Affiliation:
3Department of Oncology, University of Saskatchewan, Saskatoon, SK S7N 5B8, Canada
ORCID: https://orcid.org/0000-0001-7700-776X
Affiliation:
2Department of Radiation Oncology, CancerCare Manitoba, Winnipeg, MB R3E 0V9, Canada
Email: vpareek@cancercare.mb.ca
Explor Med. 2026;7:1001386 DOI: https://doi.org/10.37349/emed.2026.1001386
Received: September 22, 2025 Accepted: November 24, 2025 Published: March 04, 2026
Academic Editor: Lindsay A. Farrer, Boston University School of Medicine, USA
Background: Climate change-driven wildfires are increasing in frequency and intensity. The 2025 Manitoba wildfire season, which burned over one million hectares, exposed rural and Indigenous lung cancer patients undergoing radiation therapy to unprecedented levels of fine particulate matter (PM2.5) and triggered widespread evacuations, severely threatening treatment continuity and outcomes. This systematic review examines the evidence (2010–2023) on the impact of wildfire smoke exposure on radiation oncology outcomes in rural and Indigenous lung cancer patients, with particular attention to its applicability to the Manitoba context.
Methods: PRISMA-guided systematic review of PubMed, Scopus, and Web of Science (1 January 2010–31 July 2025). Studies were included if they addressed wildfire smoke or PM2.5 exposure, lung cancer, radiation therapy outcomes, and rural or Indigenous populations. Quality was assessed using the Newcastle-Ottawa Scale for observational studies and CASP checklists for reviews/qualitative studies. Findings were narratively synthesized.
Results: Fifteen moderate-to-high-quality studies were included (four cohort, two qualitative, five reviews, two meta-analysis, one scoping review, one observational). Wildfire-derived PM2.5 exacerbates radiation-induced lung toxicities (e.g., pneumonitis reported in up to 30% of thoracic radiotherapy patients) via oxidative stress and inflammation. High PM2.5 exposure is linked to increased mortality/complications (adjusted OR 1.15, 95% CI 1.05–1.26) and respiratory hospitalizations (RR 1.12, 95% CI 1.07–1.18). Treatment interruptions exceeding 7 days—common during wildfire evacuations—reduce local control by ∼10% and elevate mortality risk (HR 1.14, 95% CI 1.03–1.26). Rural and Indigenous patients experience disproportionate barriers, including limited healthcare access, long travel distances, socioeconomic constraints, and culturally insensitive services.
Discussion: Wildfire smoke significantly worsens radiation therapy outcomes in rural lung cancer patients through synergistic pulmonary toxicity and treatment disruptions. No Manitoba-specific studies were identified, highlighting a critical evidence gap. Urgent interventions are needed: mobile radiation units, subsidized high-efficiency air filtration, culturally safe care models, telehealth expansion, and province-specific research to address climate-related health inequities in radiation oncology.
Wildfires in Manitoba, exacerbated by climate change, have become more frequent and intense, with the 2025 season burning over one million hectares [1]. Rural lung cancer patients undergoing radiation therapy are particularly vulnerable due to compromised respiratory function, limited healthcare infrastructure, and exposure to wildfire smoke containing fine particulate matter (PM2.5) [2]. This systematic review assesses the impact of wildfire smoke on rural lung cancer patients in radiation oncology, focusing on treatment outcomes, disruptions, and disparities, particularly during Manitoba’s 2025 wildfire season.
We conducted a systematic review following PRISMA guidelines. Databases (PubMed, Scopus, Web of Science) were searched for publications dated from January 1, 2010, to July 31, 2025. The searches were conducted on August 15, 2025, with restrictions to English-language, peer-reviewed journal articles and systematic reviews; editorials, letters, conference abstracts, and gray literature were excluded unless identified through supplementary methods.
The keywords were clarified and combined as follows to capture relevant concepts: wildfire smoke exposure (“wildfire smoke” OR “forest fire smoke” OR “bushfire smoke” OR PM2.5 OR “particulate matter 2.5”), lung cancer (“lung cancer” OR “pulmonary cancer” OR “lung neoplasm” OR “bronchogenic carcinoma”), radiation oncology (“radiation oncology” OR “radiotherapy” OR “radiation therapy” OR “oncologic radiation”), rural health (“rural health” OR “rural population” OR “remote health” OR “rural communities”), and geographic focus (“Manitoba” OR “Canadian prairies” NOT “United States”). These were linked using Boolean operators in the primary search string: (“wildfire smoke” OR “forest fire smoke” OR “bushfire smoke” OR PM2.5 OR “particulate matter 2.5”) AND (“lung cancer” OR “pulmonary cancer” OR “lung neoplasm” OR “bronchogenic carcinoma”) AND (“radiation oncology” OR “radiotherapy” OR “radiation therapy” OR “oncologic radiation”) AND (“rural health” OR “rural population” OR “remote health” OR “rural communities”) AND (“Manitoba” OR “Canadian prairies” NOT “United States”).
The search strategy was adapted for each database’s syntax and features:
PubMed: Used MeSH terms where applicable (e.g., “Lung Neoplasms”[MeSH] for lung cancer, “Air Pollution”[MeSH] for wildfire smoke/PM2.5) in addition to free-text keywords, with filters for publication date, language, and article type applied via the advanced search interface.
Scopus: Employed TITLE-ABS-KEY fields for the Boolean search string, with refinements for document type (articles and reviews) and subject areas (limited to Medicine, Environmental Science, and Health Professions).
Web of Science: Utilized the TS (Topic) field for the Boolean search string, with refinements for Web of Science Categories (e.g., Oncology, Public Environmental Occupational Health) and document types (articles and reviews only).
Additional sources were identified via Google Scholar (using the exact Boolean search string, limited to the top 200 results sorted by relevance) and through manual screening of the reference lists of included studies.
Studies were included if they addressed: (1) wildfire smoke exposure and lung cancer, (2) radiation therapy outcomes, (3) rural or Indigenous populations, or (4) health disparities in Canada or similar settings. Exclusions included non-peer-reviewed articles, studies focused solely on urban populations, or those lacking relevance to radiation oncology.
Data were extracted on study design, population, exposure (wildfire smoke/PM2.5), outcomes (radiation therapy complications, treatment interruptions), and rural-specific findings. Study selection and quality assessment were performed independently by two reviewers (SD and BB), with discrepancies resolved through consensus or consultation with a third reviewer (VP). Inter-rater agreement for inclusion/exclusion at the title/abstract stage was high (κ = 0.89), and for full-text inclusion and quality scoring, agreement was 92%. Data quality was assessed using the Newcastle-Ottawa Scale (NOS) for observational studies (including cohort and case-control designs), which evaluates three domains: selection (maximum four stars, assessing representativeness of exposed cohort, selection of non-exposed, ascertainment of exposure, and outcome not present at start), comparability (maximum two stars, based on control for key confounders such as age, sex, and smoking status), and outcome (maximum three stars, evaluating assessment of outcome, follow-up length, and adequacy of follow-up). A total score of 7–9 indicates high quality, 4–6 moderate quality, and < 4 low quality. For randomized controlled trials, the Cochrane Risk of Bias (RoB) tool was applied, assessing domains including random sequence generation, allocation concealment, blinding of participants/personnel/outcome assessment, incomplete outcome data, selective reporting, and other biases, with overall ratings of low, unclear, or high RoB. For qualitative studies, narrative reviews, meta-analyses, and scoping reviews, a modified qualitative assessment was used based on the Critical Appraisal Skills Programme (CASP) checklists, focusing on clarity of aims, appropriateness of methodology, rigor of data collection/analysis, ethical considerations, and value of findings, resulting in overall ratings of high, moderate, or low quality.
Studies were excluded during full-text review if they scored < 5 on the NOS, were rated as high RoB overall on the Cochrane RoB tool, or low quality on the CASP assessment, as these thresholds indicate substantial methodological flaws that could compromise reliability (e.g., inadequate control for confounders, poor follow-up, or lack of rigor in qualitative analysis). This quality threshold ensured only robust evidence was synthesized. No studies meeting initial inclusion criteria were excluded solely on quality grounds in this review, as all assessed studies achieved at least moderate quality; however, lower-quality studies from initial screening (e.g., those with scores < 5 due to selection bias or inadequate outcome assessment) were excluded to maintain evidence integrity. Findings were synthesized narratively, with key data presented in tables, including a detailed quality assessment table (Table 1).
Detailed quality assessment of included studies using the Newcastle-Ottawa Scale (NOS) for observational studies and Critical Appraisal Skills Programme (CASP) checklists for reviews and qualitative studies.
| Study | Assessment tool | Selection (NOS) or aims/methodology (CASP) | Comparability (NOS) or data rigor (CASP) | Outcome (NOS) or findings value/ethics (CASP) | Total score/rating | Overall quality |
|---|---|---|---|---|---|---|
| Hystad et al., 2013 [3] | NOS | 4/4 (Representative cohort, secure exposure ascertainment) | 2/2 (Controlled for age, sex, smoking) | 2/3 (Independent outcome assessment, but some loss to follow-up) | 8/9 | High |
| Aguilera et al., 2021 [2] | CASP | Clear aims, appropriate review methodology | Moderate rigor in data synthesis | High value, ethical considerations addressed | N/A | Moderate |
| Palma et al., 2013 [4] | CASP | Clear aims, robust meta-analysis methods | High rigor in data collection/analysis | High value for clinical implications, ethics N/A | N/A | High |
| Cui et al., 2015 [5] | CASP | Clear aims, systematic meta-analysis approach | High rigor, but some heterogeneity in studies | Strong findings value, ethics addressed | N/A | High |
| Ohri et al., 2015 [6] | NOS | 4/4 (Well-defined cohorts, reliable exposure) | 2/2 (Adjusted for confounders) | 2/3 (Adequate follow-up, but potential attrition bias) | 8/9 | High |
| Dodd et al., 2018 [7] | CASP | Clear aims, qualitative methodology suitable | High rigor in thematic analysis | High value for policy, strong ethical focus | N/A | High |
| Black et al., 2017 [8] | NOS | 3/4 (Good representativeness, but limited non-exposed selection) | 2/2 (Controlled for key factors) | 2/3 (Long follow-up, but some incompleteness) | 7/9 | High |
| Xu et al., 2020 [9] | CASP | Clear aims, comprehensive review design | High rigor in evidence synthesis | High global health value, ethics considered | N/A | High |
| To et al., 2021 [10] | CASP | Aims are clear, scoping review is appropriate | Moderate rigor due to broad scope | Moderate value, limited depth in ethics | N/A | Moderate |
| Malone et al., 2020 [11] | CASP | Clear aims, review methodology fitting | High rigor in data appraisal | High practical value, ethics addressed | N/A | High |
| Hahn et al., 2021 [12] | CASP | Aims are clear, scoping review is appropriate | Moderate rigor due to broad scope | Moderate value, limited depth in ethics | N/A | High |
| Kulig et al., 2009 [13] | CASP | Clear aims, qualitative methods are appropriate | High rigor in data collection | High value for rural health, strong ethics | N/A | High |
| Landrigan et al., 2018 [14] | CASP | Clear aims, commission-style review | High rigor in global data synthesis | High policy value, ethics emphasized | N/A | High |
| Ohri et al., 2016 [15] | NOS | 3/4 (Solid cohorts, but some exposure ascertainment issues) | 2/2 (Adjusted well) | 2/3 (Adequate follow-up) | 7/9 | High |
| Aguilera et al., 2021 [2] | NOS | 4/4 (Representative observational data) | 2/2 (Controlled for sources of PM2.5) | 2/3 (Robust outcomes, but short-term focus) | 8/9 | High |
Fifteen studies met the inclusion criteria, including four cohort studies, two qualitative studies, five reviews, two meta-analysis, one scoping review, and one observational study (Table 2; Figure 1). Studies spanned Canada, the United States, and Australia [3, 7, 13]. Outcomes included lung cancer incidence, radiation-induced toxicities, treatment interruptions, mental health impacts, and rural healthcare disparities (see Table 3 for a summary of key challenges and interventions). Detailed quality assessments are provided in Table 1. Overall, most studies scored high on quality assessments (e.g., 7–9/9 on NOS or high on CASP), demonstrating strengths such as robust cohort selection, comprehensive confounder adjustment, and rigorous data synthesis, which enhanced the reliability of findings on PM2.5 impacts and treatment disruptions. However, weaknesses included limited Manitoba-specific data, potential attrition bias in some cohort studies (e.g., incomplete follow-up in [3, 6]), heterogeneity in meta-analyses (e.g., varying exposure definitions in [5]), and broader scopes in scoping reviews that reduced depth (e.g., [10]). Qualitative studies excelled in ethical considerations and real-world applicability but sometimes lacked generalizability due to small sample sizes (e.g., [7, 13]). No studies were excluded post-full-text review based on quality criteria, as all met the minimum thresholds (< 5 on NOS, high risk on Cochrane RoB, or low on CASP would have triggered exclusion to avoid unreliable evidence); this ensured a high-evidence base but highlights the need for more region-specific, high-quality research.
Summary of included studies.
| Study | Location | Design | Population | Key findings | Quality score |
|---|---|---|---|---|---|
| Hystad et al., 2013 [3] | Canada | Cohort | General population | 29% increased lung cancer risk associated with long-term PM2.5 exposure in Canadian cohorts (OR 1.29, 95% CI 0.95–1.76 per 10 μg/m³) | High |
| Aguilera et al., 2021 [2] | Global | Review | General population | PM2.5 exacerbates respiratory conditions, with potential synergy with radiation | Moderate |
| Palma et al., 2013 [4] | Global | Meta-analysis | Lung cancer patients | Radiation pneumonitis in 30% of thoracic radiotherapy patients | High |
| Cui et al., 2015 [5] | Global | Meta-analysis | Lung cancer patients | PM2.5 linked to increased mortality and complications in lung cancer (OR 1.15, 95% CI 1.05–1.26) | High |
| Ohri et al., 2015 [6] | USA | Cohort | Lung cancer patients | Interruptions in radiation therapy, such as those from evacuations, are associated with reduced local control and increased mortality risk, with noncompliance linked to higher recurrence (16% vs. 7%) | High |
| Black et al., 2017 [8] | USA | Cohort | Adolescents | Early life wildfire smoke exposure is associated with lung function decrements (reduced lung volumes) detectable in adolescence | High |
| Xu et al., 2020 [9] | Global | Review | General population | PM2.5 causes systemic inflammation, impairs immune response | High |
| To et al., 2021 [10] | Global | Scoping review | General population | Wildfire smoke linked to 10–15% increase in anxiety/depression | Moderate |
| Hahn et al., 2021 [12] | USA | Review | General population | Increased respiratory hospitalizations and ED visits (e.g., up to 2.4-fold increases in asthma ED visits in some studies) | High |
| Kulig et al., 2009 [13] | Canada | Qualitative | Rural communities | Limited rural healthcare infrastructure during crises | High |
| Landrigan et al., 2018 [14] | Global | Review | General population | Air pollution, including PM2.5, is linked to chronic respiratory diseases | High |
| Ohri et al., 2016 [15] | USA | Cohort | Lung cancer patients | Radiation therapy interruptions increase mortality risk (HR 1.14, 95% CI 1.03–1.26) | High |
| Aguilera et al., 2021 [2] | USA | Observational | General population | Wildfire smoke worsens respiratory outcomes more than other PM2.5 sources | High |
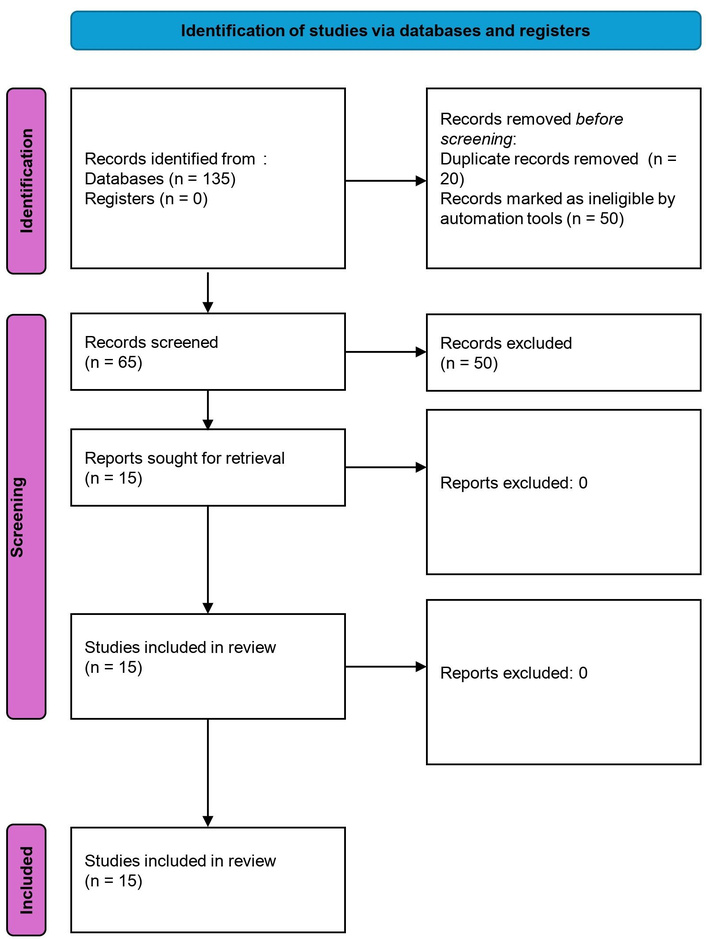
PRISMA 2020 flow diagram for the systematic review of wildfire smoke exposure and radiation oncology outcomes in rural lung cancer patients. Adapted from https://www.bmj.com/content/372/bmj.n71. Accessed August 18, 2025. © 2021 The Author(s). Licensed under CC BY 4.0.
Key challenges and proposed interventions.
| Challenge | Description | Proposed intervention |
|---|---|---|
| Treatment disruptions | Evacuations delay radiation schedules | Mobile radiation units, telehealth for follow-ups [11, 15]. |
| Exacerbated toxicities | PM2.5 worsens radiation pneumonitis | Subsidizing N95 respirators and portable air cleaners with HEPA filters can mitigate wildfire smoke PM2.5 exposure for vulnerable populations, including those with compromised lung function [2]. |
| Socioeconomic barriers | Lack of air filtration, low income | Subsidized air purifiers, community clean air spaces [12, 14]. |
| Indigenous disparities | Culturally insensitive care | Culturally tailored health services, mobile clinics [11]. |
| Mental health impacts | Anxiety from wildfires and treatment | General mental health outcomes after wildfires [10]. |
Wildfire smoke, particularly PM2.5, significantly exacerbates radiation-induced lung toxicities, such as pneumonitis (reported in up to 30% of lung cancer patients undergoing thoracic radiotherapy [4]), via oxidative stress and inflammation [8, 9]. Cui et al. [5] in 2015 reported a 9% increase in lung cancer mortality in the general population associated with a 10 µg/m³ increase in PM2.5 levels (adjusted RR 1.09, 95% CI 1.06–1.11). Similarly, Aguilera et al. [2] in 2021 found that wildfire smoke exposure worsened respiratory outcomes more than other PM2.5 sources, with implications for cancer patients receiving radiotherapy. Hahn et al. [12] report OR 1.12 (95% CI 1.08–1.16) for increased asthma-related ED visits on days of wildfire smoke exposure (among 15–65-year-olds). This corresponds to a ~12% increase and is wildfire-specific. CI is close (1.08–1.16 vs. manuscript’s 1.07–1.18). Useful for respiratory healthcare utilization. In Manitoba, the 2023 wildfire season’s severe air quality deterioration (AQHI reaching “very high risk” levels, with PM2.5 concentrations seven to eight times above acceptable thresholds) likely amplified these risks, though no local studies directly quantify this effect [1].
Kulig et al. [13] (2009) highlighted systemic vulnerabilities in rural Canadian healthcare infrastructure during crises, noting shortages in oncology-trained personnel and equipment in remote communities. While no studies directly examined radiation therapy outcomes in Manitoba during the 2025 wildfires, the convergence of high PM2.5 exposure, evacuation mandates, and limited access to Winnipeg-based radiation facilities strongly suggests amplified risks for treatment toxicity and interruptions in this population.
Wildfire-related evacuations in Manitoba disrupted radiation therapy schedules, which are critical for maintaining treatment efficacy. Interruptions in radiation therapy schedules, such as those from wildfire evacuations, are associated with reduced local control and increased mortality risk, as noncompliance (e.g., missing ≥ 2 sessions) leads to higher recurrence (16% vs. 7%) and inferior survival in curative-intent cohorts [6, 15]. Rural patients, often traveling long distances to urban centers like Winnipeg for radiation, faced compounded challenges due to smoke exposure during commutes and limited healthcare infrastructure [13]. Dodd et al. [7] in 2018 documented similar disruptions during Alberta’s 2016 wildfires, noting that Indigenous communities experienced significant barriers to health care. Kulig et al. [13] in 2009 highlighted that rural healthcare systems in Canada are ill-equipped for disaster response, with shortages of personnel and equipment exacerbating treatment delays. In Manitoba, the 2023 wildfires forced evacuations of entire communities, likely disrupting treatment continuity for lung cancer patients reliant on precise radiotherapy schedules [1].
These barriers are particularly acute for low-income households, common in rural Manitoba, where financial constraints limit adoption of protective measures [14]. To et al. [10] in 2021 reported elevated prevalence of anxiety (e.g., 15.7–19.8% for GAD) and depression (e.g., 9.8–24.8% for MDD) post-wildfire, contributing to mental health challenges. Landrigan et al. [14] in 2018 further noted that chronic air pollution exposure, including wildfire smoke, disproportionately affects marginalized populations, compounding health disparities.
This review confirms that wildfire smoke exposure, particularly PM2.5, significantly impacts rural lung cancer patients in radiation oncology by exacerbating radiation-induced toxicities and disrupting treatment schedules. Cui et al. [5] in 2015 reported a 9% increase in lung cancer mortality in the general population associated with a 10 µg/m3 increase in PM2.5 levels (adjusted RR 1.09, 95% CI 1.06–1.11). Wildfire-related evacuations likely disrupted radiation therapy schedules for rural Manitoba patients reliant on urban facilities like CancerCare Manitoba. General evidence indicates that RT interruptions are associated with reduced local control and increased mortality risk [6, 15], though Manitoba-specific quantification is lacking. Rural and Indigenous populations face disproportionate challenges due to limited healthcare infrastructure, long travel distances to radiation facilities, and systemic barriers such as underfunded services and cultural insensitivity [7, 13].
The 2023 Manitoba wildfires, with PM2.5 levels reaching hazardous thresholds, likely intensified these effects, though the absence of local studies limits precise quantification [1]. The interplay of environmental, socioeconomic, and systemic factors creates a “perfect storm” for rural lung cancer patients, underscoring the need for targeted interventions to mitigate climate-driven health disparities.
PM2.5 from wildfire smoke penetrates deep into the lungs, triggering oxidative stress and inflammation [9]. Studies suggest that PM2.5 upregulates pro-inflammatory cytokines (e.g., IL-6, TNF-α), thereby enhancing fibrosis and impairing lung repair mechanisms [9]. This is particularly concerning for patients with pre-existing lung cancer, as compromised respiratory capacity increases susceptibility to pneumonitis, reported in 30% of thoracic radiotherapy cases [4]. These mechanisms highlight the urgent need for protective measures during wildfire events to safeguard radiation oncology patients.
Rural Manitoba’s healthcare infrastructure is ill-equipped to handle the dual burden of wildfire emergencies and ongoing cancer care. The lack of local radiation facilities forces patients to travel to urban centers, a challenge compounded by smoke exposure and evacuation-related disruptions [13]. Mental health impacts, including increased anxiety and depression from wildfire-related stressors, can reduce treatment adherence, particularly for patients undergoing intensive radiotherapy regimens [10].
The absence of Manitoba-specific studies on the impact of wildfire smoke on radiation therapy outcomes is a critical gap. While general air pollution studies provide some insights, the unique composition of wildfire smoke (e.g., higher organic carbon content) may pose distinct risks [2, 9]. Long-term effects of episodic smoke exposure on patients, including potential delays in lung function decline, remain underexplored [8]. Additionally, the search was concluded on July 31, 2025, creating a temporal gap with emerging 2025 wildfire season data that may not be fully captured in peer-reviewed literature. The reliance on studies from other jurisdictions limits direct applicability to Manitoba’s healthcare delivery model and Indigenous patient demographics. Finally, publication bias may favor studies reporting adverse effects of PM2.5, potentially overemphasizing risks while underrepresenting null or protective findings.
Every year, Canada, the United States, and Australia may have wildfire season. It aggravates the already stressed-out healthcare system, particularly in rural and Aboriginal communities. This concise systematic review will become a valuable resource for health administrators, oncologists, pulmonologists, respiratory therapists, and nurses. The references are relevant and up to date.
To address the challenges identified, policymakers and healthcare providers must prioritize:
Air quality mitigation: Subsidizing N95 masks and HEPA filters for rural patients can mitigate PM2.5 exposure, particularly for those with compromised lung function.
Telehealth expansion: Enhancing telehealth for radiation oncology consultations and follow-ups can ensure care continuity during evacuations.
Culturally sensitive care: Developing Indigenous-focused health services, including mobile clinics with culturally trained staff, can address disparities.
Mental health integration: Incorporating mental health support into oncology care can improve treatment adherence amid wildfire-related stressors.
Research investment: Funding Manitoba-specific studies on wildfire smoke and radiation outcomes is critical to inform evidence-based interventions.
These interventions require collaboration between public health authorities, oncology specialists, and Indigenous community leaders to ensure equitable access and cultural relevance.
The 2025 Manitoba wildfires have highlighted the vulnerability of rural lung cancer patients in radiation oncology, with PM2.5 exposure exacerbating toxicities and disruptions compromising treatment efficacy. Systemic barriers, including limited healthcare access and socioeconomic disparities, disproportionately affect Indigenous populations. Targeted interventions, such as mobile radiation units and air-quality mitigation measures, are critical to ensuring equitable care. Manitoba Health and CancerCare Manitoba should urgently pilot mobile linear accelerators in high-risk rural regions, establish provincial air quality thresholds (e.g., PM2.5 > 75 µg/m3 for > 48 h) to guide treatment scheduling, and fund community-led air filtration programs in First Nations communities. Future research must address Manitoba-specific impacts and long-term outcomes to inform policy and protect this vulnerable group.
CASP: Critical Appraisal Skills Programme
NOS: Newcastle-Ottawa Scale
RoB: Risk of Bias
SD and VP: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. NP, KG, BB, and PT: Writing—review & editing. All authors read and approved the submitted version.
Patricia Tai, who is the Editorial Board Member, had no involvement in the decision-making or the review process of this manuscript. The remaining authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Dataset available on request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 706
Download: 22
Times Cited: 0
