Affiliation:
1Department of Biochemistry, Saraswati Dental College and Hospital, Lucknow 226028, Uttar Pradesh, India
Email: ghoshdevlin6@gmail.com
ORCID: https://orcid.org/0000-0003-1167-6009
Affiliation:
2Department of Basic Science, Saraswati Dental College and Hospital, Lucknow 226028, Uttar Pradesh, India
ORCID: https://orcid.org/0009-0009-9752-7565
Affiliation:
2Department of Basic Science, Saraswati Dental College and Hospital, Lucknow 226028, Uttar Pradesh, India
ORCID: https://orcid.org/0009-0001-9611-6256
Explor Immunol. 2026;6:1003238 DOI: https://doi.org/10.37349/ei.2026.1003238
Received: October 28, 2025 Accepted: February 13, 2026 Published: March 11, 2026
Academic Editor: Roberto Paganelli, G. d’Annunzio University, Italy
Short-chain fatty acids (SCFAs), primarily acetate, propionate, and butyrate, are microbial metabolites generated through the fermentation of dietary fibers by the gut microbiota and are increasingly recognized as critical regulators of host immune homeostasis. Beyond their metabolic roles, SCFAs exert potent immunomodulatory effects across mucosal tissues, including the periodontium, by shaping both innate and adaptive immune responses. This review synthesizes current evidence on the dual roles of SCFAs in periodontal health and disease, with particular emphasis on the contrasting effects of systemically derived versus locally accumulated SCFAs within periodontal tissues. Gut-derived SCFAs absorbed into the circulation modulate immune function through activation of G protein-coupled receptors (GPR41, GPR43, and GPR109A) and inhibition of histone deacetylases. These pathways promote regulatory T cell differentiation, skew macrophage polarization toward anti-inflammatory M2 phenotypes, and regulate neutrophil and dendritic cell activity. These effects support immune tolerance, epithelial barrier integrity, and resolution of inflammation, thereby contributing to periodontal homeostasis. In contrast, SCFAs produced locally within periodontal pockets can reach millimolar concentrations that disrupt epithelial and fibroblast function, induce oxidative stress, and amplify inflammatory signalling, ultimately driving tissue destruction and disease progression. Emerging evidence links gut microbial composition and systemic SCFA availability to periodontal inflammation through immune and neuroimmune crosstalk, supporting a biologically plausible gut-oral axis. Translational strategies aimed at harnessing the immunoregulatory properties of SCFAs are critically evaluated in this review. While these approaches are promising, challenges related to dosing, delivery, inter-individual variability, and limited human interventional data remain, underscoring the need for rigorously designed translational studies.
Short-chain fatty acids (SCFAs), notably acetate (C2), propionate (C3), and butyrate (C4), are metabolic byproducts of anaerobic fermentation of dietary polysaccharides by gut microbiota. These metabolites serve as essential energy sources for colonocytes, contribute to mucosal barrier maintenance, regulate luminal pH, and exert broad effects on host metabolic and immune pathways [1]. Predominant SCFA-producing taxa include Faecalibacterium, Bacteroides, Prevotella, Lactobacillus, and Akkermansia [2]. Among the gut-derived SCFAs, butyrate plays a particularly important role in immune regulation by enhancing regulatory T cell (Treg) function through histone deacetylase (HDAC) inhibition and GPR109A signalling, leading to an anti-inflammatory cytokine profile characterized by interleukin (IL)-10 secretion [3–5].
In the context of periodontal disease, which arises from microbial dysbiosis and host inflammatory responses to pathogens such as Porphyromonas gingivalis (P. gingivalis) and Fusobacterium nucleatum, SCFAs have a dual role. Locally, within periodontal pockets, SCFAs accumulate at millimolar concentrations, especially butyrate and propionate, derived from bacterial metabolism. These elevated levels can be cytotoxic to epithelial and fibroblast cells, compromise junctional complexes, and promote inflammatory signalling and apoptosis [6, 7]. Conversely, systemically derived SCFAs are absorbed into the circulation from the colon, interact with immune receptors to dampen pro-inflammatory cytokines, enhance barrier integrity at distal sites, and potentially foster periodontal tissue homeostasis [8]. Emerging evidence suggests that gut-derived SCFAs may influence periodontal inflammation through neuroimmune pathways in addition to direct systemic immunomodulation (Figure 1). SCFAs such as acetate, propionate, and butyrate can enter systemic circulation and have been shown to cross the blood-brain barrier via monocarboxylate transporters, where they modulate neuroinflammatory responses, microglial activation, and blood-brain barrier integrity. By regulating central immune signalling and autonomic outflow, SCFAs may indirectly shape peripheral immune homeostasis, including immune responses within periodontal tissues. Neuroimmune crosstalk can influence the trafficking, activation, and polarization of immune cells such as macrophages, dendritic cells, neutrophils, and T cells, thereby contributing to a shift toward anti-inflammatory phenotypes, including enhanced Treg responses and M2 macrophage polarization. Although direct evidence linking SCFA-mediated neuroimmune signalling to periodontal outcomes remains limited, this axis represents a biologically plausible and emerging mechanism connecting gut microbial metabolites to periodontal immune regulation and warrants further investigation.
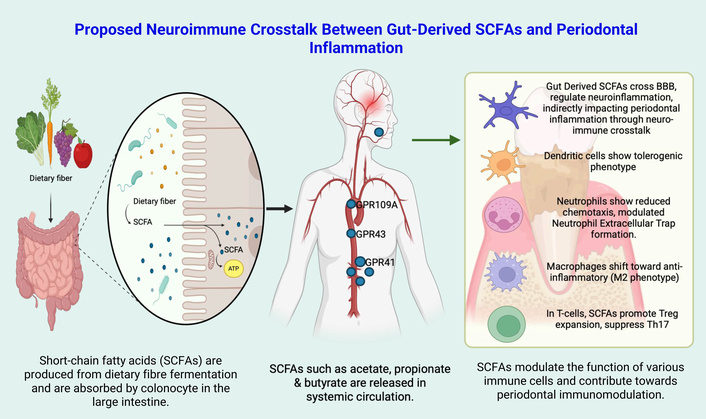
Schematic representation of gut-derived SCFAs influencing periodontal inflammation through systemic immune modulation and potential neuroimmune crosstalk via the blood-brain barrier (BBB). “Proposed Neuroimmune Crosstalk Between Gut-Derived SCFAs and Periodontal Inflammation” created in BioRender. Kumar, A. (2026) https://BioRender.com/ywvm97a is licensed under CC BY 4.0. Treg: regulatory T cell.
Recognizing this dichotomy between detrimental local effects and beneficial systemic actions is essential. This review explores how gut-derived SCFAs shape innate and adaptive immune responses, contrasts them with the pathological influence of locally generated SCFAs in the periodontium, and evaluates emerging strategies to leverage systemic SCFA activity for improving periodontal health.
Macrophages in periodontal tissues adopt a spectrum of activation states, with classically-activated (M1) and alternatively-activated (M2) subsets dominating the pro-inflammatory and reparative phases of periodontitis, respectively. In healthy tissues, resident macrophages contribute to homeostasis and microbial surveillance, but in disease conditions, dysbiotic biofilms trigger shifts toward an M1-dominant milieu that exacerbates inflammation and bone loss [9, 10]. Conversely, M2 macrophages secrete anti-inflammatory mediators and pro-resolving factors that restrain tissue destruction, promote efferocytosis, and support osteoblast activity to restore the periodontium [11, 12].
Under the influence of interferon-γ (IFN-γ), lipopolysaccharide (LPS), and periodontal pathogens, most notably P. gingivalis, monocytes differentiate into M1 macrophages characterized by high expression of inducible nitric oxide synthase (iNOS), CD86, and secretion of tumor necrosis factor-α (TNF-α), IL-1β, and IL-6 [9, 10]. In vitro and animal model studies demonstrate that M1 skewed macrophages amplify osteoclastogenesis via RANKL upregulation on osteoblasts and stromal cells, accelerating alveolar bone resorption [10, 13]. Clinically, elevated M1/M2 ratios in gingival biopsies correlate with probing depth and attachment loss, underscoring the link between M1 predominance and disease severity [14].
M2 macrophages, polarized by IL-4, IL-10, and IL-13, express arginase-1 (Arg-1), CD206, and secrete IL-10 and transforming growth factor beta (TGF-β), fostering resolution of inflammation and matrix remodeling [10, 11]. In murine periodontitis models, localized injections of ex vivo-generated M2 macrophages attenuated osteoclastic activity and preserved alveolar bone height, highlighting their therapeutic potential [11]. More recently, systemic treatment with dimethyl fumarate (DMF), which activates NRF2 signalling, has been shown to promote a shift in macrophages toward the anti-inflammatory M2 phenotype, enhance mitophagy, and significantly reduce bone loss in rodent models. These findings suggest that small-molecule therapies like DMF can help restore the balance between M1 and M2 macrophage responses [12].
Quantitative immunohistochemistry of human periodontal biopsies reveals that chronic periodontitis sites show significantly higher densities of CD86⁺ M1 macrophages relative to CD206⁺ M2 cells, compared to gingivitis or healthy controls [13]. Mapping of transcriptional profiles of periodontal macrophages identified Akt2 as a key regulator that promotes M1 polarization; Akt2-deficient mice exhibited reduced bone loss and lower M1/M2 ratios, pointing to intracellular signalling nodes as intervention targets [10].
SCFAs signal through distinct GPCRs. Acetate primarily activates GPR43 [free fatty acid receptor 2 (FFAR2)], whereas propionate and butyrate also engage GPR41 (FFAR3), with butyrate showing preferential activity at GPR109A [5]. These receptors are expressed across multiple immune cell populations, including neutrophils, monocytes/macrophages, dendritic cells, and T lymphocytes, where they initiate anti-inflammatory signalling cascades [15]. Activation of GPR43 enhances neutrophil movement to sites of infection and aids microbial clearance, while at the same time reducing pro-inflammatory cytokines such as IL-6, IL-12, and TNF-α through macrophage-dependent mechanisms [16]. Similarly, activation of GPR109A by butyrate promotes tolerogenic dendritic cells that support Treg development and help limit systemic inflammation. In addition, SCFA signalling through GPCRs regulates inflammasome activity, as GPR43-dependent calcium signalling controls NOD-, LRR-, and pyrin domain-containing protein 3 (NLRP3) inflammasome assembly and IL-1β release without triggering pyroptotic cell death under normal conditions [17].
Due to their small size and ability to enter cells and the nucleus, SCFAs—especially butyrate—act as inhibitors of class I and II HDACs. This leads to increased histone acetylation at genes involved in immune regulation, such as forkhead box P3 (FOXP3) and IL-10, promoting the differentiation of naïve CD4⁺ T cells into Tregs with anti-inflammatory functions [3]. At the same time, butyrate suppresses Th17 and Th1 cell development, reducing IL-17 and IFN-γ production in inflammatory settings. In innate immune cells, including macrophages and dendritic cells, HDAC inhibition decreases the expression of pro-inflammatory mediators such as nitric oxide synthase 2 (Nos2), IL-6, IL-12, and TNF, thereby favouring a more tolerogenic, anti-inflammatory phenotype [4].
SCFAs, particularly butyrate, play a central role in preserving the integrity of the intestinal epithelial barrier, the body’s first line of defence against invading pathogens and luminal antigens. One mechanism involves the stabilization of hypoxia-inducible factor-1α (HIF-1α), a transcription factor that supports epithelial survival under hypoxic conditions [18]. Stabilized HIF-1α enhances the expression of genes critical for barrier maintenance, including those regulating tight junction proteins such as claudin-1, occludin, and zonula occludens-1 (ZO-1) [19]. By reinforcing these junctional structures, SCFAs reduce microbial translocation and the risk of systemic inflammation [20]. Beyond barrier stabilization, butyrate directly influences host microbe interactions by inducing antimicrobial peptides (AMPs), such as RegIIIγ and β-defensins, without disrupting commensal communities. This induction occurs via GPR43 signalling, which activates downstream mitogen-activated protein kinase (MAPK) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathways in epithelial cells [21].
SCFAs also fine-tune innate immune sensors by modulating inflammasome activity. Through calcium-dependent signalling downstream of GPR41/43, they regulate NLRP3 inflammasome assembly. This regulation prevents excessive IL-1β production, a potent pro-inflammatory cytokine capable of driving tissue injury and systemic inflammation if unchecked. In this way, SCFAs function as molecular brakes on innate immune hyperactivation, preserving their protective roles while limiting pathological outcomes [22].
SCFAs also influence neutrophil biology, a cornerstone of the innate immune response. Neutrophils are among the first immune cells recruited to infection or injury sites, where they eliminate pathogens. Acetate, propionate, and butyrate activate GPR43 (FFAR2), a receptor abundantly expressed on neutrophils, to regulate their migration and effector functions [23]. Engagement of GPR43 enhances neutrophil chemotaxis, allowing precise navigation toward chemokine gradients and microbial signals within inflamed tissues [24]. This targeted recruitment facilitates rapid deployment of innate defence at the site of infection [25]. In addition to recruitment, SCFAs boost neutrophil effector functions. They increase phagocytic activity and enhance the oxidative burst by promoting reactive oxygen species (ROS) production, thereby strengthening microbial killing [26]. SCFAs also stimulate the formation of neutrophil extracellular traps (NETs), web-like DNA structures coated with antimicrobial proteins that immobilize and kill microbes [27]. However, excessive neutrophil activation and ROS release can damage host tissues, disrupting epithelial integrity and perpetuating inflammation. This duality is evident in inflammatory bowel disease (IBD), where uncontrolled neutrophil responses contribute to mucosal erosion and tissue injury. SCFAs appear to balance these opposing outcomes by supporting efficient pathogen clearance while restraining pathological inflammation, thereby sustaining neutrophil function in a protective, rather than destructive, manner [28].
Elevated levels of SCFAs, particularly propionate and butyrate, have been detected in the gingival crevicular fluid (GCF) of patients with periodontitis, often reaching millimolar concentrations and correlating with the presence of periodontal pathogens such as P. gingivalis, Treponema denticola [29]. At these local concentrations, butyrate compromises epithelial integrity by disrupting tight junctions and adhesion proteins (e.g., cadherins), while also activating caspase-3 and pyroptotic pathways in gingival epithelial cells, thereby accelerating tissue destruction [30]. Gingival fibroblasts similarly respond to high SCFA exposure with increased production of TNF-α, ROS, matrix metalloproteinases (MMPs), and enhanced apoptotic activity, all of which contribute to periodontal tissue degradation [31]. These observations highlight the dual nature of SCFA activity; at pathological, millimolar levels within the periodontal pocket, they exert cytotoxic effects on epithelial and stromal cells, weakening barrier function and amplifying pro-inflammatory mediator release. In contrast, at systemic or gut-derived concentrations, typically within the micromolar to low millimolar range, SCFAs promote anti-inflammatory immunity, reinforce epithelial barrier function, and support the induction of regulatory immune phenotypes [32]. Accordingly, the impact of SCFAs depends on their concentration and tissue context, resulting in either pathogenic or homeostatic immune effects.
Beyond their local effects, SCFAs also contribute to systemic immune regulation through the gut-oral axis. Experimental studies show that oral infection with P. gingivalis alters gut microbial composition, leading to expansion of SCFA-producing taxa and increased fecal and systemic SCFA levels. These systemic changes are associated with enhanced Treg frequencies, reduced inflammatory responses, and even improved outcomes such as prolonged skin graft survival in murine models, demonstrating that gut-derived SCFAs can modulate immunity at distal mucosal sites, including the oral cavity [33]. Furthermore, interventions designed to boost gut SCFA production, such as administration of Clostridium butyricum or dietary fiber enrichment, attenuate alveolar bone loss and systemic inflammatory markers in experimental periodontitis models, underscoring their therapeutic potential in periodontal disease [34].
Although several clinical studies report altered gut microbiota and elevated SCFAs in subjects with periodontitis, the human evidence to date is predominantly associative and does not establish causality between gut-derived SCFAs and oral disease progression. Clinical measurements show that elevations of propionate and butyrate in GCF correlate with probing depth and attachment loss, but these local increases most likely reflect pocket microbiology rather than systemic SCFA flux alone [35].
In mice, fecal microbiota transplantation and targeted microbiome manipulation show that dysbiotic gut communities can worsen systemic inflammation and alveolar bone loss [36]. Conversely, interventions that enhance butyrate availability, such as supplementation with Clostridium butyricum or systemic SCFA administration, reduce inflammatory responses and bone loss in rodent periodontitis models, supporting a causal role for gut-derived metabolites in shaping periodontal outcomes [37]. These findings are mechanistically supported by studies showing that SCFAs regulate bone-relevant immune pathways, including osteoclast differentiation and macrophage-driven inflammatory processes, ultimately influencing bone resorption in vivo. Together, these systemic immunometabolic effects provide a biologically plausible mechanism through which gut-derived SCFAs can modulate immune responses and tissue remodelling at distal periodontal sites [38].
Important caveats remain, particularly the marked compartmental differences in SCFA concentrations. Circulating SCFAs are generally present at low, micromolar levels, whereas GCF and plaque can contain millimolar concentrations that directly damage epithelial and stromal cells through cytotoxic and barrier-disruptive mechanisms. Consequently, systemic SCFA elevations that support immune modulation are modest relative to the locally generated, pathogenic SCFA levels within periodontal pockets, complicating their therapeutic translation [29]. So far, human interventional studies testing whether modulation of gut-derived SCFAs (through diet, probiotics, FMT, or SCFA supplementation) confers meaningful periodontal benefit are limited. To move beyond correlative evidence, we emphasize the importance of clearly distinguishing local cytotoxic from systemic immunomodulatory SCFA effects, highlighting causal preclinical and clinical studies summarized in Table 1, and calling for early-phase human trials that integrate systemic and site-specific SCFA measurements with immune and bone-related endpoints to determine their true impact on periodontal biology.
Recent preclinical and clinical evidence linking short-chain fatty acids (SCFAs) to periodontal outcomes.
| Model/population | SCFA-related intervention | Periodontal-associated outcomes | Evidence level | Reference |
|---|---|---|---|---|
| Mouse FMT study | Feces from high-fat diet/obese donor mice transferred to recipients | Recipients developed elevated serum uric acid, systemic inflammation, and increased alveolar bone loss in ligature-induced periodontitis compared with controls | Preclinical (causal) | [36] |
| Mouse periodontitis model | Clostridium butyricum administration | Reduced alveolar bone loss and inflammatory markers in diabetes mellitus-associated periodontitis. | Preclinical (interventional) | [37] |
| Mouse ligature-induced periodontitis model | Dietary fiber-induced SCFA elevation | Attenuation of periodontal inflammation and modulation of systemic inflammatory and metabolic markers associated with altered gut microbiota. | Preclinical (dietary intervention) | [39] |
| Human periodontitis patients vs. healthy controls | Quantification of GCF SCFAs (propionic acid and butyric acid) | Elevated butyrate/propionate correlated with disease severity | Human (observational) | [35] |
| Human gingival epithelial cells (Ca9-22) in vitro | High local butyrate/propionate exposure as periodontopathic bacterial metabolites | Butyrate/propionate induced epithelial cell death via HDAC inhibition, associated with enhanced autophagy and reactive oxygen species, leading to barrier disruption and cytotoxicity | In vitro (pathological) | [40] |
Summary of recent preclinical and clinical evidence on SCFAs in periodontal disease. The table outlines experimental models, SCFA-related interventions or measurements, key periodontal outcomes, and levels of evidence. SCFA effects reflect either beneficial systemic immunomodulation or pathological local effects at elevated periodontal concentrations. FMT: fecal microbiota transplantation; GCF: gingival crevicular fluid; HDAC: histone deacetylase.
Through receptor-mediated signalling and HDAC inhibition, SCFAs promote anti-inflammatory and tolerogenic responses that collectively contribute to reduced tissue destruction and bone resorption.
Experimental studies using RAW 264.7 macrophages and primary human monocytes demonstrate that exposure to SCFAs, particularly butyrate, significantly modulates their inflammatory responses. Butyrate suppresses LPS or P. gingivalis-induced expression of Nos2, IL-1β, TNF-α, and CXCL2, key mediators driving periodontal tissue destruction. These findings suggest that SCFAs dampen inflammatory cascades that would otherwise accelerate alveolar bone loss and connective tissue breakdown. Interestingly, butyrate does not act in a strictly suppressive manner; at low concentrations, it can paradoxically elevate IL-1β production, indicating a biphasic, dose-dependent effect that underscores the fine balance between protective and pathogenic immune outcomes in the periodontal niche [41, 42]. SCFAs also shape macrophage polarization, favoring a shift from a pro-inflammatory M1-like phenotype toward an anti-inflammatory M2-like state. This reprogramming is mediated by HDAC inhibition and GPCR signalling, which attenuate NF-κB activity while enhancing IL-10 secretion. These epigenetic and metabolic changes drive a transition from glycolysis to oxidative phosphorylation, reinforcing tolerogenic and tissue-protective functions [43]. Nevertheless, persistently high levels of SCFAs, as observed in advanced periodontitis, may exert cytotoxic or dysregulatory effects, complicating their protective role [44]. Figure 2 illustrates how gut microbial balance influences macrophage-mediated immune regulation through SCFA signalling. Under eubiotic conditions, SCFAs foster an anti-inflammatory M2 phenotype, reinforcing neuroimmune stability and mitigating periodontal tissue damage. Conversely, dysbiosis disrupts SCFA availability, skewing macrophages toward a pro-inflammatory M1 state that amplifies systemic inflammation and periodontal disease progression.
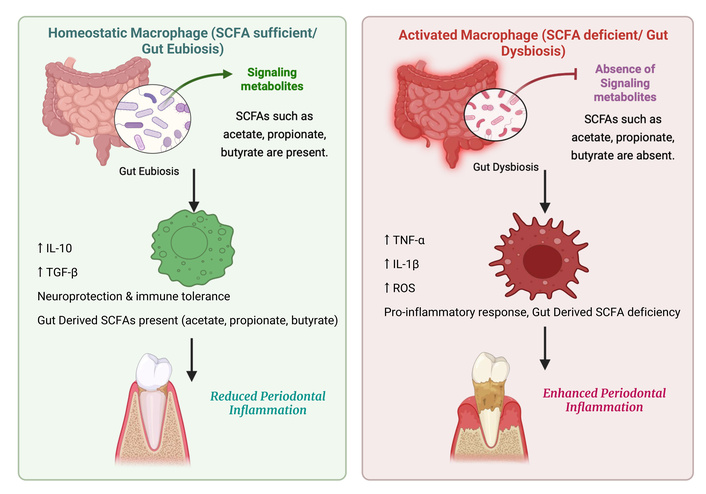
Comparative illustration of macrophage responses under gut eubiosis versus dysbiosis. During eubiosis, microbial fermentation of dietary fibers produces SCFAs that promote M2 macrophage polarization through GPR43/GPR109A signalling and HDAC inhibition, enhancing IL-10 and TGF-β secretion. These anti-inflammatory mediators support neuroprotection and contribute to reduced periodontal inflammation. In contrast, gut dysbiosis and the consequent depletion of SCFAs drive macrophage M1 polarization, leading to elevated TNF-α, IL-6, and IL-12 levels that exacerbate systemic and periodontal inflammation. SCFA: short-chain fatty acid; IL: interleukin; TGF-β: transforming growth factor beta; TNF-α: tumor necrosis factor-α; ROS: reactive oxygen species; HDAC: histone deacetylase. “Comparative illustration of macrophage responses under gut eubiosis versus dysbiosis” created in BioRender. Kumar, A. (2026) https://BioRender.com/ulv3rl0 is licensed under CC BY 4.0.
Fibroblasts and epithelial cells, essential for maintaining periodontal structure and barrier integrity, are highly susceptible to SCFA toxicity. At concentrations in the 1–5 mM range, butyrate and propionate impair fibroblast proliferation by inducing G0/G1 or G2/M cell cycle arrest through downregulation of cyclin B1, Cdc2, and Cdc25C, thereby disrupting tissue repair and regenerative capacity [45]. SCFA exposure also increases ROS generation, causing DNA and protein damage, thus triggering apoptosis through mitochondrial and caspase-dependent pathways. Recent reports further implicate butyrate in ferroptosis induction via ferritinophagy, linking iron-dependent cell death to periodontal oxidative stress [40]. Epithelial keratinocytes display similar vulnerabilities. In Ca9-22 cells, elevated SCFAs induce apoptosis, pyroptosis, and autophagy, accompanied by loss of tight junction proteins such as occludin and E-cadherin. This barrier disruption facilitates deeper bacterial penetration into the gingival tissues. Clinical studies confirm that butyrate levels in GCF are higher in periodontitis patients compared to healthy individuals, directly correlating with epithelial barrier breakdown [46, 47].
SCFAs strongly influence neutrophil function through GPR43 activation. Acetate, propionate, and butyrate act as ligands to drive chemotaxis via PI3Kγ, Rac2, and MAPK signalling, enhancing recruitment to infected sites. While this supports microbial clearance, excessive signalling leads to hyper recruitment, elevated ROS release, and protease activity; factors contributing to periodontal tissue injury [29]. Supporting this, GPR43-deficient mice exhibit uncontrolled neutrophil accumulation, elevated cytokine release, and aggravated inflammation during microbial challenge, emphasizing SCFA GPR43 signalling as a checkpoint for neutrophil homeostasis [48]. NETs are released during NETosis and serve as an important barrier against periodontal pathogens, but when NET formation is exaggerated or dysregulated, it contributes to tissue destruction by releasing proteases, histones, and ROS that damage epithelium and alveolar bone [49, 50].
In dendritic cells, butyrate induces a tolerogenic phenotype characterized by reduced major histocompatibility complex class II (MHC-II) and co-stimulatory molecule expression alongside increased IL-10 production. These tolerogenic dendritic cells promote Treg differentiation while suppressing Th1 and Th17 responses, contributing to immune tolerance within the periodontium. Thus, SCFAs orchestrate dual outcomes, enhancing localized innate defence yet promoting systemic immune regulation through dendritic cell programming [51].
Gut-derived SCFAs also regulate adaptive immunity by influencing CD4⁺ T cell differentiation. Butyrate enhances the generation of Tregs via HDAC inhibition, upregulating FOXP3 and IL-10 expression, hallmark features of an anti-inflammatory phenotype. Simultaneously, SCFAs attenuate Th1 and Th17 polarization, thereby reducing secretion of IL-17 and IFN-γ [3, 4]. Epigenetically, SCFAs increase acetylation at the FOXP3 promoter while repressing RORγt, the master regulator of Th17 differentiation, thus stabilizing Treg development and limiting pro-inflammatory effector responses in periodontal tissues. Importantly, these protective effects are Treg-dependent; in Treg-deficient settings, SCFAs fail to confer immunoregulation [4].
To translate these findings therapeutically, novel delivery approaches such as SCFA-loaded nanoparticles or liposomal butyrate are under development. These systems sustain Treg-promoting and anti-inflammatory effects while minimizing cytotoxicity, offering promise as adjunctive strategies in managing periodontal and systemic inflammatory diseases [52]. Thus, SCFAs have a significant impact on key immune cell populations relevant to periodontal health, as summarized in Table 2.
Immunomodulatory effects of gut-derived SCFAs on periodontal immune responses.
| Immune cell | Mechanism/receptor | SCFA effect | Periodontal outcomes | Key references |
|---|---|---|---|---|
| Macrophages | GPR43, GPR109A; HDAC inhibition | M2 polarization ↑; TNF-α, IL-6, IL-12 ↓; IL-10 ↑ | Anti-inflammatory shift; reduced bone resorption | [12, 41] |
| Neutrophils | GPR43; NET regulation | Chemotaxis ↑; excessive NETosis ↓ | Balanced pathogen control; limited tissue damage | [29, 49, 50] |
| Dendritic cells | GPR109A; HDAC inhibition | Tolerogenic phenotype; IL-12 ↓; Treg induction ↑ | Reduced adaptive overactivation | [4, 29, 52, 53] |
| T cells (Treg/Th17) | HDAC inhibition; FOXP3 acetylation | Tregs ↑; Th17/Th1 ↓; IL-17, IFN-γ ↓ | Restored immune tolerance | [4, 52] |
| Gingival fibroblasts | GPR41, GPR43; HDAC inhibition | IL-6, IL-8 ↓; collagen synthesis ↑; oxidative stress ↓ | Enhanced tissue repair; reduced inflammatory signaling | [31, 44, 48] |
| Gingival epithelial cells | GPR43; barrier modulation; HDAC inhibition | Tight junction integrity ↑; pro-inflammatory cytokines ↓ | Strengthened epithelial barrier; reduced microbial invasion | [30, 54, 55] |
Summary of the immunomodulatory effects of gut-derived SCFAs on periodontal immune responses, highlighting their role in promoting anti-inflammatory macrophage phenotypes and maintaining tissue homeostasis. SCFAs: short-chain fatty acids; HDAC: histone deacetylase; TNF-α: tumor necrosis factor-α; IL: interleukin; NET: neutrophil extracellular trap; Treg: regulatory T cell; FOXP3: forkhead box P3; IFN-γ: interferon-γ. ↑: upregulated; ↓: downregulated.
Increasing dietary intake of fermentable fibers, such as inulin and resistant starch, enhances colonic SCFA production, reinforcing barrier integrity and anti-inflammatory immune responses [56]. In preclinical models, high fiber diets reduce systemic inflammation and attenuate experimental periodontal bone loss by elevating butyrate levels. Similarly, administration of butyrate-producing probiotics, including Faecalibacterium prausnitzii and Clostridium butyricum raise systemic SCFA concentrations and diminishes inflammatory pathology in models of arthritis and periodontitis, highlighting their potential as adjuncts in periodontal care [57].
Direct SCFA delivery is being explored to bypass variability in host microbiota composition. Encapsulation techniques and prodrugs such as tributyrin allow systemic butyrate release while minimizing local cytotoxicity. In vitro, lipid-encapsulated butyrate supports dose-dependent Treg differentiation and reduces epithelial cell damage. Although human studies in periodontal disease remain limited, early phase trials in other inflammatory conditions suggest that SCFA supplementation is both safe and immunomodulatory [58].
SCFA levels may serve as useful biomarkers for both local and systemic immune homeostasis. In periodontal pockets, elevated butyrate and propionate in gingival crevicular fluid reflect microbial dysbiosis and tissue activity, whereas fecal or serum SCFA levels correlate with gut microbial resilience and lower inflammatory burden. Longitudinal studies are required to validate these signatures and to develop microbiome-targeted, personalized strategies for managing periodontal disease [59].
Although several routes exist to harness SCFAs for periodontal benefit, their comparative feasibility and readiness for clinical translation differ markedly. Dietary fiber/prebiotic approaches are the most advanced in humans. Animal and small clinical studies show that increasing fermentable fiber reliably raises systemic SCFA production and can reduce inflammatory biomarkers, but effects on periodontal endpoints are variable and slow to appear because of individual differences in baseline microbiota and adherence [60]. Probiotic strategies (including Clostridium butyricum strains) have demonstrated efficacy in rodent periodontitis models and show promise for restoring butyrogenic taxa, yet human evidence is limited and strain-, dose-, and formulation-dependent [37]. Direct systemic SCFA supplementation or prodrugs offers controlled dosing and proof-of-principle immunomodulation in preclinical models (including bone-protective effects), but rapid host absorption/metabolism and uncertain safety at therapeutic doses complicate translation [38]. Local, targeted periodontal delivery (mucoadhesive hydrogels, microparticles, nanoparticles) is attractive because it limits systemic exposure and can concentrate agents at the diseased site; however, the periodontal pocket’s fluid flow, salivary washout, and the small, highly diffusible nature of SCFAs require advanced encapsulation chemistries and mucoadhesive strategies to achieve sustained, sub-toxic release—most such platforms are still at the preclinical/proof-of-concept stage [61]. Engineered SCFA-bearing nanocarriers and enzyme-responsive SCFA pro-materials are emerging preclinical options that promise tuned pharmacokinetics and site-specific release but face formulation complexity, biocompatibility testing, and regulatory hurdles before human trials [62].
SCFAs, primarily butyrate, propionate, and acetate, are key microbial metabolites derived from dietary fiber fermentation. Beyond their role in host energy metabolism, they exert potent immunoregulatory effects across mucosal sites, including the oral cavity and gut [18]. Their anti-inflammatory activity is mediated in part by GPCR signalling. SCFAs activate receptors such as GPR41, GPR43, and GPR109A expressed on dendritic cells, neutrophils, macrophages, and epithelial cells. This signalling cascade reduces pro-inflammatory cytokines (IL-6, IL-1β, TNF-α) while enhancing IL-10 production, thereby promoting immune tolerance at barrier sites [53]. In addition, SCFAs function as HDAC inhibitors, leading to chromatin relaxation and transcriptional activation of immunoregulatory genes. This epigenetic effect fosters Treg differentiation and function, which restrains excessive immune activation against commensals and prevents tissue-damaging inflammation [63]. These properties are highly relevant to periodontitis, a chronic inflammatory disease driven by dysregulated host responses to biofilm. By limiting destructive immunity, SCFAs may simultaneously mitigate periodontal destruction and reduce systemic inflammatory burden, thereby influencing comorbidities such as cardiovascular disease and diabetes [64–66].
Reduced SCFA levels are consistently observed in conditions marked by dysbiosis and chronic inflammation, including inflammatory bowel disease, obesity, and metabolic syndrome [67, 68]. This suggests that SCFA insufficiency undermines mucosal immune tolerance, leaving tissues, including the gingiva, more susceptible to inflammatory pathology. IL-10 upregulation by SCFAs is particularly central, restraining macrophage and dendritic cell overactivation while curbing TNF-α and IL-6 production [52, 69, 70].
Butyrate is particularly effective in preserving epithelial barrier integrity. By upregulating tight junction proteins such as claudin-1, occludin, ZO-1, and junctional adhesion molecule (JAM), it reinforces epithelial cohesion and prevents paracellular leakage of bacterial components such as LPS [55, 71, 72]. Experimental studies demonstrate that butyrate protects intestinal epithelial cells from LPS induced disruption through activation of survival pathways, including Akt, AMPK, and mammalian target of rapamycin (mTOR), thereby sustaining transepithelial resistance and energy balance [73–75]. SCFAs also exhibit antimicrobial properties, suppressing bacterial virulence gene expression and reducing pathogenicity. While much of this evidence derives from gut models, similar mechanisms are likely in the oral cavity, given the shared mucosal biology [76]. In the context of periodontal disease, SCFAs may attenuate the virulence of keystone pathogens such as P. gingivalis while simultaneously strengthening epithelial defenses [77, 78].
This dual action, reinforcing the barrier while reducing microbial aggression, positions SCFAs as both preventative and therapeutic agents. By reducing microbial translocation and systemic inflammation, they may help lower the risk of systemic comorbidities linked to periodontitis, including obesity, cardiovascular disease, and type 2 diabetes [79–81]. Importantly, diet plays a pivotal role: high-fiber and prebiotic intake (e.g., inulin, resistant starch, oligosaccharides) selectively expands butyrate-producing taxa such as Faecalibacterium prausnitzii and Roseburia spp., effectively transforming dietary substrates into anti-inflammatory metabolites [82–84].
Macrophage polarization represents a central immunological mechanism underlying periodontal inflammation and tissue remodeling. This review highlights the emerging concept that restoring the balance between pro-inflammatory M1 macrophages and pro-resolving M2 phenotypes constitutes a promising host-modulatory strategy in periodontal therapy. SCFAs, particularly butyrate, propionate, and acetate, act as immunometabolic regulators capable of influencing macrophage polarization, attenuating destructive inflammation, and promoting resolution pathways within periodontal tissues [8, 85]. Beyond macrophages, SCFAs exert broader immunomodulatory effects on adaptive immunity. Notably, butyrate has been shown to enhance CD8⁺ T-cell effector function and support memory T-cell differentiation, suggesting that SCFAs may coordinate innate and adaptive immune responses relevant to periodontal homeostasis and protective immunity [7]. These pleiotropic effects position SCFAs as integrative immune modulators rather than single-target therapeutic agents.
Despite encouraging mechanistic and preclinical evidence, the clinical translation of SCFA-based strategies remains challenging. A major limitation lies in achieving sustained, physiologically relevant SCFA concentrations at periodontal sites while avoiding systemic adverse effects. Approaches such as dietary modulation, probiotics, direct supplementation, and emerging nanoparticle- or biomaterial-based delivery systems are under investigation, but most remain at preclinical or early translational stages [61, 62, 77]. Additionally, host heterogeneity, including genetic background, metabolic status, diet, and microbiome composition, may substantially influence responsiveness to SCFA-based interventions, underscoring the need for stratified and precision-oriented therapeutic frameworks [86, 87]. Importantly, the regenerative potential of SCFAs remains insufficiently explored. By modulating macrophage-driven osteoimmune signalling, angiogenesis, and epithelial barrier function, SCFAs may contribute not only to inflammation resolution but also to alveolar bone preservation and periodontal tissue repair [85, 86, 88].
In summary, SCFAs represent compelling immunometabolic candidates for periodontal host modulation. Advancing their clinical utility will require integration of mechanistic insights with optimized delivery strategies and well-designed human interventional studies.
FFAR: free fatty acid receptor
FOXP3: forkhead box P3
GCF: gingival crevicular fluid
GPCRs: G protein-coupled receptors
HDAC: histone deacetylase
HIF-1α: hypoxia-inducible factor-1α
IFN-γ: interferon-γ
IL: interleukin
LPS: lipopolysaccharide
MAPK: mitogen-activated protein kinase
NETs: neutrophil extracellular traps
NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells
NLRP3: NOD-, LRR-, and pyrin domain-containing protein 3
Nos2: nitric oxide synthase 2
ROS: reactive oxygen species
SCFAs: short-chain fatty acids
TNF-α: tumor necrosis factor-α
Treg: regulatory T cell
ZO-1: zonula occludens-1
DG: Conceptualization, Visualization, Writing—original draft, Writing—review & editing, Supervision. AY: Writing—original draft, Writing—review & editing. DP: Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1973
Download: 30
Times Cited: 0
