Affiliation:
Centro de Investigación y Desarrollo en Ciencia y Tecnología de los Alimentos (CIDCA-CONICET-UNLP-CIC), La Plata, Buenos Aires 1900, Argentina
Email: cdini@biol.unlp.edu.ar
ORCID: https://orcid.org/0000-0002-2780-6261
Explor Foods Foodomics. 2026;4:1010128 DOI: https://doi.org/10.37349/eff.2026.1010128
Received: December 17, 2025 Accepted: March 05, 2026 Published: March 25, 2026
Academic Editor: Javad Sharifi-Rad, Universidad del Azuay, Ecuador
Edible canna is the common name given to Canna indica L., also known as Canna edulis Ker Gawl. This Andean crop has been gaining attention due to some characteristics of its rhizome starch that distinguish it from those found in other roots and tubers. Canna starch is currently used in some regions of Latin America for producing traditional baked products and desserts. In Asian countries such as China and Vietnam, it is industrially produced mainly for the elaboration of starch noodles. This review summarizes the up-to-date knowledge about edible canna as a starch source for the food industry. The composition, granule morphology, and molecular structure of canna starch are described and related to the functional properties displayed as a food ingredient. The thermal and pasting properties, gel stability, digestibility, and susceptibility to acid hydrolysis are also addressed, as well as recent reports on physical and chemical modifications to expand its applications in the food industry.
Edible canna (Canna indica L.) is a perennial herb native to the Andean region in South America. Archaeological findings in Peru suggest that its cultivation dates back to 2500 BC [1]. This crop is distributed across many regions, throughout the tropical and sub-tropical America, Asia, and Africa, and is typically cultivated in highlands, at 1,000–2,500 m.a.s.l. [1–3].
Taxonomically, it belongs to the order of the Zingiberales, the Cannaceae family, for which more than 100 species have been described. The Cannas are cultivated as garden flowers, and some species, such as Canna indica L., are used to obtain starch. The species has the following synonyms: Canna edulis Ker Gawl., Canna coccinea Mill., and Canna orientalis Roscoe, among others, but Canna indica L. is the currently accepted botanical name [4, 5].
Canna indica L. is known by several common names worldwide, with edible canna the most widespread term [4]. It is also known as “Indian shot” or “African arrowroot”. In South America, it is known as “Achira”, but has other names such as “Chisgua”, “Sagu”, and “Capacho” in Colombia and Venezuela, and “Imbirg” in Brazil. “Sagu” is also the name given in Thailand. In the West Indies, it is known as “Tous-les-mois”, “Queensland arrowroot” in Australia, “Ganyong” in Indonesia, and “Zembu” in the Philippines [2, 6].
This plant has abundant large starchy rhizomes, which can be spherical, cylindrical, or spinning top-shaped, from 5 to 20 cm in length and 3 to 12 cm in width [1] (Figure 1). Occasionally, lengths up to 50–60 cm have been reported [7, 8]. On their surface, they present transverse grooves that mark the base of the scales that cover it; white and cylindrical rootlets are usually seen at the lower part of the rhizome, and the pseudostem, the leaves, and the floral stem grow from the apex. The flowers are organized in clusters, with colors ranging from red to yellow (Figure 2A). The fruits are three-celled capsules with spherical black seeds (Figure 2B) [1].
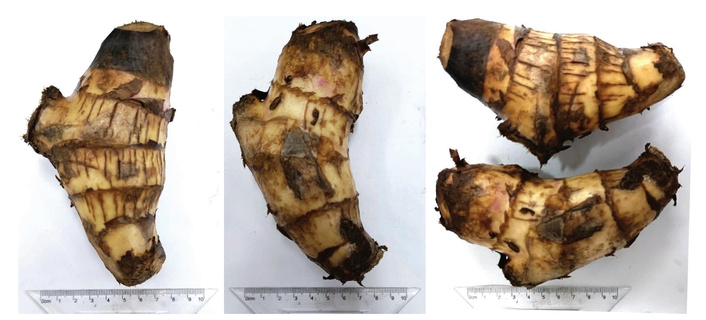
Canna indica L. rhizomes. Taken from Lv et al. [9] without modification. © 2023 The Authors. Licensed under a Creative Commons Attribution license 4.0.

Canna indica L. (A) flowers and (B) fruits. Taken from Dini C [10] without modification. © 2026 The Author. Licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 2.5 Unported.
Achira plants can be cultivated from sea level to 2,700 meters above sea level, requiring frost-free areas during the period of maturation of the rhizomes. It develops roughly from 9 to 30–32°C. It thrives in a wide range of soil types and requires moderate and well-distributed rainfall. The plant resists drought, while excessive moisture causes abnormal development [1].
Harvesting must be done between 6 and 9 months, depending on the cultivar, the altitude, and the type of soil. The rhizomes tend to emerge to the surface (negative geotropism) and are harvested manually by uprooting the plant with a shovel or crowbar and cutting the base of the stems to separate the rhizomes. Young rhizomes or parts of them with functional buds are used as propagation material [1].
The cultivars are regularly classified into two main groups according to the color of the plant, one group includes green or white varieties, which stand for the color of the rhizomes and leaves, respectively; and the other includes the purple varieties, for plants that have a purple color at the tip of rhizomes, and the rim of the leaves [1, 3].
Rhizome yields range from 15 to 50 t/ha, depending on the variety and harvesting time, with an average fresh weight of 5.3 kg per plant in seven-month-old crops [1, 2, 11–14]. The rhizomes have 2.4–3.3% protein, 0.25–1.63% fat, 2.9–3.2% fiber, and 5.1–5.7% ash (dry basis, db), and up to 18.0% starch (wet basis, wb) [2, 12]. The peeled rhizome has a moisture content of 85.2% and contains 60.3% starch db [15], lower than other more widespread root and tuber starch sources such as potato (79% db) or cassava (up to approximately 80% db) [16]. Available evidence indicates that starch accumulation in Canna indica rhizomes increases progressively with physiological maturity, peaking at around eight months, followed by a subsequent decline in starch content [17].
This crop is currently cultivated for starch production in small-scale factories in China and Vietnam [18]. In Latin America, canna starch is mostly produced in an artisanal way by small rural communities, and a few small production plants [19, 20].
The extraction of canna starch follows a process similar to that used for other Andean root and tuber crops, such as ahipa (Pachyrhizus ahipa), oca (Oxalis tuberosa), and arracacha (Arracacia xanthorrhiza), and is largely based on industrial cassava starch extraction methods [21]. The process begins with cleaning, peeling, and slicing the rhizomes. The resulting pieces are subjected to wet milling with water to obtain a slurry; antioxidants such as sodium metabisulfite may be added to improve starch purity. The slurry is allowed to stand for several hours (up to 12 h) and is then sieved to separate the fibrous residue from the starch-rich liquid. Starch is subsequently recovered by sedimentation, purified through repeated washing with water, and finally dried at 30–40°C before being milled into a fine powder [12, 22]. Scientific information on extraction efficiency, processing-induced structural changes, and storage stability is scarce. Future research should focus on optimizing extraction and processing technologies, evaluating their effects on starch functionality.
In Asian countries, canna starch is mainly used for making glassy noodles (a.k.a glass noodles, cellophane noodles or starch noodles) [3]. Glassy noodles require starches with high amylose content, restricted swelling, and C-type Brabender viscosity. Canna starch has good characteristics for this purpose, while mung bean starch is ideal but has higher cost.
Within Latin America, canna starch is used in Colombian gluten-free baked products such as “bizcocho de achira”, a crunchy biscuit made with curd and egg yolk [8]. It is also used to prepare “colada de achira”, which is made by adding canna starch to boiling water with raw sugarcane, clove, and cinnamon. The mixture is cooled, chopped, and served with warm milk. In the Bolivian Yungas (Coroico), achira starch is used for making “biscochuelos”, sweet cakes baked in paper and offered during All-Saints festivity [8].
This Review analyzes the composition as well as the molecular structure and thermal characteristics of Canna indica L. starch to provide a deeper understanding of its functional properties, thereby supporting the expansion of its gastronomic applications. Data from both early and recent studies were considered in order to compile a comprehensive dataset, allowing a realistic assessment of the range of values reported for this starch.
Collectively, the available literature reveals a shift from basic physicochemical characterization toward starch modification strategies aimed at enhancing technological performance. While early studies established foundational compositional and structural data, inconsistencies among reported values remain evident. These discrepancies likely arise from limited sample numbers and the absence of comprehensive, multi-factorial analyses integrating ecotype variability, cultivation conditions, post-harvest and extraction processing, as well as starch molecular architecture, crystalline organization, and granule size distribution. A systematic and comparative evaluation of these variables is still lacking. Addressing these fundamental gaps is essential to clarify structure-function relationships, support reproducibility, and ultimately enable the targeted large-scale production of starches tailored for specific culinary and industrial applications.
Canna starch can be extracted with high purity, typically above 94%. However, starch whiteness ranges between 84.5 and 91.5, which is low compared to other unrefined root and tuber starches such as cassava, oca (Oxalis tuberosa), or arracacha (Arracacia xanthorrhiza), which range between 93.5–95.6 [2, 18–21, 23, 24]. The application of acids for starch bleaching must be approached with caution due to the susceptibility of canna starch to acid hydrolysis (Susceptibility to acid hydrolysis). In particular, ascorbic acid (0.1%) applied during extraction showed evidence of rupture in the extracted starch granules [25].
No specific feature was detected that may distinguish starches from both origins. The low protein (0.06 to 0.93%) and lipid (up to 0.4%) contents of this starch agree with those expected for root and tuber starches [26].
Regarding the mineral composition, the ash content ranges between 0.18 and 1.69%, with phosphorus being the predominant element. Informed values of the phosphorus content of canna starch are mostly between 300 and 400 ppm, but concentrations from 100 to 779 ppm have been reported [3, 12, 15, 27–30]. This value is high compared to other sources such as cassava, arrowroot, sweet potato, yam, and ginger [28]. The high phosphorus content is one characteristic that canna starch shares with potato starch, the latter exhibiting even higher phosphorus levels, typically ranging from around 300 to 1,200 ppm [31].
In potato starch, phosphorus is primarily found as phosphate monoester, with ~61% located at the C-6 and ~38% at the C-3 glucosyl residue [32]. Similar results have been reported for canna starch, from which 82.2% of the total phosphorus corresponds to starch-bound phosphorus via ester linkage [33], with 69–71% at C-6 of the glucosyl residue [18], and the remainder at C-3 [34]. Most of the phosphate groups in canna starch are found in the long B-chains of amylopectin, which can exist as both phosphorylated and non-phosphorylated chains [18].
The phosphorus content in starch is linked to increased hydration capacity of the granule, influencing paste rheology and stability, and it is also associated with increased paste clarity (Paste clarity) [35]. The composition of canna starches from different regions of Asia and Latin America is shown in Table 1.
Chemical composition of canna starches from different origins.
| Origin | Variety/ecotype | Moisture (%) | Protein (%) | Fat (%) | Ash (%) | Amylose (%) | Starch purity (%) | Reference |
|---|---|---|---|---|---|---|---|---|
| Bolivia | Purple | 8.8 | 0.07 | - | - | 48.0* | 99.1 | [30] |
| Brazil | - | 9.8 | 0.60 | 0.15 | 0.53 | 39.0* | 98.8 | [36] |
| Brazil | - | - | 0.13 | 0.19 | 0.24 | 31.7* | - | [28] |
| Colombia | Nativa, Negra, Verde, and Morada | 13.6–14.2 | 0.20–0.50 | < 0.005 | 0.18–0.34 | 21.2–31.7 | - | [23] |
| Colombia | Verde | 8.3 | 0.80 | 0.05 | 0.22 | 40.7* | 99.8 | [36] |
| Venezuela | - | 18.2 | 0.67 | 0.05 | 0.32 | 13.8* | - | [27] |
| India | - | 11.0 | 0.93 | 0.40 | 0.06 | 38.0* | - | [15] |
| Indonesia | Ganyong merah | 12.3 | 0.80 | - | - | 35.0* | 88.1 | [37] |
| Thailand | Vietnam-purple (different rhizome segments) | - | 0.06 | Nd | 0.18–1.69 | 30.9–36.2* | - | [12] |
| Thailand | Japanese-green, Thai-green, and Thai-purple | 9.4–10.0 | 0.07–0.08 | 0.01–0.02 | 0.25–0.33 | 21.0–28.0*19.0–25.0 | - | [3] |
| Thailand | Vietnam | ~15.0 | 0.06 | Nd | 0.34 | 34* | - | [38] |
| Thailand | Japanese-green, Thai-green, Thai-purple, and Chinese-purple | - | 0.05–0.20 | 0.01–0.15 | 0.70–0.90 | - | 94.0–96.6 | [2] |
*: Apparent amylose; Nd: not detected; -: not informed.
Other minerals reported to be present in canna starch are sodium 27–443 ppm, potassium 35–272 ppm, calcium 24–421 ppm, magnesium 17–204 ppm, iron 19–50 ppm, and zinc 22.1 ppm [3, 12, 27].
Canna starch shows round, oval, and disk-shaped granules, with smooth surfaces, and generally exhibits unimodal distribution (Figure 3A) [2, 3, 15, 23, 37]. This starch stands out because of having the largest granules among the known starch sources. Canna starch granules show medium (10–30 μm) and large-sized (30–100 μm) particles, with mean sizes typically between 35–63 μm (Figure 3) [27, 37, 39–41]. Small granules (< 10 um) are either absent or represent a minimal fraction of the starch [2, 42]. Ren [42] reported that canna starch granules, with an average size of 48.99 μm, were the largest among the 18 starch sources analyzed, followed by potato starch (42.34 μm), which is the largest among the commonly used starch sources. Canna starch shares similarities in shape, size, and smooth surface with potato starch.

Scanning electron micrographs of Canna Indica L. starch. (A) magnification 2,000× and (B) magnification 8,000×. Taken from Dini et al. [43] without modification. © 2024 The Authors. Licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 2.5 Unported.
Andrade-Mahecha et al. [36] compared the starches extracted from 7-month Brazilian rhizomes and from 12-month Colombian rhizomes. The Brazilian starch exhibited a unimodal distribution, with 90% of the granules ranging between 7.5 and 90.5 μm, while the Colombian starch showed a bimodal distribution with one population ranging between 19 and 121 μm, and the other above 200 μm. The presence of unusually large particles in the Colombian starch was corroborated by SEM images [36]. Besides the differences inherent to crop variety, it is necessary to determine the extent to which agroecological growing conditions influence starch granule size.
Rhizome maturity stage also defines the granule size distribution of starch. Puncha-arnon et al. [12] studied the size and morphology of canna starch granules from different segments of the rhizome: the mother segment (initial stage of rhizome development), immature (second stage), premature (third stage) and mature (final stage) segments, and found that the mother segment had lower proportion of large granules (80–140 μm) compared to the others.
The immature, premature, and mature segments had 13.7–19.5% of starch, with smooth granules, while the mother segment showed lower starch content (7.9%). Some defects were observed on the granules from the mother segment, including parallel-striations or wave-like patterned surface, holes, and indentations, indicating starch degradation (Figure 3B) [12]. As the plant grows, the starch granules in the mother segment make up a smaller proportion of the total starch content [12].
Irrespective of the origin, variety, and segment of the rhizome from which it was extracted, canna starch displays a B-type crystalline structure [3, 12, 23, 44].
The X-ray diffractogram of B-type starches is characterized by a small peak at 5.6°, one peak at 17°, and a doublet at 22° and 24° [45]. This crystalline arrangement is typical of root, tuber, and rhizome starches [26] in which six parallel-stranded double helices are arranged in a hexagonal unit cell with water molecules filling the channel inside the hexagonal arrangement [46]. This crystalline form is naturally assembled under relatively high moisture and cool temperature, which are the growth conditions for several underground organs, while in dryer and warmer conditions, such as those in which cereal grains grow, the A-type allomorph is favored [46]. In A-type starch, the double helices are arranged in a monoclinic lattice, more densely packed than in the B-type crystalline structure. There is a third crystalline type, named C-type allomorph, which is a mixture of A- and B-type structures.
The length of the chains involved in the formation of the crystalline lamellae also determines the type of crystal formed, with larger chains tending to yield the B-type form [46].
Informed values for the crystallinity degree of canna starch calculated from the XRD analyses range between 17 and 37%, but are typically around 26–27% [14, 16, 22, 24, 36, 39, 47, 48], similar to kudzu root and potato starches [42].
Despite being influenced by multiple factors, the reported crystallinity values are consistent with a B-type structure typical of starches with relatively high amylose content, in which amylose contributes to amorphous regions within the granules [49]. Phosphate groups may further reduce crystallinity by disrupting the intramolecular hydrogen bonding required for helical crystal formation [35].
The chain length distribution of canna amylopectin shows two main populations: a larger peak, with relative abundance of 43.9–45.3%, located at degree of polymerization (DP) 14–16.6, and a smaller peak (26.8–29.3%) between DP 40 and 57.9 [18, 44, 50–53]. Particularly, the presence of a shoulder in the main peak of canna starch amylopectin at DP 22–24 suggests a defective crystalline structure of the starch granules, which results in lower gelatinization temperatures [44, 51].
The highest detectable DP of canna starch amylopectin chains is between 82 and 85, which is within the values expected for B-type starches, and higher than those of A-type starches (DP 71–80) [51].
The distribution and average chain length (DP 21.9–28.9) of canna amylopectin is almost identical to that of potato starch (DP 21.4–31.3) [18, 34, 50, 51, 53]. Both starches show a lower average length of short (DP ≤ 38) and long (DP ≥ 39) chains than other B-type starches such as lesser yam and shoti [34], which partially explains the lower gelatinization temperature of canna and potato starches compared to other B-type starches (Thermal properties).
Amylopectin chains fractionation usually shows peaks every 12-residue intervals. Therefore, the relative abundance of amylopectin chains is normally presented as fractions of 12 residues: 1–12, 13–24, etc [54]. However, one particular feature that canna starch shares with yam starch is that they apparently exhibit a periodicity of 15 residues in the chain length distribution [52, 54].
Canna starch is rich in amylose, with absolute values typically between 20 and 30% (Table 1). Apparent amylose values have been reported to exceed the absolute ones by up to 20–25% [50, 51]. The overestimation of the amylose content by iodine affinity assay is generally observed in B-type starches due to the high proportion of long branch chains (DP ≥ 37) in their amylopectin [51]. These long chains can bind iodine, resulting in the formation of a single helical complex and the development of a blue color [3], providing higher blue values than other root and tuber starches from A- or C-crystalline type [28].
Regarding the size of amylose, Thitipraphunkul et al. [18] reported two populations for canna starch analyzed by fluorescence labeling and HPLC fractionation: one peak at DP 580–700 and the other at DP 3,010–3,760, with the predominant one of lower molecular size. Bertoft et al. [53] reported two populations for canna amylose separated by gel permeation chromatography and quantified by the phenol-sulfuric acid method, being predominant that of higher molecular weight, but the proportion of short-chain amylose in canna starch resulted considerably higher (39%) than that of other roots and tubers such as cassava or potato (19% and 28%, respectively) [53]. The average DP of canna starch amylose ranges between 1,590 and 1,650, lower than that of mung bean (DP 2,200) and cassava (DP 3,220), and can be classified as small-to-medium size compared to potato (DP 4,370) or sweet potato (DP 3,230) [18].
Unlike the highly branched amylopectin molecules, amylose is mostly linear, showing in some cases a limited number of branching points. The mol percentage of branched amylose in canna starch is 13–16%, and the average number of chains per amylose molecule is between 17 and 21, with chain lengths ranging between DPn 350 and 471 [18]. Canna starch showed a slightly lower percentage of branched amylose than potato, and both were lower than shoti and lesser yam starches (both from B-type). Less branched amylose, along with the shorter amylopectin chains, was associated with the lower gelatinization temperature of canna and potato starches compared to other B-type starches [34].
The phosphorus content in starch is known to be related to its swelling capacity. The negatively charged phosphate groups within the granules create repulsion forces, which facilitate the separation of starch chains and encourage the formation of hydrogen bonds with water molecules [55]. Conversely, amylose tends to inhibit granule swelling [28]. Therefore, the phosphorus and amylose content in canna starch have opposing effects on starch swelling and hydration. Granule size and amylopectin structure also greatly influence starch hydration properties [56]. Therefore, the balance of all these characteristics determines the behavior of each specific sample, for which diverse results have been reported for the swelling power of canna starch compared to other root and tuber starches.
Peroni et al. [28] studied the swelling power of starches from different underground organs between 60 and 90°C, and canna starch showed intermediate behavior among starches from cassava, yam, sweet potato, ginger, and arrowroot, showing values from ~10 to 20 g/g when the temperature increased from 60 to 90°C. Other works informed even lower swelling power for canna starch at 90°C (~12 g/g), lower than potato, cassava, and sweet potato starches (~18–22 g/g) [2, 45]. Canna starches with restricted swelling are suitable for glass noodles production [45].
On the contrary, other works found that canna starch showed the highest swelling power at 90°C compared to sweet potato, konjac, yam, cassava, arrowroot, and taro starch, reaching approximately 55 g/g [37, 40]. Additional studies are required to elucidate which of these structural and compositional characteristics most strongly determine the swelling behavior of canna starch.
Despite variations in the swelling values, numerous studies reported consistent temperature-related swelling profiles for canna starch. These profiles typically involve minimal to no swelling between 55 and 65°C, a notable increase in swelling between 65 and 75°C, followed by a gradual increase up to 80–85°C. From then on, some samples increase steadily up to 90–95°C [37, 45] while others show a sharp increase between approximately 80 and 95°C [15, 29, 57].
A particular case was that reported by Andrade-Mahecha et al. [36], who observed that a starch obtained from 12-month Colombian canna rhizomes swelled significantly between 55 and 65°C. This starch was reported to contain exceptionally large granules, which may be linked to the superior swelling power of this sample [36]. Understanding the relationship between cultivation conditions and rhizome maturity on starch granule size would enable the optimization of growing practices to obtain starches with high cold swelling capacity, without the need for physical or chemical modifications.
Much like the swelling behavior, the solubility of canna starch is notably low in the temperature range of 50 to 60–65°C, typically ranging from 2% to 5% [2, 23]. As the temperature increases, solubility gradually rises, reaching values of 8% to 20% at 90–95°C [2, 15, 22, 23, 29].
Canna exhibits higher solubility than cassava and arrowroot starch between 65 and 90°C [2, 27], and is approximately 3% higher than maize starch at 95°C, which is attributed to its high amylose content [15, 36]. The high proportion of short-chain amylose may also contribute to the solubility of this starch. However, despite being higher than that of other native starches, the solubility of canna starch is not sufficiently high to confer distinctive technological functionality in its native form.
Regarding the pH, the solubility of canna starch exhibits a particular dependency. Increasing the pH from 2 to 4 significantly raises starch solubility from 8.85 to 14.25%. A subsequent increase of pH from 4 to 6 reduces solubility to 3.76%, and from then on, the starch solubility rises with the pH to reach a value of 9.36% at pH 12 [25]. No conclusive explanation for this behavior was provided by the authors, but they hypothesized it was related to the phosphate groups present in the starch [25].
The gelatinization parameters of canna starches from different origins are shown in Table 2. The informed peak temperatures range from 61.2 to 74.2°C, and the gelatinization enthalpy (ΔHG) ranges between 9.4 and 20.1 J/g db. No differences were observed in the thermal properties of canna starch granules obtained from segments of different growth stages of the rhizome [12]. Furthermore, no direct association was detected between the amylose content of canna starches from different varieties or origins and their thermal properties [3, 23, 36].
Thermal properties of canna starches from different origins measured by differential scanning calorimetry.
| Origin | Variety/ecotype | Cultivated in (region/city) | Altitude (m.a.s.l) | Starch:water ratio (w/w) | T0 (°C) | Tp (°C) | Tc (°C) | ΔH (J/g) | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Bolivia | Purple | Cochabamba | 2,300 | 1:3 | 61.5 | 65.6 | 70.0 | 15.4 | [30] |
| Brazil | - | Conchal | 591 | 1:3 | 67.6 | 71.2 | 77.4 | 14.5 | [36] |
| Brazil | - | Botucatu | 645* | 1:3 | 65.4 | 70.1 | 74.8 | 14.2 | [28] |
| Colombia | Nativa, Negra, Verde, and Morada | Huila, Nariño, and Cundinamarca | 1,746–2,240 | 1:2 | 61.2–63.1 | 63.3–65.6 | 67.6–71.1 | 10.6–13.5 | [23] |
| Colombia | Verde | Cundinamarca | 2,291 | 1:3 | 59.9 | 63.7 | 71.6 | 14.0 | [36] |
| Ecuador | -→† | Quito | 2,930* | 1:3 | 56.8 | 61.2 | 67.7 | 15.7 | [44] |
| Venezuela | - | Trujillo, Venezuelan Andes | 923* | - | 58.9 | 63.6 | 72.1 | 9.4 | [27] |
| China | - | - | - | 1:3.5 | 59.7 | 64.2 | 71.9 | 9.5 | [48] |
| China | - | - | - | 1:3 | 60.0–60.6 | 66.8–67.8 | 73.9–75.7 | 14.8–15.1 | [22] |
| Indonesia | Ganyong merah | Tulung Agung (East Java) | 263* | 3:11 | 64.1 | 69.8 | 77.5 | 11.4 | [37] |
| Indonesia | - | Java | 39* | 1:2 | 67.4 | 70.0 | 78.9 | 18.7 | [39] |
| Sri lanka | Buthsarana | Peradeniya | 519* | 1:3.3 | 68.7 | 71.3 | - | 20.1 | [40] |
| Thailand | Vietnam-purple (different rhizome segments) | Rayong | 38* | 1:2 | 70.8–71.5 | 73.6–74.2 | 76.9–77.6 | 16.6–18.0 | [12] |
| Thailand | Vietnam | Rayong | 38* | 1:2 | 69.2 | 71.9 | 74.8 | 15.1 | [38] |
| Thailand | Japanese-green, Thai-green, and Thai-purple | Bangkok | 4* | 1:2 | 65.8–66.8 | 67.7–68.9 | 70.3–71.6 | 17.6–18.4 | [3] |
| Vietnam | - | Northern part of Vietnam (unspecified region) | - | 3:10 | 67.4 | 70.8 | 76.1 | 14.5 | [45] |
| Vietnam | - | Hanoi | 41* | 1:3 | 66.4 | 70.1 | 74.0 | 16.9 | [58] |
-: Not informed; *: not specified in the referenced work, indicative average values from https://en-gb.topographic-map.com/; †: CIP cultivar MH-1173.
Srichuwong et al. [39] studied the thermal properties of starches from different botanical sources and crystalline types. The ΔHG and gelatinization temperature, which measure the unwinding of the double helices in the crystalline lamellae and the melting of the lamellae, resulted independent from the granule size or shape, or the apparent amylose content, but were positively correlated with the length of the chains forming the crystalline domain. This is why these features tend to be higher in B-type than in A-type starches [59]. However, canna and potato starches show the high ΔHG proper of B-type starches, but much lower gelatinization temperature than that expected for their structure.
According to Aprianita et al. [37], the peak temperature of canna starch was one of the lowest among starches from sweet potato, konjac, yam, cassava, arrowroot, and taro, while the ΔHG was among the highest.
The low gelatinization temperature of canna and potato starches is probably related to a combination of high phosphorus content and a high proportion of amylopectin chains with DP ≤ 12 compared to other B-type starches, which may have a detrimental effect on the stability of crystalline lamellae [34], and may enable the gelatinization process to initiate earlier than other starches from the same crystalline type.
Gelatinized canna starch retrogrades to a great extent despite phosphate groups in starch tending to reduce starch chains’ reassociation [35]. The high retrogradation tendency of canna starch has been mainly associated with its high amylose content [60].
The retrogradation percentage of canna starch, calculated from the informed retrogradation and gelatinization enthalpies (retrogradation enthalpy (ΔHR) and ΔHG, respectively) as ΔHR/ΔHG × 100, ranges between 36.1 and 55.6% [12, 28, 39, 45], higher than cassava (2.4–17.6%) and sweet potato (13.0%) [39, 45], but lower than yam and ginger (74.1% and 68.6%, respectively) [28]. When compared to potato starch, retrogradation percentages are similar or higher for canna starches with higher amylose content [39, 45] and lower for those with lower amylose values [50].
High retrogradation tendency of starch is related to reduced shelf life in some food products, but is also associated with lower starch digestibility. The mechanisms underlying the unusually low gelatinization temperature of canna starch despite its B-type crystalline structure, as well as its pronounced retrogradation tendency despite a relatively high phosphorylation degree, are not yet fully understood. Further integrative studies combining advanced structural techniques with thermal, rheological, and hydration analyses are required to establish causal relationships and predictive models.
Canna starch swelling is governed by the counterposed effects of the high amylose content, which restricts it, and the phosphate monoester content, which improves starch hydration and swelling. Starches that exhibit high swelling often show high peak viscosity (PV) in the Rapid Visco Analyzer (RVA) profile, due to the friction of the swollen granules, and a pronounced drop of viscosity (high breakdown) during the holding stage [61]. The pasting profile of canna starch is often characterized by high PV (Table 3), higher than ginger, sweet potato, konjac, yam, cassava, arrowroot, and taro starches [28, 37, 42], but exceptions have been reported for samples that show restricted swelling [45].
Pasting parameters of canna starch measured by Rapid Visco Analyzer.
| Origin | Variety/ecotype | Starch concentration | Pasting T (°C) | PV (RVU) | BD (RVU) | SB (RVU) | FV (RVU) | Reference | |
|---|---|---|---|---|---|---|---|---|---|
| - | - | 5% w/w | 71.1 | 183 | 2* | 113** | 294 | [51] | |
| Sri lanka | Buthsarana | 6% w/w | 73.5 | 348 | 172 | 40 | 217 | [40] | |
| Thailand | Japanese-green, Thai-green, and Thai-purple | 6% w/w | 71–72 | ~170–258 | 18–115 | 65–122 | ~240–300 | [3] | |
| Thailand | Thai-purple and Thai-green | 6% w/w | 70–70.6 | 140.6–154.1 | 7.2–10 | 103.7–115.7 | 237.1–259.8 | [62] | |
| Thailand | Vietnam | 6% w/w | 73.7 | 145.8 | 24.1 | 154.6 | 276.2 | [38] | |
| Thailand | Vietnam-purple (different rhizome segments) | 6% w/w | 74–76 | 120–123 | 17–21 | 114–126 | 216–233 | [12] | |
| - | - | 8% w/w | 72.4 | 397.2 | 139.8 | 123.8 | 381.2 | [29] | |
| Brazil | - | 9% w/w | 71.2 | 413 | 198 | 120 | 335 | [28] | |
PV: peak viscosity; BD: breakdown; SB: setback; FV: final viscosity; RVU: rapid visco units; -: not informed. *: Calculated from the informed values as: hot paste viscosity – peak viscosity. **: Calculated from the informed values as: final viscosity – hot paste viscosity.
Canna starch shows pasting temperatures between 70 and 76°C (Table 3) in agreement with the increased swelling observed between ~60 and 70°C. There are disparate reports about the breakdown values of canna starch, with some studies indicating high breakdown values [28, 37] and others reporting low or exceedingly low breakdown values, associated with excellent hot paste stability (Table 3).
This discrepancy could be partially explained by the paste concentration used in different studies. Srichuwong et al. [29] reported a significant increase in breakdown of different starch sources when the starch concentration was raised from 6% to 8% w/w. This increase was attributed to heightened friction and rupture of the swollen granules. Notably, among the 15 starch sources examined, this effect was particularly pronounced for elephant yam, new cocoyam, and edible canna starches [29]. This observation aligns with the trend observed in Table 3, where more concentrated canna starch pastes generally exhibited higher breakdown values. However, assays using different concentrations of the same canna starch sample are needed to categorically accept or refuse this hypothesis.
During the cooling stage of the RVA, many studies consistently report a substantial setback (SB) for canna starch, which aligns with the starch’s high retrogradation tendency observed in DSC analyses. This retrogradation leads to final viscosities considerably higher than the PV in most cases (Table 3) [2, 3, 41]. Final viscosity of canna starch has been reported to be higher than sweet potato, konjac, yam, arrowroot, and taro starches, and more than double that of maize starch [51]. Furthermore, among canna starches derived from different ecotypes, those with higher amylose content tend to exhibit higher SB values [3, 41]. No differences were observed in the pasting profile of canna starches isolated from different segments (immature, premature, mature, and mother) of canna rhizomes [12].
High SB values, indicative of a strong tendency toward retrogradation, are associated with bread staling and reduced gel stability. Consequently, starches exhibiting this behavior are traditionally used in products such as crunchy biscuits like “bizcocho de achira”, freshly prepared and non-refrigerated gels as in “colada de achira”, and glassy noodles, which differ fundamentally from wheat-based pasta. Glass noodles are manufactured from starch suspensions that undergo controlled gelatinization followed by extrusion or dripping and subsequent amylose retrogradation, being this last step essential for the development of a transparent, cohesive noodle structure [63].
Acidification significantly modifies the RVA profile of canna starch pastes and could be used for tuning the behavior during heat processing. In the work of Chuenkamol et al. [64], a marked decrease in PV was observed when the pH of the medium was lowered from 5.6 to 4, indicating that starch granules became fragile and prone to breakage, but no changes were observed in the SB. This agrees with the higher solubility observed by Zhang et al. [25] at pH 4 compared to pH 6. Further reductions to pH 3 and 2 did not produce noticeable modification in the PV, as starch solubility was reported to be decreased [25], but greatly reduced starch retrogradation. This reduction was probably related to the presence of shorter starch chains resulting from acid hydrolysis, which could not form highly ordered crystalline structures [64].
Paste clarity is regularly expressed as the transmittance percentage of a 1% gelatinized starch suspension. This feature is crucial because a transparent or glossy appearance is critical in foods where the starch paste is visible. It is also a crucial feature for the elaboration of glass noodles [56].
Phosphate content in native starches is directly related to starch hydration, and the separation of starch chains consequently leads to a reduction in paste transmittance [55]. There is a threshold level of phosphorylation, beyond which the transmittance of the pastes begins to decrease due to a weakening effect on the hydrogen bonds between starch and water molecules, for which synthetically phosphorylated starches may show lower paste clarity than the native ones [55].
Another factor affecting paste clarity is granule size. Starches with larger granules tend to have better swelling and hydration capacity [55]. In this regard, both the phosphorus content and granule size of canna starch have a positive impact on its paste clarity, while no relationship was observed between amylose percentage and transmittance of freshly prepared pastes [3, 23, 37]. Canna starch forms clear pastes, but due to its fast and extensive retrogradation, the opacity increases rapidly with storage time. Canna starch from cultivars Japanese-green, Thai-green, and Thai purple, all with similar phosphorus contents (362–399 ppm), showed similarly high paste clarity, between 78 and 82%, which decreased to 46–64% after 60 min [3]. Comparable values were informed for the aforementioned canna ecotypes plus the Chinese-purple after 60 min storage (43.1–51.3%), and for ecotypes nativa, verde, negra and morada after 30 min (43.1 to 58.9%) [2, 23].
Fonseca-Santanilla and Betancourt-Lopez [14] found that canna starch displayed higher paste clarity, measured after 15 min (approximately 35.5%), compared to sweet potato (19.5%) and oca (Oxalis tuberosa) starch (approximately 24.1%). Hung and Morita [45] reported that the paste clarity of canna starch was similar to that of cassava starch. However, in a study involving the storage of starch pastes, canna showed considerably lower paste clarity (~60%) than cassava (~70%) after 1 hour and was less clear during the whole storage period (7 days). Both starches displayed a reduction in paste transmittance over time, but this decline was more pronounced for canna starch, reaching a final value of around 30%, much lower than that of cassava starch (~55%) [37]. Furthermore, canna starch showed similar or higher paste transmittance than taro, yam, sweet potato, and arrowroot starches after 1 hour storage, while after 7 days, canna starch displayed the lowest value [37].
Canna starch tends to form firm gels mainly due to its high amylose content. The firmness of canna starch gels plays a crucial role in maintaining the texture of gel pieces in the “colada de achira” dessert.
At low concentration (2% w/v), gelatinized canna starch exhibits the pseudoplastic behavior typical of starch pastes [41]. The texture analysis of more concentrated starch gels (8% w/w) showed the peak puncture force of canna starch was almost 4 times higher than that of arrowroot starch, and 11 times higher than cassava starch [65]. In another work, the hardness of 8% w/w gelatinized starch gels from two Peruvian canna ecotypes was compared to that of potato and corn starches. The San Gabán canna ecotype showed significantly higher hardness (34.6 N) than the potato and corn gels (27.6 N and 20.4 N, respectively), while the Sandia ecotype had significantly lower hardness (8.0 N) [41]. The authors attributed this difference to the higher amylose content of the San Gabán ecotype compared to Sandia [41]. However, the phosphorylation degree also influences the stability of the starch gel and should be considered.
Canna starch gels show low stability during refrigerated storage, which makes them unsuitable for refrigerated or frozen foods. A 10% (w/w) canna starch gel stored at 4°C for 7 days exhibited lower freeze-thaw stability, measured as the exuded water (syneresis), than mung bean starch, and much lower than cassava starch [3]. The reduced stability can be attributed to the substantial retrogradation (67–82%) observed in canna starch during the storage period, as determined by DSC analysis [3]. The high content of amylose is the main cause for the elevated retrogradation and syneresis of canna starch gels [37, 45].
The stability of canna starch gel during cooling was similarly low to that of yam and konjac starches and was all less stable than starches with lower amylose contents such as sweet potato, cassava, arrowroot, and taro [37]. Similarly, Hung and Morita [45] demonstrated that the stability of refrigerated and frozen gelatinized canna starch was considerably lower than that of sweet potato, potato, and cassava starch. In the first week of storage at 4°C, canna starch showed 42.3% syneresis at 4°C, similar to the value informed by Jane et al. [51] (45.2%), while those of potato, cassava, and sweet potato were between 3.8 and 7.3%. At –18°C, canna starch showed 72.0% syneresis, while potato, sweet potato, and cassava showed values of 59.2, 40.4, and 2.9%, respectively [45]. When subjected to several freeze-thaw cycles, a syneresis % of 73.5 was observed in the first cycle, while the second and third cycles only slightly increased the syneresis to 74.7 and 74.9%, respectively [66].
The freeze-thaw stability of starches from different sources was negatively correlated with the apparent amylose content and positively correlated with the proportion of short chains (DP 6–12) in amylopectin [66]. Canna starch has high amylose content, and, despite having a higher proportion of short chains in amylopectin than other B-type starches, the proportion is low compared to A-type starches, both of which are associated with its low stability to freezing and thawing.
Starches with B-type crystalline structure often have lower digestibility than A-type and C-type starches [39]. The hydrolysis degree of B-type starches from water yam, canna, and potato with α-amylase from porcine pancreas did not significantly change with the incubation time, reaching only 2.5–7.2% of hydrolysis after 72 h, while rice, yam bean, and cassava starches (A-type) were hydrolyzed to 88.4–89.0% after the same period [39].
Lower differences in the degree of hydrolysis of B-type starches (canna and potato) compared to A-type (cassava) and C-type (sweet potato) starches were observed by Hung and Morita [45]. Canna and potato starches treated with α-amylase for 48h showed degrees of hydrolysis of 51 and 48%, respectively, while cassava and sweet potato reached 93.6 and 88.5% of hydrolysis, respectively. SEM observation of the hydrolyzed canna starch revealed that some of the large granules were broken into fragments or exhibited disc-like depressions on their surfaces, while others were still intact with no evidence of α-amylase attack [45]. Puncha-arnon et al. [12] suggested that the primary mode of α-amylase attack on canna starch was through surface corrosion.
The resistant starch (RS) content, measured using an enzymatic kit (K-RSTAR, Megazyme, Ireland), was notably high for canna starch (70.8%) compared to taro, yam, sweet potato, arrowroot, konjac, and cassava starches, which exhibited values between 3.3 and 20.9% [37]. Furthermore, when exposed to a mixture of α-amylase and amyloglucosidase, canna starch showed significantly lower digestion 31.3–44.5% compared with cassava starch (54.6%) [2]. Raw canna starch also showed high resistance to digestion with glucoamylase from Rhizopus sp. compared to yam, sweet potato, taro, arrowroot, and cassava, and this was attributed to their larger granules compared with the other sources [40].
Among granules from the same crystalline type, granule size and phosphate monoester content are both negatively related to starch digestibility [39, 67]. The digestibility of raw starch with α-amylase also showed a negative correlation with the blue value, final viscosity, pasting temperature, and starch ΔHG, and was positively correlated with the SB in the RVA profile [41]. Canna starch shows most of the characteristics associated with low digestibility, which explains its high resistance to enzymatic hydrolysis.
Gelatinization of the starch for 5 min at 100°C reduced the RS of Vietnam and Thai green canna varieties from 88.1–88.2% to 26.1% and 22.9%, respectively [68]. Huang et al. [22] and Wandee et al. [60] reported higher RS values in native canna starch: 91.3–93.8% and 97.3%, respectively, and lower values for the gelatinized starch: 9.4% and 1.9%, respectively. Both studies reported that the RS content was significantly increased to 12.7–16.8% in the retrograded starch, for which canna starch is a favorable starting material for type-3 RS production [22, 60].
When incorporated in the formulation of wheat flour noodles, substitutions from 8% to 40% of wheat flour with canna starch significantly reduced the rapidly digestible starch (RDS) and increased the RS in the cooked paste [69]. Canna type-3 RS proved to have prebiotic activity, promoting the growth of beneficial genera such as Bifidobacterium, Faecalibacterium, and Agathobacter in the human gut [70, 71] and produced a hypoglycemic effect and increased high-density lipoprotein (HDL) levels in broilers [72].
While promising, the available evidence remains largely limited to in vitro studies and animal models. Human intervention trials assessing glycemic response, gut microbiota modulation, and metabolic outcomes are still lacking. In addition, the effects of realistic food processing and cooking conditions on starch digestibility and RS retention have not been sufficiently investigated. Addressing these gaps is essential to substantiate health-related claims and to support future regulatory approval.
Piyachomkwan et al. [2] studied the susceptibility of canna and cassava starches to acid hydrolysis using sulfuric acid 1.35 N. The authors found that canna starch underwent faster hydrolysis, with degradation rates ranging from 43.1 to 51.3% after 24 hours, compared to cassava starch, which showed a degradation rate of 38.0%. After 72 h, canna starch reached degradation levels between 89.9 and 91.5%, whereas cassava starch experienced a degradation rate of 93.4% [2].
Mendez-Montealvo et al. [47] assayed the acid hydrolysis of canna starch (1/20 w/v) in hydrochloric acid (7.5% w/w) at 25°C. The authors also reported a rapid degradation of canna starch in the first 3 days. After 15 days of treatment, the hydrolysis percentage of canna starch was 40.3%, higher than that of normal and high amylose maize starches (18.8 and 13.8%, respectively), but much lower than that reported for potato starch at 40°C (89.8%). This suggests that both canna and potato starches have a poorer internal structure with crystalline regions more susceptible to acid attack compared to A-type starches.
Besides the crystalline type, this susceptibility was also attributed to the presence of phosphate groups covalently bound to amylopectin chains, weakening the interaction between chains in the crystalline domain [47].
Acid hydrolysis reduced the swelling power and increased the solubility and RS content of canna starch [47, 73] and reduced the viscosity of the starch paste [74]. As mentioned above, acid treatment offers a relatively simple strategy to modulate starch functionality and adapt it to different applications.
Several cross-cutting features are consistently observed among all samples and underpin properties characteristic of canna starch. Table 4 summarizes the main structural features of Canna indica starch and relates them to its technological and functional properties. Building upon these foundational characteristics, recent research has increasingly focused on modification strategies to enhance technological performance and promote RS formation.
Relationship between Canna indica starch features and technological properties.
| Starch feature | Related technological/functional property |
|---|---|
| High organic phosphorus content (371–420 ppm) | Paste clarity and consistency: High phosphorus is directly linked to the production of exceptionally transparent pastes and high initial consistency. |
| Exceptional granule size (very large, typically 10 to 100–140 µm) | High peak viscosity: Larger granules generally correlate with higher peak viscosity and swelling power. |
| High amylose content (reported ranges: 13.7% to ~40%) | Strong retrogradation: High amylose levels drive the rapid reassociation of starch chains, creating firm, elastic gels. Noodle quality: essential for providing the high tensile strength and “bite” required for high-quality transparent noodles. |
| B-type crystalline pattern | Enzyme resistance: low enzymatic susceptibility, resulting in high resistant starch content. |
Some physical and chemical modifications have been assayed on canna starch to improve its characteristics as a food ingredient, such as pasting properties, gel stability, or digestibility. The main results of these modifications and their potential implications for the food industry are summarized in Table 5.
Properties of modified canna starches and their potential implications for the food industry.
| Modification | Main results | Potential implications for the food industry | References |
|---|---|---|---|
| Heat-moisture treatment | Reduced swelling, solubility, peak viscosity, breakdown, and retrogradation. Changes were more evident when higher moisture was used in the treatment | Softer gels with improved hot paste stability and stability during storage. Reduced staling in bakery products | [25, 38, 68] |
| Cross-linking | Reduced swelling and digestibility of raw starch. Changes in the viscosity profile | Better suitability for starch noodles production | [68, 75] |
| Acetylation | Reduced peak viscosity. Improved gel stability towards retrogradation, and enhanced paste clarity | Softer gels with improved stability during storage. Useful in translucent products such as fruit fillings | [62] |
| Hydroxypropylation | Increased peak viscosity and reduced setback. Reduced gelatinization temperature and enthalpy. Improved gel stability towards retrogradation, and enhanced paste clarity | Suitable as a thickening agent. Improved storage stability and lower cooking temperature. Reduced the opacity of the final product | [64] |
| Octenylsuccinilation | Increased paste viscosity. Reduced the digestibility of both raw and cooked starch | Suitable as a thickening agent with reduced glycemic response | [68, 76] |
Heat-moisture treatment (HMT) refers to the exposure of starch granules to a temperature below the gelatinization temperature but above the glass transition temperature, at restricted moisture content (below 35%). This process leads to a reorganization of the starch chains, resulting in modifications to the crystalline structure and overall starch characteristics [68].
Canna starch treated at moisture levels from 15 to 25% at 100°C showed no alteration in granule size or shape, or the crystalline structure [38], but moisture levels of 18–27% at 110°C produced a transition from B-type to A-type crystalline structure [25], more pronounced as higher moisture levels were used in the treatment. Transitions to A-type structures were also observed by Chatpapamon et al. [77].
HMT reduced the swelling power and solubility of canna starch due to strengthened interactions between starch chains. This change was also more pronounced when higher moisture was used in the treatment [25]. HMT also reduced the water binding and oil binding capacity, increased the acid susceptibility and the gelatinization temperature, and slightly decreased the ΔHG of canna starch [25].
Regarding the pasting properties, the HMT-starch showed remarkably lower paste viscosity than the native starch, in agreement with the reduced granule swelling [77, 78]. The HMT-starch also showed improved paste stability, showing no breakdown, attributed to the higher granules’ resistance to heat and shear, and a noticeable decrease in the SB viscosity compared to the native starch [25, 38]. These changes were similar to those reported for other HMT-modified root and tuber starches such as potato, cassava, true yam, and taro [38].
No clear trend was observed in the digestibility of HMT-starches. According to Watcharatewinkul et al. [79], the starches treated at moisture levels of 20 and 22% showed lower susceptibility to α-amylase than the native starch, while those treated with 15 and 25% moisture showed a higher percentage of hydrolysis than the native starch.
Juansang et al. [68] reported that raw HMT canna starches obtained at moisture percentages of 18%, 22%, and 25% contained considerably higher amounts of RS (92.2–94.8%) than the native starch (88.1%), but cooking markedly reduced the RS to values similar or lower than those of the cooked native starch. Controlled heating of HMT-modified canna starch suspensions, followed by freezing the swollen granules, reduced the rate of digestion of the HMT-starch [80].
Cross-linking is a chemical modification in which functional groups are introduced into the starch chains, strengthening the interactions between them.
Mung bean starch is considered ideal for manufacture of glass noodles. Lii and Chang [75] found that substituting 30–50% of mung bean starch with crosslinked canna starch resulted in glass noodles that met the desired quality in sensory tests. Cross-linking restricted swelling and changed the Brabender viscosity profile of canna starch, making it more similar to mung bean starch [75]. These characteristics of crosslinked canna starch resemble those of HMT-starch, for which HMT-canna starch could be an interesting alternative to explore in the production of glass noodles.
Regarding the digestibility, cross-linked raw canna starch has significantly higher proportion of RS (94.1%) than the native starch (88.2%). However, the RS content diminishes rapidly during cooking, reaching 19.8% after 40 min of gelatinization [68].
The introduction of acetyl groups into starch reduces the interaction strength between starch molecules, thereby increasing the swelling power and solubility, lowering the gelatinization temperature, decreasing starch retrogradation, and improving paste clarity and freeze-thaw stability.
Saartrat et al. [62] assayed the acetylation of canna starch at low degrees of substitution (1.5–2.5%) to reduce its retrogradation tendency. Granule morphology and size were not altered by the treatment. The PV of the acetylated starches was 13–20% lower than that of the native starch, making them less suitable as thickening agents. Nevertheless, acetylation also lowered the gelatinization temperature by 3–5°C and reduced the retrogradation and syneresis during freeze-thaw studies, resulting in softer and clearer gels.
Hydroxypropylation is an etherification with propylene oxide in alkaline medium, which replaces hydroxyl groups with -O-CH2CH2CH2-OH groups, providing hydrophilicity and enabling the disruption and weakening of the granule internal structure [11].
Hydroxypropylation was assayed on canna starch with propylene oxide (0.5 to 10% v/w) at 40°C to reach molar substitutions of 0.01 to 0.11 [64]. The treatment did not significantly change the granule size or shape, or the X-ray diffraction pattern of the starch. However, in RVA assays, increasing molar substitutions resulted in a decrease in pasting temperature and SB, and increased PV. These changes reflect a weakening of the associative bonding forces within the granule and the hindrance of chain reassociation and alignment during retrogradation due to the voluminous hydroxypropyl groups introduced [64].
Hydroxypropylation reduced the gelatinization parameters (To, Tp, Tc, and ΔH) of canna starch, which is also related to the disruption of hydrogen bonds between starch chains. Hydroxypropylated starch produced softer gels, and the paste clarity was maintained after 7 days at 4°C due to reduced retrogradation. The freeze-thaw stability was significantly improved, and no syneresis was observed for the starch with the highest molar substitution [64].
Octenyl succinic anhydride (OSA) is used to modify starch surface by esterification, providing an amphiphilic character and conferring emulsifying properties to starch. OSA-modification can also change the rheological properties and the digestibility of starch [76].
Octenylsuccinylation did not change the crystalline structure and the morphology of canna starch granules, except for the appearance of some pores on the surface [76]. However, it greatly restricted enzymatic attack, significantly reducing starch digestibility [76]. OSA-starch showed significantly lower digestibility than the native starch in both raw and gelatinized form [68]. The resistant and slowly digestible starch fractions in the gelatinized starch paste increased from 19.2 to 52.2% and from 2.8 to 14.5%, respectively, while that of RDS decreased from 78.0 to 33.3% when the degree of esterification increased from 0 to 0.0121 [76]. OSA-modification also increased the phosphorus content, swelling power and paste viscosity compared to the native starch [74, 76].
The most outstanding features of canna starch are its extremely large granule size, exhibiting B-type crystalline structure, elevated phosphorus content and low digestibility, which resemble those of potato starch. A high amylose content has likewise been consistently reported across most samples and contributes to the technological properties of this starch.
Canna starch is characterized by a low gelatinization temperature and high PV. However, discrepancies have been reported regarding hot paste stability, showing some canna varieties low or exceedingly low breakdown values, which would typically make them suitable as thickening agents, particularly for instant foods requiring short cooking times, whereas other samples show high breakdown values associated with poor hot paste stability. It remains unclear whether these differences are attributable to intrinsic differences among starch samples or to the starch concentration used in pasting profile analyses. pH also plays a crucial role in determining the pasting behavior. The high susceptibility of canna starch to acid hydrolysis is reflected in significant changes in the RVA profile, even with slight variations in the medium pH.
A high retrogradation tendency is a recurrent feature across all samples, suggesting that native canna starch is primarily suitable for foods consumed hot and freshly prepared, limiting its application in refrigerated or frozen products, while making it particularly promising for the production of type-3 RS.
A major knowledge gap lies in the absence of a systematic framework linking genotype, agroecological conditions, rhizome developmental stage, and starch extraction and purification processes to starch composition and, ultimately, functionality. Future research should prioritize harmonized analytical protocols and multivariate approaches to identify robust structure-property relationships. Establishing functional quality criteria tailored to specific end uses will be critical for both scientific comparability and technological valorization.
Drastic changes in the rheology of starch pastes, retrogradation behavior, and digestibility can be achieved through physical and chemical modifications, which may reduce syneresis and help maintain paste clarity during storage, thereby expanding its potential applications in the food industry. However, a deeper understanding of the structure-function relationships of native starch would facilitate the prediction of how such modifications influence its technological performance.
There is a need for systematic evaluation of the industrial feasibility, scalability, and cost-effectiveness of these modification approaches, as well as comparative performance studies against commercially established starches in real food systems. Addressing these aspects is crucial for expanding the applicability of canna starch beyond its traditional food uses.
db: dry basis
DP: degree of polymerization
HMT: heat-moisture treatment
OSA: octenyl succinic anhydride
PV: peak viscosity
RDS: rapidly digestible starch
RS: resistant starch
RVA: Rapid Visco Analyzer
SB: setback
wb: wet basis
ΔHG: gelatinization enthalpy
ΔHR: retrogradation enthalpy
The use of images under Creative Commons licenses is gratefully acknowledged.
CD: Conceptualization, Writing—original draft. The author read and approved the submitted version.
The author declares no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
All datasets generated for this study are included in the manuscript.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1079
Download: 70
Times Cited: 0
