long-term mortality and lower disabling stroke with VKAs, whereas rates of major bleeding were similar.Net clinical benefit composite** and primary safety composite. |
*: Composite endpoint in Schipper et al. [27]: ischemic CVE or major bleeding (Bleeding Academic Research Consortium ≥ 3); **: net clinical benefit in Alaour et al. [28]: composite of all-cause mortality, myocardial infarction, stroke, or life-threatening/major bleeding. CVE: cerebrovascular event; DOAC: direct oral anticoagulant; HR: hazard ratio; KCCQ: Kansas City Cardiomyopathy Questionnaire; MVARC: Mitral Valve Academic Research Consortium; NYHA: New York Heart Association; VA: ventricular arrhythmias; VKA: vitamin K antagonist.
Two studies [17, 25] found that MitraClip implantation resulted in high success rates of MR reduction, with procedural success rates exceeding 95% in most cases. However, Tendyne and Tiara devices showed more challenges in procedural outcomes, including device migration and valve leakage, suggesting that some devices may carry a higher risk of complications, potentially affecting long-term results.
Moreover, Nathan et al. [20] investigated anticoagulation strategies after TMVR and found that VKAs and DOACs were associated with a reduced risk of thromboembolic events. However, the optimal anticoagulation strategy after TMVR remains a topic of ongoing debate. These findings highlight the importance of tailoring anticoagulation therapy to individual patient needs, particularly in high-risk populations (Table 3).
The risk of bias across the 15 studies was assessed using two different tools: the Cochrane risk of bias tool for RCTs and the NOS for other non-randomized studies. This RCT demonstrated a low risk of bias in all key domains, including random sequence generation, allocation concealment, and blinding, making it a high-quality study. The remaining 15 non-randomized studies were assessed using the NOS, where most of the studies scored six stars, indicating moderate quality. These studies provided valuable data on treatment outcomes, complications, and effectiveness but were limited by their observational nature and the absence of control groups, which introduced potential biases due to confounding factors. Overall, an RCT had the lowest risk of bias, while the non-randomized studies, despite their limitations, provided important evidence on the effectiveness and safety of various interventions (Table 4).
Newcastle-Ottawa Scale (NOS) risk of bias assessment.
| Study | Selection (max 4★) | Comparability (max 2★) | Outcome (max 3★) | Total score (max 9★) |
|---|---|---|---|---|
| El Bèze et al. [15] | ★★★★ | ★★ | ★★★ | 9 |
| Guerrero et al. [16] | ★★★★ | ★★ | ★★★ | 9 |
| Giustino et al. [17] | ★★★★ | ★★ | ★★★ | 9 |
| Körber et al. [18] | ★★★ | ★ | ★★ | 6 |
| Paukovitsch et al. [19] | ★★★ | ★ | ★★ | 6 |
| Nathan et al. [20] | ★★★★ | ★★ | ★★★ | 9 |
| Li et al. [21] | ★★ | ★ | ★★ | 5 |
| Geis et al. [22] | ★★★ | ★ | ★★ | 6 |
| Malaisrie et al. [23] | ★★★★ | ★★ | ★★★ | 9 |
| Mentias et al. [14] | ★★★★ | ★★ | ★★★ | 9 |
| Ludwig et al. [24] | ★★ | ★ | ★★ | 5 |
| Hohmann et al. [25] | ★★★ | ★★ | ★★★ | 8 |
| Tichelbäcker et al. [26] | ★★★ | ★ | ★★ | 6 |
| Schipper et al. [27] | ★★★ | ★★ | ★★★ | 8 |
| Alaour et al. [28] | ★★★★ | ★★ | ★★★ | 9 |
NOS star rating system for cohort studies. The maximum score is 9. Studies are graded as: low risk of bias (7–9 stars), moderate risk (4–6 stars), high risk of bias (0–3 stars).
This systematic review synthesizes evidence on antithrombotic strategies following transcatheter mitral valve intervention, encompassing both repair (TEER) and replacement procedures. The principal challenge is balancing thromboembolic and bleeding risks across a heterogeneous patient and procedural landscape [27]. Our findings underscore that optimal therapy is not uniform but must account for procedure type, device, and individual patient factors.
The challenges of antithrombotic management following TMVR are compounded by the heterogeneous nature of the patient population and the multitude of TMVR procedures and devices. TMVR is performed in patients with various clinical profiles, including degenerative mitral valve disease, functional MR due to heart failure, and ≤ 50% of surgical candidates [4]. These patients are often elderly and have several comorbidities [6, 9], including AF, chronic kidney disease, and a history of bleeding, making the decision-making process even more challenging. The increasing complexities and comorbidities highlight the need for personalized risk assessments and antithrombotic strategies. Therefore, a comprehensive review of the existing data is required to inform clinical decision-making and optimize outcomes for this high-risk group.
A key finding of this review is the critical distinction between TEER and replacement. TEER with the MitraClip involves native valve tissue with a lower intrinsic thrombotic risk; thus, antithrombotic therapy is often dictated by concomitant indications like AF [14, 18]. In contrast, TMV replacement implants a prosthetic valve, carrying a higher risk of valve thrombosis analogous to surgical bioprostheses or TAVR, typically mandating at least temporary OAC [15, 28]. Similarly, valve-in-valve or valve-in-ring procedures have unique risk profiles influenced by the underlying substrate [16]. Pooling these populations is problematic, and our stratified synthesis highlights the need for procedure-specific guidelines.
Evidence suggests that DOACs also demonstrate a superior safety profile regarding bleeding prevention compared to VKAs. Nonetheless, considering its efficacy against thromboembolic events and safety profile, the best antithrombotic regimen in TMVR patients is still an essential choice.
DOACs have replaced VKAs. They are preferentially chosen because of their ease of use, fewer dietary restrictions, and lower risk of drug interactions, thus facilitating management and improving patient compliance [15, 16]. Using a recent analysis of the National Inpatient Sample national dataset between 2016 and 2020, malignancy did not increase mortality, length of stay, or procedural costs for both TMVR and TEER [29]. However, the number of bleeding events was lower in the DOAC arm, suggesting a possible survival benefit with newer anticoagulants. This is especially important in the older population, who often undergo TMVR procedures, where bleeding complications result in worse outcomes and increased mortality. In addition, the study by Mentias et al. [14] confirmed these findings, demonstrating lower mortality rates in DOACs versus VKAs patients, potentially due to lower bleeding risk. Schipper et al. [27] reported no difference in stroke or bleeding between DOAC and VKA after mitral valve-TEER, but higher mortality with VKA. Likewise, Alaour et al. [28] found equivalent composite outcomes after TAVR under DOAC vs. VKA (with higher long-term mortality on VKAs).
However, a recent meta-analysis by Zorman et al. [29] has indicated that VKAs may be more effective in preventing bioprosthetic mitral valve thrombosis than their safety. VKAs, with their more exhaustive coverage of anticoagulation, are likely to yield better protection against valve thrombosis than DOACs, which inhibit only particular clotting factors. Given these contradictory observations, a more sophisticated view must consider the prospect of both subclinical and clinical thromboses.
The role of antiplatelet therapy, particularly DAPT, in addition to anticoagulation after TMVR, remains a matter of debate. Although DAPT is frequently used to reduce device-related thrombosis risk compared with SAPT, several studies have demonstrated that DAPT is associated with an increased risk of bleeding complications without a significant protective effect against stroke [30, 31].
DAPT was implemented to prevent device-related thrombosis but was consistently associated with increased bleeding. Data from studies such as POPular transcatheter aortic valve implantation (TAVI) [32] and those by Mentias et al. [14] demonstrated that although DAPT has the potential to reduce thrombosis in the device profile, its risk of bleeding outweighs any theoretical benefit that may be observed compared with single-agent therapy (acetylsalicylic acid alone). The POPular TAVI trial [33] demonstrated that DAPT following TAVR did not reduce stroke outcomes but was associated with a higher rate of bleeding than SAPT. Based on these results, the current recommendation for the management of post-TMVR patients leans towards SAPT as the standard regimen, and only patients with additional cardiovascular risk factors (e.g., recent myocardial infarction, stent implantation) should receive DAPT.
In post-TMVR patients with high thromboembolic event risk, such as those with AF, triple therapy (OAC plus DAPT) is occasionally employed. However, multiple studies, such as those conducted by Paukovitsch et al. [19] and Zorman et al. [29], have cast some doubt on the high bleeding risk associated with triple therapy with no concomitant reduction in thromboembolic events. Given the relative lack of benefits and increased bleeding risk, this approach should be limited to the highest-risk patients and for the minimum time possible [30].
In the post-TMVR care landscape, subclinical valve thrombosis, presenting as leaflet thickening and reduced leaflet motion on imaging, has surfaced as an important consideration. Even if asymptomatic, it may cause device dysfunction and systemic embolization, affecting long-term outcomes [16, 29].
Diagnosing and treating subclinical valve thrombosis are challenging; otherwise, they can lead to clinical events. Recent studies have shown that this augments transvalvular gradients and accelerates valve degeneration, requiring repeated interventions [34]. The early detection and treatment of subclinical thrombosis may enhance long-term valve durability and reduce adverse events [35].
Subclinical thrombosis detection often relies on the use of advanced imaging modalities. Transthoracic echocardiography (TTE) is the standard initial imaging modality; however, it has limited sensitivity for detecting subtle leaflet abnormalities. High-resolution transesophageal echocardiography (TEE) combined with four-dimensional computed tomography (4D-CT) has also been integrated into pathways for early leaflet thickening and decreased leaflet mobility [36]. For example, four-dimensional CT provides enhanced spatial resolution and the capability to quantify leaflet motion to enhance the detection of subclinical thrombosis. Specifically, four-dimensional CT allows evaluating various structural features to improve device assessment.
The antithrombotic regimen chosen significantly influenced both stroke and mortality outcomes in patients with TMVR. Stroke and mortality rates differ among anticoagulation strategies, and this quality of evidence underlines the necessity of refining treatment goals. Mentias et al. [14]. demonstrated that patients on DOACs experienced significantly lower mortality rates than their VKA counterparts, suggesting a potential survival advantage with newer anticoagulants. This can be ascribed to the lower risk of bleeding associated with DOACs, as bleeding events have been shown to predict all-cause mortality in this cohort strongly.
However, DOACs may increase asymptomatic thromboembolism, which could offset some of their advantages in stroke prevention. Tomšič et al. [37] noted the importance of weighing the thromboembolic prevention benefits of DOACs against the subclinical thrombosis risk. Patient selection and monitoring are critical for optimizing the outcomes, highlighting the inherent limitations of this study.
The complex balance of thrombotic and bleeding risks in patients undergoing TMVR makes it almost certain that a one-size-fits-all approach to antithrombotic therapy will not be optimal. Clinicians should adopt a tailored approach that addresses patient-specific traits, procedural considerations, and device-specific properties.
Careful pretreatment risk stratification and regimen tailoring to the individual patient profile are crucial for the successful management of antithrombotic therapy. This involves assessing patient-specific risk factors, such as age, comorbidities (e.g., AF, chronic kidney disease, and previous bleeding), frailty, and concomitant medications. Additionally, procedural considerations should be considered, such as the type of TMVR procedure performed (e.g., valve-in-valve, valve-in-ring, and valve-in-native annulus), access site, and procedural success [38–40].
Equally important are device-specific characteristics, including valve design, material, and implantation techniques. These factors influence the risk of thromboembolic events and bleeding; therefore, the choice of antithrombotic therapy must be personalized based on a comprehensive assessment of these variables.
This approach includes risk assessment tools, such as the HAS-BLED score to assess bleeding risk and the CHA2DS2-VASc score to assess stroke risk, which can assist in rational decision-making [41, 42]. These tools can help assess each patient’s risk and guide the most appropriate antithrombotic strategy choice. Nonetheless, it is important to note that these scores have not yet been validated in the TMVR population. They should be used with clinical judgment and considering individual patient-specific variables.
Despite the growing body of literature, there are multiple unanswered questions regarding antithrombotic therapy following TMVR. The rarity of substantial RCTs comparing various antithrombotic strategies has made it difficult for many clinical providers to base their decisions on high-level evidence. Additionally, current guidelines are often not tailored to patients undergoing TMVR, resulting in diverse practices and possibly inadequate outcomes [7].
This review has several limitations. First, the studies included are highly heterogeneous, encompassing both transcatheter repair and replacement procedures with fundamentally different thrombotic risks. Although we have narratively synthesized findings by procedure type where possible, the inability to perform a quantitative meta-analysis due to this heterogeneity limits the strength of pooled conclusions. Second, all but one study was observational, susceptible to unmeasured confounding and selection bias. The reported HRs are derived from individual studies, not from a pooled meta-analysis. Third, definitions for bleeding and thromboembolic outcomes varied (e.g., BARC, Mitral Valve Academic Research Consortium).
The ideal antithrombotic regimen after TMVR remains an active area of investigation. Although DOACs have a better safety profile with a lower risk of bleeding and greater convenience for the patient, VKAs remain the drug of choice for thromboembolic prevention in high-risk populations. The roles of DAPT and triple therapy remain debatable, with data indicating that these regimens should be reserved for carefully selected patients. Subclinical valve thrombosis, the most recent complication of TMVR, requires attention and imaging after device implantation. This highlights the principle of a personalized approach to antithrombotic management that focuses on the particularities of individual patient characteristics, procedural factors, and device properties. Filling these gaps will allow clinicians to make informed decisions and improve the safety and effectiveness of TMVR. It can also be useful for highlighting the emerging biomarker image modalities, such as transesophageal echocardiography for subclinical thrombosis.
AF: atrial fibrillation
BARC: Bleeding Academic Research Consortium
DAPT: dual antiplatelet therapy
DOACs: direct oral anticoagulants
HR: hazard ratio
MR: mitral regurgitation
NOS: Newcastle-Ottawa Scale
OAC: oral anticoagulant
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
QoL: quality of life
RCTs: randomized controlled trials
SAPT: single antiplatelet therapy
TAVI: transcatheter aortic valve implantation
TAVR: transcatheter aortic valve replacement
TEER: transcatheter edge-to-edge repair
TMVR: transcatheter mitral valve repair
VKAs: vitamin K antagonists
TP: Conceptualization, Data curation, Formal analysis, Writing—original draft. MF: Conceptualization, Data curation, Formal analysis, Writing—original draft. AS: Data curation, Formal analysis, Writing—original draft. AD: Data curation, Methodology, Formal analysis, Writing—original draft. SSK: Data curation, Methodology, Formal analysis, Writing—original draft. DNH: Methodology, Formal analysis, Writing—original draft. TJ: Methodology, Formal analysis, Writing—original draft. MD: Conceptualization, Data curation, Methodology, Formal analysis, Writing—original draft. SAS: Data curation, Formal analysis, Writing—original draft. KYA: Data curation, Formal analysis, Writing—original draft. A Alkassar: Methodology, Formal analysis, Writing—original draft. SA: Data curation, Formal analysis, Writing—original draft. NAM: Methodology, Formal analysis, Writing—original draft. A Awosika: Supervision, Conceptualization, Formal analysis, Writing—original draft. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
All relevant data is contained within the manuscript.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 874
Download: 20
Times Cited: 0
