Affiliation:
1Department of Physical Education and Sport Sciences, Faculty of Humanities and Social Sciences, University of Kurdistan, Sanandaj 6617715175, Iran
ORCID: https://orcid.org/0009-0002-1816-8584
Affiliation:
1Department of Physical Education and Sport Sciences, Faculty of Humanities and Social Sciences, University of Kurdistan, Sanandaj 6617715175, Iran
Email: a.saeidi@uok.ac.ir
ORCID: https://orcid.org/0000-0003-2458-6256
Affiliation:
2Department of Biological Sciences, College of Science, King Faisal University, Al-Ahsa 36362, Saudi Arabia
ORCID: https://orcid.org/0000-0003-2288-089X
Affiliation:
3Faculty of Medicine, University of Tunis El Manar, Tunis 2092, Tunisia
4International Institute of Sport Sciences (2I2S), 35000 Rennes, France
ORCID: https://orcid.org/0000-0001-8160-3871
Affiliation:
5Department of Family and Community Medicine, College of Medicine, King Faisal University, Al-Ahsa 36362, Saudi Arabia
ORCID: https://orcid.org/0000-0001-6035-1230
Affiliation:
6Department of Sport Sciences, Faculty of Sport Sciences, Hakim Sabzevari University, Sabzevar 9617976487, Iran
ORCID: https://orcid.org/0000-0002-4590-8018
Affiliation:
4International Institute of Sport Sciences (2I2S), 35000 Rennes, France
Email: hassane.zouhal@gmail.com
ORCID: https://orcid.org/0000-0001-6743-6464
Explor Immunol. 2026;6:1003253 DOI: https://doi.org/10.37349/ei.2026.1003253
Received: November 06, 2025 Accepted: March 24, 2026 Published: May 09, 2026
Academic Editor: Bernhard Ryffel, University of Orleans, France
The article belongs to the special issue Physical Activity and Immune System in Chronic Diseases: Mechanisms and Insights
Obesity is a major global health challenge characterized by chronic low-grade inflammation and metabolic dysfunction. Among lipid-derived mediators involved in inflammation resolution, maresin-1 (MaR1)—a specialized pro-resolving mediator derived from docosahexaenoic acid (DHA) and produced mainly by M2 macrophages—has attracted increasing attention due to its potent anti-inflammatory and metabolic regulatory properties. MaR1 promotes the resolution of inflammation by limiting neutrophil infiltration, enhancing macrophage efferocytosis, and shifting cytokine profiles toward an anti-inflammatory phenotype. In addition, it modulates metabolic pathways related to insulin sensitivity and skeletal muscle glucose uptake through signaling mechanisms involving Akt and AMP-activated protein kinase (AMPK). Reduced circulating levels of MaR1 have been consistently associated with metabolic disorders, including obesity, type 2 diabetes, and cardiovascular disease, highlighting its potential as a biomarker of metabolic health. Exercise is a cornerstone non-pharmacological strategy for obesity management and activates molecular pathways—such as AMPK and Sirtuin 1 (SIRT1)—that overlap with those regulated by MaR1. However, human studies examining how different exercise modalities influence MaR1 production remain scarce. This perspective highlights the mechanistic links between exercise and MaR1 biology and proposes a translational research agenda to investigate how aerobic, resistance, and high-intensity interval training modulate MaR1 levels. Understanding this exercise–MaR1 axis may help establish MaR1 as a biomarker of exercise responsiveness and support the development of targeted lifestyle interventions for metabolic disease management.
Obesity is a global public health crisis with significant metabolic and inflammatory consequences, making its control a public health priority [1]. In recent years, attention has shifted to lipid-derived mediators and adipokines as key regulators of inflammatory and metabolic responses. Among these mediators, maresin-1 (MaR1) has emerged for its potent anti-inflammatory properties. The term ʻMaresinʼ stands for Macrophage Resolution Signaling Intermediator, reflecting its primary biosynthetic origin in pro-resolving macrophages. As a specialized pro-resolving mediator (SPM) produced from the omega-3 fatty acid docosahexaenoic acid (DHA) via the 12-lipoxygenase (12-LOX) pathway, MaR1 plays a pivotal role in limiting neutrophil infiltration and enhancing the clearance of apoptotic cells [2].
MaR1 is a potent SPM primarily biosynthesized by M2-polarized macrophages from the precursor fatty acid DHA [3, 4]. Beyond its classic anti-inflammatory role, MaR1 exerts multi-organ benefits, including stimulating tissue regeneration, providing neuroprotection in neurodegenerative conditions, and enhancing the phagocytic clearance of apoptotic cells (efferocytosis). Crucially, it promotes a phenotypic switch in macrophages from M1 to M2, thereby orchestrating the transition from the inflammatory phase to the resolution phase of healing [5]. Among the Maresin family, MaR1 stands out as the most promising biomarker for exercise interventions. While other types like MaR2 share similar biosynthetic origins, MaR1 exhibits a unique potency in modulating skeletal muscle metabolism and enhancing glucose uptake via Akt and AMP-activated protein kinase (AMPK) signaling [6]. This mechanistic overlap with exercise-induced adaptations, combined with its established clinical relevance in human obesity and type 2 diabetes, provides a strong rationale for prioritizing MaR1 over other isoforms in translational exercise physiology research. MaR1 accelerates the resolution of inflammation by limiting neutrophil infiltration, enhancing macrophage efferocytosis and phagocytosis, and shifting cytokine profiles toward an anti-inflammatory phenotype. Preclinical and clinical data indicate that MaR1 can suppress pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) while increasing anti-inflammatory mediators. Additionally, lower circulating concentrations of MaR1 have been associated with metabolic disorders, including type 2 diabetes, hypertension, and cardiovascular disease [2, 7, 8].
Despite its robust preclinical profile, MaR1 has not yet progressed to phase I–III clinical trials, primarily due to the inherent pharmacological challenges of lipid-derived mediators, including rapid metabolic inactivation and the need for stabilized analogs. However, recent observational clinical studies have firmly established its relevance in human pathophysiology; lower circulating concentrations of MaR1 are consistently associated with metabolic disorders such as obesity [9], type 2 diabetes [10], and cardiovascular diseases. These findings underscore the current status of MaR1 as a potent mechanistic biomarker for monitoring chronic inflammation and metabolic health [11]. A critical question in translational research is whether direct MaR1 administration offers advantages over DHA supplementation. Although DHA is the essential precursor, its conversion into bioactive MaR1 can be significantly compromised in individuals with metabolic syndrome due to impaired 12-LOX signaling. Direct use of MaR1 overcomes this ʻenzymatic bottleneck,ʼ providing a more targeted and potent resolution of inflammation. However, successful translation into clinical practice faces hurdles, including the rapid metabolic inactivation of natural lipids and the complexity of synthesizing stable, bioavailable mimetics that can withstand the systemic environment [12, 13].
Mechanistically, multiple pathways have been proposed to mediate the beneficial effects of MaR1. These include the activation of the PI3K/Akt signaling pathway, which leads to Akt phosphorylation and subsequent endothelial nitric oxide synthase (eNOS) activation, resulting in increased nitric oxide production and improved endothelial function. Additionally, MaR1 attenuates oxidative stress by upregulating antioxidant enzymes and activating metabolic regulators such as the AMPK/Sirtuin 1 (SIRT1) axis. It has also been implicated in promoting thermogenic programs by upregulating uncoupling protein 1 (UCP1), peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), and other browning markers, thereby enhancing brown adipose tissue activity and promoting the browning of white adipose depots. These changes are expected to increase energy expenditure and improve insulin sensitivity [14, 15]. A critical advantage of MaR1 over conventional anti-inflammatory drugs, such as non-steroidal anti-inflammatory drugs (NSAIDs), lies in its ʻpro-resolvingʼ rather than ʻimmunosuppressiveʼ nature. While traditional therapies often inhibit cyclooxygenase enzymes and can lead to adverse effects or delayed tissue repair, MaR1 promotes the active resolution of inflammation by enhancing macrophage phagocytosis and tissue regeneration without impeding host defense mechanisms. This distinction positions MaR1 as a potentially superior therapeutic target for chronic metabolic inflammation [5].
Exercise is the most widely recommended non-pharmacological intervention for obesity, exerting broad systemic effects on inflammation, energy metabolism, insulin sensitivity, endothelial function, and body composition. Aerobic exercise, resistance training, and high-intensity interval training (HIIT) have all been shown to activate signaling hubs such as AMPK and SIRT1, increase mitochondrial biogenesis and fatty acid oxidation, shift adipokine profiles toward reduced pro-inflammatory signaling, and stimulate brown and beige adipose activity [16–19]. Given the clear mechanistic overlap between exercise-induced pathways and those modulated by MaR1, a critical and currently unanswered question is how different exercise modalities (acute vs. chronic; aerobic vs. resistance vs. HIIT) influence MaR1 biosynthesis and circulating levels in humans, and to what extent changes in MaR1 mediate the cardiometabolic benefits of physical activity. Addressing this question has important translational implications: MaR1 could serve as a biomarker for responsiveness to exercise interventions, and understanding the exercise–MaR1 axis might enable the design of targeted exercise prescriptions or combined lifestyle-nutritional strategies (e.g., exercise plus DHA supplementation) to enhance MaR1 production and improve the resolution of adipose inflammation.
A search of the literature reveals a surprising lack of focused human studies that directly quantify the effects of defined exercise programs on MaR1 in clinically relevant populations, such as healthy individuals, people with obesity, and those with type 2 diabetes or metabolic syndrome. This represents a significant knowledge gap and a promising avenue for further research. Therefore, we propose a coordinated research agenda that includes cross-sectional and longitudinal observational studies to characterize correlations between habitual physical activity, objectively measured fitness, and circulating MaR1, along with inflammatory and metabolic biomarkers. Additionally, we recommend randomized controlled trials to compare the effects of different exercise modalities and intensities on MaR1 and downstream metabolic outcomes. Furthermore, we suggest mechanistic sub-studies employing sensitive analytical methods (e.g., LC-MS/MS for MaR1 quantification), adipose tissue biopsies to assess local expression and pathway activation, imaging modalities to evaluate brown adipose tissue activity (such as PET-CT or MRI), and concurrent measurement of a panel of cytokines, oxidative stress, and insulin sensitivity indices, including clamp studies where feasible.
Additionally, factorial trials that combine exercise interventions with DHA supplementation could determine whether substrate availability limits MaR1 production, and whether nutritional augmentation works synergistically with exercise stimuli to enhance pro-resolving mediator responses. Clinically, clarifying the interaction between exercise and MaR1 may inform precision exercise prescriptions for patients with obesity, diabetes, and hypertension, while also identifying MaR1 as a mechanistic biomarker for monitoring therapeutic response. Given the current lack of direct evidence, we urge research funding bodies and investigators to prioritize rigorous clinical and translational studies that explore the role of exercise in regulating MaR1 and its contribution to the metabolic and cardiovascular benefits of physical activity. Such work has the potential not only to advance our mechanistic understanding of exercise biology but also to support the development of optimized, mechanism-guided lifestyle interventions aimed at reducing the global burden of metabolic diseases.
Future studies should prioritize distinguishing between the acute biosynthetic response to a single bout of exercise and the chronic adaptations in MaR1 baseline levels following long-term training. It is hypothesized that high-intensity modalities like HIIT may trigger a more robust immediate release of MaR1 to counteract acute inflammatory stress, whereas aerobic training might foster a sustained anti-inflammatory environment by enhancing basal MaR1 production. The multifaceted biological roles of MaR1 and the strategic framework for future clinical investigations are summarized in Figure 1. This visual representation emphasizes the transition from basic physiology to precision exercise prescription. This schematic representation illustrates the key physiological functions of MaR1 on the left, including the promotion of macrophage polarization (M1 to M2), tissue regeneration (muscle and neural), enhancement of efferocytosis, and improving insulin sensitivity via AMPK and Akt signaling pathways. On the right, the conceptual model for transitioning from basic physiology to clinical application is depicted, emphasizing the need to investigate the effects of various exercise modalities (aerobic, resistance, and HIIT training) on MaR1 production. The mechanistic overlap between contraction-induced signaling and MaR1 biology provides a rationale for developing precise biomarkers and tailored exercise prescriptions for managing metabolic diseases.
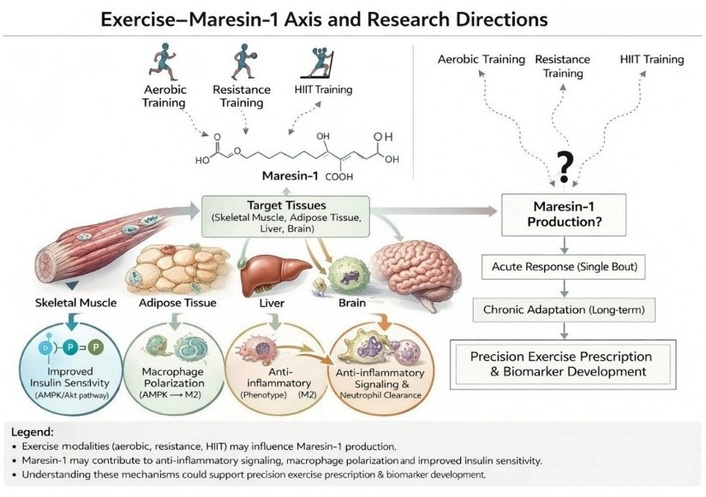
The multifaceted biological roles of Maresin-1 and the proposed framework for future exercise investigations. HIIT: high-intensity interval training.
12-LOX: 12-lipoxygenase
AMPK: AMP-activated protein kinase
DHA: docosahexaenoic acid
HIIT: high-intensity interval training
MaR1: maresin-1
SIRT1: Sirtuin 1
SPM: specialized pro-resolving mediator
MA, AS, MNA, KA, AA, KH, and HZ: Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
Hassane Zouhal, who is the Guest Editor of Exploration of Immunology, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 609
Download: 37
Times Cited: 0
Francisco Pitanga
Marios Hadjicharalambous ... Nikolaos Zaras
