Affiliation:
1Department of Biochemistry, School of Life Sciences, Federal University of Technology, Akure 340001, Nigeria
ORCID: https://orcid.org/0000-0001-9767-1844
Affiliation:
1Department of Biochemistry, School of Life Sciences, Federal University of Technology, Akure 340001, Nigeria
2Department of Medical Biochemistry, School of Basic Medical Sciences, Federal University of Technology, Akure 340001, Nigeria
Email: adoolamide@gmail.com; owfasakin@futa.edu.ng
ORCID: https://orcid.org/0000-0002-1352-3252
Affiliation:
1Department of Biochemistry, School of Life Sciences, Federal University of Technology, Akure 340001, Nigeria
Affiliation:
1Department of Biochemistry, School of Life Sciences, Federal University of Technology, Akure 340001, Nigeria
ORCID: https://orcid.org/0000-0001-5167-9779
Explor Foods Foodomics. 2026;4:1010132 DOI: https://doi.org/10.37349/eff.2026.1010132
Received: July 20, 2025 Accepted: March 18, 2026 Published: April 13, 2026
Academic Editor: Amnart Poapolathep, Kasetsart University, Thailand
The article belongs to the special issue Neuroprotective Potential of Natural Products from Foods
Aim: This study evaluated the comparative effects of commonly consumed artificial and natural sweeteners on cognitive function, neurotransmitter-related enzyme activities, and oxidative stress status in the brains of Wistar rats to elucidate their potential neurotoxic or neuroprotective properties under sub-chronic dietary exposure.
Methods: Seventy-two male Wistar rats were randomly assigned to twelve groups and fed composite biscuits formulated with sucrose (15% and 30%), aspartame (3.5% and 7.0%), date sugar (10% and 20%), erythritol (15% and 30%), or stevia (2.5% and 5.0%) for 21 days. Control groups received either a basal diet or plain wheat biscuits. Spatial working memory was assessed using the Y-Maze spontaneous alternation test. Hippocampal tissue was harvested to determine monoamine oxidase (MAO), acetylcholinesterase (AChE), and butyrylcholinesterase (BChE) activities, lipid peroxidation (TBARS, thiobarbituric acid-reactive substances), reactive oxygen species (ROS) production, and antioxidant enzyme activities (superoxide dismutase, catalase, glutathione-S-transferase (GST), and glutathione peroxidase).
Results: Sucrose (15% and 30%) and aspartame (3.5% and 7.0%) significantly reduced spontaneous alternation performance, indicating impaired working memory. Both sucrose and aspartame dosages markedly elevated MAO, AChE, and BChE activities, increased TBARS and ROS levels, and suppressed antioxidant enzyme activities in the hippocampus. In contrast, diets containing date sugar and erythritol preserved cognitive performance and maintained neurochemical and redox homeostasis. Notably, stevia (5.0%) significantly reduced MAO and AChE activities, attenuated oxidative stress markers, and enhanced endogenous antioxidant defenses.
Conclusions: Sub-chronic consumption of sucrose and aspartame induces hippocampal neurotransmitter dysregulation and oxidative stress, contributing to cognitive impairment, whereas natural sweeteners, particularly stevia and date sugar, exhibit neuroprotective effects. These findings support the preferential use of natural sweeteners as safer dietary alternatives for maintaining cognitive and neurochemical health.
In recent decades, dietary habits have undergone significant changes, primarily due to the growing consumption of sweetened products that are both appealing and readily available. A key aspect of this dietary shift is the rising use of natural and artificial sweeteners, which enhance flavor while adjusting caloric content. Traditionally, these sweeteners have been assessed primarily for their effects on metabolic health, including glycemic control and weight management [1]. However, increasing evidence suggests they also significantly impact neurophysiology, cognitive function, and oxidative balance. Since the brain is a metabolically active and redox-sensitive organ, it is particularly susceptible to dietary changes, especially those that affect neurotransmission and oxidative homeostasis [2].
Sweeteners are generally categorized as nutritive (caloric) and non-nutritive (non-caloric). Sucrose, a disaccharide of glucose and fructose, is the most common caloric sweetener. Concerns about its links to metabolic syndrome, type 2 diabetes, and cognitive decline have increased interest in low-calorie and natural alternatives [3]. Artificial sweeteners like aspartame, a dipeptide methyl ester, are intensely sweet and low in calories. Despite regulatory approval, aspartame has been linked to neurological side effects, including headaches and memory issues, due to its metabolites, such as phenylalanine, aspartic acid, and methanol [4]. Natural alternatives such as dates (Phoenix dactylifera), erythritol, and stevia (Stevia rebaudiana) are gaining interest for their health benefits beyond their sweetness. Dates contain polyphenols, carotenoids, flavonoids, and dietary fiber, offering antioxidant, anti-inflammatory, and neuroprotective effects [5]. Erythritol, a four-carbon sugar alcohol, is non-glycemic and poorly metabolized, preventing post-meal glucose and insulin spikes [6]. Stevia, from the Stevia plant, contains steviol glycosides that are sweet and have antioxidant and anti-inflammatory properties in vitro and in vivo [7]. However, there is still much to learn about their neurological safety and effectiveness, especially under real conditions.
Recent studies reveal the neurochemical and oxidative effects of prolonged exposure to sweeteners. Animal research shows high sucrose intake harms hippocampal memory, reduces neurogenesis, and increases oxidative stress, likely due to elevated MAO (monoamine oxidase) activity, decreased acetylcholine via cholinesterase upregulation, and excess reactive oxygen species (ROS) [8]. Aspartame in rodents disrupts neurotransmission, exacerbates oxidative damage, and impairs learning through glutamatergic excitotoxicity and mitochondrial dysfunction [9]. Understanding how sweeteners affect brain chemistry is vital as consumption rises among populations at risk for neurodegenerative diseases like Alzheimer’s, Parkinson’s, and age-related decline. These conditions involve neurotransmitter imbalance and redox dysregulation, with MAO enzymes breaking down dopamine, norepinephrine, and serotonin, reducing their levels and increasing oxidative stress. Elevated cholinesterases, including acetylcholinesterase (AChE) and butyrylcholinesterase (BChE), impair acetylcholine and are found in dementia [10–12].
Considering the widespread use of sweeteners in diets and their varying metabolic effects, this study evaluates their influence on cognitive function, neurotransmitter activity, and oxidative stability. It examines sucrose, aspartame, date sugar, erythritol, and stevia in biscuits, focusing on behavioral outcomes, enzyme functions, and antioxidant systems in Wistar rats. The study’s novelty lies in its systematic, integrative approach to assessing diverse sweeteners under consistent conditions, with an emphasis on neurochemical and redox biomarkers. Unlike prior research on individual sweeteners, this work offers a comprehensive view of their impact on brain function across behavioral, enzymatic, and molecular levels. By analyzing both artificial and natural sweeteners, the findings aim to guide public health policies, dietary guidelines, and functional food development with minimal neurotoxicity.
Aspartame, dates, erythritol, and sucrose were sourced from the Akure South Local Government Area of Ondo State, Nigeria. Meanwhile, stevia was purchased from a verified store in Lagos State, Nigeria. Refined white wheat flour (Triticum aestivum) with a low fiber content, suitable for biscuit production, was purchased from a local commercial supplier in Akure, Nigeria.
The chemicals and reagents used included bovine serum albumin, acetylthiocholine, and Ellman’s reagent from Sigma-Aldrich (Germany) and Sigma Chemical Co., St. Louis, MO, USA, and Organon Limited, Kolkata, India. All the kits used for the bioassays were sourced from Randox Laboratories Ltd., Crumlin, Co. Antrim, UK. All other chemicals and reagents were of analytical grade, and the water was glass-distilled.
The sweeteners (stevia, aspartame, dates, erythritol, and sugar) were blended with white wheat flour in different ratio percentages [sucrose (30% and 15%), aspartame (7.0% and 3.5%), dates (20% and 10%), erythritol (30% and 15%), and stevia (5.0% and 2.5%)] to produce a composite flour for biscuit making. The proportion of sweeteners to wheat flour (%) was determined by varying the inclusion percentages of each sweetener. The flours were subsequently mixed with additional ingredients such as cocoa butter (4%), orange peel flour (3%), aspartame (0.5%), egg albumin (0.5%), and skimmed milk (0.5%) to enhance their nutritional value and to meet the specific dietary standards outlined by Oguntuase et al. [13]. The sweetener-wheat flour mixtures were combined with water, baking powder, salt, and baking fat until the desired dough consistency was reached, then kneaded on a rolling table to achieve a thickness of approximately 3–4 mm. The dough was then cut into rounds using a biscuit cutter and baked in a mechanized oven for 10 minutes at 200°C. After baking, the biscuits were cooled and packaged [14]. All composite biscuit diets were formulated to be approximately isocaloric and comparable in macronutrient composition, with the primary experimental variable being the type and inclusion level of the sweetener.
Seventy-two male albino Wistar rats weighing 202 to 214 g were obtained from the Animal House of the Department of Biochemistry at the Federal University of Technology, Akure, Ondo State, Nigeria. The animals were acclimatized in plastic cages and kept at room temperature (25–27°C), with relative humidity (60–70%) and a controlled light cycle (12 h dark/12 h light) for two weeks, during which they had free access to commercial rat chow and water ad libitum before their use. The exploitation of animals complies with the ethical guidelines established by the US National Institutes of Health (NIH). Ethical approval for animal use (FUTA/ETH/24/675) was obtained from the Animal Ethical Committee, Centre for Research and Development (CERAD), Federal University of Technology, Akure. After two weeks of acclimatization, the experimental animals were randomly divided into twelve groups as presented in Table 1, each comprising six animals. The experimental animals were fed 15 g of composite biscuits daily for twenty-one days. The 21-day feeding period was selected to model sub-chronic dietary exposure and is sufficient to elicit measurable behavioral, neurochemical, and oxidative alterations in rodent models.
Experimental procedure.
| S/N | Groups | Administration procedure |
|---|---|---|
| 1 | Control | Rats were fed basal diets (standard chow). |
| 2 | Plain | Rats were fed plain biscuits (made only from wheat flour). |
| 3 | Sucrose (15%) | Rats were fed sucrose (15%) biscuits. |
| 4 | Sucrose (30%) | Rats were fed sucrose (30%) biscuits. |
| 5 | Aspartame (3.5%) | Rats were fed aspartame (3.5%) biscuits. |
| 6 | Aspartame (7.0%) | Rats were fed aspartame (7.0%) biscuits. |
| 7 | Date (10%) | Rats were fed date (10%) biscuits. |
| 8 | Date (20%) | Rats were fed date (20%) biscuits. |
| 9 | Erythritol (15%) | Rats were fed erythritol (15%) biscuits. |
| 10 | Erythritol (30%) | Rats were fed erythritol (30%) biscuits. |
| 11 | Stevia (2.5%) | Rats were fed stevia biscuits (2.5%). |
| 12 | Stevia (5.0%) | Rats were fed biscuits containing stevia (5.0%). |
The Y-Maze test was conducted a week before the animals received composite biscuits, allowing them to familiarize themselves with the behavioral paradigm. On day 21, the Y-Maze test was reassessed. This paradigm is a reliable and quick method for evaluating the impact of drugs on rodent memory. The maze was built, and short-term memory was analyzed according to the procedure outlined by Oyeleye et al. [15]. The memory index was plotted to assess cognitive impairments, with higher values indicating greater cognitive impairment.
After the behavioral study, the experimental animals were first anesthetized with ketamine (80 mg/kg) and xylazine (5 mg/kg) given intraperitoneally to ensure deep anesthesia and lack of pain perception. Euthanasia was then carried out via cervical dislocation while still under anesthesia, which is a NIH-approved method that ensures rapid and humane death [16, 17]. This combined approach of anesthesia followed by cervical dislocation reduces distress, ensures consistency across experimental groups, and is a safe, widely accepted standard for laboratory animal studies. The whole brain was removed and homogenized in ice-cold 0.1 M phosphate buffer (pH 7.4), then centrifuged at 12,000 rpm for 10 min at 4°C. The clear supernatants obtained were used for biochemical analyses [18].
The method of Green and Haughton [19], as adapted by Fasakin et al. [20], was used to evaluate MAO activity. The reactants included 0.025 mol/L phosphate buffer at pH 7.0, 0.0125 mol/L semicarbazide, 10 mmol/L benzylamine, and 100 µL of hippocampal homogenate. This mixture was incubated for 30 minutes, then acetic acid was added, and the mixture was further incubated in a hot water bath for 3 minutes. A 1 mL solution was combined with an equal volume of 2,4-dinitrophenylhydrazine. Next, 1.25 mL of benzene was added, and the mixture was reacted at room temperature for 10 minutes. The benzene layer was separated and mixed with 0.1 N NaOH. The alkaline layer was decanted and incubated at 80°C for 10 minutes. Finally, the color intensity was measured at 450 nm.
The cholinesterase assay on AChE and BChE used the Ellman et al. [21] spectrophotometric method as described by Fasakin et al. [22]. The reaction solution consisted of 100 mM phosphate buffer (pH 7.5), 1 mM 5,5'-dithiobisnitrobenzoic acid, and brain homogenate, which generated the yellow anion 5,5'-dithiobis-acidnitrobenzoic, measured by absorbance at 412 nm. The reaction began with 0.8 mmol/L acetylthiocholine iodide for AChE and butyrylthiocholine iodide for BChE. Enzyme activity was quantified in micromoles of substrate per hour per milligram of protein.
Lipid peroxidation was assessed by measuring the TBARS [23]. Typically, a mixture of 300 μL of hippocampal homogenate, 300 μL of 8.1% sodium dodecyl sulfate (SDS), 500 μL of 0.8% TBA, and 500 μL of a 2.5 M HCl solution (pH 3.4) was incubated at 100°C for 1 hour and then analyzed at 532 nm.
ROS production was measured using the method of Hayashi et al. [24], as adapted by Fasakin et al. [25]. In general, 50 mL of hippocampal tissue homogenate and 1,400 mL of sodium acetate buffer were placed into a cuvette. Subsequently, 1,000 mL of the reagent mixture (N, N-diethyl-para-phenylenediamine, DEPPD, 6 mg/mL) was combined with 4.37 mM ferrous sulfate dissolved in 0.1 M sodium acetate buffer (pH 4.8). The solution was then incubated at 37°C for 5 minutes before being read at 505 nm.
The activity of superoxide dismutase (SOD) in the tissue homogenate was assessed by measuring its ability to inhibit the autoxidation of epinephrine at pH 10.2 and 30°C [26]. Specifically, SOD activity was quantified as the amount needed to achieve a 50% inhibition of adrenaline autoxidation. Catalase (CAT) activity in the hippocampal homogenate was evaluated using the method reported by Ademosun et al. [27], which involved monitoring the absorbance of hydrogen peroxide at 240 nm, at pH 7.0, and 25°C. The activity of glutathione-S-transferase (GST) in the brain homogenate was measured using the procedure outlined by Habig et al. [28], which employed 1-chloro-2,4-dinitrobenzene (CDNB) as a substrate. To assay glutathione peroxidase (GPx) activity in the brain homogenate, a method based on Rotruck et al. [29] was employed, which involves the catalytic oxidation of glutathione (GSH) by hydrogen peroxide. Hydrogen peroxide swiftly converts GSH to its oxidized form (GSSG) in this reaction.
The Shapiro-Wilk test was used to assess the normality of the data patterns. The sample size was established through Power Analysis [30]. Each outcome represents a replicate of six. The results were consolidated and presented as the mean ± standard error of the mean (SEM) (n = 6). Statistical significance was determined using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc multiple comparison test, and was significantly different at p ˂ 0.05. All p-values reported in the text and figures reflect adjusted values obtained after multiple comparisons. All statistical analyses were conducted using GraphPad Prism (version 8.0.2) software.
The Y-Maze test assessed spatial working memory and spontaneous alternation, both sensitive to diet-induced neurocognitive changes. Various sweeteners (sucrose, 15% and 30%; aspartame, 3.5% and 7.0%; date, 10% and 20%; erythritol, 15% and 30%; and stevia, 2.5% and 5.0%) were evaluated in animals for 21 days. The neuro-modulatory effects, shown in Figure 1, indicated dose-dependent responses. Date, erythritol, and stevia had no significant effect on the preference index, but 15% and 30% sucrose, and 3.5% and 7.0% aspartame doses significantly reduced spontaneous alternation (p < 0.01, p < 0.001, p < 0.05, respectively) compared to controls.
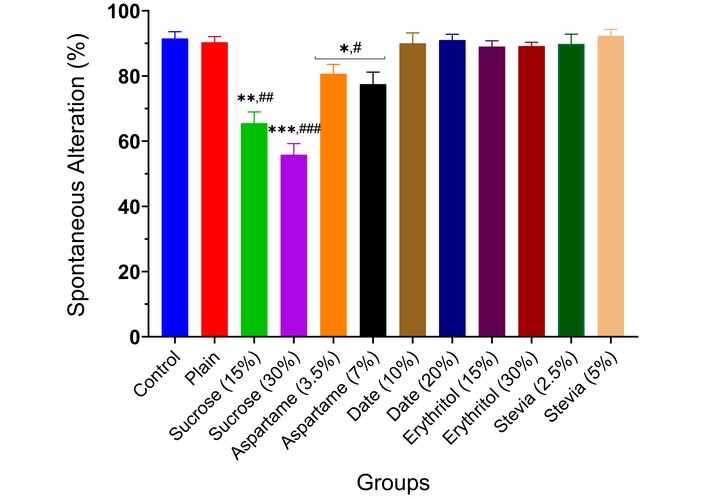
Spontaneous alternation (memory index) of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)] using the Y-Maze task. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
The results showed that MAO activity rose significantly in the brains of animals given low and high doses of sucrose (p < 0.01) and aspartame (p < 0.05), depending on inclusion percentage, compared to controls (Figure 2). Date and erythritol did not alter MAO activity from controls. Stevia (5.0%) biscuits significantly reduced MAO activity (p < 0.05). AChE activity increased significantly in animals given 15% sucrose (p < 0.01), 30% sucrose (p < 0.001), and 7.0% aspartame (p < 0.05), depending on inclusion. However, 20% date (p < 0.05) and 5.0% stevia (p < 0.05) decreased enzyme activity. Other sweeteners did not affect AChE activity compared to controls (Figure 3). BChE activity increased significantly in animals given 15% sucrose (p < 0.01), 30% sucrose (p < 0.001), and 7.0% aspartame (p < 0.05), depending on inclusion. Other sweeteners had no effect, except for 30% erythritol, which significantly decreased BChE activity (p < 0.05) (Figure 4).
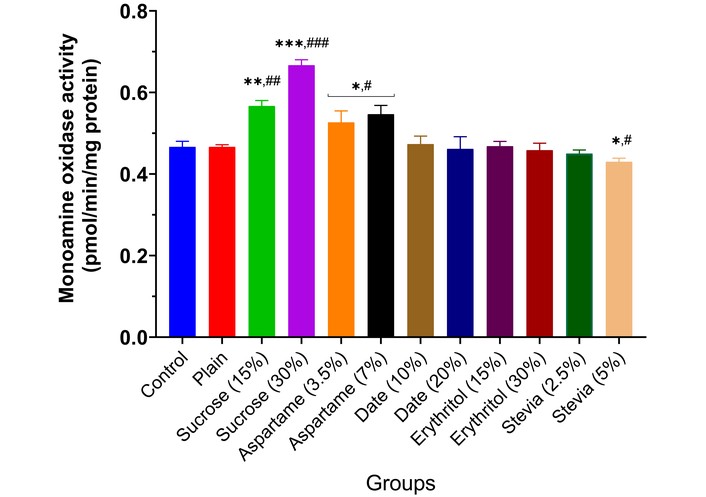
Monoamine oxidase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
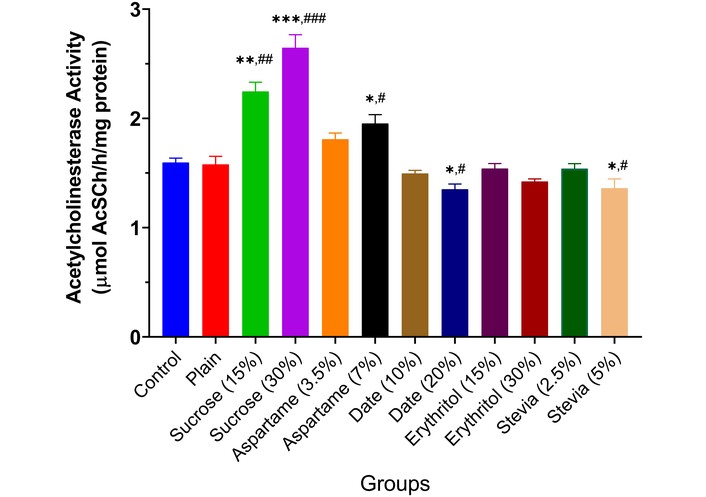
Acetylcholinesterase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
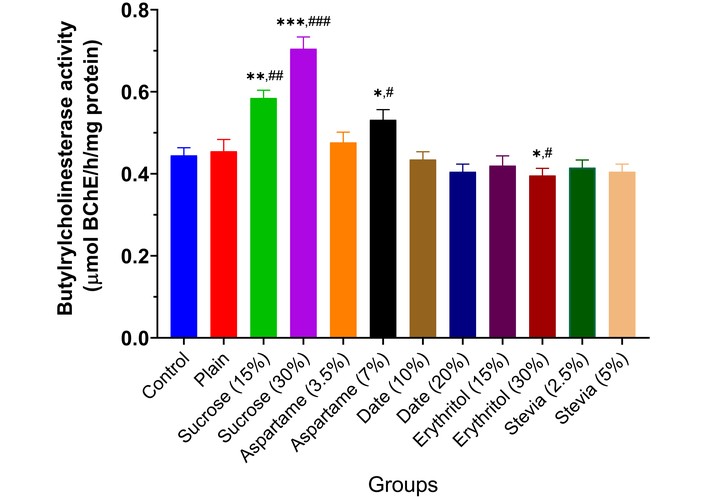
Butyrylcholinesterase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
Administration of 15% and 30% sucrose (p < 0.01 and p < 0.001) and 3.5% and 7.0% aspartame (p < 0.05) significantly increased brain thiobarbituric acid levels compared to controls (Figure 5). Other sweeteners showed no significant effects, except stevia (5.0%), which reduced thiobarbituric acid (p < 0.05).
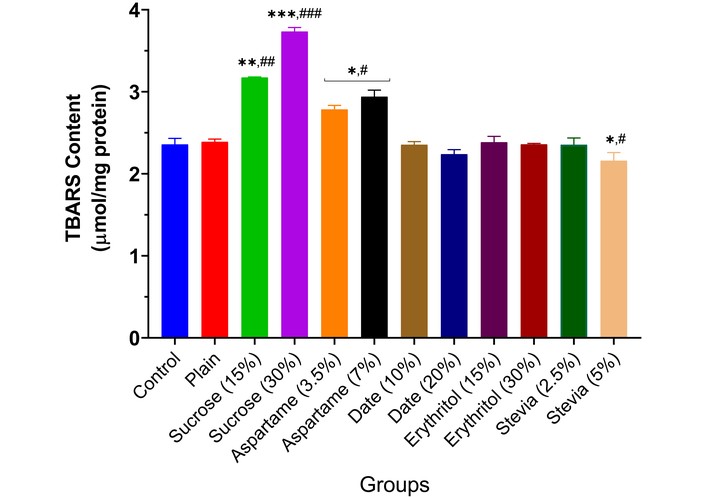
Lipid peroxidation in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
Different sweeteners—sucrose, aspartame, date, erythritol, and stevia—also elevated ROS levels, with sucrose (15% and 30%) notably increasing ROS (p < 0.01, p < 0.001), while date (20%) decreased ROS (p < 0.05) (Figure 6).
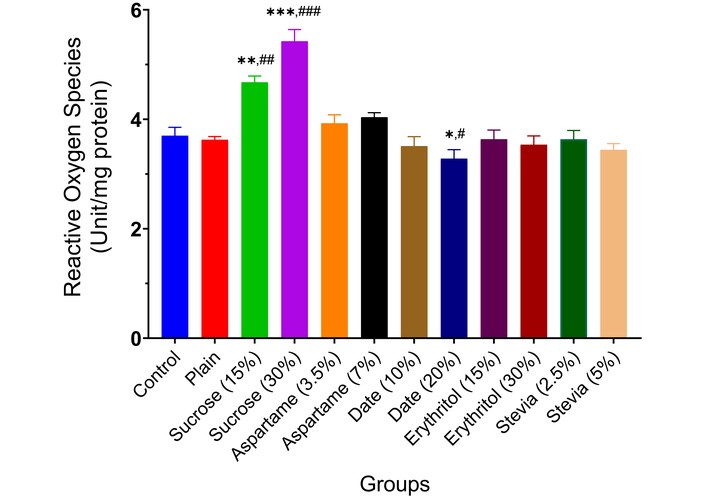
Reactive oxygen species production in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
The effect of the various sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)] on the activities of enzymatic antioxidants (SOD, CAT, GST, and GPx) in the brain of experimental animals is reported in Figures 7, 8, 9 and 10. Typically, the administration of sucrose (15% and 30%) and aspartame (3.5% and 7.0%) significantly depleted the activities of SOD (p < 0.01, p < 0.001, p < 0.05), CAT (p < 0.01, p < 0.001, p < 0.05), GST (p < 0.01, p < 0.001, p < 0.05), and GPx (p < 0.01, p < 0.001, p < 0.05) in the brain of experimental animals compared to the normal control and wheat-biscuit-fed groups. Remarkably, stevia (5.0%) improved the activities of the enzymatic antioxidants (p < 0.01) in the brain of experimental animals compared to the normal control and wheat-biscuit-fed groups.
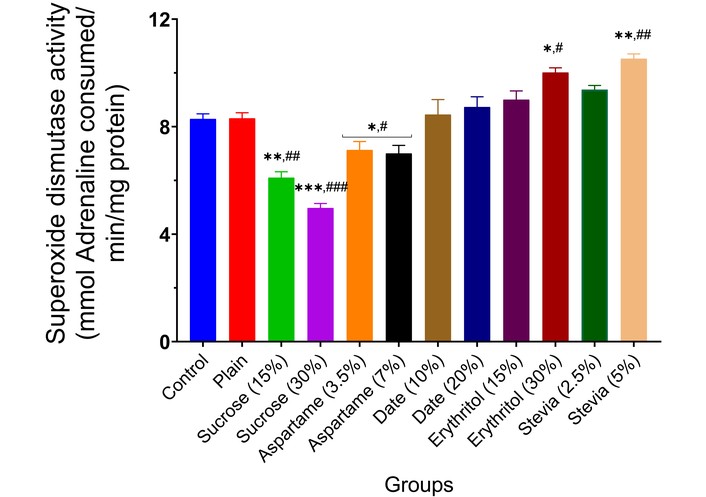
Superoxide dismutase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
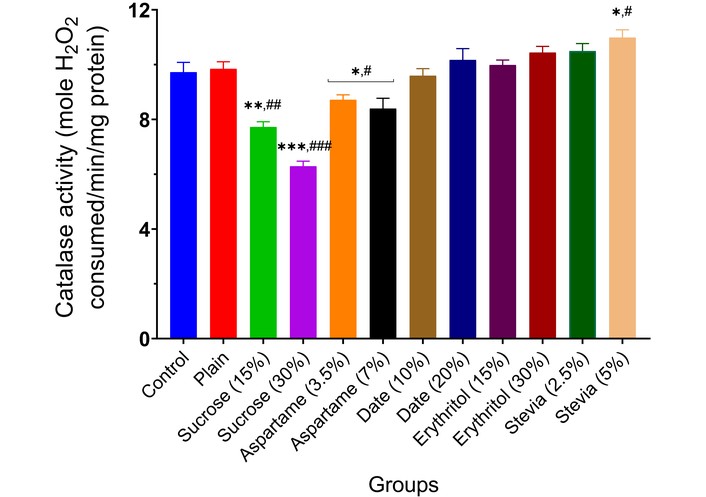
Catalase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
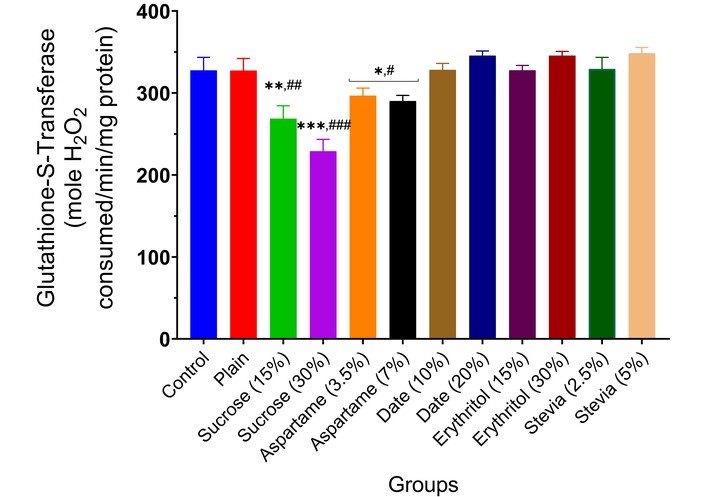
Glutathione-S-transferase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
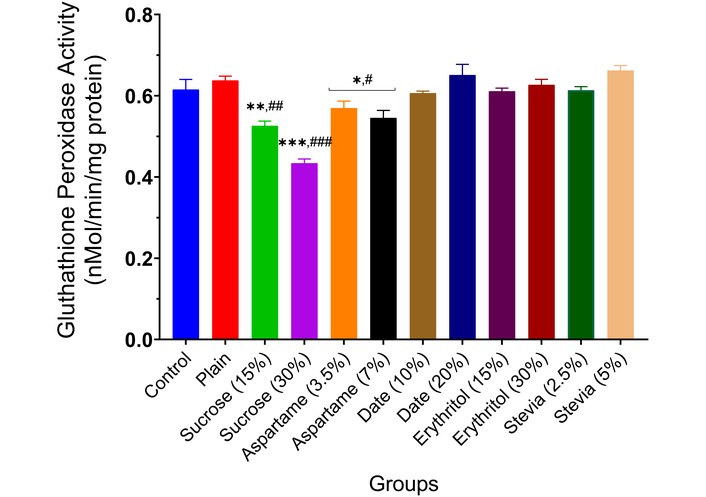
Glutathione peroxidase activity in the brain of experimental animals administered sweeteners [sucrose (15% and 30%), aspartame (3.5% and 7.0%), date (10% and 20%), erythritol (15% and 30%), and stevia (2.5% and 5.0%)]. Results are expressed as mean ± SEM (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the normal control group. # p < 0.05, ## p < 0.01, ### p < 0.001 compared with the plain (wheat supplemented) biscuit fed group.
The present study systematically investigated the neurobehavioral, neurochemical, and antioxidant effects of various sweeteners, including sucrose, aspartame, dates, erythritol, and stevia, on experimental rats over 21 days. The findings reveal intricate, differential effects of the sweeteners across behavioral paradigms and biochemical analyses, offering crucial insights into their safety and potential neurotoxicity. The Y-Maze test, a reliable indicator of spatial working memory [11], showed a significant reduction in spontaneous alternation behavior in rats administered sucrose and aspartame, indicating impaired cognitive function. In contrast, date, erythritol, and stevia did not significantly alter cognitive performance, demonstrating their relative safety in preserving memory function.
The impairments observed with sucrose and aspartame are consistent with previous reports suggesting that high sugar intake can detrimentally impact hippocampal-dependent memory processes. Contreras-Rodriguez et al. [31] demonstrated that high-energy diets rich in simple sugars impair hippocampal function, critical for memory and learning. Similarly, aspartame consumption has been linked to cognitive dysfunctions. Dar [9] highlighted that aspartame metabolites, particularly aspartic acid and phenylalanine, may induce excitotoxicity, potentially disrupting neural circuits involved in memory. Meanwhile, the relatively mild cognitive effects of dates, erythritol, and stevia align with prior evidence suggesting a neuroprotective or neutral role. Dates, rich in polyphenolic antioxidants, have been shown to enhance cognitive function by counteracting oxidative stress [32]. Erythritol and stevia, being non-glycemic sweeteners, have minimal impact on metabolic pathways associated with cognitive decline [33].
The neurochemical assessments supported these behavioral findings. MAO activity was significantly elevated following the consumption of sucrose and aspartame, suggesting increased degradation of monoaminergic neurotransmitters, which may contribute to memory impairments. As Behl et al. [34] indicated, elevated MAO activity suggests increased degradation of monoamine neurotransmitters such as dopamine and serotonin, which could underlie the observed memory impairments. These findings corroborate the study by Witek et al. [35], which reported that high-sugar diets can dysregulate monoaminergic systems, predisposing individuals to mood and cognitive disorders. Conversely, MAO activity remained unaffected in rats fed diets containing date or erythritol, and was significantly reduced by high-dose stevia, indicating a potential neuroprotective effect.
Further analysis of cholinergic markers showed that AChE and BChE activities were significantly increased by sucrose and aspartame, reinforcing their potential to impair cognitive processing by reducing acetylcholine availability. Elevated ChE activities typically reduce acetylcholine levels, a neurotransmitter vital for memory consolidation [11]. Increased BChE has been associated with cholinergic dysfunction in neurodegenerative diseases like Alzheimer’s disease [36]. These results echo findings by Spagnuolo et al. [8], who linked high-sugar diets to decreased hippocampal brain-derived neurotrophic factor (BDNF) expression and impaired cholinergic transmission. Notably, diets containing 20% date and 5.0% stevia significantly reduced AChE activity, while a 30% erythritol diet lowered BChE activity, suggesting improved cholinergic function and supporting their neuroprotective profile. Notably, the polyphenolic content of dates might inhibit cholinesterase activity, aligning with studies demonstrating the inhibitory effects of natural antioxidants on AChE [15, 37].
Oxidative stress markers, notably TBARS and ROS production, were significantly elevated by sucrose administration, suggesting lipid peroxidation and increased free-radical generation. Interestingly, stevia at 5.0% also significantly reduced TBARS levels, demonstrating antioxidative capacity at this dose. Dates and erythritol showed no pro-oxidative effects, maintaining an oxidative balance comparable to that of the controls. Researchers such as Zhang et al. [38] and Li et al. [39] have demonstrated that high sugar consumption exacerbates oxidative damage, disrupts mitochondrial function, and contributes to cognitive deficits. Mechanistically, excessive glucose metabolism generates ROS via the polyol pathway and glycation reactions, thereby impairing neuronal function.
In terms of endogenous antioxidant defenses, sucrose and aspartame significantly reduced the activities of key antioxidant enzymes, including SOD, CAT, GST, and GPx, confirming an oxidative burden on neural tissues. This depletion aligns with data from Bacil et al. [40], which indicates that sugar-enriched diets impair endogenous antioxidant defenses and exacerbate oxidative injury. In contrast, stevia (5.0%) significantly improved the enzyme activities of SOD and CAT. At the same time, erythritol and date also maintained or slightly enhanced enzymatic antioxidant status, supporting their roles in redox homeostasis.
The neuroprotective properties of stevia and dates are likely due to their high levels of flavonoids and polyphenols, which influence essential cellular processes. Flavonoids such as quercetin and kaempferol, present in dates and stevia, can activate the Nrf2 (nuclear factor erythroid 2-related factor 2) pathway, boosting the production of antioxidant enzymes like SOD, CAT, and GPx [41]. This process enhances the body’s natural defenses against oxidative stress. Furthermore, these phytochemicals exhibit cholinesterase-inhibitory effects, helping maintain acetylcholine levels and supporting synaptic function [42]. By alleviating oxidative stress and promoting cholinergic activity, stevia and dates offer a dual approach to neuroprotection, helping combat cognitive decline and neurodegeneration. Notably, the present study highlights the non-disruptive nature of these natural sweeteners, further supporting the safety profiles suggested by Echegaray et al. [43], Han et al. [44], and Soetan et al. [45].
This study highlights the impact of diet on neurodegenerative processes. Elevated levels of MAO, AChE, and BChE in rats consuming sucrose and aspartame suggest a more rapid breakdown of neurotransmitters, such as dopamine and serotonin, which are essential for cognitive function and mood stability. Increased enzyme activity correlates with neurodegenerative diseases like Alzheimer’s and Parkinson’s. Furthermore, rising oxidative stress indicators and reduced antioxidant enzymes point to a disrupted redox state, resulting in mitochondrial dysfunction, neuronal death, and inflammation, hallmarks of neurodegeneration. Conversely, supplementation with dates and stevia shows neuroprotective benefits by reducing cholinesterase activity, maintaining MAO balance, and enhancing antioxidant defenses, potentially lowering the risk of neurodegeneration. Stevia’s role in restoring redox balance and regulating neurotransmitter enzymes underscores its potential as a dietary neuroprotective agent. These findings highlight the importance of nutritional choices in influencing neurochemical pathways and oxidative balance, providing strategies to mitigate cognitive decline. Using natural sweeteners with antioxidant and neuromodulatory benefits may offer a non-invasive approach to enhancing brain health.
Conclusively, this study contributes to the growing body of evidence that sweetener choice has a profound impact on brain health. Sucrose and aspartame, widely consumed globally, demonstrate potential neurotoxic effects even over short durations. In contrast, natural sweeteners like dates and erythritol pose minimal risk and may confer neuroprotection. Given the widespread consumption of these substances, especially among vulnerable populations such as children and older adults, these findings highlight the urgent need for public health policies promoting healthier sweetening alternatives. Further studies, particularly long-term and translational human research, are warranted to validate these observations and guide evidence-based recommendations.
Despite strong behavioral and neurochemical evidence, this study has some limitations. It did not include histopathological analysis of brain tissue, which could confirm structural changes related to neurochemical and oxidative shifts. Systemic metabolic markers such as blood glucose, lipid profile, and lipoprotein levels were also not measured, limiting understanding of peripheral metabolic influences on neurobiological effects. Additionally, only male Wistar rats were studied, so sex-dependent effects could not be evaluated. Future research should include detailed brain histopathology, a broader range of behavioral tests, longer exposure periods, both sexes, and systemic metabolic assessments to gain a comprehensive view of how sweeteners affect neurobiology and to improve relevance to humans.
AChE: acetylcholinesterase
ANOVA: analysis of variance
BChE: butyrylcholinesterase
BDNF: brain-derived neurotrophic factor
CAT: catalase
CDNB: 1-chloro-2,4-dinitrobenzene
DEPPD: N, N-diethyl-para-phenylenediamine
GPx: glutathione peroxidase
GSH: glutathione
GST: glutathione-S-transferase
MAO: monoamine oxidase
NIH: National Institutes of Health
Nrf2: nuclear factor erythroid 2-related factor 2
ROS: reactive oxygen species
SEM: standard error of the mean
SOD: superoxide dismutase
TBARS: thiobarbituric acid-reactive substances
AOA: Conceptualization, Supervision, Formal analysis, Writing—original draft, Visualization. OWF: Investigation, Writing—review & editing, Supervision, Formal analysis. MA: Investigation, Writing—review & editing. GO: Conceptualization, Supervision, Formal analysis. All authors read and approved the manuscript.
All the authors of the present study declare no conflicts of interest.
The exploitation of animals complies with the ethical guidelines established by the US National Institutes of Health (NIH). Ethical approval for animal use (FUTA/ETH/24/675) was obtained from the Animal Ethical Committee, Centre for Research and Development (CERAD), Federal University of Technology, Akure.
Not applicable.
Not applicable.
The results section provides the summarized data necessary to support the conclusions of this manuscript. The raw data are available upon reasonable request from the corresponding author.
This study received no funding or financial support from any organization.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 495
Download: 22
Times Cited: 0
