Affiliation:
1Department of Food Science and Nutrition, Kyungpook National University, Daegu 41566, Korea
ORCID: https://orcid.org/0000-0002-0518-3390
Affiliation:
2Department of Food and Nutrition, Yeonsung University, Anyang 14011, Korea
Email: bumsik@yeonsung.ac.kr
ORCID: https://orcid.org/0000-0001-8793-7696
Explor Foods Foodomics. 2026;4:1010122 DOI: https://doi.org/10.37349/eff.2026.1010122
Received: February 02, 2026 Accepted: February 27, 2026 Published: March 13, 2026
Academic Editor: Zuhaib F Bhat, SKUAST-Jammu, India
Aim: This study aimed to compare the antioxidant and anti-inflammatory properties of Sparassis crispa (S. crispa) extracts prepared using different extraction methods and to evaluate how extraction conditions influence bioactive component profiles and biological activities.
Methods: S. crispa was extracted using hot water (SC-HWE), high-temperature and high-pressure water (SC-HPWE), and 70% ethanol (SC-EE). Total polyphenol and flavonoid contents, β-glucan content, and antioxidant activities [2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), ferric reducing antioxidant power (FRAP), superoxide dismutase (SOD)-like activity, and catalase-related activity] were evaluated. Anti-inflammatory effects were assessed in lipopolysaccharide (LPS)-stimulated RAW 264.7 (murine macrophage cell line) macrophages by measuring cell viability, nitric oxide (NO) production, and pro-inflammatory cytokine [interleukin (IL)-1β, IL-6, and tumor necrosis factor alpha (TNF-α)] production using enzyme-linked immunosorbent assay (ELISA). Cytokine levels were expressed as a percentage of the LPS-treated control.
Results: Extraction methods significantly affected the composition and bioactivities of S. crispa extracts. SC-EE exhibited the highest total polyphenol and flavonoid contents and showed higher DPPH radical scavenging activity and NO inhibitory effects. SC-HPWE contained the highest β-glucan content and demonstrated superior FRAP values along with notable NO inhibitory activity. All extracts reduced LPS-induced IL-1β, IL-6, and TNF-α production; IL-1β showed greater responsiveness to extract treatment, whereas TNF-α exhibited relatively modest changes. At higher concentrations, the suppressive effect on cytokine production was attenuated, indicating a modulatory rather than a strictly monotonic response. Under the present experimental conditions, quercetin showed a limited reduction in cytokine production.
Conclusions: These results demonstrate that S. crispa extracts exhibit extraction method-dependent antioxidant and anti-inflammatory activities. The observed effects may reflect the combined contributions of phenolic compounds and β-glucan rather than a single bioactive component. S. crispa extracts may serve as promising natural materials for functional applications related to oxidative stress and inflammation regulation.
In recent years, functional foods and bioactive materials derived from natural sources have received increasing global attention as strategies for health promotion and disease prevention [1]. In addition, concerns regarding the safety of long-term use of synthetic antioxidants and anti-inflammatory agents have emphasized the importance of exploring natural product-based functional materials and scientifically validating their biological efficacy across the food, pharmaceutical, and cosmetic industries [2]. Within this context, mushrooms have attracted attention as natural resources that combine edibility with biological activity, and they have been reported to contain various functional components, including polysaccharides, phenolic compounds, and triterpenoids [3–5].
Excessive production of reactive oxygen species (ROS) can disrupt cellular redox homeostasis, leading to oxidative stress that contributes to cellular damage and inflammatory responses. In macrophages, oxidative stimulation promotes nitric oxide (NO) production and the secretion of pro-inflammatory cytokines, which are key mediators of chronic inflammation. Accordingly, antioxidant and anti-inflammatory activities are closely interconnected physiological responses, and integrated evaluation of these activities has been widely applied in assessing the functional efficacy of natural materials. However, the biological activities of naturally derived functional materials are influenced not only by the types of constituent compounds but also by extraction processes that affect the release characteristics of these components [6, 7]. In particular, for complex matrices such as mushrooms, where polysaccharides coexist with low-molecular-weight phenolic compounds, extraction solvent polarity, temperature, and pressure conditions are key factors determining the composition of functional components and the resulting biological activities [8].
Sparassis crispa (S. crispa) is an edible mushroom naturally distributed in coniferous forests of East Asia, and its value as a functional mushroom has increased following the establishment of artificial cultivation techniques. S. crispa is rich in polysaccharides, particularly β-glucan, which have been reported to exhibit immunomodulatory, anticancer, and anti-inflammatory activities [9–11]. In addition, phenolic compounds such as veratric acid have been identified in S. crispa, suggesting its potential as an antioxidant-related functional material [12, 13]. Because S. crispa contains functionally distinct components such as polysaccharides and phenolic compounds, extraction conditions may differentially influence the release of these components and the expression of biological activities. However, systematic comparative studies directly evaluating the effects of different extraction solvents and conditions on the antioxidant and anti-inflammatory activities of S. crispa remain limited. Therefore, the present study prepared hot water, high-temperature and high-pressure water, and ethanol extracts of S. crispa and comparatively evaluated their antioxidant and anti-inflammatory activities according to the extraction processes. Through this approach, the study provides quantitative evidence on extraction method-dependent functional properties of S. crispa, contributing to the development of functional foods and naturally derived bioactive materials.
S. crispa used in this study was provided by Gyeongshin Bio Co., Ltd. (Korea) in 2024. Reagents used for antioxidant activity assays, including 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), Folin-Ciocalteu’s phenol reagent, gallic acid, pyrogallol, ferrous chloride, ferrozine, and 2,4,6-tris(2-pyridyl)-s-triazine (TPTZ), as well as lipopolysaccharide (LPS, Escherichia coli O111:B4) and quercetin, were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Mouse interleukin (IL)-1β, IL-6, and tumor necrosis factor alpha (TNF-α) enzyme-linked immunosorbent assay (ELISA) kits used for pro-inflammatory cytokine analysis were obtained from R&D Systems (Minneapolis, MN, USA). RPMI-1640 medium, fetal bovine serum (FBS), and penicillin-streptomycin used for cell culture were purchased from Gibco (Grand Island, NY, USA). All other reagents were of analytical reagent grade. RAW 264.7 (murine macrophage cell line) macrophages were obtained from the Korean Cell Line Bank (KCLB, Seoul, Republic of Korea). The cell line was authenticated by the supplier prior to distribution and routinely tested for mycoplasma contamination. Cells were used at low passage numbers for all experiments.
S. crispa extracts were prepared using three different extraction methods: hot water extraction, high-temperature and high-pressure water extraction, and ethanol extraction. For hot water extraction, distilled water equivalent to 10 times the sample weight was added, and extraction was performed under reflux at 90°C for 5 h. For high-temperature and high-pressure water extraction, distilled water equivalent to 10 times the sample weight was added, and extraction was conducted using an autoclave at 121°C for 30 min. For ethanol extraction, 70% ethanol equivalent to 10 times the sample weight was added, and extraction was carried out under reflux at 50°C for 3 h. All extracts were filtered through filter paper (Whatman No. 2, Whatman, Maidstone, UK) and concentrated under reduced pressure using a rotary vacuum evaporator (R-210, BUCHI, Kyudo, Japan). The concentrated extracts were freeze-dried at –70°C using a freeze dryer (FD8512, Ilshin, Seoul, Korea), and the freeze-dried samples were stored in an ultra-low-temperature freezer until use. The hot water extract, high-temperature and high-pressure water extract, and ethanol extract were designated as SC-HWE, SC-HPWE, and SC-EE, respectively, throughout the study.
Total polyphenol and total flavonoid contents of the S. crispa extracts were determined using modified methods described by Folin and Denis [14] and Moreno et al. [15], respectively. For total polyphenol analysis, each sample solution was mixed with Folin-Ciocalteu’s phenol reagent and sodium carbonate solution, followed by incubation in the dark for 2 h. Absorbance was measured at 750 nm using a microplate reader. Gallic acid was used as the standard, and results were expressed as mg gallic acid equivalents (GAE)/g extract.
Total flavonoid content was measured by reacting the sample solution with aluminum nitrate and potassium acetate in 80% ethanol. After incubation at room temperature for 40 min, absorbance was recorded at 510 nm. Quercetin was used as the standard, and results were expressed as mg quercetin equivalents (QE)/g extract.
β-Glucan content was analyzed using a commercial β-glucan assay kit (Mushroom and Yeast β-Glucan Assay Procedure, K-YBGL; Megazyme International, Wicklow, Ireland) according to the manufacturer’s instructions. For total glucan determination, 100 mg of each sample was hydrolyzed with concentrated hydrochloric acid, followed by heating, neutralization, and dilution to prepare the test solution.
α-Glucan content was determined by treating the sample with potassium hydroxide, followed by enzymatic hydrolysis using the α-glucan enzyme mixture provided in the kit. After centrifugation, the supernatant was used for analysis. Total glucan and α-glucan contents were quantified using the GOPOD (glucose oxidase/peroxidase) reagent, and absorbance was measured at 510 nm. β-Glucan content was calculated as the difference between total glucan and α-glucan contents and expressed as g/100 g on a dry weight basis.
DPPH radical scavenging activity was evaluated according to the method of Blois [16]. Sample solutions (40 μL) were mixed with 160 μL of 0.2 mM DPPH solution in a 96-well plate and incubated at 37°C for 30 min in the dark. Absorbance was measured at 517 nm using a microplate reader (Infinite M Nano, TECAN, Switzerland). L-Ascorbic acid was used as a positive control. DPPH radical scavenging activity was expressed as the percentage reduction in absorbance compared with the control.
ABTS radical scavenging activity was measured following a modified method of Re et al. [17]. ABTS+ radicals were generated by reacting 7 mM ABTS with 2.45 mM potassium persulfate in the dark for 12–14 h. The resulting solution was diluted with 5 mM potassium phosphate buffer (pH 7.4) to obtain an absorbance of 0.70–0.80 at 734 nm. Sample solutions (40 μL) were mixed with 180 μL of diluted ABTS+ solution and allowed to react for 1 min before absorbance measurement at 734 nm.
Ferric reducing antioxidant power (FRAP) was determined according to the method of Benzie and Strain [18]. The FRAP reagent was prepared by mixing acetate buffer, TPTZ solution, and FeCl3·6H2O solution in a ratio of 10:1:1 (v/v/v). Sample solution (0.5 mL) was mixed with 3.5 mL of FRAP reagent, incubated at 37°C for 5 min, and absorbance was measured at 593 nm. Results were expressed as μM FeSO4·7H2O equivalents per g extract.
Superoxide dismutase (SOD)-like activity was measured using a modified method of Marklund S and Marklund G [19]. Sample solutions were reacted with Tris-HCl buffer and pyrogallol, incubated at 25°C for 45 min, and absorbance was measured at 420 nm. Results were expressed as the percentage inhibition of pyrogallol autoxidation relative to the control.
Catalase activity was measured according to a modified method of Aebi [20] using a UV-transparent 96-well microplate. Sample solutions (50 μL) were mixed with catalase solution (50 μL) prepared in 50 mM potassium phosphate buffer (pH 7.0) and pre-incubated for 5 min at room temperature. The reaction was initiated by adding 100 μL of 30 mM hydrogen peroxide, and the decrease in absorbance at 240 nm was monitored immediately using a microplate reader. Catalase activity was expressed as relative activity (%) compared with the catalase + H2O2 control.
RAW 264.7 macrophages were cultured in RPMI-1640 medium supplemented with 10% FBS and 1% penicillin-streptomycin at 37°C in a 5% CO2 incubator. Cell viability was assessed using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay [21]. Cells were seeded at a density of 1 × 105 cells/well in 96-well plates and incubated for 24 h. After treatment with sample solutions for 48 h, 20 μL of MTT solution (0.5 mg/mL) was added to each well and incubated for an additional 4 h. Formazan crystals were dissolved with a dimethyl sulfoxide (DMSO)/ethanol mixture (1:1, v/v), and absorbance was measured at 550 nm using a microplate reader. Cell viability was expressed as a percentage relative to the untreated control.
NO production was measured using the Griess reaction. RAW 264.7 cells were seeded at 1 × 105 cells/well and pretreated with SC-HWE, SC-HPWE, or SC-EE at various concentrations, followed by stimulation with LPS (1 μg/mL) for 24 h. Culture supernatants were reacted with Griess reagent, and absorbance was measured at 540 nm. NO inhibition was calculated relative to the LPS-treated control.
RAW 264.7 cells were treated with SC-HWE, SC-HPWE, or SC-EE (50–300 μg/mL) or quercetin (positive control) for 1 h, followed by stimulation with LPS (1 μg/mL) for 24 h. Levels of IL-1β, IL-6, and TNF-α in the culture supernatants were quantified using ELISA kits according to the manufacturer’s instructions. Absorbance was measured at 450 nm using a microplate reader, and results were expressed as a percentage of the LPS-treated control (% of control).
All experiments were performed in triplicate, and data are presented as the mean ± standard deviation (SD). Statistical analyses were performed using IBM SPSS Statistics (version 25). For cell viability data obtained from the MTT assay, statistical analysis was conducted using two-way analysis of variance (two-way ANOVA) with extract type and concentration as independent variables, followed by Duncan’s multiple range test. For NO production and cytokine (IL-1β, IL-6, and TNF-α) data, differences among groups were evaluated by one-way ANOVA, followed by Duncan’s multiple range test. A p-value of less than 0.05 was considered statistically significant.
The total polyphenol and flavonoid contents of S. crispa extracts are presented in Table 1. Total polyphenol content differed significantly according to extraction method (p < 0.05). SC-EE exhibited the highest total polyphenol content (62.63 mg GAE/g), which was significantly higher than that of SC-HPWE and SC-HWE. Total flavonoid content also varied significantly among the extracts (p < 0.05). SC-EE and SC-HPWE showed relatively higher flavonoid contents, whereas SC-HWE exhibited the lowest value.
Total polyphenol, flavonoid, and β-glucan contents of Sparassis crispa extracts.
| Sample | Total polyphenol contents(mg GAE/g) | Total flavonoid contents(mg QE/g) | β-Glucan content (g/100 g) |
|---|---|---|---|
| SC-HWE | 48.35 ± 1.81a | 12.26 ± 0.23a | 48.98 ± 2.32b |
| SC-HPWE | 52.22 ± 0.40b | 15.83 ± 0.65b | 56.80 ± 1.48a |
| SC-EE | 62.63 ± 0.60c | 16.31 ± 0.88b | 16.94 ± 0.84c |
Values are expressed as mean ± standard deviation (SD). Different superscript letters within the same column indicate significant differences (p < 0.05). GAE: gallic acid equivalents; QE: quercetin equivalents; SC-EE: Sparassis crispa ethanol extract; SC-HPWE: Sparassis crispa high-temperature and high-pressure water extract; SC-HWE: Sparassis crispa hot water extract.
The β-glucan content of S. crispa extracts differed significantly depending on the extraction method (Table 1). SC-HPWE showed the highest β-glucan content (56.80 g/100 g), followed by SC-HWE (48.98 g/100 g), whereas SC-EE exhibited a markedly lower β-glucan content (16.94 g/100 g) (p < 0.05).
DPPH radical scavenging activity of all extracts increased significantly with increasing concentration (Figure 1A, p < 0.05). Within each extract, significantly higher scavenging activity was observed at 500–700 μg/mL compared with 50–100 μg/mL. At the same concentration, SC-EE showed significantly higher DPPH radical scavenging activity than SC-HWE and SC-HPWE at 200 μg/mL and above (p < 0.05). SC-HPWE also exhibited higher activity than SC-HWE at most concentrations, with statistically significant differences observed at concentrations ≥ 200 μg/mL.
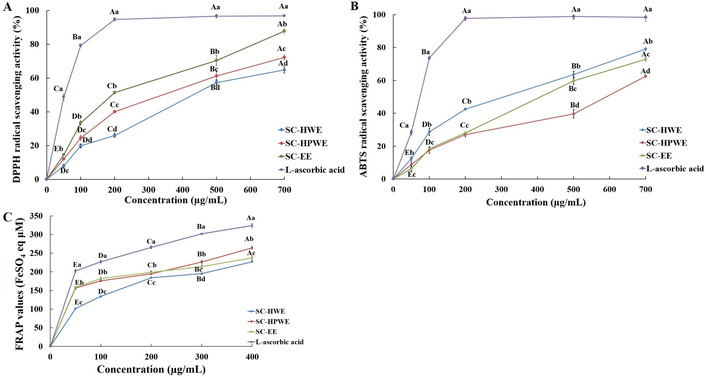
Antioxidant activities of Sparassis crispa extracts measured by radical scavenging and reducing power assays. (A) DPPH radical scavenging activity, (B) ABTS radical scavenging activity, and (C) FRAP of Sparassis crispa extracts prepared by hot water extraction (SC-HWE), high-temperature and high-pressure water extraction (SC-HPWE), and ethanol extraction (SC-EE), compared with L-ascorbic acid as a positive control. All values are expressed as the mean ± standard deviation (SD) of three independent experiments (n = 3). Different lowercase letters (a–d) indicate significant differences among samples at the same concentration (p < 0.05), whereas different uppercase letters (A–E) indicate significant differences among concentrations within the same sample (p < 0.05), based on Duncan’s multiple range test. ABTS: 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); DPPH: 2,2-diphenyl-1-picrylhydrazyl; FRAP: ferric reducing antioxidant power.
ABTS radical scavenging activity increased significantly with concentration in all extracts (Figure 1B, p < 0.05). No significant differences among extracts were observed at 50–100 μg/mL. However, at concentrations ≥ 200 μg/mL, significant differences were detected among the extracts (p < 0.05). SC-HWE exhibited significantly higher ABTS radical scavenging activity than SC-HPWE and SC-EE at concentrations ≥ 200 μg/mL, whereas no significant difference was observed between SC-HPWE and SC-EE at most concentrations.
The FRAP values of all extracts increased significantly with increasing concentration (Figure 1C, p < 0.05). SC-HPWE showed significantly higher FRAP values than SC-HWE and SC-EE at 300 and 400 μg/mL (p < 0.05). SC-EE also exhibited significantly higher FRAP values than SC-HWE at the same concentrations.
SOD-like activity and catalase activity are summarized in Table 2. For SOD-like activity, significant differences among extracts were observed at 100 and 200 μg/mL (p < 0.05). At 500 μg/mL, SC-HWE and SC-EE exhibited significantly higher SOD-like activity than SC-HPWE (p < 0.05). Catalase activity increased significantly with increasing concentration in all extracts (p < 0.05). At 100 and 200 μg/mL, significant differences among extracts were observed, and significant differences among extracts were also detected at 500 μg/mL (p < 0.05).
SOD-like activity and catalase activity of Sparassis crispa extracts.
| Sample | SOD-like activity (%) | Catalase activity (%) | ||||
|---|---|---|---|---|---|---|
| 100 μg/mL | 200 μg/mL | 500 μg/mL | 100 μg/mL | 200 μg/mL | 500 μg/mL | |
| SC-HWE | 26.54 ± 1.62Cc | 47.12 ± 0.49Bc | 67.21 ± 0.37Ab | 60.59 ± 0.92Cb | 65.01 ± 1.00Bc | 81.60 ± 0.47Ab |
| SC-HPWE | 20.32 ± 0.91Cd | 39.30 ± 1.09Bd | 60.66 ± 2.27Ac | 50.21 ± 0.48Cd | 61.91 ± 0.30Bd | 66.74 ± 0.15Ad |
| SC-EE | 46.10 ± 0.74Cb | 55.91 ± 0.75Bb | 67.90 ± 0.23Ab | 56.13 ± 0.74Cc | 73.13 ± 0.15Bb | 76.96 ± 0.15Ac |
| Ascorbic acid | 61.84 ± 0.55Ca | 68.43 ± 0.41Ba | 77.29 ± 0.23Aa | 67.85 ± 0.67Ba | 94.32 ± 0.34Aa | 95.41 ± 1.09Aa |
Different lowercase letters (a–d) indicate significant differences among samples at the same concentration (p < 0.05), whereas different uppercase letters (A–C) indicate significant differences among concentrations within the same sample (p < 0.05), based on Duncan’s multiple range test. SOD: superoxide dismutase; SC-EE: Sparassis crispa ethanol extract; SC-HPWE: Sparassis crispa high-temperature and high-pressure water extract; SC-HWE: Sparassis crispa hot water extract.
The effects of S. crispa extracts on RAW 264.7 cell viability are shown in Figure 2A. Cell viability decreased gradually with increasing concentration for all extracts. At concentrations of 100–300 μg/mL, cell viability remained above approximately 80% for all extracts (p < 0.05). At concentrations ≥ 400 μg/mL, cell viability decreased significantly, with a marked reduction observed at 500 and 600 μg/mL (p < 0.05).
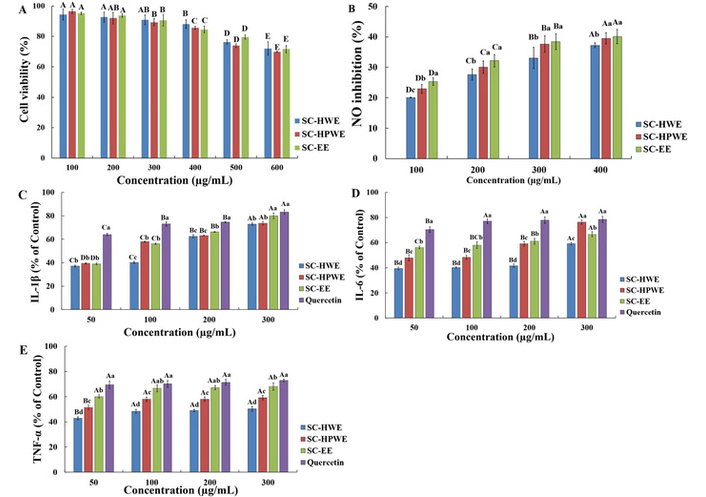
Anti-inflammatory effects of Sparassis crispa extracts in LPS-stimulated RAW 264.7 macrophages. (A) Cell viability determined by the MTT assay. (B) Inhibition of NO production. (C) IL-1β production expressed as a percentage of the control. (D) IL-6 production expressed as a percentage of the control. (E) TNF-α production expressed as a percentage of the control. All values are expressed as the mean ± standard deviation (SD) of three independent experiments (n = 3). For cell viability data (A), statistical analysis was performed using two-way ANOVA with extract type and concentration as independent variables, followed by Duncan’s multiple range test. For NO and cytokine data (B–E), one-way ANOVA followed by Duncan’s multiple range test was applied. For panels (B–E), different lowercase letters (a–d) indicate significant differences among samples at the same concentration (p < 0.05), whereas different uppercase letters (A–D) indicate significant differences among concentrations within the same sample (p < 0.05). IL: interleukin; LPS: lipopolysaccharide; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; NO: nitric oxide; SC-EE: Sparassis crispa ethanol extract; SC-HPWE: Sparassis crispa high-temperature and high-pressure water extract; SC-HWE: Sparassis crispa hot water extract; TNF-α: tumor necrosis factor alpha.
The inhibitory effects of S. crispa extracts on NO production in LPS-stimulated RAW 264.7 macrophages are shown in Figure 2B. All extracts exhibited concentration-dependent inhibition of NO production in the range of 100–400 μg/mL (p < 0.05). At the same concentration, SC-EE and SC-HPWE showed significantly higher NO inhibitory activity than SC-HWE, particularly at 300 and 400 μg/mL (p < 0.05).
The effects of S. crispa extracts on pro-inflammatory cytokine production in LPS-stimulated RAW 264.7 macrophages are shown in Figure 2C–E. Treatment with SC-HWE, SC-HPWE, and SC-EE resulted in reduced production of IL-1β, IL-6, and TNF-α compared with the LPS-treated control. IL-1β production was markedly reduced at lower concentrations, with % of control values of approximately 37–39% at 50 μg/mL, and remained at a similar level at concentrations ≥ 200 μg/mL. IL-6 production showed a relatively consistent reduction at 50–200 μg/mL, whereas an increase in % of control values was observed at 300 μg/mL, particularly in the SC-HPWE and quercetin-treated groups. TNF-α production exhibited smaller changes compared with IL-1β and IL-6, with % of control values gradually increasing as the concentration increased. At 50 μg/mL, extract-treated groups showed % of control values ranging from approximately 43–60%, whereas the quercetin-treated group showed values of approximately 69–73% across all concentrations.
The extraction conditions applied in this study were selected to enable a comparative evaluation of antioxidant and anti-inflammatory activities under extraction environments commonly used for edible and medicinal mushrooms. Accordingly, high-temperature aqueous extraction was included to allow comparison with hot water and ethanol extraction methods applied under practically relevant conditions, while 70% ethanol was selected as an extraction solvent because it is widely used to efficiently recover phenolic compounds and medium-polar bioactive constituents from mushroom matrices.
Under these extraction conditions, the total polyphenol and flavonoid contents of S. crispa extracts differed significantly depending on the extraction solvent and conditions, with the highest levels observed in the ethanol extract (SC-EE). This result can be attributed to the structural diversity of phenolic compounds, which exhibit a wide range of molecular weights and polarities and may exist in free forms or as bound forms associated with cell wall components through ionic, ester, ether, or carbon-carbon linkages [7]. Ethanol, as a solvent of intermediate polarity, is capable of solubilizing both hydrophilic and relatively low-polarity phenolic compounds, thereby facilitating the extraction of diverse low-molecular-weight phenolics [7, 22]. In addition, the relatively high polyphenol and flavonoid contents observed in the high-temperature and high-pressure water extract (SC-HPWE) suggest that elevated temperature and pressure conditions may partially disrupt the cell wall matrix, promoting the release of bound phenolic compounds [6]. Similar trends have been reported for S. crispa, where higher extraction temperatures resulted in increased recovery of phenolic compounds and antioxidant-related indices [23]. However, excessively harsh extraction conditions may also lead to substrate over-degradation or reduced selectivity, indicating that extraction parameters should be optimized according to target compounds and intended applications.
The β-glucan content of S. crispa extracts showed marked differences according to extraction method, with SC-HPWE exhibiting the highest level. β-Glucan is a high-molecular-weight polysaccharide that exists as a structural component of fungal cell walls, where it forms stable complexes with chitin, proteins, and other polysaccharides. Its (1→3), (1→6)-β-D-glucan structure is known to adopt a triple-helix conformation, which may limit its solubilization under conventional hot-water extraction conditions. Accordingly, high-temperature and high-pressure extraction appears to be more effective in disrupting the cell wall matrix and releasing bound β-glucan, which explains the significantly higher β-glucan content observed in SC-HPWE compared with SC-HWE. In contrast, the markedly lower β-glucan content in SC-EE is consistent with previous reports indicating that ethanol is unsuitable for the extraction of highly hydrophilic, high-molecular-weight polysaccharides [7, 22]. Notably, the lack of a direct correspondence between β-glucan content and radical scavenging activity suggests that β-glucan may contribute to bioactivity through mechanisms distinct from direct electron donation, highlighting the need to consider the physicochemical properties of bioactive components when interpreting antioxidant assays [24].
The antioxidant activities of S. crispa extracts varied depending on the assay employed, reflecting differences in the chemical nature of antioxidant components extracted under different conditions. The relatively strong DPPH radical scavenging activity observed in SC-EE can be explained by the preferential extraction of low-molecular-weight phenolic compounds, which are effective hydrogen or electron donors in hydrophobic radical systems [16, 24].
In contrast, ABTS radical scavenging activity was more pronounced in water-based extracts, particularly SC-HWE, suggesting a greater contribution of hydrophilic antioxidant components. ABTS assays are known to reflect the activity of both phenolic compounds and other water-soluble reducing substances present in complex food matrices [24]. Since mushrooms lack the typical flavonoid biosynthetic pathways found in plants, the observed radical scavenging activities are more likely attributable to phenolic acids and other non-flavonoid antioxidant constituents. Indeed, previous studies on edible mushrooms have reported correlations between ABTS or DPPH activities and phenolic acids such as caffeic, protocatechuic, and p-coumaric acids [25].
Similarly, the higher FRAP values observed for SC-HPWE at higher concentrations indicate that high-temperature and high-pressure extraction may favor the recovery of hydrophilic reducing agents or bound phenolic compounds, which contribute to electron transfer-based antioxidant capacity [6, 26].
Differences in SOD-like activity among extracts further suggest that extraction conditions influence the composition and relative proportions of antioxidant constituents. Mushrooms are known to contain a mixture of phenolic compounds, low-molecular-weight antioxidants, and water-soluble components, whose extraction efficiencies vary with solvent and processing conditions [8, 27].
Catalase-related activity increased with concentration in all extracts; however, differences among extracts diminished at higher concentrations. This trend may indicate that catalase-mediated reactions approach saturation at elevated substrate levels, thereby reducing the apparent influence of compositional differences among extracts.
The absence of marked cytotoxicity at concentrations up to 400 μg/mL indicates that the observed inhibitory effects on inflammatory mediators are unlikely to be attributable to nonspecific cell damage. Under LPS stimulation, all S. crispa extracts inhibited NO production in a concentration-dependent manner, with SC-EE and SC-HPWE showing relatively stronger inhibitory effects than SC-HWE.
Phenolic compounds have been reported to modulate oxidative stress-related signaling pathways and suppress inducible nitric oxide synthase (iNOS) expression, which may partly explain the NO inhibitory effects observed for SC-EE [24]. Meanwhile, the enhanced activity of SC-HPWE may be associated with the higher content of β-glucan and other water-soluble bioactive components released under high-temperature and high-pressure conditions. Previous studies have demonstrated that hot-water or pressurized extracts of edible mushrooms can exert anti-inflammatory effects through the combined actions of phenolic compounds and polysaccharides [8, 27].
Regarding cytokine production, IL-6 exhibited a more gradual concentration-dependent response compared with IL-1β, which may reflect its regulation by multiple signaling pathways, including NF-κB, STAT3, and MAPK [28]. TNF-α showed relatively modest changes, consistent with reports that TNF-α expression is tightly regulated through strong feedback mechanisms following rapid induction during early inflammatory responses [29]. Although quercetin is a well-known anti-inflammatory flavonoid, its relatively weaker cytokine suppression compared with S. crispa extracts under the present conditions suggests that the combined action of polysaccharides and phenolic compounds in mushroom extracts may provide broader modulation of inflammatory responses than a single isolated compound [10, 30].
ABTS: 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)
ANOVA: analysis of variance
DPPH: 2,2-diphenyl-1-picrylhydrazyl
ELISA: enzyme-linked immunosorbent assay
FBS: fetal bovine serum
FRAP: ferric reducing antioxidant power
GAE: gallic acid equivalents
IL: interleukin
LPS: lipopolysaccharide
MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
NO: nitric oxide
SC-EE: Sparassis crispa ethanol extract
SC-HPWE: Sparassis crispa high-temperature and high-pressure water extract
SC-HWE: Sparassis crispa hot water extract
SOD: superoxide dismutase
TNF-α: tumor necrosis factor alpha
TPTZ: 2,4,6-tris(2-pyridyl)-s-triazine
MHP: Conceptualization, Investigation, Methodology, Data curation, Formal analysis, Visualization, Writing—original draft. BK: Conceptualization, Supervision, Writing—review & editing. Both authors read and approved the submitted version of the manuscript.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 862
Download: 75
Times Cited: 0
