Affiliation:
1Department of Life Science, Health and Health Professional, Link Campus University, 00165 Roma, Italy
2Department of Biomedical, Surgical and Dental Sciences, Milan University, 20122 Milan, Italy
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
†These authors share the first authorship.
ORCID: https://orcid.org/0000-0002-5947-8987
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
†These authors share the first authorship.
ORCID: https://orcid.org/0000-0002-6366-1039
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
ORCID: https://orcid.org/0009-0007-8701-3734
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
ORCID: https://orcid.org/0009-0000-9426-9454
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
ORCID: https://orcid.org/0009-0008-7035-4548
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
ORCID: https://orcid.org/0009-0000-2382-3016
Affiliation:
4Department of Experimental Medicine, University of Salento, 73100 Lecce, Italy
ORCID: https://orcid.org/0000-0002-3288-490X
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
Email: francesco.inchingolo@uniba.it
ORCID: https://orcid.org/0000-0003-3797-5883
Affiliation:
5Department of General Surgery and Surgical-Medical Specialties, School of Dentistry, University of Catania, 95124 Catania, Italy
Email: gaetano.isola@unict.it
ORCID: https://orcid.org/0000-0003-4267-6992
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
ORCID: https://orcid.org/0000-0002-0521-1363
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
#These authors share the last authorship.
ORCID: https://orcid.org/0000-0001-9163-2350
Affiliation:
2Department of Biomedical, Surgical and Dental Sciences, Milan University, 20122 Milan, Italy
3Department of Interdisciplinary Medicine, University of Bari Aldo Moro, 70121 Bari, Italy
#These authors share the last authorship.
ORCID: https://orcid.org/0000-0003-0104-6337
Explor Med. 2026;7:1001387 DOI: https://doi.org/10.37349/emed.2026.1001387
Received: November 01, 2025 Accepted: January 27, 2026 Published: March 08, 2026
Academic Editor: Luca Testarelli, Sapienza University of Rome, Italy
Background: The increasing use of electronic cigarettes and heated tobacco products has raised concerns regarding their potential effects on periodontal health. The objective of this systematic review was to assess the impact of these alternative nicotine delivery systems on periodontal inflammation, oral microbiota, and outcomes of non-surgical periodontal therapy.
Methods: A comprehensive literature search was conducted in PubMed, Web of Science, and Scopus, in accordance with PRISMA guidelines.
Results: The findings indicate that although electronic cigarettes and heated tobacco products may reduce exposure to certain harmful constituents compared with conventional cigarettes, they are not free of adverse effects. Their use has been associated with increased levels of pro-inflammatory cytokines, reduced anti-inflammatory mediators, delayed periodontal healing, and alterations in oral microbiota composition, which may contribute to the progression of periodontal disease. These biological changes may negatively influence the response to non-surgical periodontal therapy.
Discussion: From a clinical perspective, individuals using electronic cigarettes or heated tobacco products tend to exhibit less favorable treatment outcomes, often comparable to those observed in conventional cigarette smokers. Despite being promoted as harm-reduction alternatives, current evidence suggests that electronic cigarettes and heated tobacco products still pose significant risks to periodontal tissues. Overall, the available biological and clinical evidence supports the need for further investigation into the underlying mechanisms of tissue damage and reinforces the importance of promoting complete smoking cessation, including avoidance of electronic cigarettes and heated tobacco products, to preserve long-term oral health.
Despite the abundance of information on the risks associated with traditional cigarette smoking, electronic cigarettes, or “E-Cs”, are considered a safer alternative [1, 2].
E-Cs are becoming more and more popular as a smoking cessation tool [3, 4]. Smoking tobacco is a significant risk factor for oral health and for a number of dental disorders, including periodontitis.
Teenagers, young adults, and adults particularly enjoy E-Cs and heated tobacco products (HTPs), and their use has increased dramatically [5, 6]. Additionally, it has been noted that vaping may contribute to a return to traditional smoking.
But it’s important to know how E-Cs could affect the user’s health, particularly in the oral cavity, where they can have a big impact on your teeth and gums [7, 8]. An important measure of general health, oral health reveals the state of the microbiota, the strength of the soft and hard tissues, and the existence of inflammatory conditions such as periodontitis.
Numerous harmful substances, such as carbonyl compounds, volatile organic compounds, carcinogenic nitrosamines, and heavy metals, were found in the aerosol of different E-Cs [9–11]. The levels of these pollutants were 9–450 times lower than those found in cigarette smoke. According to recent research, superheating glycol-based e-liquid solvents, such as propylene glycol and glycerol, which is possible with more recent, higher voltage “tank” devices, increases the production of carbonyl chemicals that may cause cancer [12–14].
Exposure to the major constituents of e-liquids, such as nicotine, propylene glycol, and vegetable glycerin, can cause microbial dysbiosis, which is a shift in the oral bacterial flora’s equilibrium that favours harmful strains over helpful ones [15, 16]. Nitrosamines, aldehydes, metals, and volatile organic compounds in E-C aerosol can change the oral microbiome and cause dysbiosis, which can lead to the development of periodontal disorders [17, 18].
E-C and heated tobacco devices may release or create other harmful substances, such as carcinogens, which are not commonly found in e-liquids. These chemicals have been found in several electronic nicotine delivery system (ENDS) aerosols at low concentrations [19–21].
Some of these toxicants are thought to be formed during the vaporisation process, but they can also come from heated structural elements such as the heating coil, wick, and metallic parts, when air is drawn through an ENDS device [20–22].
Recently, measurements of specific particulate matter, heavy metals, and hazardous carbonyl compounds have also been conducted in aerosols generated by ENDS, including E-Cs [23–25].
Together with the chemicals found in the aerosol, the changes in the microbiota might cause inflammatory processes that impair the condition of the gingival and periodontal tissues, increasing the risk of periodontal disorders [26, 27].
Certain ingredients, like nicotine and flavourings, may also increase the production of proinflammatory cytokines like TNF-α and interleukin-6 (IL-6), making the mouth more susceptible to harm. People may be more susceptible to periodontal diseases and other oral pathologies as a result of this microbiota change, which has been linked to increased gingival inflammation and a weakened local immune response [28, 29].
For instance, the cytotoxic effects of aerosols derived from e-liquids on human gingival fibroblasts, including apoptosis induction, may contribute to tissue damage and compromised oral health. Additionally, E-C use has been shown to enrich periodontal pathogens, further promoting dysbiosis and inflammatory responses in the oral cavity [30, 31].
Changes in antibacterial properties of saliva due to exposure to E-Cs can also impair the oral immune defense, creating an environment conducive to bacterial overgrowth. Moreover, longitudinal studies have identified specific biomarkers of tissue degradation and disease progression in E-C users, highlighting their susceptibility to periodontitis [32].
Lastly, evidence from a systematic review [33] has reinforced the association between E-C smoking and compromised periodontal health, including increased risks of inflammation and tissue damage.
The aerosol from E-Cs, which contains harmful chemicals and nanoparticles, can change cellular processes and encourage the development of bacterial plaque, creating an adverse oral environment.
In a similar vein, aerosols derived from HTPs pose certain dangers even though they create less harmful compounds than regular smoking [33].
In addition to heavy metal nanoparticles, which have recently been demonstrated to be present in E-C aerosols at comparable amounts per puff as traditional cigarette smoke, which contains about 1015 free radicals/puff. By increasing ROS generation, heavy metals can undergo redox cycling and alter the cell’s oxidation state [34–36].
According to data, the chemical compounds that are released when tobacco is heated have the potential to harm oral tissues and change the microbiota, which could hasten the development of periodontal disorders. Chronic exposure to HTPs, in particular, has been associated with decreased wound healing and increased tissue necrosis, underscoring the detrimental effects on oral tissues [37–40].
The evidence does, however, indicate that the emitted aerosol can still have detrimental effects on oral cells and gingival fibroblasts, jeopardizing the possibility of periodontal and peri-implant healing. In vitro studies have demonstrated that HTPs produce inflammatory reactions that may result in tissue damage comparable to that produced by traditional smoking, but are less hazardous than combustible products [41–44].
It’s also critical to take into account how these devices might affect taste perception and tooth sensitivity, two factors that can influence people’s eating habits and quality of life. The purpose of this review is to examine a critical topic: the harm and mechanisms of action that HTPs and E-Cs cause to oral health [45].
There is currently little information available, but mounting indications that these items may have detrimental effects on the oral microbiota, raise inflammation, and reduce the efficacy of periodontal treatments [46–49].
This review is particularly timely given the growing scientific and clinical interest in the oral health consequences of vaping and heated tobacco use, as emphasized by recent research initiatives. It aims to address significant gaps in the current literature, particularly the lack of systematic and direct comparisons between the periodontal effects of HTPs, E-Cs, and conventional smoking [50–53]. The findings can assist consumers in making better decisions regarding the usage of HTPs and E-Cs.
Improving consumer quality of life and lowering the prevalence of periodontal disorders in the general population requires a better knowledge of how these products affect oral health.
The current systematic review followed the PRISMA and International Prospective Register of Systematic Review Registry procedures (ID: CRD420250616785).
The following databases: PubMed, Web of Science, and Scopus, were examined from 1 October 2014 to 21 November 2024, to search articles of the last 10 years. The search strategy was created by combining terms relevant to the study’s purpose. The following Boolean keywords were applied: (Vaping) OR (Cigarette) OR (Smoking) OR (e-cigarette) OR (Electronic cigarettes) AND (Periodontitis).
The reviewers worked in groups to assess all relevant studies that analyzed or compared the impact of smoking traditional cigarettes, E-Cs, and HTPs on periodontal health, with a focus on inflammatory markers and oral tissue response, according to the following inclusion criteria:
Studies that did the research “in vivo” or in “humans”;
Case-control studies, cohort studies, RCTs;
Studies that were published in the last 10 years;
Studies that fulfill any of the following exclusion criteria were excluded: reviews, case reports and series, letters to the authors, animal models, in vitro studies, and surveys.
The PICOS question addressed in this systematic review is summarized in Table 1.
PICOS criteria used for study selection.
| Criterion | Description |
|---|---|
| Population | Adult patients with periodontal disease |
| Intervention | The use of electronic cigarettes (E-Cs) or heated tobacco products (HTPs) |
| Comparison | Compared to traditional smokers or non-smokers |
| Outcome | How does it affect the response to non-surgical periodontal treatments in terms of inflammation, oral microbiome, and tissue healing |
| Study design | Systematic review of clinical and observational studies |
It shows the components of the PICOS criteria, as well as their use in this evaluation. PICOS: population, intervention, comparison, outcome, study design.
Four independent reviewers (B.F.P.P., V.C., D.C., R.V.G.) assessed the quality of the included studies using specified criteria, such as selection criteria, methods of outcome evaluation, and data analysis.
This enhanced ‘risk of bias’ tool additionally includes quality standards for selection, performance, detection, reporting, and other biases. Any differences were settled through conversation or collaboration with other researchers (G. D., A.D.I., V.C., R.V.G., B.F.P.P., D.C., F.I., A.P., A.M.I.). The reviewers (B.F.P.P., V.C., D.C., R.V.G.) screened the records according to the inclusion and exclusion criteria.
Doubts have been resolved by consulting the senior reviewer (F.I.). The selected articles were downloaded into Zotero 8.0.2, a reference management software used to organize and manage bibliographic data.
Figure 1 shows the flow diagram of a systematic review carried out using the PRISMA reporting criteria. The diagram describes the search strategy, inclusion, and exclusion of publications at each stage of detection. A total of 2,277 publications were identified in three databases, including PubMed (1,957), Web of Science (320), and Scopus (0), obtaining 1,022 records after the duplicates (30) and records marked as ineligible by automation tools, e.g., software such as Excel used to identify duplicates or clearly irrelevant records, and 1,225 articles were deleted. The title and abstract analysis resulted in the exclusion of 796 articles because they were off topic. The remaining 226 records were read, deleting 214 articles that did not fulfil the inclusion criteria. This resulted in a total of 12 publications for qualitative analysis (Table 2).
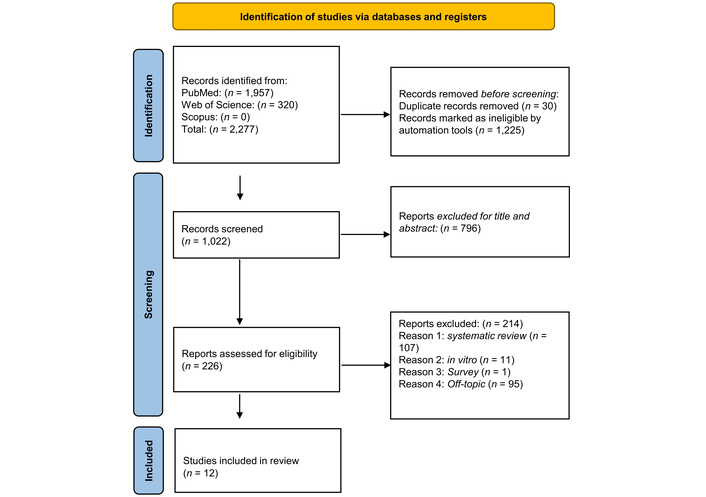
Literature search Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram and database search indicators. Adapted from [54]. © Author(s) 2021. Distributed under a Creative Commons CC BY 4.0 license.
Featured research in the qualitative analysis and its characteristics.
| Authors (year) | Type of study | Aim of the study | Material and methods | Results |
|---|---|---|---|---|
| Xu et al. (2021) [55] | Longitudinal study/comparative clinical study | To evaluate the impact of E-C aerosol on periodontal health in periodontitis patients | 140 participants (cigarette smokers, E-C users, non-smokers) followed for 6 months, monitoring periodontal parameters and smoking behaviors. | E-C users and smokers had more severe periodontal disease progression compared to non-smokers. |
| Shah et al. (2023) [56] | Retrospective clinical study | To compare periodontal treatment responses in E-C users, non-smokers, and smokers | A retrospective analysis of 220 periodontitis patients’ baseline charting, plaque removal, and re-evaluation. Linear models are used to analyze clinical outcomes based on smoking status. | E-C users had less favorable responses to treatment compared to non-smokers, similar to current smokers, with increased need for surgery and deeper probing depths. |
| Kumar et al. (2023) [57] | Exploratory clinical study | To evaluate how E-C use affects response to N-SPT | Patients underwent N-SPT, and clinical parameters were measured before and after treatment to assess the effect of E-C use on periodontal outcomes. | E-C users showed less improvement in clinical periodontal parameters compared with non-smokers. |
| Holliday et al. (2019) [58] | Feasibility pilot RCT | To assess the feasibility of E-Cs for smoking cessation in patients with periodontitis | Pilot randomized controlled trial involving patients using E-C toquit smoking, monitoring periodontal health and smoking cessation outcomes over time. | The study showed that E-Cs could be a feasible smoking cessation tool for periodontitis patients, but further larger studies are needed. |
| Karaaslan et al. (2020) [59] | Observational study | To examine the effects of vaping on periodontitis progression | Clinical evaluations of patients who vape, focusing on periodontal measurements like probing depth and attachment loss, compared to non-smokers and smokers. | Vaping was associated with increased periodontal inflammation and disease progression, similar to traditional smokers. |
| Kiiun et al. (2023) [60] | Biochemical and clinical study | To investigate oral dysbiosis and enzyme activity in E-C users with periodontitis | Biochemical tests on oral fluid for urease and lysozyme activity were performed on E-C users and compared to non-smokers and cigarette smokers with periodontal disease. | E-C users showed significant oral dysbiosis and altered enzyme activity compared to non-smokers, potentially worsening periodontitis outcomes. |
| Akram et al. (2021) [61] | Longitudinal cohort study | To evaluate periodontal disease progression biomarkers among E-C users | Clinical and biochemical markers of periodontal tissue degradation were tracked over time in smokers, E-C users, and non-smokers to compare disease progression. | E-C users showed elevated biomarkers indicating tissue degradation, similar to cigarette smokers. |
| ArRejaie et al. (2019) [62] | Cross-sectional study | To compare peri-implant parameters and cytokine levels among smokers, vapers, and non-smokers | Clinical measurements of peri-implant health and analysis of pro-inflammatory cytokines in the gingival fluid across the three groups. | Smokers and vapers both had higher levels of inflammation and worse peri-implant health compared to non-smokers. |
| ALHarthi et al. (2019) [63] | Prospective clinical study | To evaluate the effect of full-mouth ultrasonic scaling in smokers and vapers | Patients underwent full-mouth ultrasonic scaling, and gingival inflammation parameters were assessed pre- and post-treatment, comparing smokers, vapers, and non-smokers. | Vapers showed less reduction in inflammation compared to non-smokers after scaling, without coming close to smokers. |
| Mišković et al. (2024) [64] | Cross-sectional study | To assess periodontal health in adults exposed to tobacco heating systems versus cigarette smoke | Periodontal health of adults exposed to HTPs, cigarettes, and non-smokers was assessed through clinical periodontal parameters like probing depth and attachment loss. | Heated tobacco users had slightly better periodontal health than cigarette smokers, but worse than non-smokers. |
| Pouly et al. (2021) [65] | Randomized controlled multicenter study | To evaluate the effect of switching to heated tobacco on periodontitis treatment outcomes | Multicenter study with patients randomized to either switch to heated tobacco or continue smoking, assessing clinical periodontal treatment outcomes after non-surgical therapy. | Switching to heated tobacco showed improved periodontal treatment outcomes compared to continued smoking, but not as good as those of non-smokers. |
| Al-Hamoudi et al. (2020) [66] | Clinical study | To assess the anti-inflammatory cytokine response in E-C users and non-smokers | Analysis of anti-inflammatory cytokine levels (IL-4, IL-9, IL-10, and IL-13) in gingival crevicular fluid after SRP in E-C users and non-smokers. | E-C users had lower levels of anti-inflammatory cytokines compared to non-smokers, suggesting a less favorable immune response. |
E-C: electronic cigarette; N-SPT: non-surgical periodontal therapy; HTPs: heated tobacco products; IL: interleukin; SRP: scaling and root planing.
The risk of bias in the included studies is reported in Table 3.
Bias assessment by the Robins tool.
| Authors (year) | D1 | D2 | D3 | D4 | D5 | D6 | D7 | Overall |
|---|---|---|---|---|---|---|---|---|
| Xu et al. (2021) [55] |  |  |  |  |  |  |  |  |
| Shah et al. (2023) [56] |  |  |  |  |  |  |  |  |
| Kumar et al. (2023) [57] |  |  |  |  |  |  |  |  |
| Holliday et al. (2019) [58] |  |  |  |  |  |  |  |  |
| Karaaslan et al. (2020) [59] |  |  |  |  |  |  |  |  |
| Kiiun et al. (2023) [60] |  |  |  |  |  |  |  |  |
| Akram et al. (2021) [61] |  |  |  |  |  |  |  |  |
| ArRejaie et al. (2019) [62] |  |  |  |  |  |  |  |  |
| ALHarthi et al. (2019) [63] |  |  |  |  |  |  |  |  |
| Mišković et al. (2024) [64] |  |  |  |  |  |  |  |  |
| Pouly et al. (2021) [65] |  |  |  |  |  |  |  |  |
| Al-Hamoudi et al. (2020) [66] |  |  |  |  |  |  |  |  |
High:  ; low:
; low:  ; some concerns:
; some concerns:  ; no information:
; no information:  ; D1: bias due to confounding; D2: bias arising from the measurement of the exposure; D3: bias in the selection of participants into the study (or into the analysis); D4: bias due to post-exposure interventions; D5: bias due to missing data; D6: bias arising from the measurement of the outcome; D7: bias in the selection of the report result.
; D1: bias due to confounding; D2: bias arising from the measurement of the exposure; D3: bias in the selection of participants into the study (or into the analysis); D4: bias due to post-exposure interventions; D5: bias due to missing data; D6: bias arising from the measurement of the outcome; D7: bias in the selection of the report result.
Regarding bias due to confounding, most studies have a medium risk. The bias resulting from measurement is a parameter with a low risk of bias. Many studies have a low risk of bias due to participant selection. Post-exposure bias is low in most studies. Bias due to missing data is medium in many studies. The bias resulting from the outcome measurement cannot be calculated due to heterogeneity. The selection bias of the reported results is low in 7 of the studies and medium in the other 5.
Although the overall methodological quality of the included studies was acceptable, a moderate risk of bias was identified in several domains, particularly those related to confounding factors and missing data. These limitations should be considered when interpreting the reported associations between E-C or heated tobacco use and periodontal outcomes, as residual confounding cannot be completely excluded.
Different studies have come out to investigate the effect of E-Cs on the outcomes of N-SPT by determining differences in responses among traditional smokers, users of E-Cs, and non-smokers (N-S). Shah et al. (2023) [56] did an investigation of these differences and found that users of E-Cs responded differently compared to traditional smokers; the users of E-Cs showed generally retarded or less marked improvement of periodontal parameters [67–69].
Kumar et al. (2023) [57] further established that E-C users responded less favorably to scaling and root planing (SRP) than N-S, and the issues were more serious in traditional smokers [70, 71].
Al-Hamoudi et al. (2020) [66] further established that E-C users had lower anti-inflammatory cytokine levels following SRP, which could compromise the effectiveness of N-SPT by delaying the resolution of inflammation and healing [72, 73].
Both E-Cs and HTPs highly impact the oral microbiome, generally by enriching pathogenic species that may well contribute to periodontal disease exacerbation. Mišković et al. (2024) [64] presented a study about subgingival biofilm in both E-C and heated tobacco system users [74, 75]. An increased appearance of periodontopathogenic bacteria was found in these groups compared to the group of N-S. This modified composition may partly account for their higher predisposition to periodontal diseases compared with users of other methods of smoking. The enriched pathogenic species may promote tissue destruction and inflammation; thus, management of periodontitis may be more distressing as compared to traditional smokers and N-S [76–78].
E-C exposure has been associated with increased inflammatory oral responses that may negatively affect periodontal status by immune modulation.
ArRejaie et al. (2019) [62] proved that the peri-implant tissues of E-C users showed similar levels of inflammation to traditional smokers, with significantly higher levels of pro-inflammatory cytokines compared to N-S [79, 80]. This is further supported by Al-Hamoudi et al. (2020) [66], who showed that following periodontal treatment, E-C users had impaired anti-inflammatory responses, which may extend the period of inflammation. Thus, high levels of inflammatory markers among E-C users suggest chronic inflammation that can lead to the complication of periodontal management due to impaired healing and poor treatment outcomes [81, 82].
A number of longitudinal studies have evaluated the effects of E-Cs and HTPs on the progression of periodontitis. Holliday et al. (2019) [58] discussed the effects of E-Cs on smoking cessation and periodontal health in subjects [83]. It was noted that, upon cessation of smoking, periodontal health improves, but residual effects of previous smoking continue, most highly among users of E-Cs. Mišković et al. (2024) [64] noted that the periodontal health among users of E-Cs and heated tobacco showed considerably poorer health than in non-smoking subjects, though heated tobacco users showed slightly better evidence than traditional smokers [84]. Pouly et al. (2021) [65], on their part, carried out multicenter studies confirming that the periodontal improvements after switching from cigarettes to heated tobacco remained inferior to those achieved through complete cessation [61, 85, 86]. Interpretation of disease progression in E-C and heated tobacco users should be approached with caution, as the available evidence is largely derived from observational or cross-sectional studies and is characterized by limited follow-up durations. The lack of long-term data, together with substantial variability in exposure patterns, frequency, and duration of use across studies, introduces uncertainty regarding the cumulative and sustained effects of these products on periodontal tissues.
Progression among smokers and E-C users: Tissue degradation and clinical biomarkers give some insight into the process of periodontitis among smokers and E-C users. Akram et al. (2021) [61] proved that the pattern of degradation in tissues was similar to that of traditional smokers, hence establishing that both user groups are at higher risks of periodontal damage than N-S [56, 87]. According to ALHarthi et al. (2019) [63], the chronic states of periodontitis in both traditional smokers and E-C users were indeed complex to handle, as higher levels of ultrasonic scaling were needed in these groups to reduce inflammation, though the rate of healing was also slower compared to the N-S group. These findings indicate that E-C use enhances periodontitis, similar to conventional smoking, and compromises the treatment process.
Kiiun et al. (2023) [60] studied oral dysbiosis and enzymatic activity in relation to periodontal disease insubjects using E-C. In the present study, big changes in oral microbiota were found, including increased urease and lysozyme activity, in E-C smokers in accordance with a significant increase in pathogenic bacteria [88]. Thus, this microbial imbalance, together with enzymatic changes, could point out that the environment for periodontal tissues due to E-C use is even more dangerous, leading to an even higher risk of disease progression. This oral dysbiosis, therefore, adds another level of complication in the periodontal care and treatment outcome of such patients [89–91].
The use of E-Cs as an aid to smoking cessation regarding the long-term implications for periodontal health has been argued. Accordingly, though E-Cs may help in smoking cessation, the use of E-Cs does not considerably offer a long-term periodontal health benefit compared to complete smoking cessation [92–94]. There is some improvement in periodontal status among E-C users, which was not that strong compared to their N-S counterparts [95–97]. In contrast, Pouly’s 2021 study [65] reported modest gains in periodontal health among subjects who switched from cigarettes to heated tobacco; even these gains did not compare to the subjects who attained complete smoking cessation.
Full-mouth ultrasonic scaling is one of the common N-SPT. It has been noted to be variably effective in smokers and E-C users. For example, ALHarthi et al. (2019) [63] compared the healing of patients with gingival inflammation who either use or do not use E-Cs and established that the E-C users recover more slowly after scaling, just like traditional smokers. This was further supported by Al-Hamoudi et al. (2020) [66], who concluded that E-C users continuously maintained the inflammatory markers even after scaling and root planning, hence indicating a status that may affect the overall healing process and treatment success.
Mišković et al. (2024) [64] conducted a profound analysis of subgingival biofilm in users of E-C and heated tobacco systems. It was noted that in the biofilm of both groups, there was a much higher amount of pathogenic species than among traditional smokers and N-S [98, 99]. Therefore, the microbial environment that developed in both E-C and heated tobacco users is more likely to support periodontal disease development due to increased abundance of pathogens, further increasing tissue inflammation and leading to its destruction [100–102]. Results represent the microbiological aspect of the use of alternative tobacco products concerning periodontitis.
Several cross-sectional studies have assessed the periodontal status of individuals exposed to E-C and heated tobacco aerosols; these studies have presented marked differences in the various clinical parameters assessed [103, 104]. Mišković et al. (2024) [64] showed that both groups had higher pocket depth and attachment loss when compared with N-S. Karaaslan et al. (2020) [59] further illustrated that E-C users had higher peri-implant complications, while ArRejaie et al. (2019) [62] further confirmed that both smokers and E-C users showed heightened peri-implant inflammation. All these studies indicate that, in general, E-Cs and heated tobacco systems have an adverse impact on periodontal and peri-implant health, further complicating the management of periodontal diseases [105, 106].
The findings of this systematic review confirm and extend existing literature regarding the negative impact of E-Cs and HTPs on periodontal health.
In accordance with prior observational and clinical studies, the review highlights that both E-C and HTP use are associated with worsened clinical outcomes following N-SPT, showing patterns of inflammation, delayed healing, and persistent dysbiosis that are comparable to traditional cigarette smoking. Several studies reviewed also align with broader evidence indicating that E-C use may enrich periodontopathogenic bacteria and elevate inflammatory biomarkers, suggesting that the supposed “reduced harm” profile of these alternative products is not reflected in periodontal outcomes. These findings corroborate previous meta-analyses and clinical observations that suggest E-Cs and HTPs do not confer significant periodontal health benefits compared to complete smoking cessation.
An additional limitation of the current evidence lies in the considerable heterogeneity among included studies in terms of study design, population characteristics, clinical endpoints, and periodontal outcome measures. This heterogeneity precluded quantitative synthesis and meta-analysis and limits the comparability of results across studies, thereby restricting the strength of causal inferences that can be drawn from the existing literature.
Despite the growing body of evidence addressing the periodontal effects of E-Cs and HTPs, the current literature remains limited by the absence of large-scale, randomized controlled trials directly comparing these products with conventional cigarette smoking within standardized periodontal treatment protocols. Moreover, former smoking status is often insufficiently characterized, particularly with regard to time since cessation or switching, and the potential confounding impact of dual use of electronic or HTPs alongside conventional cigarettes is inconsistently reported. These limitations underscore the need for well-designed longitudinal and interventional studies to clarify the true clinical relevance of alternative tobacco products in periodontal disease progression and management.
E-Cs: electronic cigarettes
ENDS: electronic nicotine delivery system
HTPs: heated tobacco products
N-S: non-smokers
N-SPT: non-surgical periodontal therapy
PICOS: population, intervention, comparison, outcome, study design
SRP: scaling and root planing
GD: Conceptualization, Investigation, Writing—review & editing. ADI: Conceptualization, Data curation, Visualization, Funding acquisition. VC: Methodology, Writing—original draft, Supervision. BFPP: Validation, Writing—original draft, Funding acquisition. RVG: Methodology, Formal analysis, Project administration. DC: Conceptualization, Validation, Formal analysis. AP: Software, Formal analysis, Visualization. FI: Software, Resources, Supervision, Project administration. GI: Investigation, Resources. MC: Software, Visualization, Writing—original draft. GM: Methodology, Investigation, Visualization, Writing—review & editing. AMI: Validation, Data curation, Writing—review & editing. All authors read and approved the submitted version.
Gaetano Isola, who is the Associate Editor of Exploration of Medicine, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The primary data for this systematic review were sourced online from databases listed in the methods. References articles are accessible on PubMed, Web of Science, and Scopus. Additional supporting data are available from the corresponding author upon request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 3756
Download: 47
Times Cited: 0
