Affiliation:
1Clinical Immunology Unit, Department of Paediatrics, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Selangor 43400, Malaysia
2Advanced Medical Research in Allergy and Clinical Immunology (AMRAC), Hospital Sultan Abdul Aziz Shah, Universiti Putra Malaysia, Selangor 43400, Malaysia
ORCID: https://orcid.org/0009-0000-9834-8459
Affiliation:
3Department of Community Health, Advanced Medical and Dental Institute, Universiti Sains Malaysia, Bertam, Pulau Pinang 13200, Malaysia
ORCID: https://orcid.org/0000-0003-2310-6767
Affiliation:
1Clinical Immunology Unit, Department of Paediatrics, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Selangor 43400, Malaysia
2Advanced Medical Research in Allergy and Clinical Immunology (AMRAC), Hospital Sultan Abdul Aziz Shah, Universiti Putra Malaysia, Selangor 43400, Malaysia
Email: intanhakimah@upm.edu.my
ORCID: https://orcid.org/0000-0003-4614-0140
Explor Immunol. 2026;6:1003248 DOI: https://doi.org/10.37349/ei.2026.1003248
Received: July 22, 2025 Accepted: March 27, 2026 Published: April 20, 2026
Academic Editor: Calogero Caruso, University of Palermo, Italy
Recurrent infections in children often prompt evaluation for primary immunodeficiency diseases, particularly those affecting humoral immunity. Assessment of memory B cell subsets and vaccine-specific antibody responses provides critical insight into long-term protective immunity. While peripheral blood mononuclear cell (PBMC) isolation is common, direct whole blood staining offers faster processing, reduced cell loss, and better preservation of fragile B cell populations. This comprehensive protocol describes a standardised whole blood flow cytometry method for memory B cell phenotyping, alongside ELISA-based measurement of vaccine-specific antibody titres as part of assessing children with recurrent infections.
Recurrent infections in children are a common clinical challenge and may signal underlying primary immunodeficiency diseases (PIDs), particularly those affecting humoral immunity. Antibody deficiencies are the most frequently diagnosed PIDs, with many patients presenting with recurrent sinopulmonary infections such as sinusitis, otitis media, and pneumonia [1, 2]. Evaluation of these patients requires both quantification of B cell subsets and assessment of functional antibody responses to vaccines [3–5].
Memory B cells play a central role in long-term immune protection, enabling rapid recall responses upon re-exposure to pathogens or vaccines. Reduced memory B cell populations are associated with impaired humoral immunity and increased susceptibility to infections [6, 7]. Similarly, inadequate vaccine-induced antibody responses may indicate specific antibody deficiency (SAD) [8–10].
Memory B cells can be further categorized into switched memory B cells (CD19⁺CD27⁺IgM⁻IgD⁻), non-switched memory B cells (CD19⁺CD27⁺IgM⁺IgD⁺), IgM-only memory B cells (CD19⁺CD27⁺IgM⁺IgD⁻), and IgD-only memory B cells (CD19⁺CD27⁺IgM⁻IgD⁺) [11]. The activation of memory B cells could be generated by T cell-dependent pathways. Once activated, memory B cells can quickly recall and heighten antibody production against specific pathogens upon subsequent exposures, whether through natural infection or vaccination [12, 13]. Consequently, a reduced population of memory B cells can lead to increased susceptibility to recurrent infections due to impaired humoral immunity [14].
Therefore, identifying the B cell subsets and profiling of specific antibody responses to vaccines are important for assessing children with recurrent infections, particularly those suspected of having PIDs [15]. Conventional approaches typically rely on peripheral blood mononuclear cell (PBMC) isolation for B cell analysis, which is labour-intensive, time-consuming, and may result in cell loss or compromise fragile B cell populations [16]. Direct whole blood staining offers a simpler, faster, and more reliable alternative. In resource-limited countries such as Malaysia, the high cost and limited availability of serotype-specific pneumococcal antibody assays make it difficult to evaluate anti-pneumococcal polysaccharide (PCP) antibody responses in children with recurrent infections. Under these circumstances, assessing memory B cell subsets offers a valuable alternative or complementary approach within the immunological work-up to determine immune competence. This evaluation is particularly relevant for diagnosing IgG subclass deficiency (IGGSD) and SAD. Previous studies have shown that children with SAD often have lower proportions of IgM-memory and class-switched memory B cells [17], while patients with IGGSD display alterations in B cell subsets, especially within the naïve and memory B cell compartments, compared to healthy controls [18].
Despite the recognized importance of memory B cell analysis and vaccine-specific antibody measurement in the evaluation of children with recurrent infections, many published immunophenotyping workflows utilize PBMC isolation for cellular immune analysis, including immunophenotyping and immunometabolic profiling [19, 20]. PBMC-based workflows are labour-intensive, time-consuming, require larger blood volumes, and are particularly vulnerable to cell loss and pre-analytical variability, limiting their practicality in paediatric settings and resource-constrained laboratories. To address this methodological gap, we describe a standardized step-by-step protocol that integrates direct whole blood flow cytometric phenotyping of memory B cell subsets with ELISA-based quantification of pneumococcal and tetanus-specific IgG responses. Although various vaccines are available to evaluate the specific antibody responses, polysaccharide pneumococcal and tetanus vaccines have been frequently used to represent functional assessment of humoral immune competency to generate antigen-specific antibodies either through a T cell-independent or T cell-dependent pathway, respectively. This workflow provides a simpler and faster alternative to PBMC-based assays, minimizes sample manipulation, preserves fragile B cell populations, and requires smaller blood volumes. Importantly, the protocol is designed to be readily implemented by clinicians and laboratory immunologists, including those in settings where clinical immunology services are still developing, thereby supporting the establishment and expansion of reliable humoral immune assessment in both research and routine clinical laboratories, particularly for evaluating children with recurrent infections and suspected antibody deficiencies.
General consumables used in this protocol included plain gel tubes and K2 EDTA vacutainer tubes (Eppendorf, Germany), Falcon® 13 mm × 75 mm round bottom capped polystyrene test tube (Corning, United States), micropipettes (5 to 1,000 µL) with disposable tips (Eppendorf, Germany), disposable pipettes, FACS lysing solution 10× (BD Pharmingen™, United States), sheath fluid, stain buffer (0.2% bovine serum albumin in 0.09% sodium azide) (BD Pharmingen™, United States), and ultrapure water. The specific monoclonal antibodies, enzyme immunoassay kits, vaccines, instruments, and software required are listed in Table 1.
List of monoclonal antibodies, ELISA kits, vaccines, instruments, and software applied in this protocol.
| Resource | Company | Volume | Catalogue number |
|---|---|---|---|
| Monoclonal antibodies | |||
| CD19-PerCP-CY5.5 | BD Pharmingen™ | 10 µL | 340951 |
| IgD-PE | BD Pharmingen™ | 10 µL | 555779 |
| IgM-BB515 | BD Pharmingen™ | 5 µL | 564622 |
| CD24-APC-H7 | BD Pharmingen™ | 5 µL | 658331 |
| CD27-APC | BD Pharmingen™ | 5 µL | 337169 |
| CD38-PE-Cy7 | BD Pharmingen™ | 5 µL | 335790 |
| ELISA kits | |||
| VaccZyme™ anti-tetanus toxoid IgG | Binding Site, UK | 96-well plate | MK010.0 |
| VaccZyme™ anti-PCP IgG | Binding Site, UK | 96-well plate | MK012.0 |
| Vaccines | |||
| Pneumovax23, PCP vaccine | Merck Sharp & Dohme, Netherlands | 0.5 mL | - |
| Tetanus toxoid vaccine | Biofarma, Indonesia | 0.5 mL | - |
| Instruments & software | |||
| Flow cytometer (FACS Lyrics) | BD Biosciences, US | - | - |
| BD FACS Suite software | BD Biosciences, US | - | - |
| FlowJo software | BD Biosciences, US | - | - |
| Microplate reader (Multiskan FC) | Thermo Fisher Scientific, US | - | - |
| Microplate reader software (SkanIt) | Thermo Fisher Scientific, US | - | - |
| Automated plate washer (Wellwash) | Thermo Fisher Scientific, US | - | - |
PCP: pneumococcal polysaccharide.
This sampling schedule applies to participants undergoing immunological evaluation for suspected humoral immune dysfunction, rather than routine vaccination recipients.
On the day of vaccination (baseline visit), collect 3 to 5 mL of peripheral blood from each participant into two collection tubes: a plain tube for serum collection and an EDTA tube for flow cytometry analysis.
Administer the Pneumovax23 PCP vaccine and the tetanus toxoid vaccine in accordance with institutional immunization guidelines and standard clinical practice.
Process the EDTA blood sample immediately following collection for whole blood memory B cell phenotyping by flow cytometry. Samples should be maintained at room temperature and protected from light prior to processing to preserve cell integrity.
Centrifuge the plain blood tube at 3,000 rpm for 5 min within 2 hours of collection to obtain serum. Aliquot a minimum of 1 mL of serum into appropriately labelled microcentrifuge tubes for enzyme immunoassay analysis.
A follow-up visit was conducted 28 to 30 days after vaccination to collect a second peripheral blood sample using a plain tube for post-vaccination serum analysis in all vaccine recipients.
Store all pre- and post-vaccination serum aliquots at −20°C until batch analysis is performed. Repeated freeze-thaw cycles should be avoided to maintain antibody stability.
Prepare 1× FACS lysing solution by diluting the 10× stock solution in deionized water at a ratio of 1:10 (one part lysing solution to nine parts water) at room temperature.
Label polystyrene test tubes for each individual sample.
Pipette 100 µL of whole blood into each tube and add 5 mL of stain buffer. Gently mix to ensure a homogenous cell suspension.
Centrifuge the samples at 300 g for 5 min and carefully discard the supernatant (first washing step).
Repeat the washing step by resuspending the cell pellet in stain buffer, followed by centrifugation at 300 g for 5 min.
Add the appropriate volumes of fluorochrome-conjugated monoclonal antibodies as listed in Table 1 to each tube.
Gently vortex the tubes and incubate for 15 min at room temperature in the dark to allow antibody binding.
Add 2 mL of the prepared 1× FACS lysing solution to each tube.
Gently vortex and incubate for 10 min at room temperature in the dark to lyse erythrocytes.
Centrifuge the tubes at 300 g for 5 min and discard the supernatant.
Wash the cells twice with 2–3 mL of stain buffer, centrifuging at 300 g for 5 min after each wash.
Resuspend the final cell pellet in 500 µL of stain buffer.
Acquire samples by flow cytometry within 24 hours of staining. If immediate acquisition is not possible, store samples at 4°C in the dark until analysis.
Prior to sample acquisition, perform routine instrument QC and fluidics cleaning using FACS Flow™ and FACS Clean™ solutions in accordance with the manufacturer’s recommendations. Daily cytometer performance was verified using calibration beads to ensure consistent laser alignment, fluorescence sensitivity, and signal stability.
Compensation settings were established using single-stained compensation controls for each fluorochrome to correct for spectral overlap. Fluorescence-minus-one (FMO) controls were applied where appropriate to support accurate gating of dim populations.
A minimum of 50,000 lymphocyte events per sample were acquired to ensure sufficient resolution of B cell subsets.
Figure 1 demonstrates a gating strategy using the FlowJo software. Lymphocytes are first identified based on forward scatter (FSC) and side scatter (SSC) characteristics. Total B cells were defined as CD19⁺ events within the lymphocyte gate. Naïve and memory B cell compartments are initially distinguished based on IgD and CD27 expression. Further subclassification is performed using IgM, CD27, CD38, and CD24 markers to identify:
Naïve B cells (CD19⁺CD27⁻IgD⁺)
Transitional B cells (CD19⁺CD27⁻CD24++CD38++)
Total memory B cells (CD19⁺CD27⁺)
Non-switched memory B cells (CD19⁺CD27⁺IgD⁺IgM⁺)
Class-switched memory B cells (CD19⁺CD27⁺IgD⁻IgM⁻)
Plasmablasts (CD19⁺CD27⁺CD38⁺⁺)
Each cell captured will be analyzed according to the gated populations.
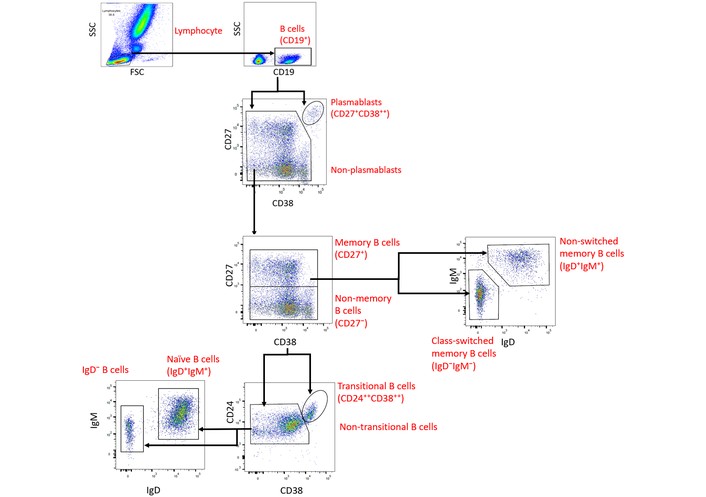
Representative flow cytometry gating strategy for B cell subsets and memory B cell phenotyping.
The frequency of each B cell subset displayed in the FlowJo software was expressed as a percentage, which could be derived in two ways.
Percentage of parent (gated) populations.
Percentage of total lymphocytes.
Based on the above gating hierarchy, the percentages for each B cell subset were calculated automatically. Although the absence of counting beads used in this protocol, the absolute counts for each B cell subset can be calculated by multiplying the percentage of gated populations by the absolute lymphocyte count obtained from the complete blood count analysis performed on the same blood sample. The formula derived for the absolute counts calculation is as follows:
Bring VaccZyme™ ELISA kit and all reagents to room temperature (20–25°C) approximately 60 min before use. The 96-well ELISA plates are pre-coated with PCP or tetanus toxoid antigens according to the manufacturer’s specifications. All reagents should be gently mixed before use.
Serum samples, calibrators, and manufacturer-provided high and low controls are run in duplicate. Serum samples are diluted 1:100 using the supplied diluent (10 µL serum + 1,000 µL diluent). The wash buffer is prepared by diluting the concentrated wash solution 1:20 with distilled water.
A total of 100 µL of calibrators, controls, and diluted samples were added to the designated wells and incubated for 30 min at room temperature. Plates are washed three times with 250–350 µL wash buffer per well using an automated plate washer. Subsequently, 100 µL of conjugate solution is added to each well and incubated for 30 min at room temperature, followed by three additional wash cycles. Then, 100 µL of tetramethylbenzidine (TMB) substrate was added and incubated for 30 min at room temperature in the dark. The reaction is stopped by adding 100 µL of stop solution (3 M phosphoric acid), and optical density (OD) is read at 450 nm using a microplate reader within 30 min.
Strict QC criteria are applied to every assay run. Calibrators and manufacturer-provided high and low controls are included in every batch run. Measured values for the high and low controls of anti-PCP or anti-tetanus IgG must fall within the acceptable ranges specified in the QC certificate supplied with each kit.
For each calibrator, sample, and control, the mean OD value of duplicate wells is calculated, and the coefficient of variation (CV) between duplicates is determined. Duplicate measurements are accepted only when the CV is < 15%; samples or controls exceeding this threshold are repeated.
Calibration curves for anti-PCP IgG and anti-tetanus toxoid IgG were generated using a four-parameter logistic (4PL) model in SkanIt™ microplate reader software (Thermo Fisher Scientific) to obtain the best fit, as illustrated in Figure 2. The standard curve must closely match the sigmoidal shape provided in the manufacturer’s QC certificate, and assay runs are accepted only when all calibrator points fall within the specified ranges, and the coefficient of determination (R2) is ≥ 0.98.
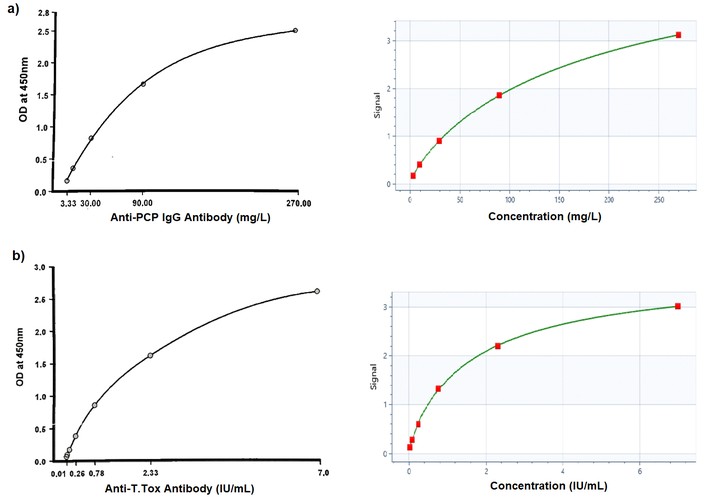
Representative calibration curves for specific antibody responses are shown: (a) anti-PCP IgG antibody and (b) anti-tetanus toxoid IgG antibody. The four-parameter logistic (4PL) curve generated using SkanIt™ microplate reader software (Thermo Fisher Scientific) is displayed on the right, while the manufacturer’s reference curve is shown on the left. The similar curve shape between the two graphs demonstrates that a valid and reliable standard curve was achieved. PCP: pneumococcal polysaccharide. Adapted from Binding Site with permission. © 2026 Thermo Fisher Scientific Inc and its subsidiaries. All rights reserved. Unauthorized use, reproduction or distribution is prohibited.
Any calibrator or sample point deviating markedly from the fitted curve is treated as an outlier and excluded from analysis. Calibrator values are pre-adjusted for the 1:100 sample dilution; therefore, no additional dilution correction is required.
Antibody concentrations for anti-PCP and anti-tetanus IgG in samples and controls were calculated directly from the fitted calibration curve. Results are expressed as mg/L for pneumococcal antibodies and IU/mL for tetanus antibodies. Pre- and post-vaccination antibody concentrations are compared within subjects to determine the adequacy of antibody responses. Protective status is determined based on the post-vaccination antibody titre.
A protective pneumococcal antibody response is defined as an antibody concentration ≥ 110 µg/mL, representing an adequate overall pneumococcal IgG response, while a protective tetanus IgG response is defined as > 0.15 IU/mL. In this protocol, deficient pneumococcal or tetanus responses are indicated by a post-vaccination antibody concentration that fails to reach at least a fourfold increase compared with the pre-vaccination level.
This protocol integrates direct whole blood staining for memory B cell phenotyping with ELISA-based quantification of vaccine-specific IgG responses, enabling simultaneous assessment of cellular and functional humoral immunity in children with recurrent infections or suspected antibody deficiencies. When performed according to the described QC parameters, the workflow is expected to generate biologically plausible B-cell subset distributions consistent with previously reported age-dependent distributions of B-cell subsets in healthy paediatric populations [21–24]. Interpretation of results requires age-stratified reference ranges, as B-cell subset composition varies throughout childhood. Population-specific reference intervals are recommended for clinical applications. Where external reference intervals are adopted, they should be verified in the local population to account for demographic and methodological differences.
Multiple studies have reported that naïve B cells typically comprise approximately 60–70% of circulating B cells in healthy children, while class-switched memory B cells account for approximately 10–20% and non-switched memory B cells for 10–15%, with plasmablasts generally representing less than 2% of total B cells under non-inflammatory conditions [21–24]. In children with recurrent infections or suspected humoral immunodeficiency, deviations from these distributions may be observed. Reduced proportions of class-switched memory B cells have been associated with impaired long-term immune memory and increased susceptibility to recurrent respiratory infections [14]. Similarly, increased proportions of transitional B cells may reflect delayed or defective B cell maturation, which has been described in patients with antibody deficiencies and IgG subclass deficiencies [7, 18]. These phenotypic patterns are consistent with previous reports demonstrating alterations in the memory B cell compartments in children with SAD and related disorders [7, 17].
It is important to acknowledge that the child’s age may influence B-cell subset composition and immune responsiveness. The paediatric immune system undergoes dynamic maturation, with younger children exhibiting higher proportions of naïve B cells and progressively increasing memory B cell compartments with advancing age [21–24]. This age-dependent variation is particularly relevant when interpreting the naïve and memory B cell populations. The present protocol does not specifically address age-related immunological differences but instead provides a standardized methodological approach applicable across paediatric age groups. Consequently, interpretation of B cell subset frequencies should be performed using age-matched reference ranges whenever available. Table 2 in this manuscript is intended as an example of how a laboratory can display the B cell subset results accordingly.
Example of a results template displaying the percentage and absolute count of B cell subsets.
| Patient’s result | Percentage (%) | Absolute count (× 106/L) | Age-specific reference ranges* |
|---|---|---|---|
| Total lymphocytes | |||
| Total B cells | |||
| Transitional B cells | |||
| Naïve B cells | |||
| Total memory B cells | |||
| Switched memory B cells | |||
| Non-switched memory B cells | |||
| Plasmablasts |
* Reference ranges should be established for population-specific and age-dependent conditions to avoid any potential bias in interpreting the results.
ELISA-based measurement of PCP and tetanus toxoid-specific IgG provides a functional assessment of humoral immune competence. In immunologically healthy children, post-vaccination antibody concentrations are expected to reach protective thresholds and demonstrate a robust rise compared with baseline levels [8, 9, 25, 26]. Protective responses are typically defined as pneumococcal IgG concentrations ≥ 110 µg/mL and tetanus IgG concentrations > 0.15 IU/mL, and normal antibody responses by a minimum fourfold increase from pre-vaccination levels [9, 27, 28]. Table 3 represents how the outcome can be displayed in formal laboratory results. The antibody response findings must correlate clinically, especially in evaluating vaccine responsiveness in children with recurrent infections.
Example of displaying the result of specific antibody responses to pneumococcal and tetanus vaccines.
| Test | Result | Unit | Ref. range | Assays |
|---|---|---|---|---|
| Pneumococcal | VaccZyme™ PCP IgG | |||
| Pre | mg/L | - | ||
| Post | mg/L | > 110 | ||
| Tetanus | VaccZyme™ Tetanus Toxoid IgG | |||
| Pre | IU/mL | - | ||
| Post | IU/mL | > 0.15 | ||
PCP: pneumococcal polysaccharide.
Children with SAD fail to achieve adequate protective antibody concentrations or demonstrate a sufficient post-vaccination increment despite normal total immunoglobulin levels [7–9, 27]. Such impaired vaccine responses have been consistently reported in paediatric cohorts with recurrent infections and are often accompanied by abnormalities in memory B cell subsets, particularly reduced IgM-memory and class-switched memory B cells [7, 17]. Therefore, the integration of memory B cell phenotyping with vaccine-specific antibody quantification in this protocol supports a more comprehensive assessment of humoral immunity and enhances interpretive confidence by linking cellular immune involvement with functional antibody production.
Overall, the workflow is intended to facilitate reproducible implementation, particularly for use in clinical and research laboratories establishing or expanding immunological assessment of children with recurrent infections.
This protocol provides a standardized workflow for measuring memory B cells and vaccine-specific antibody responses in children with recurrent infections. However, several limitations should be acknowledged. First, this protocol is designed to provide supportive immunological insights and may help indicate the presence of an underlying humoral immune abnormality; however, it is not intended to serve as a stand-alone diagnostic tool for primary immunodeficiency. All results should therefore be interpreted in the context of clinical findings and routine immunological investigations, in accordance with established diagnostic guidelines. Second, the method requires fresh whole blood; delayed handling or transport may compromise B-cell viability and staining quality, and the protocol is not suitable for settings where PBMC cryopreservation is preferred. Third, flow cytometry results may vary depending on instrument configuration and fluorochrome compensation, necessitating local optimization. Finally, the manuscript does not include representative comparative data between healthy controls and patients with confirmed PID. While this limits demonstration of disease-specific patterns, the primary aim of the study is to present a standardized methodological framework rather than diagnostic validation. Future studies applying this protocol to well-characterized patient cohorts will be essential to establish disease-associated immunophenotypic patterns.
This protocol presents a standardized workflow integrating whole blood memory B cell phenotyping with vaccine-specific antibody quantification to facilitate comprehensive evaluation of humoral immunity, especially among children with recurrent infections. By combining cellular immunophenotyping with functional serological assessment, the protocol provides complementary insights into both the structural integrity of the B cell compartment and the capacity to mount antigen-specific antibody responses. Such an integrated approach is particularly relevant in the clinical investigation of suspected PID, where isolated phenotypic or serological abnormalities may not fully capture immune competence, hence leading to missed/under interpretation of PID.
Flow cytometric analysis using the described gating strategy allows clear discrimination of major B cell subsets, including naïve B cells, transitional B cells, class-switched memory B cells, non-switched memory B cells, and plasmablasts. In healthy children, naïve B cells typically constitute the predominant population, reflecting ongoing B cell development, while memory B cells represent a smaller but stable fraction of circulating B cells [22, 29]. Memory B cell subsets, particularly class-switched memory B cells have been increasingly recognized as important indicators of long-term humoral immune competence and correlates of vaccine responsiveness. Several studies have demonstrated that children with SAD or IGGSD frequently exhibit diminished memory B cell compartments, particularly reduced class-switched memory B cells and IgM memory B cells, which have been consistently associated with impaired vaccine responsiveness and increased susceptibility to recurrent infections in paediatric populations [17, 30]. In common variable immunodeficiency (CVID) patients, reduced frequencies of CD27⁺ memory B cells are associated with impaired antigen-specific memory responses and suboptimal vaccine serology outcomes [31]. In this context, the combined assessment of B cell subset composition alongside post-vaccination antibody titres strengthens clinical interpretation by enabling differentiation between defects in B cell maturation and functional antibody production, enabling a more comprehensive interpretation of immune dysfunction than either approach alone.
PBMC isolation using Ficoll density gradient centrifugation remains a widely used method for immunophenotyping. While PBMC-based assays provide high-quality cellular preparations, they are labour-intensive, time-consuming, and prone to cell loss, particularly of fragile populations such as plasmablasts [32]. Delayed processing can further compromise cell viability and functionality, which is particularly challenging in paediatric settings. In contrast, whole blood lyse-wash staining offers several practical advantages. It minimizes sample handling, reduces processing time, and allows same-day flow cytometric acquisition [16]. Although the relative frequencies of most B cell subsets are generally preserved following PBMC isolation, the whole blood approach enhances operational efficiency and is more feasible in routine clinical laboratories, especially in resource-limited environments. This protocol therefore prioritizes practicality and reproducibility while maintaining reliable immunophenotypic resolution.
In future studies, several enhancements may further strengthen the utility of this protocol. Expansion of the antibody staining panel to include additional immunoglobulin subclasses, such as IgA1, IgA2, IgG1, IgG2, IgG3, IgG4, and IgE, would enable more detailed characterization of humoral immune defects, particularly in patients with subclass deficiencies. Furthermore, incorporation of antigen-specific memory B cell staining represents a promising direction for directly assessing vaccine-induced B cell memory. Antigen-specific probes are already available for tetanus toxoid and could potentially be extended to other clinically relevant vaccines, providing a direct cellular correlate of vaccine responsiveness. Such advancements would deepen mechanistic understanding of humoral immune dysfunction and further enhance the clinical relevance of the integrated workflow.
In summary, this protocol offers a practical and clinically meaningful framework for evaluating of memory B cell subsets and vaccine-specific antibody responses in children with recurrent infections. While not intended to replace established serotype-specific antibody assays, the protocol provides a feasible and reproducible framework that is particularly valuable in routine clinical immunology laboratories and resource-limited settings. In such settings, memory B cell subset profiling can serve as an important complementary marker of immune competence. The protocol also supports rapid turnaround time, facilitating timely clinical decision-making in paediatric patients presenting with recurrent infections. Importantly, the combined cellular and serological approach enhances diagnostic confidence. Future studies incorporating age-stratified reference data and patient cohorts with defined immunodeficiencies will further enhance its clinical applicability and diagnostic utility.
4PL: four-parameter logistic
CV: coefficient of variation
IGGSD: IgG subclass deficiency
OD: optical density
PBMC: peripheral blood mononuclear cell
PCP: pneumococcal polysaccharide
PIDs: primary immunodeficiency diseases
QC: quality control
SAD: specific antibody deficiency
We would like to thank the Clinical Immunology Centre (CIC) team from Hospital Sultan Abdul Aziz Shah (HSAAS), Universiti Putra Malaysia for their excellent technical assistance.
JJ: Methodology, Writing—original draft. SMM: Writing—review & editing, Supervision. IHI: Conceptualization, Methodology, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
This study protocol was reviewed and approved by the National Medical Research Register (NMRR-19-2299-49077), Medical Research & Ethics Committee (MREC), Ministry of Health Malaysia and Ethics Committee for Research Involving Human Subject, Universiti Putra Malaysia (JKEUPM-2023-337), in accordance with the Declaration of Helsinki (2013).
Written informed consent will be obtained from all the parents/guardians of the children prior to any procedures.
Not applicable.
All data presented or cited in this study were traceable directly to the reference sources.
This protocol was funded by the Fundamental Research Grant Scheme (FGRS/1/2018/SKK02/UPM/02/1, 04-01-18-1973FR), Ministry of Higher Education, Malaysia and Geran Inisiatif Putra Siswazah (GP-IPS/2022/9739900), Universiti Putra Malaysia. Both funders had no role in protocol setup, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 453
Download: 8
Times Cited: 0
