Affiliation:
Department of Microbiology, SRM Medical College Hospital and Research Centre, SRM Institute of Science and Technology, Chengalpattu 603203, Tamil Nadu, India
ORCID: https://orcid.org/0009-0005-5675-7197
Affiliation:
Department of Microbiology, SRM Medical College Hospital and Research Centre, SRM Institute of Science and Technology, Chengalpattu 603203, Tamil Nadu, India
Email: manonmoj@srmist.edu.in
ORCID: https://orcid.org/0000-0002-1165-7599
Affiliation:
Department of Microbiology, SRM Medical College Hospital and Research Centre, SRM Institute of Science and Technology, Chengalpattu 603203, Tamil Nadu, India
ORCID: https://orcid.org/0009-0004-7002-9324
Affiliation:
Department of Microbiology, SRM Medical College Hospital and Research Centre, SRM Institute of Science and Technology, Chengalpattu 603203, Tamil Nadu, India
ORCID: https://orcid.org/0000-0003-3288-1335
Explor Immunol. 2026;6:1003239 DOI: https://doi.org/10.37349/ei.2026.1003239
Received: September 08, 2025 Accepted: February 13, 2026 Published: March 11, 2026
Academic Editor: Francois Niyonsaba, Juntendo University, Japan
Atopic dermatitis is a chronic inflammatory skin condition characterized by immune dysregulation and dysfunction of the epidermal barrier. Although occludin, a vital tight junction protein, is essential for preserving barrier integrity, very little is known about its potential as a serum biomarker in atopic dermatitis. This pilot case-control study measured serum occludin levels in 30 adult atopic dermatitis patients and 30 age- and sex-matched healthy controls between May and August 2025, using a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kit. Findings showed serum occludin levels were significantly higher in atopic dermatitis patients (mean: 2.50 ± 1.10 ng/mL) compared to healthy controls (mean: 0.68 ± 0.69 ng/mL; p < 0.001), with levels showing a decreasing trend with age. Serum occludin exhibited high diagnostic performance [area under the curve (AUC) = 0.92] and represents a sensitive and specific, non-invasive biomarker for atopic dermatitis. These preliminary findings lay the groundwork for future studies exploring serum occludin as a potential biomarker for atopic dermatitis assessment.
Atopic dermatitis (AD) is a multifactorial disease resulting from a complex interplay between genetic predisposition, immune dysregulation, environmental factors, and defects in epidermal barrier integrity. Clinically, it presents with xerosis, lichenification, and recurrent episodes of itchy, inflamed skin, predominantly affecting the flexural areas in adults and the face or extensor surfaces in infants. A hallmark perpetuating skin damage and inflammation of AD is the “itch-scratch” cycle [1–3].
One of the critical factors in maintaining skin barrier integrity is the epidermal tight junction proteins, which regulate paracellular permeability. Occludin is a key transmembrane tight junction protein that plays an essential role in sealing the spaces between adjacent keratinocytes, thereby preventing transepidermal water loss and entry of allergens or pathogens [4, 5].
In AD, there is a compromise in the epidermal barrier, which reduces the function of the tight junction proteins like occludin.
The investigation of serum occludin as a biomarker has thus far been confined primarily to neurological and psychiatric disorders, with its role in AD remaining unexplored at the serum level [6–9]. This pilot study aims to fill this gap by assessing serum occludin levels in adult AD patients compared to matched healthy controls. Due to the limited sample size, the findings are preliminary and intended to lay the groundwork for future, more extensive analyses. Additionally, this study examines differences in serum occludin by age and sex to identify demographic influences and to better understand its potential as a non-invasive biomarker reflecting skin barrier impairment in AD.
Study sample: A pilot case-control study, consisting of 30 AD adult patients and 30 age- and sex-matched healthy controls, from the dermatology outpatient department (OPD) at SRM Medical College Hospital and Research Centre, was conducted between May 2025 and August 2025. The inclusion criterion was the enrolment of adult patients with clinically diagnosed AD. The patients with any other coexisting dermatological conditions, systemic diseases, or treatment that could interfere with the result were excluded.
Materials: Venous blood samples (5 mL) were collected by venipuncture without anticoagulant under aseptic conditions. All samples were centrifuged at 1,000 rpm for 5 minutes and then stored immediately at −80°C until analysis. Serum occludin levels assay was performed using a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) (Elabscience®, Human Occludin ELISA Kit), according to the manufacturer’s instructions and measured at 450 nm with a detection range of 0.16–10 ng/mL.
Statistical analysis: The data were analysed using IBM SPSS version 27.0 software. The values were calculated and expressed as mean ± SD. Statistical significance (p-value) was assessed using a t-test.
A total of 30 adult AD patients, with 15 (50%) males and 15 (50%) females, were tested. The mean age of AD patients was 24.77 ± 5.86 years. For healthy controls, age and sex were matched with the cases.
The highest level of serum occludin in AD patients was 4.32 ng/mL, and in healthy controls was 2.84 ng/mL, whereas the lowest level of serum occludin in AD patients was 0.24 ng/mL, and in healthy controls was 0.16 ng/mL, as shown in Figure 1. The serum occludin levels were significantly elevated in AD patients compared to the healthy controls. The mean was 2.50 ± 1.10 ng/mL in AD patients, but in healthy controls it was 0.68 ± 0.69 ng/mL, which was markedly lower. The p-value for comparison between serum occludin levels between AD patients and control groups is < 0.001, which is statistically significant.
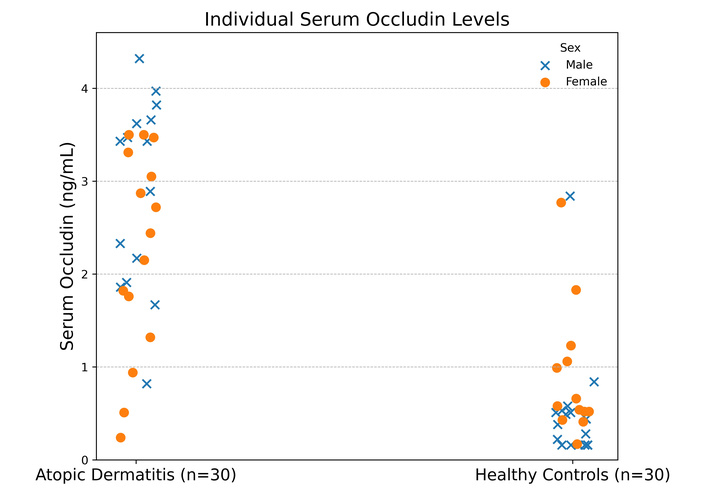
Serum occludin levels (ng/mL) of atopic dermatitis patients (n = 30) and healthy controls (n = 30).
The mean serum occludin level in AD patients age groups 18–20 years was 3.07 ± 0.87 ng/mL, 21–25 years was 2.77 ± 0.88 ng/mL, 26–30 years was 2.61 ± 1.07 ng/mL, 31–35 years was 1.14 ± 0.88 ng/mL and for ages more than 35 years it was 0.59 ± 0.49 ng/mL as seen in Table 1. There is a declining trend in the serum occludin levels in AD with an increase in age, indicating higher occludin levels in younger patients, which gradually decrease in older groups. This corresponds to the immune dysregulation seen as age progresses in AD. But there is no specific trend of serum occludin levels seen in the healthy controls over different age groups.
The distribution of atopic dermatitis and healthy controls across different age groups and their mean serum occludin levels.
| Age in years | Atopic dermatitis patients | Healthy controls | p-value | ||
|---|---|---|---|---|---|
| Total number | Mean serum occludin (ng/mL) | Total number | Mean serum occludin (ng/mL) | ||
| 18–20 | 8 | 3.07 ± 0.87 | 3 | 0.53 ± 0.08 | < 0.001 |
| 21–25 | 12 | 2.77 ± 0.88 | 6 | 0.83 ± 1.00 | < 0.01 |
| 26–30 | 6 | 2.61 ± 1.07 | 7 | 0.68 ± 0.94 | < 0.01 |
| 31–35 | 2 | 1.14 ± 0.88 | 8 | 0.74 ± 0.56 | 0.64 |
| > 35 | 2 | 0.59 ± 0.49 | 6 | 0.50 ± 0.40 | 0.84 |
The mean serum occludin level in male AD patients was 2.89 ± 1.11 ng/mL, which was higher than that of female AD patients, which was 2.24 ± 1.03 ng/mL. The mean serum occludin level in male healthy controls was 0.50 ± 0.63 ng/mL, which was lower than female healthy controls, which was 0.90 ± 0.71ng/mL, as shown in Table 2.
The distribution of atopic dermatitis and healthy controls across males and females and their mean serum occludin levels.
| Sex | Atopic dermatitis patients | Healthy controls | p-value | ||
|---|---|---|---|---|---|
| Total number | Mean serum occludin (ng/mL) | Total number | Mean serum occludin (ng/mL) | ||
| Male | 15 | 2.89 ± 1.11 | 13 | 0.50 ± 0.63 | < 0.001 |
| Female | 15 | 2.24 ± 1.03 | 17 | 0.90 ± 0.71 | < 0.001 |
Receiver operating characteristic (ROC) curve analysis was done to assess the diagnostic performance and area under the curve (AUC), which was 0.92, as seen in Figure 2. The curve is near the top left corner, which suggests that the test has a high true positive rate and a low false positive rate across various threshold levels.
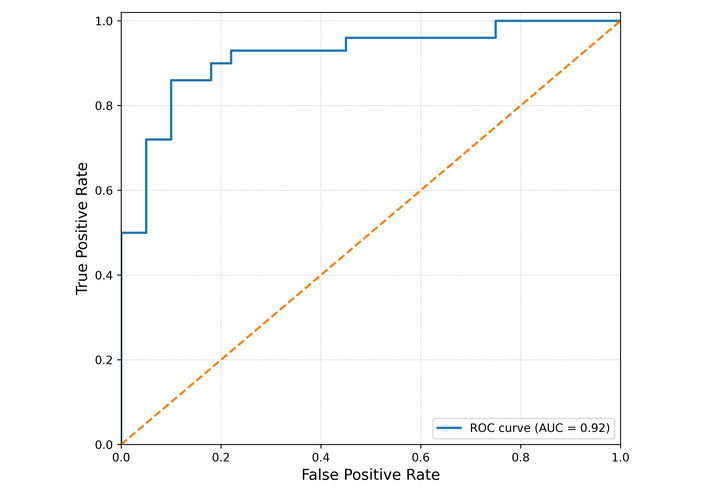
ROC curve indicating the diagnostic accuracy (AUC = 0.92). AUC: area under the curve; ROC: receiver operating characteristic.
Occludin has been previously investigated as a serum biomarker in neurological and psychiatric disorders, where it reflects blood-brain barrier disruption [6, 7]. Previous literature has explored serum occludin levels in psoriasis and their association with skin barrier dysfunction [10–13]. However, similar studies in AD are notably lacking. Its detection and significance in the serum of AD patients have not been examined until this pilot study.
Our findings indicate that occludin leaks or sheds into the bloodstream in AD, likely reflecting the compromised epidermal barrier characteristic of the disease [11, 12, 14, 15].
In this study, we observed a clear elevation of serum occludin levels in AD patients compared to healthy individuals whose occludin levels consistently remained in a lower range. This difference shows the extent of skin barrier disruption in AD, altering occludin, a key tight junction protein. This suggests that serum occludin could serve as a reliable non-invasive biomarker for assessing barrier integrity in AD.
Furthermore, the consistent decrease in serum occludin levels with age in AD patients demonstrates the immunogenic dysregulation seen in AD, which is consistent with the decrease in symptoms as age progresses. This suggests that occludin can be used as a biomarker to assess the severity, prognosis, and therapeutic response.
But the lower number of participants in each age and sex group suggests that the differences observed are not statistically significant. This calls for a study with a larger sample size to draw conclusions about the findings.
The ROC curve with an AUC of 0.92 suggests diagnostic potential, but these exploratory results should be interpreted cautiously due to the pilot study’s limited sample size.
This study was limited by its small exploratory sample size (n = 30). Future studies with larger sample sizes, paediatric patients, and longitudinal follow-up will be useful to validate these findings and further help to understand the role of occludin in AD.
According to our study, serum occludin levels were significantly higher in AD patients, suggesting that occludin is a sensitive biomarker for AD and can be useful as an adjunct diagnostic and severity-assessment tool. With further research, it could serve as a non-invasive biomarker for predicting disease progression and for better therapeutic approaches.
AD: atopic dermatitis
AUC: area under the curve
ELISA: enzyme-linked immunosorbent assay
ROC: receiver operating characteristic
STJ: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. MJ: Conceptualization, Writing—review & editing, Validation. PT: Writing—review & editing. KVL: Validation, Supervision. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not required for this pilot study.
Informed consent to participate in the study was obtained from all participants.
Not applicable.
The data will not be shared due to confidentiality.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 527
Download: 18
Times Cited: 0
