Affiliation:
1Department of Physical Education and Sport Sciences, Islamic Azad University, Mahabad 1955847781, Iran
ORCID: https://orcid.org/0000-0002-0953-1102
Affiliation:
2School of Sport, Exercise and Nutrition, Massey University, Turitea 4410, New Zealand
ORCID: https://orcid.org/0000-0002-3372-3008
Affiliation:
3Faculty of Psychology Education and Sport Sciences-Blanquerna, University Ramon Llull, 08022 Barcelona, Spain
ORCID: https://orcid.org/0000-0002-9197-3179
Affiliation:
4Faculty of Health Sciences-Blanquerna, University Ramon Llull, 08025 Barcelona, Spain
ORCID: https://orcid.org/0000-0001-9531-9580
Affiliation:
3Faculty of Psychology Education and Sport Sciences-Blanquerna, University Ramon Llull, 08022 Barcelona, Spain
4Faculty of Health Sciences-Blanquerna, University Ramon Llull, 08025 Barcelona, Spain
ORCID: https://orcid.org/0000-0002-0396-4185
Affiliation:
5Area of Radiology, Faculty of Health Sciences, Soria Campus, University of Valladolid, 42004 Soria, Spain
6Neurobiology Research Group, University of Valladolid, 47005 Valladolid, Spain
ORCID: https://orcid.org/0000-0001-6521-2053
Affiliation:
7Department of Applied Biology-Nutrition, Institute of Bioengineering, University Miguel Hernández, 03202 Elche, Spain
8Alicante Institute for Health and Biomedical Research (ISABIAL), 03010 Alicante, Spain
9CIBER Physiopathology of Obesity and Nutrition (CIBEROBN), Health Institute Carlos III (ISCIII), 28029 Madrid, Spain
10Research Group “Nutrition and Physical Activity”, Spanish Nutrition Society “SEÑ”, 28010 Madrid, Spain
ORCID: https://orcid.org/0000-0001-5128-1672
Affiliation:
10Research Group “Nutrition and Physical Activity”, Spanish Nutrition Society “SEÑ”, 28010 Madrid, Spain
11Faculty of Health Sciences, University of Burgos, 09001 Burgos, Spain
ORCID: https://orcid.org/0000-0002-6554-4602
Affiliation:
6Neurobiology Research Group, University of Valladolid, 47005 Valladolid, Spain
12Doctoral School, University of Leon, 24007 Leon, Spain
Email: amcelorrio@educa.jcyl.es
ORCID: https://orcid.org/0009-0003-6099-2021
Affiliation:
6Neurobiology Research Group, University of Valladolid, 47005 Valladolid, Spain
10Research Group “Nutrition and Physical Activity”, Spanish Nutrition Society “SEÑ”, 28010 Madrid, Spain
13Area of Histology, Faculty of Health Sciences, Soria Campus, University of Valladolid, 42004 Soria, Spain
14Biomedical Research Institute of Leon (IBIOLEÓN), University Hospital Complex of Leon, 24071 Leon, Spain
#These authors share the last authorship.
Email: diego.fernandez.lazaro@uva.es
ORCID: https://orcid.org/0000-0002-6522-8896
Affiliation:
15Department of Physical Education and Sport Sciences, CT.C., Islamic Azad University, Tehran 1955847781, Iran
#These authors share the last authorship.
ORCID: https://orcid.org/0000-0002-3502-7487
Explor Endocr Metab Dis. 2026;3:101469 DOI: https://doi.org/10.37349/eemd.2026.101469
Received: December 31, 2025 Accepted: March 31, 2026 Published: April 30, 2026
Academic Editor: Tzong-Shyuan Lee, National Taiwan University, Taiwan, China
The article belongs to the special issue Metabolic Syndrome in Menopause
Aim: The aim of this study was to compare the effects of high-intensity interval training (HIIT) and concurrent training (CONC) on body composition and metabolic profile in postmenopausal women.
Methods: Forty-five postmenopausal women with overweight or obesity (50–65 years) were randomly assigned to one of three groups: HIIT group, CONC group, or control group (CG). All groups underwent a 12-week intervention, consisting of three training sessions per week, scheduled on alternate days. All dependent variables (body composition, lipid, or glycemic markers) were assessed before and after a 12-week intervention period.
Results: The CONC and HIIT groups showed anthropometric improvements compared to the CG. In this regard, CONC and HIIT significantly (P < 0.05) reduced their body weight and body mass index (BMI) compared with the CG. This reduction was due to a decrease in fat mass, mainly in the abdominal area, as reflected by the reduction in the waist-to-hip ratio. Participants in the CONC group showed greater reductions in body weight, BMI, and body fat percentage compared with the HIIT group, while both exercise interventions produced similar improvements in metabolic biomarkers. Regarding circulating parameters, CONC and HIIT significantly (P < 0.05) reduced circulating triglycerides, total cholesterol, and LDL-cholesterol, which resulted in a decrease in the atherogenic index of plasma (AIP, calculated as log[TG/HDL-C]) compared with CG. Blood glucose and insulin levels also decreased significantly (P < 0.05) in CONC and HIIT compared with CG, with a consequent reduction in the homeostatic model assessment-insulin resistance (HOMA-IR).
Conclusions: A 12-week program of HIIT or CONC training represents an effective strategy for improving body fat reduction and metabolic profile in sedentary postmenopausal women with overweight or obesity [ClinicalTrials.gov, identifier (ID NCT07302191)].
Menopause is a natural biological transition marked by the permanent cessation of menstrual periods, resulting from the loss of ovarian function and the end of oocyte production. This process typically occurs between the ages of 45 and 55 years [1]. In many countries, the life expectancy of women approaches 80 years, meaning that a substantial proportion of women live several decades beyond menopause. This extended postmenopausal period is associated with an increased risk of chronic conditions, including osteoporosis, cardiovascular diseases (CVD), and metabolic disorders, such as obesity, dyslipidemia, high blood pressure, insulin resistance (IR), and type 2 diabetes (T2D) [2]. These risks are largely attributed to declining estrogen levels and other hormonal changes that occur during and after menopause [3].
Menopause-related estrogen deficiency contributes to central obesity (abdominal fat) and adverse changes in lipid metabolism [2]. The decline in estrogen during menopause is a key factor in body fat (BF) redistribution, shifting from peripheral body regions (hips and thighs) to central abdominal areas, particularly visceral fat. This transition is accompanied by increases in BF mass and body mass index (BMI), with postmenopausal women often exhibiting larger waist circumferences (WC) [4]. The combination of menopause and obesity is associated with an atherogenic lipid profile, characterized by elevated levels of triglycerides (TG) and low-density lipoprotein-cholesterol (LDL-C) and low levels of high-density lipoprotein-cholesterol (HDL-C), which collectively contribute to an increased risk of CVD [5]. Sedentary behavior further exacerbates weight gain and obesity in this population [6]. Estrogen deficiency also plays a central role in impairing glucose regulation and insulin sensitivity [7]. Furthermore, the shift in fat distribution toward the abdomen promotes the release of free fatty acids, which interfere with insulin signaling [8]. In addition, sleep disturbances common during menopause may elevate cortisol levels, further disrupting glycemic control [9]. Altogether, the combination of all these factors increases the risk of IR, leading to higher homeostatic model assessment (HOMA)-IR scores and a higher susceptibility to prediabetes and other cardiometabolic disorders in postmenopausal women. Lifestyle interventions, such as diet modifications and structured exercise, are instrumental for mitigating these risks and promoting overall health in this population.
Among the various behavioral strategies, traditional exercise training interventions have proven effective for weight maintenance, weight loss, and promoting favorable changes in body composition and lipid metabolism, evidenced by improvements in lipid biomarkers [4]. However, evidence specifically targeting sedentary postmenopausal women with overweight or obesity remains limited, and the physiological responses to different exercise modalities may differ substantially in this population. More recently, alternative exercise modalities have gained attention for their potential to enhance health outcomes, particularly in sedentary populations. One of these approaches is the concept of “exercise snacks”, based on short bouts of high-intensity interval activity, which have demonstrated health benefits comparable to longer workouts, especially in physically inactive adults [10]. In this context, high-intensity interval training (HIIT) has emerged as a time-efficient strategy capable of eliciting significant metabolic adaptations and improving biomarkers related to physical fitness and overall health in older adults [11]. Several studies [12–14] in postmenopausal women have shown that HIIT can reduce visceral fat and improve aspects of metabolic health, although findings remain heterogeneous and sample sizes are often small. Concurrent training (CONC), which combines resistance and endurance exercises within the same program, optimizes body composition, muscle strength, and cardiovascular fitness. This exercise modality represents an effective strategy for individuals with obesity to reduce %BF and improve physical function. Evidence suggests that CONC training may be more effective than single-mode training in these populations [15]. In postmenopausal women, CONC has been associated with reductions in fat mass, improvements in circulating lipid profile, and enhanced glycemic control [16], although direct comparisons with HIIT routines are scarce. While both exercise training programs (HIIT and CONC) represent promising non-pharmacological interventions for improving health, physical fitness, mental well-being, and independence in older adults, further research is needed to clarify their specific adaptations and safety considerations in individuals with coexisting medical conditions [17].
Despite growing interest in exercise-based interventions, research directly comparing HIIT and CONC in the context of postmenopausal women remains relatively limited. Only a few randomized studies have evaluated these exercise modalities in this population [15, 18–20]; however, no study has directly contrasted their effects on body composition and circulating parameters related to lipid and glucose metabolism in postmenopausal women with overweight or obesity. Today, this is increasingly relevant given that women tend to live longer and have a longer postmenopausal period. This gap is increasingly relevant, as women tend now to live longer and therefore have a longer postmenopausal phase, spending a substantial portion of their lives in this stage. This demographic shift underscores the importance of promoting healthy lifestyles and implementing comprehensive policies to support women’s health and well-being during this period. Exercise modality is a key component of the FITT principle (frequency, intensity, time, and type) [21], with each training program eliciting different physiological adaptations. A deeper understanding of the potential differential effects of HIIT and CONC may inform clinical decision-making and guide sports medicine physicians in tailoring exercise prescriptions for postmenopausal women, particularly regarding BF reduction and improvements in lipid profile outcomes. Therefore, the primary objective of this study was to assess the comparative efficacy of HIIT and CONC as isolated interventions on body composition in sedentary postmenopausal women. The secondary objective was to evaluate their effects on biochemical markers related to lipid and glucose metabolism. To our knowledge, no previous randomized controlled trials have directly compared these exercise modalities (HIIT vs. CONC) in this population.
The trial protocol was approved by the Research Ethics Committee of the Islamic Azad University Mahabad Branch (IR.IAU.MAHABAD.REC.1404.012). The study was conducted in accordance with the principles of the Declaration of Helsinki [22]. All participants signed an informed consent prior to enrollment in the study, and each patient received a copy of the signed consent form. The sample trial was registered on ClinicalTrials.gov (NCT07302191).
A randomized controlled trial was conducted to evaluate the effects of 12 weeks of exercise interventions, comprising HIIT and CONC training, on health biomarkers in postmenopausal women with overweight or obesity. Participants were randomly allocated to one of the three groups using a stratified block design [23]. An independent statistician generated the randomization sequence, assigning participants to: a) HIIT group, b) CONC group, and c) control group (CG). CG did not receive any exercise program. Randomization followed the Consolidated Standards of Reporting Trials (CONSORT) 2025 guidelines for parallel-group randomized trials (Table S1) [24]. Baseline anthropometric and clinical characteristics of participants were assessed prior to the intervention and are presented in Table 1, ensuring comparability between groups.
Descriptive characteristics of subjects at baseline.
| Variables | HIIT group (n = 13) | CONC group (n = 13) | CG (n = 14) | F | P |
|---|---|---|---|---|---|
| Age (years) | 54.82 ± 3.81 | 55.72 ± 3.76 | 57.80 ± 2.55 | 2.730 | 0.078 |
| Weight (kg) | 80.92 ± 5.65 | 82.87 ± 6.13 | 83.37 ± 6.55 | 0.595 | 0.557 |
| Height (cm) | 163.15 ± 6.02 | 164.07 ± 6.50 | 162.59 ± 7.80 | 0.158 | 0.854 |
| BMI (kg/m2) | 30.37 ± 0.52 | 30.76 ± 1.05 | 31.53 ± 1.14a | 5.160 | 0.011* |
| Body fat (%) | 32.28 ± 4.90 | 35.06 ± 3.67 | 36.57 ± 2.37 | 2.710 | 0.080 |
BMI: body mass index; CG: control group; CONC: concurrent training; F: frequency; HIIT: high-intensity interval training; %: percentage. *: significant differences between groups (P < 0.05); a: significant differences with the HIIT group (P < 0.05). Values are expressed as mean ± standard deviation (SD) for quantitative variables and as frequency (%) for categorical variables; P-values calculated using one-way analysis of variance.
The sample size was calculated using G*Power version 3.1, following Faul’s guidelines [25]. A total of 36 participants were required. To account for an anticipated 20% dropout rate, the final sample size was set at 45 participants (15 per group). In addition, a power analysis (1 – β error probability) indicated that a sample size of 36 participants would be sufficient to detect a minimum difference of 1% in lipid biomarkers (TG, LDL-C, or HDL-C). The statistical power was set at 0.95, with an estimated effect size of 0.8.
Each participant underwent two testing sessions under identical conditions, separated by a 12-week period (September 2, 3, and 4, 2025, and November 25, 26, and 27, 2025). Tests were standardized to begin at 9:00 am to minimize circadian variability, with procedures conducted following the same protocol, sequence, and timing. Anthropometric biomarkers, physical parameters, oxidative stress status, and cardiac metabolic indicators were assessed. Five days prior to the start of the study, participants received detailed instructions regarding the exercise protocols and the type of tests to be performed, ensuring appropriate preparation and progression.
Recruitment was conducted in July and August 2025 using convenience sampling through community advertisements in health centers, sports centers, and community networks in Mahabad (Iran). Postmenopausal women with overweight or obesity who responded to the invitations were contacted by phone call and provided with detailed information about the study, including its objectives, methodology, potential benefits, and associated risks.
A total of 45 postmenopausal women (aged 50–65 years) were included in the study. Eligibility criteria comprised BMI > 25 and ≤ 40 kg/m2, sedentary behavior defined as less than 150 min/week of moderate physical activity according to World Health Organization (WHO) guidelines [26], being in generally good health and able to safely engage in exercise protocols and having at least one year since their last menstrual period, in accordance with standard clinical criteria for menopause. Exclusion criteria included medical contraindications to strenuous physical activity, joint pain, current use of hormone replacement therapy, missing more than three exercise sessions during intervention, or illness during the study period. Additionally, women taking drugs or supplements known to significantly affect weight, metabolism, or exercise response, as well as those with substance use disorders, were also excluded.
Accordingly, 45 women were selected and allocated in a 1:1:1 ratio to the HIIT group (n = 15), CONC group (n = 15), and CG (n = 15). However, 2 women from each training group (HIIT and CONC) were excluded due to excessive absenteeism, and one participant from the CG did not complete the post-test assessment (Figure 1). Absenteeism withdrawals were unrelated to exercise adverse events. Four participants discontinued the intervention due to scheduling constraints, preventing them from completing 3 consecutive training sessions. Randomization was performed using a computer-generated sequence created using the Research Randomizer software (version 4.0) [27] (random number generator software), concealed in sequentially numbered, sealed, opaque envelopes (SNOSE method), and kept by the principal researcher. In addition, due to the nature of the intervention, participant and trainer blinding was not possible. However, outcome data were coded, and the statistician remained blinded to group assignment throughout the analysis.
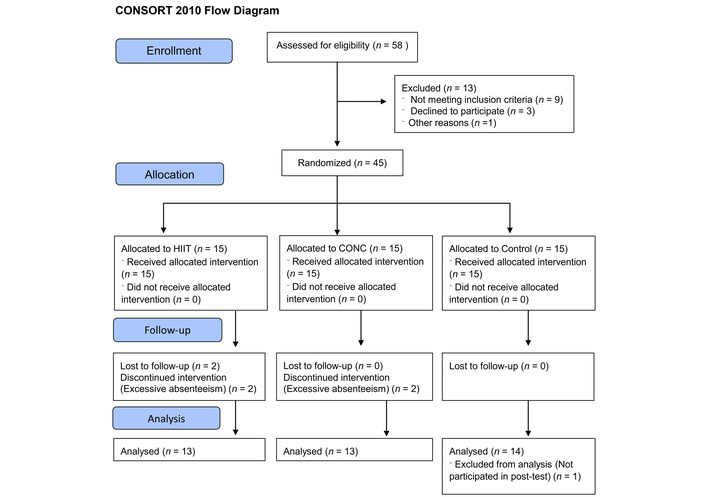
Flow-chart of the study participants. Adapted from [24]. © 2025 The Author(s). Distributed/Licensed under a license. CONC: concurrent training; HIIT: high-intensity interval training.
Both the HIIT and CONC groups followed a supervised 12-week exercise intervention, performed 3 times per week on alternate days. All training sessions were conducted in the Exercise Physiology Laboratory of the Islamic Azad University (Mahabad Branch) and adjacent training facilities under standardized environmental conditions (temperature 20–22°C; humidity 50–60%). All sessions were supervised by certified exercise specialists to ensure participant safety and protocol fidelity. Each session consisted of a 10-min warm-up (light aerobic activity and dynamic stretching), the main training component, and a 10-min cool-down (low-intensity walking and stretching). Attendance, heart rate responses, and perceived exertion were recorded in each session to verify compliance. Participants missing > 20% of sessions (i.e., more than seven of 36) were classified as non-completers. The exercise mode consisted of treadmill or outdoor walking/running, selected according to participant preference to minimize impact on the lower limbs. In this context, joint load was carefully considered in the design of all selected exercises, particularly in this last exercise (running/walking). Nevertheless, the possibility of adverse effects was systematically monitored at each session, and no joint-related issues were reported during intervention.
The HIIT group training program included 60 seconds of bouts at maximal intensity [90–95% of individual maximum heart rate (HRmax)], separated by 60 seconds at lower intensity (55–60% HRmax) [28]. HRmax was estimated individually using the Tanaka et al. equation [29] (HRmax = 208 − 0.7 × age), which has been validated in middle-aged women. Session duration ranged from approximately 25 to 35 min (excluding warm-up and cool-down).
Progression was achieved by gradually increasing the number of high-intensity bouts across the intervention period: i) week 1: 6 intervals; ii) week 2: 8 intervals; iii) weeks 3–8: 10 intervals; iv) weeks 9–12: 12 intervals. This progression model was adapted from Afrasyabi et al. [30], who applied a similar HIIT framework in adults with metabolic impairments.
To monitor exercise intensity during HIIT sessions, the women wore heart rate monitors (Polar Vantage M, POLAR, Oulu, Finland) throughout all training sessions. Heart rate data were recorded in an online personal training diary (https://flow.polar.com/diary) and used to assess adherence and intensity. Data were downloaded and reviewed weekly to ensure that the correct prescriptions of intensity thresholds were maintained.
To ensure consistent exertion, the Borg 6–20 rating of perceived exertion (RPE) scale was recorded at the end of each interval and maintained between 15 and 17 (“hard” to “very hard”), confirming alignment with the prescribed HR range.
This HR-RPE correspondence followed the intensity prescription criteria outlined by the American College of Sports Medicine [31] for older adult populations.
The CONC group participated in a combined resistance and endurance training program. Resistance training involved 3 sets of selected exercises at designated stations, with one-min rest intervals between sets. During the first week, CONC participants completed 10–15 repetitions at 55% of their one-repetition maximum (1RM). By week 12, participants progressed to 8–12 repetitions at 75% of 1RM. To ensure appropriate training load, 1RM was reassessed every three weeks and adjusted accordingly. Perceived exertion was recorded (Borg 6–20 scale), maintaining RPE 13–15 (“somewhat hard”) to confirm compliance with relative intensity.
The aerobic component of the CONC program consisted of 20 min of running. Intensity was gradually increased from 55% HRmax in the first week to 75% HRmax by the final week, following the protocol described by Kargarfard et al. [32]. The average total duration of each CONC session, including warm-up and cool-down, was approximately 50–60 min.
To maintain comparable total training volume across groups, energy expenditure per session (kcal/session) was estimated from heart rate recordings and adjusted weekly to equalize cumulative training load between HIIT and CONC.
The exercise mode consisted of treadmill or outdoor walking/running, selected according to participant preference to minimize impact on the lower limbs. In this context, joint load was carefully considered in the design of all selected exercises, particularly in this last exercise (running/walking). Nevertheless, the possibility of adverse effects was systematically monitored at each session, and no joint-related issues were reported during intervention.
Finally, participants in CG continued their usual daily activities without any follow-up during the 12-week intervention period. To minimize differences in participant-researcher contact, the CG received biweekly phone calls similar in frequency to the intervention groups. During these calls, participants completed a brief standardized checklist regarding daily activity, health status, and medication changes. Sedentary postmenopausal women do not routinely receive structured exercise programs, and maintaining their usual lifestyle does not expose them to additional risk. All participants were informed during consent that the CG would not receive supervised training during the study but would be offered individualized exercise guidance after completion. This approach was approved by the institutional Ethics Committee to avoid contamination between groups and preserve the internal validity of the trial.
Body composition measurements were taken before and after the 12-week intervention, under fasting conditions. A stadiometer (SECA 213, Hamburg, Germany) was used to assess the heights and weights of participants, wearing minimal clothing. BMI was calculated according to the formula: weight (kg)/height2 (m2). WC, hip circumference (HC), and waist-hip ratio (WHR) were measured using a flexible measuring tape. To determine %BF using the Jackson et al. equation [33], subcutaneous fat thickness was measured at the triceps, suprailiac, and thigh sites using a skinfold caliper (Yagami, Japan; accuracy: 1 mm). All measurements were taken on the right side of the body three times, with 20-second intervals between each measurement. These values, along with age, were used to calculate body density, which was subsequently converted to %BF using the Siri equation.
Blood samples were collected in a laboratory setting at the beginning and at the end of the 12-week training program. Following a 12-h fast, venous blood was drawn from the left arm while participants were seated. After centrifugation (12 min at 3,000 rpm in a Hettich centrifuge, Germany), the resulting serum was aliquoted, frozen, and stored at –70°C for subsequent analysis.
Lipid profile indices, including total cholesterol (TC), TG, and HDL-C, were measured with an enzymatic method (Pars Azmun, Iran). LDL-C was calculated using the Friedwald equation [34], which estimates LDL-C by subtracting HDL-C and one-fifth of TG from TC. The atherogenic index of plasma (AIP) was determined using the logarithmic ratio of TG to HDL-C (log[TG/HDL-C]) [35].
Fasting glucose levels were measured using the Pars Azmun glucose kit (Pars Azmun, Iran), based on the glucose oxidase method, with a sensitivity of 5 mg/mL. Serum insulin concentrations were determined using an ELISA kit (Monobind, United States), with a sensitivity of 0.4 IU/mL. The HOMA-IR was calculated using the following formula with glycemia expressed in mmol/L and insulin levels in mIU/L [36]: HOMA-IR = (glucose × insulin)/22.5.
Participants were instructed to maintain their habitual diet throughout the intervention. Habitual diet was not strictly controlled, but participants were advised to avoid any significant dietary changes during the study period. No specific nutritional counseling was provided, as the primary objective of this study was to isolate the effects of exercise training. To minimize potential confounding, participants were asked to replicate their dietary intake during the 24 h preceding each testing session, ensuring consistency. Additionally, to reduce the impact of dietary factors on oxidative stress and inflammatory biomarkers, participants were instructed to avoid antioxidant supplementation (e.g., vitamin C, E, polyphenols) and alcohol consumption for at least 48 h before each testing session. These instructions aim to minimize potential interference with biomarker measurements sensitive to dietary or exogenous antioxidant fluctuations. Compliance with these dietary instructions was verbally confirmed before each testing session and recorded in standardized pre-assessment checklists. Baseline dietary patterns (energy and macronutrient distribution) were qualitatively similar across groups according to the initial screening questionnaire, minimizing potential bias from habitual diet differences.
Statistical analyses were performed using SPSS software version 24.0 (SPSS, Inc., Chicago, IL, USA). Data are presented as means ± standard deviations. A P-value < 0.05 was considered statistically significant. Kolmogorov-Smirnov and Levene’s tests were used to evaluate the normal distribution of the data and homogeneity of variances, respectively. Analyses were conducted following a per-protocol approach. Participants who missed 3 consecutive training sessions, according to the predefined withdrawal criteria, were excluded from the final analysis. An intention-to-treat analysis was not performed. Additionally, sample size was estimated a priori, calculated using G*Power software for repeated measures analysis of variance (ANOVA) (within-between interaction), based on an expected medium effect size, α = 0.05, and power (1 – β) = 0.80.
At baseline, group characteristics were compared using one-way ANOVA to ensure that initial outcomes did not differ significantly across groups. A two-way repeated measures ANOVA was performed to examine the main effects of group and time, as well as their interaction. When significant effects were observed, pairwise comparisons were conducted using the Bonferroni post hoc test. Data are presented as mean (SD). Effect sizes were calculated from the ANOVA output by converting partial eta-squared (η²p) to Cohen’s d. Cohen’s d effect size was calculated using the formula: d = (mean difference between pre- and post-intervention)/(pooled standard deviation). According to Cohen’s conventions, effect sizes were interpreted as small (Cohen’s d = 0.2), medium (d = 0.5), and large (d = 0.8) [37]. For η²p, effect sizes were interpreted as small (η²p = 0.01), medium (η²p = 0.06), and large (η²p = 0.14) [38]. In addition to effect size calculations, the percentage of changes in each outcome was calculated and reported. Relationships between changes in study outcomes (pre-post differences) were examined using Pearson’s correlation coefficient.
Baseline characteristics of participants across groups are presented in Table 1. No significant differences were observed among the three groups in baseline values and characteristics, except for BMI. BMI was significantly higher in the CG compared to the HIIT group (P = 0.011).
ANOVA showed significant time effects for all anthropometric parameters (P < 0.001 for all, d = 1.06 to 1.28). There were significant time-group effects for body weight (F2, 37 = 24.18, P = 0.001, η²p = 0.567, d = 1.12), BMI (F2, 37 = 24.54, P = 0.001, η²p = 0.570, d = 1.13), %BF (F2, 37 = 11.87, P = 0.001, η²p = 0.391, d = 0.79), WC (F2, 37 = 14.30, P = 0.001, η²p = 0.436, d = 0.86) and WHR (F2, 37 = 11.92, P = 0.001, η²p = 0.392, d = 0.79). Neither the time effect (P = 0.085) nor the time-group effect (P = 0.458) was significant for HC (data not shown in the table).
Body weight values in the HIIT group (–2.97%, P = 0.001, d = 0.40) and CONC group (–4.74%, P = 0.001, d = 0.65) were reduced significantly from baseline at the end of intervention, while no significant change was observed in the CG (+0.37%, P = 0.160, d = 0.04). The weight loss rate in the CONC group was significantly higher than in the HIIT (P = 0.014) and CG (P = 0.001) groups. Weight loss in the HIIT group was also significantly higher than in the CG (P = 0.004).
BMI also decreased significantly in the HIIT group (–2.99%, P = 0.001, d = 1.32) and CONC group (–4.71%, P = 0.001, d = 1.45) compared to baseline values. The change of BMI in CG did not show a significant difference (+0.38%, P = 0.132, d = 0.10). In the CONC group, BMI loss was significantly greater than in the HIIT group and CG (P = 0.02). HIIT also lost more BMI than CG (P = 0.004). %BF in the HIIT group (–6.37%, P = 0.001, d = 0.45) and CONC group (–9.81%, P = 0.001, d = 1.21) was reduced significantly in comparison to baseline values, while no significant change was observed in CG (–0.16%, P = 0.565, d = 0.02).
The %BF loss rate in the CONC group was significantly higher than in HIIT (P = 0.048) and CG (P = 0.001). There was also a significant reduction in the %BF in the HIIT group compared to the CG (P = 0.004).
WC decreased significantly in the HIIT group (–2.88%, P = 0.001, d = 0.63) and CONC group (–2.60%, P = 0.001, d = 0.75) compared to baseline values. No significant difference was observed in CG (–0.09%, P = 0.278, d = 0.002). There was a higher reduction in WC in the CONC (P = 0.001) and HIIT (P = 0.001) groups than in the CG. However, no significant difference was observed between the CONC and HIIT groups (P = 0.990).
No significant differences were observed for HC in the three groups: HIIT group (–0.13%, P = 0.147, d = 0.03), CONC group (–0.22%, P = 0.231, d = 0.02), and CG (–0.01%, P = 0.278, d = 0.002) compared to baseline.
WHR decreased significantly in the HIIT group (–3.12%, P = 0.001, d = 1.17) and CONC group (–2.10%, P = 0.001, d = 0.66) compared to baseline values. At the same time, no significant difference was observed in CG (–0.10%, P = 0.104, d = 0.003). In the CONC (P = 0.001) and HIIT (P = 0.001) groups, WHR was reduced compared to the CG. In contrast, there was no significant difference between the CONC and HIIT groups (P = 0.997) (see Table 2 and Figure 2).
Effect of concurrent training and high-intensity interval training on anthropometric characteristics in sedentary postmenopausal women with overweight or obesity (mean ± SD) at baseline (T1) and at the end of the study (T2).
| Variable | Group | T1 | T2 | Change (%) | Cohen’s d | ANOVA. P-value (Cohen’s d) | ||
|---|---|---|---|---|---|---|---|---|
| Interaction | Group | Time | ||||||
| Body weight (kg) | CONC | 82.87 ± 6.13 | 78.94 ± 5.84†*‡ | –4.74 | 0.65 | 0.001 (1.12)** | 0.260 (0.27) | 0.001 (1.28) |
| HIIT | 80.92 ± 5.65 | 78.51 ± 6.13†* | –2.97 | 0.40 | ||||
| CG | 83.37 ± 6.55 | 83.68 ± 6.37 | +0.37 | 0.04 | ||||
| BMI (kg/m2) | CONC | 35.06 ± 3.67 | 29.31 ± 0.94†*‡ | –4.71 | 1.45 | 0.001 (1.13)** | 0.001 (0.84) | 0.001 (1.28) |
| HIIT | 32.28 ± 4.90 | 29.46 ± 0.82†* | –2.99 | 1.32 | ||||
| CG | 31.53 ± 1.14 | 31.65 ± 1.21 | +0.38 | 0.10 | ||||
| BF (%) | CONC | 35.06 ± 3.36 | 31.62 ± 2.20†*‡ | –9.81 | 1.21 | 0.001 (0.79)** | 0.005 (0.57) | 0.001 (1.06) |
| HIIT | 33.28 ± 4.90 | 31.16 ± 4.42†* | –6.37 | 0.45 | ||||
| CG | 36.57 ± 2.37 | 36.51 ± 2.55 | –0.16 | 0.02 | ||||
| WC (cm) | CONC | 97.03 ± 3.65 | 94.5 ± 3.03†* | –2.60 | 0.75 | 0.001 (0.86)** | 0.025 (0.46) | 0.001 (1.27) |
| HIIT | 95.02 ± 4.35 | 92.28 ± 4.31†* | –2.88 | 0.63 | ||||
| CG | 98.14 ± 4.59 | 98.05 ± 4.35 | –0.09 | 0.002 | ||||
| HC (cm) | CONC | 101.16 ± 4.39 | 100.93 ± 4.43 | –0.22 | 0.02 | 0.458 (0.20) | 0.398 (0.22) | 0.085 (0.28) |
| HIIT | 98.86 ± 3.49 | 98.73 ± 3.51 | –0.13 | 0.03 | ||||
| CG | 100.56 ± 5.12 | 100.55 ± 5.04 | –0.01 | 0.002 | ||||
| WHR | CONC | 0.95 ± 0.03 | 0.93 ± 0.03†* | –2.10 | 0.66 | 0.001 (0.79)** | 0.051 (0.41) | 0.001 (1.15) |
| HIIT | 0.96 ± 0.02 | 0.93 ± 0.03†* | –3.12 | 1.17 | ||||
| CG | 0.976 ± 0.03 | 0.975 ± 0.02 | –0.10 | 0.003 | ||||
BF: body fat; BMI: body mass index; CG: control group; CONC: concurrent training; HC: hip circumference; HIIT: high-intensity interval training; SD: standard deviation; WC: waist circumference; WHR: waist-hip ratio; %: percentage. †: significant difference from pre to post (P < 0.05); *: significant difference from CG (P < 0.05); ‡: significant difference from HIIT (P < 0.05); **: significant time × group interaction. Two-way ANOVA with repeated measures, followed by the post hoc test of Bonferroni. Values are expressed as mean (SD).
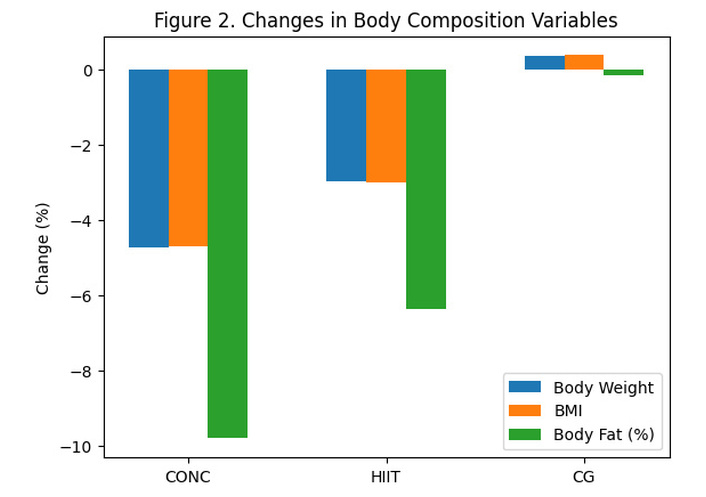
Changes in body composition variables after the 12-week intervention. Percentage changes in body weight, BMI, and body fat across groups (CONC, HIIT, and CG). BMI: body mass index; CONC: concurrent training; CG: control group; HIIT: high-intensity interval training.
The percentage changes in body composition variables are illustrated in Figure 2.
ANOVA showed significant time effects for all lipid profile parameters (P < 0.001 for all, d = 0.65 to 1.03) and time-group effects for TG (F2, 37 = 3.32, P = 0.047, η²p = 0.153, d = 0.41), TC (F2, 37 = 13.31, P = 0.001, η²p = 0.418, d = 0.83), HDL-C (F2, 37 = 8.42, P = 0.001, η²p = 0.313, d = 0.66), LDL-C (F2, 37 = 12.48, P = 0.001, η²p = 0.403, d = 0.81) and AIP (F2, 37 = 6.25, P = 0.005, η²p = 0.253, d = 0.57).
Serum TG concentrations were significantly reduced in the HIIT group (–5.76%, P = 0.011, d = 0.33) and CONC group (–6.47%, P = 0.001, d = 0.44) compared with baseline. No significant differences were observed in CG (–0.93%, P = 0.516, d = 0.05). Despite a significant decrease in serum TG concentration in the CONC and HIIT groups, no significant difference was observed between pre-post-test differences in serum TG concentration before and after the test comparing the three groups (P = 0.250).
Serum TC concentrations were significantly reduced in the HIIT group (–7.53%, P = 0.001, d = 0.53) and CONC group (–7.42%, P = 0.001, d = 0.60) compared with baseline values. No significant differences were observed in CG (+0.59%, P = 0.407, d = 0.03). The serum TC reduction in the CONC (P = 0.001) and HIIT (P = 0.005) groups is significant compared with the CG. However, no significant difference was observed between the CONC and HIIT groups (P = 0.891).
Serum LDL-C concentrations were significantly reduced in the HIIT group (–14.02%, P = 0.001, d = 0.52) and CONC group (–8.31%, P = 0.001, d = 0.63) compared with baseline values. No significant differences were observed in CG (+1.48%, P = 0.252, d = 0.05). Compared to CG, CONC (P = 0.004) and HIIT (P = 0.005) showed reduced serum LDL-C. However, no significant differences were observed between the CONC and HIIT groups (P = 0.998).
Serum HDL-C concentration increased significantly in the HIIT group (+7.74%, P = 0.001, d = 0.45) and CONC group (+6.21%, P = 0.001, d = 0.43) at the end of intervention. No significant differences were observed in the CG (–1.65%, P = 0.110, d = 0.11). HDL-C presented higher values in the CONC (P = 0.029) and HIIT (P = 0.002) groups than in the CG. However, no significant difference was observed between the CONC and HIIT groups (P = 0.589).
The AIP index values in the HIIT group (–11.88%, P = 0.001, d = 0.40) and CONC group (–12.18%, P = 0.001, d = 0.48) decreased significantly compared to baseline values. In contrast, it increased significantly in the CG (+1.00%, P = 0.001, d = 0.02). AIP index values were significantly reduced in CONC (P = 0.040) and HIIT (P = 0.045) groups compared to the CG. However, no significant difference was observed between the CONC and HIIT groups (P = 0.655) (see Table 3).
Effect of concurrent training and high-intensity interval training on lipid profile in sedentary postmenopausal women with overweight or obesity (mean ± SD) at baseline (T1) and at the end of the study (T2).
| Variable | Group | T1 | T2 | Change (%) | Cohen’s d | ANOVA. P-value (Cohen’s d) | ||
|---|---|---|---|---|---|---|---|---|
| Interaction | Group | Time | ||||||
| TG (mg/dL) | CONC | 155.88 ± 24.31 | 145.78 ± 20.97†* | –6.47 | 0.44 | 0.001 (0.75) | 0.172 (0.31) | 0.047 (0.41)** |
| HIIT | 163.06 ± 28.24 | 153.66 ± 27.78†* | –5.76 | 0.33 | ||||
| CG | 170.90 ± 28.89 | 169.30 ± 28.84 | –0.93 | 0.05 | ||||
| TC (mg/dL) | CONC | 197.88 ± 27.66 | 183.18 ± 20.04†* | –7.42 | 0.60 | 0.001 (1.03) | 0.172 (0.32) | 0.001 (0.83)** |
| HIIT | 188.51 ± 26.21 | 174.30 ± 27.39†* | –7.53 | 0.53 | ||||
| CG | 200.84 ± 30.51 | 202.04 ± 31.33 | +0.59 | 0.03 | ||||
| LDL-C (mg/dL) | CONC | 112.12 ± 28.12 | 102.80 ± 20.8†* | –8.31 | 0.63 | 0.001 (0.95) | 0.229 (0.28) | 0.001 (0.81)** |
| HIIT | 112.12 ± 28.12 | 96.40 ± 20.08†* | –14.02 | 0.52 | ||||
| CG | 122.27 ± 30.23 | 124.09 ± 31.59 | +1.48 | 0.05 | ||||
| HDL-C (mg/dL) | CONC | 45.50 ± 6.39 | 48.33 ± 6.73 | +6.21 | 0.43 | 0.001 (0.65) | 0.640 (0.15) | 0.001 (0.66)** |
| HIIT | 43.77 ± 6.91 | 47.16 ± 7.93 | +7.74 | 0.45 | ||||
| CG | 44.82 ± 6.72 | 44.08 ± 6.67 | –1.65 | 0.11 | ||||
| AIP | CONC | 3.53 ± 0.98 | 3.10 ± 0.77†* | –12.18 | 0.48 | 0.001 (0.72) | 0.293 (0.25) | 0.005 (0.57)** |
| HIIT | 3.87 ± 1.16 | 3.41 ± 1.11†* | –11.88 | 0.40 | ||||
| CG | 3.97 ± 1.24 | 4.01 ± 1.27 | +1.00 | 0.02 | ||||
AIP: atherogenic index of plasma; CG: control group; CONC: concurrent training group; HDL-C: high-density lipoprotein cholesterol; HIIT: high-intensity interval training group; LDL-C: low-density lipoprotein cholesterol; SD: standard deviation; TG: triglycerides; TC: total cholesterol; %: percentage. †: significant difference from pre to post (P < 0.05); *: significant difference from CG (P < 0.05); ‡: significant difference from HIIT (P < 0.05); **: Significant time × group interaction. Two-way ANOVA with repeated measures, followed by the post hoc test of Bonferroni. Values are expressed as mean (SD).
ANOVA showed significant time effects for all glycemic biomarker parameters (P < 0.001 for all, d = 0.935–1.17). Significant time-group effects were observed for insulin (F2, 37 = 9.70, P = 0.001, η²p = 0.344, d = 0.69), glucose (F2, 37 = 5.36, P = 0.009, η²p = 0.225, d = 0.53) and HOMA-IR (F2, 37 = 11.68, P = 0.001, η²p = 0.387, d = 0.78).
Serum insulin concentrations were significantly reduced in the HIIT group (–18.25%, P = 0.001, d = 0.85) and CONC group (–19.27%, P = 0.002, d = 1.18) compared to baseline values. No significant differences were observed in CG (–0.14%, P = 0.840, d = 0.007). The insulin concentration reduction in the CONC (P = 0.001) and HIIT (P = 0.003) groups was significantly lower than in the CG. However, no significant difference was observed between the CONC and HIIT groups (P = 0.778).
Serum glucose concentrations were significantly reduced in the HIIT group (–11.96%, P = 0.001, d = 0.66) and CONC group (–4.16%, P = 0.002, d = 0.58) compared to baseline values. No significant difference was observed in CG (–0.89%, P = 0.840, d = 0.11). No significant difference was found in glucose changes comparing all study groups. HOMA-IR index concentrations were significantly reduced in the HIIT group (–23.14%, P = 0.001, d = 1.34) and CONC group (–23.88%, P = 0.001, d = 1.24) compared to baseline values. No significant difference was observed in CG (+0.61%, P = 0.366, d = 0.05). The HOMA-IR index reduction in the CONC (P = 0.001) and HIIT (P = 0.001) groups is significant compared to the CG. However, no significant difference was observed between the CONC and HIIT groups (P = 0.970) (see Table 4).
Effect of concurrent training and high-intensity interval training on glycemic biomarkers in sedentary postmenopausal women with overweight or obesity (mean ± SD) at baseline (T1) and at the end of the study (T2).
| Variable | Group | T1 | T2 | Change (%) | Cohen’s d | ANOVA. P-value (Cohen’s d) | ||
|---|---|---|---|---|---|---|---|---|
| Interaction | Group | Time | ||||||
| Insulin (μg/L) | CONC | 14.27 ± 2.11 | 11.52 ± 2.53†* | –19.27 | 1.18 | 0.001 (0.75) | 0.172 (0.31) | 0.047 (0.41)** |
| HIIT | 12.71 ± 2.85 | 10.39 ± 2.27†* | –18.25 | 0.85 | ||||
| CG | 13.75 ± 2.46 | 13.73 ± 2.63 | –0.14 | 0.007 | ||||
| Glucose (mg/dL) | CONC | 106.05 ± 9.27 | 101.63 ± 10.86†* | –4.16 | 0.58 | 0.001 (1.03) | 0.172 (0.32) | 0.001 (0.83)** |
| HIIT | 109.05 ± 7.97 | 96.00 ± 8.32†* | –11.96 | 0.66 | ||||
| CG | 106.05 ± 9.27 | 105.10 ± 7.79 | –0.89 | 0.11 | ||||
| HOMA-IR index | CONC | 3.60 ± 0.72 | 2.74 ± 0.66†* | –23.88 | 1.24 | 0.001 (0.95) | 0.229 (0.28) | 0.001 (0.81)** |
| HIIT | 3.37 ± 0.82 | 2.59 ± 0.06†* | –23.14 | 1.34 | ||||
| CG | 3.26 ± 0.79 | 3.28 ± 0.78 | +0.61 | 0.05 | ||||
CG: control group; CONC: concurrent training; HIIT: high-intensity interval training; HOMA-IR: homeostatic model assessment-insulin resistance; SD: standard deviation; %: percentage. †: significant difference from pre to post (P < 0.05); *: significant difference from CG (P < 0.05); ‡: significant difference from HIIT (P< 0.05); **: significant time × group interaction. Two-way ANOVA with repeated measures, followed by the post hoc test of Bonferroni. Values are expressed as mean (SD).
The percentage changes in metabolic biomarkers are presented in Figure 3.
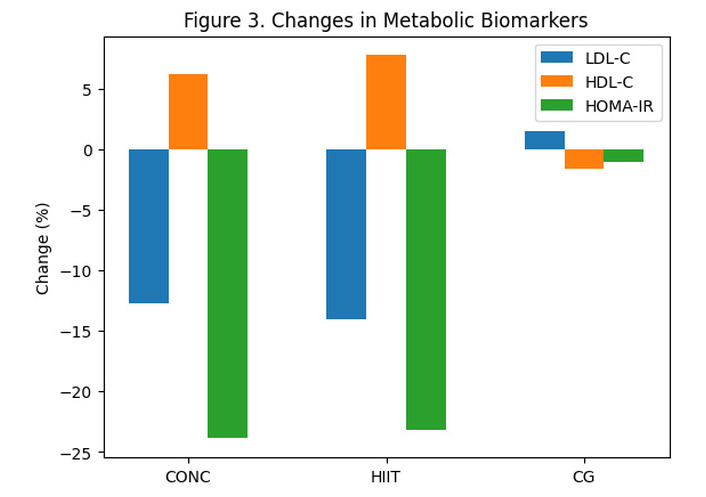
Changes in metabolic biomarkers after the 12-week intervention. Percentage changes in LDL-C, HDL-C, and HOMA-IR across groups (CONC, HIIT, and CG). CONC: concurrent training; CG: control group; HDL-C: high-density lipoprotein-cholesterol; HIIT: high-intensity interval training; HOMA-IR: homeostatic model assessment-insulin resistance; LDL-C: low-density lipoprotein-cholesterol.
The results of the Pearson correlation analysis showed that weight loss was significantly associated with reductions in BMI (r = 0.992, P = 0.001), %BF (r = 0.695, P = 0.001), WC (r = 0.652, P = 0.001), WHR (r = 0.651, P = 0.001), LDL-C (r = 0.548, P = 0.001), insulin (r = 0.468, P = 0.001), and HOMA-IR (r = 0.420, P = 0.002), and with an increase in HDL-C (r = 0.447, P = 0.001).
BMI reduction had a significant positive relationship with a decrease in %BF (r = 0.703, P = 0.001), WC (r = 0.651, P = 0.001), WHR (r = 0.644, P = 0.001), LDL-C (r = 0.561, P = 0.001), insulin (r = 0.480, P = 0.002), HOMA-IR (r = 0.436, P = 0.005) and with an increase in HDL-C (r = 0.475, P = 0.002).
Decreased %BF also presented a significant positive relationship with decreased WC (r = 0.805, P = 0.001), WHR (r = 0.746, P = 0.001), TC (r = 0.678, P = 0.001), LDL-C (r = 0.667, P = 0.001), insulin (r = 0.462, P = 0.003), HOMA-IR index (r = 0.491, P = 0.001) and with increased HDL-C (r = 0.611, P = 0.001) (see Table 5).
The Pearson correlation coefficient between the study outcomes.
| Variables | Weight | BMI | %BF |
|---|---|---|---|
| Weight | - | - | - |
| BMI | r = 0.992P = 0.001 | -- | -- |
| %BF | r = 0.695P = 0.001 | r = 0.703P = 0.001 | -- |
| WC | r = 0.652P = 0.001 | r = 0.651P = 0.001 | r = 0.805P = 0.001 |
| WHR | r = 0.651P = 0.001 | r = 0.644P = 0.001 | r = 0.746P = 0.001 |
| LDL-C | r = 0.548P = 0.001 | r = 0.561P = 0.001 | r = 0.667P = 0.001 |
| Insulin | r = 0.468P = 0.001 | r = 0.480P = 0.002 | r = 0.462P = 0.003 |
| HOMA-IR | r = 0.420P = 0.002 | r = 0.436P = 0.005 | r = 0.491P = 0.001 |
| HDL-C | r = 0.447P = 0.001 | r = 0.475P = 0.002 | r = 0.611P = 0.001 |
BF: body fat; BMI: body mass index; HDL-C: high-density lipoprotein cholesterol; HOMA-IR: homeostatic model assessment-insulin resistance; LDL-C: low-density lipoprotein cholesterol; WC: waist circumference; WHR: waist-hip ratio.
No adverse events, musculoskeletal injuries, or exercise-related complications were reported throughout the 12-week intervention, confirming the safety and feasibility of both exercise modalities in sedentary postmenopausal women with overweight or obesity.
The main findings of the study indicate that both HIIT and CONC, executed over 12 weeks, significantly improved anthropometric parameters, lipid profile, and glycemic biomarkers in postmenopausal women with overweight or obesity compared to CG. The two-way repeated-measures ANOVA (time × group) analyses suggest that exercise (HIIT and CONC) induced a favorable change in anthropometric and circulating parameters over 12 weeks of intervention, while no improvement occurred in sedentary women. Participants in the CONC group experienced significantly greater weight loss, a more pronounced reduction in BMI, and a significant decrease in %BF compared to the HIIT group participants. No other statistically significant differences were observed between CONC and HIIT groups for the remaining biomarkers. HC did not change significantly from baseline to the end of the study, nor did it differ among the three groups: CONC, HIIT, and CG.
Supporting our findings, a systematic review of 13 clinical trials demonstrated that HIIT was associated with improvements in various health markers, including body composition, cardiovascular fitness, and metabolic parameters [39]. Similarly, Mengistu et al. [40] provide strong evidence for the beneficial effects of CONC exercise on metabolic and cardiovascular health markers. CONC training has been shown to improve several biomarkers of general health status, aiding in the management of weight-related conditions in adults with concomitant overweight/obesity [41, 42]. In contrast to these findings, some studies report that HIIT does not significantly affect lipid profiles, specifically TG and HDL-C [43], nor does it improve blood markers of endothelial function, cardiovascular risk [44], and metabolic outcomes [45]. Such discrepancies could be related to interindividual variability in response to identical training protocols (HIIT or CONC) or can be attributed to differences in participant characteristics, training intensity, type, and duration. In addition, individual genetics, environmental influences, and molecular mechanisms may substantially influence individual adaptation to exercise [46].
Previous studies have demonstrated [47–49] that different types of exercise could significantly influence body composition indicators in obese and postmenopausal women. A systematic review and meta-analysis of 101 studies involving 5,697 postmenopausal women confirmed that exercise training effectively improves body composition [15]. Specifically, the meta-analysis identified CONC exercise as the most viable strategy for improving body composition and promoting overall benefits in anthropometric parameters among postmenopausal women [15]. These findings align with our results, which indicated that CONC exercise was significantly more effective than HIIT in reducing weight, BMI, and %BF. Supporting this, Shabani et al. [50] reported that an 8-week CONC training significantly improved body composition indicators in obese/overweight postmenopausal women. Moreover, although various training modalities, including resistance, endurance, and CONC, have been associated with improvements in body composition among obese individuals, CONC exercise appears to yield greater benefits in particular indicators such as weight and %BF. This may be attributed to the synergistic effects and overlap between endurance and resistance components inherent in CONC routines [51]. Mechanistically, CONC exercise may promote fat loss through enhanced mitochondrial biogenesis, improved insulin sensitivity, and increased energy expenditure [52].
Alternatively, HIIT may be particularly effective in reducing abdominal and visceral fat, potentially outperforming low- and moderate-intensity continuous aerobic exercise, by enhancing fat metabolism and reducing fat accumulation [53]. HIIT’s impact on abdominal fat is supported by activation of physiological pathways that promote increased fat metabolism, including enhanced mitochondrial capacity and content, elevated growth hormone levels, and heightened sympathetic nervous system activity. This involves the secretion of catecholamines such as epinephrine and norepinephrine, which stimulate lipolysis through β-adrenergic receptor activation, contributing to increased fat metabolism [54]. These mechanisms may stimulate lipolytic enzyme activity, increasing fat oxidation and energy expenditure, and suppressing appetite after exercise, thereby contributing to improved body composition in obese and sedentary individuals [54]. While both HIIT and CONC training are effective for enhancing health and body composition, CONC training is frequently reported to be more beneficial for beneficial improvements such as increasing lean muscle and reducing fat mass [55]. Although HIIT offers notable advantages in cardiovascular fitness and time efficiency [56], the combination of strength and endurance components of CONC programs often yields superior outcomes in %BF reduction and muscle gain, particularly for individuals aiming to optimize fat reduction.
Both CONC and HIIT significantly improved WHR, with reductions of 2.10% and 3.12%, respectively, primarily through decreases in BF and increases in muscle mass. Although both modalities were effective, HIIT showed a slightly greater reduction in abdominal fat (–2.88%) compared to CONC exercise (–2.60%). The mechanisms underlying these effects differ: HIIT enhances body composition predominantly through its strong influence on fat metabolism, including increased fat oxidation and elevated post-exercise oxygen consumption (EPOC), alongside favorable adaptations in metabolic pathways [54, 57]. These adaptations contribute to healthier body composition and a more favorable fat distribution. In contrast, CONC training promotes body recomposition by simultaneously building muscle and reducing fat, while also improving cardiovascular fitness. Resistance components within CONC protocols stimulate muscle hypertrophy, thereby increasing resting metabolic rate and facilitating greater caloric expenditure. Meanwhile, the aerobic component supports cardiovascular health and contributes to fat loss [55, 56].
Menopause is associated with atherogenic dyslipidemia, a condition characterized by an unhealthy lipid profile that increases the risk of CVD [58]. During exercise, skeletal muscles increase their uptake and utilization of circulating free fatty acids as an energy source, contributing to reductions in plasma lipid levels. This effect is partially mediated by increased mitochondrial activity and improved intramuscular fatty acid transport [59]. Therefore, exercise would be a key strategy in both the prevention and management of obesity and serves as a non-pharmacological treatment for dyslipidemia [6]. In the present study, both HIIT and CONC exercise programs significantly improved plasma lipid profile following 12 weeks of training in sedentary postmenopausal women with overweight or obesity. In addition to significant weight loss, participants exhibited marked reductions in TC, TG, LDL-C, and AIP. A positive trend was also observed in HDL-C, suggesting that the intervention not only reduced overall body weight but also favorably influenced lipid biomarkers and CVD risk. Supporting these results, Shabani et al. [50] reported that 8-weeks of CONC training improved both body composition indicators and the lipid profile biomarkers in sedentary overweight or obese post-menopausal women. In the same line, Magalhães et al. [60] found that HIIT was more effective than moderate-intensity activity in improving AIP and lipid profile biomarkers in T2D patients.
The main activator of lipolysis during exercise is the sympathoadrenal system. The effect of beta-adrenergic receptors is mediated by stimulation of the sympathetic nervous system or by circulating epinephrine (adrenaline). This hormone is considered the main activator of hormone-sensitive lipase (HSL). HSL hydrolyzes TG stored in adipocytes, releasing free fatty acids and glycerol to be used as energy substrates (i.e., skeletal muscle) by other tissues during exercise [61]. Both HIIT and CONC training could enhance the activity of lipoprotein lipase (LPL). This enzyme is essential for the breakdown of TG within circulating lipoproteins. This process facilitates the uptake of fatty acids by muscle tissue for use as fuel. This increased LPL activity represents an adaptive response to exercise, promoting more efficient lipid utilization for energy production [62]. Therefore, these mechanisms may underlie the observed effects of HIIT and CONC on the AIP index and lipid profile biomarkers [63].
It is important to emphasize that both CONC and HIIT exercises led to a more significant reduction in LDL-C than HDL-C among the obese women in our study. Reduced levels of LDL-C appear to be more directly and strongly influenced by lifestyle changes such as exercise, which affects LDL-C hepatic production and clearance [64]. In contrast, the HDL-C response is more complex, as it is modulated by individual genetic factors as well as the intensity and duration of exercise [65]. In addition, increased HDL-C may be more closely associated with enhanced functional capacity, such as improved reverse cholesterol transport, rather than with elevated plasma concentration of this lipoprotein [66]. Therefore, in obese women undergoing CONC or HIIT protocols, a significant LDL-C reduction and an improvement in HDL-C functionality can be expected, even if HDL-C concentration does not increase significantly. These findings suggest that the cardiometabolic benefits of exercise may be more closely linked to qualitative improvements in lipid metabolism than to quantitative changes alone [67].
IR is a major metabolic dysfunction implicated in the development and progression of CVD, particularly affecting women and individuals with obesity [68]. Post-menopausal women, in particular, experience marked alterations in insulin sensitivity due to a decline in estrogen levels, which reduces the cellular response to insulin [69]. Physical activity is widely recognized as a potent tool for preventing and managing obesity, metabolic disorders, and cardiovascular conditions. Regular exercise, encompassing both aerobic and resistance training, enhances insulin sensitivity by promoting more efficient glucose uptake and utilization by peripheral tissues [70]. HIIT and CONC have gained increasing attention for their effectiveness in improving insulin sensitivity [71]. Our findings demonstrate that both HIIT and CONC significantly reduce IR, as well as plasma glucose and insulin concentrations. These results are consistent with previous findings by Banitalebi et al. [72] and Ghafari et al. [73], who reported improvements in IR indices following 10 weeks of HIIT and CONC in women. In this way, a systematic review and meta-analysis including 24 clinical trials confirmed the efficacy of these exercise modalities in improving insulin sensitivity among sedentary obese adults [18]. The reduction in insulin secretion observed during exercise is attributed to increased sympathetic and decreased parasympathetic activity, alongside increased utilization of non-esterified fatty acids [74]. Furthermore, HIIT and CONC increase GLUT4 expression in skeletal muscle [75], through activation of AMP-activated protein kinase (AMPK) [76], and improve insulin receptor signaling, optimizing insulin action and glucose metabolism [77]. Moreover, exercise-induced reductions in circulating CTRP1 levels could serve as a biomarker of improved insulin sensitivity [71]. Enhanced insulin sensitivity contributes to better glycemic control, a lower risk of IR and CVD, and overall improvements in metabolic health.
Both exercise modalities appear to improve carbohydrate metabolism by promoting glucose uptake, improving insulin sensitivity, and increasing muscle capacity to store and use carbohydrates [78]. The high intensity of HIIT quickly depletes intramuscular glycogen stores, which are the primary form of glucose storage in muscle. This depletion serves as a key stimulus for enhancing insulin sensitivity. Following a HIIT session, skeletal muscle exhibits increased sensitivity to insulin, facilitating more efficient glucose uptake from the bloodstream even in the post-exercise period [77]. On the other hand, CONC exercise improves glucose uptake through increased blood flow and expansion of capillary surface in active muscles, thereby enhancing glucose delivery for the same level of insulin [79]. Aerobic exercise also upregulates key metabolic enzymes such as pyruvate dehydrogenase (PDH), which governs the entry of carbohydrates into the Krebs cycle for ATP production [80]. This enzymatic activation accelerates the rate at which carbohydrates are oxidized for energy (ATP) production. Furthermore, CONC has been associated with improved mitochondrial function, thereby enhancing the muscle’s oxidative ability to use carbohydrate substrates efficiently [16].
In summary, both HIIT and CONC training appear to modulate key metabolic pathways in skeletal muscle and adipose tissue in postmenopausal women, contributing to a more favorable anthropometric and cardiometabolic profile. In muscle, these exercise modalities enhance mitochondrial biogenesis, increase glucose uptake, and improve substrate oxidation, helping counteract the decline in insulin sensitivity associated with estrogen deficiency in menopause. In adipose tissue, high-intensity exercise has been linked to reductions in fat mass, greater glucose uptake, and alterations in proteins regulating lipid metabolism, suggesting improved adipocyte function and a less pro-inflammatory profile. Altogether, these adaptations may explain the improvements in body composition and circulating markers observed in this population [81, 82]. Future research has to verify all suggested hormonal and molecular mechanisms.
This study presents several limitations. First, the sample size was small and recruited from a single center. Future research involving larger multicentric cohort samples would provide more robust and generalizable results. Second, it is also important to consider that postmenopausal women with overweight or obesity, who voluntarily enroll in CONC or HIIT exercise programs, may be more motivated and physically capable than the broader population of postmenopausal women with overweight or obesity, potentially contributing to higher adherence rates. Consequently, our study’s findings may not be generalizable to the general population, as the study sample represents a specific and self-selected subgroup. Third, the absence of dietary monitoring precludes accurate assessment of participants’ nutritional intake, limiting the ability to control for dietary influences on metabolic outcomes. Fourth, the duration of the study, because the lack of a follow-up period further restricts the evaluation of long-term effects. Five, the study did not incorporate complementary biochemical analyses, particularly those assessing inflammatory biomarkers and antioxidant status. These unmeasured parameters could act as confounding variables. Including such parameters in future studies will be essential to provide a more comprehensive understanding of the physiological effects of exercise interventions. Finally, the idea for the intervention study arose from a clinical observation. This led to a lack of coordination and a delay in registering the study, which was ultimately completed at the time of manuscript submission.
The findings of the present study support the implementation of HIIT and CONC programs as effective tools to improve body composition, circulating lipid profile, insulin sensitivity, and carbohydrate metabolism in postmenopausal women with overweight or obesity. These modalities of exercise can be integrated into clinical and community-based health strategies to reduce cardiometabolic risk, enhance metabolic adaptation, and promote long-term adherence to physical activity. Tailoring exercise prescriptions to individual capabilities and monitoring metabolic responses may further optimize outcomes in this vulnerable population.
Our findings suggest that 12 weeks of CONC and HIIT exercises may serve as effective interventions for obesity management, particularly in sedentary postmenopausal women. Both exercise modalities were associated with improvements in body composition, reductions in BF, and favorable changes in lipid and glycemic biomarker profiles. Based on these outcomes, CONC and HIIT exercises are recommended as viable strategies to enhance metabolic health and body composition in individuals with obesity, particularly by improving blood lipid and glucose regulation.
1RM: one-repetition maximum
AIP: atherogenic index of plasma
ANOVA: analysis of variance
BF: body fat
BMI: body mass index
CG: control group
CONC: concurrent training
CVD: cardiovascular diseases
HC: hip circumference
HDL-C: high-density lipoprotein-cholesterol
HIIT: high-intensity interval training
HOMA: homeostatic model assessment
HRmax: maximum heart rate
HSL: hormone-sensitive lipase
IR: insulin resistance
LDL-C: low-density lipoprotein-cholesterol
LPL: lipoprotein lipase
RPE: rating of perceived exertion
T2D: type 2 diabetes
TC: total cholesterol
TG: triglycerides
WC: waist circumference
WHO: World Health Organization
WHR: waist-hip ratio
η²p: partial eta-squared
The supplementary table for this article is available at: https://www.explorationpub.com/uploads/Article/file/101469_sup_1.pdf.
The authors wish to thank the Neurobiology Research Group, University of Valladolid, for their support in advising on methodological design and result interpretation. CIBEROBN is an initiative of the Instituto de Salud Carlos III, Madrid (Spain).
SA: Conceptualization, Methodology, Formal analysis, Investigation, Writing—review & editing. SRS: Methodology, Investigation. MGB: Software, Validation, Investigation. JMM: Software, Validation, Project administration. GRO: Software, Validation, Project administration. GS: Methodology, Validation, Investigation, Project administration. ER: Formal analysis, Project administration, Writing—review & editing. JMA: Validation, Formal analysis, Writing—review & editing. AMCSM: Validation, Formal analysis, Writing—review & editing. DFL: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Project administration, Funding acquisition, Writing—original draft, Writing—review & editing. MAA: Conceptualization, Formal analysis, Investigation, Project administration, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
The study was approved by the Research Ethics Committee of the Islamic Azad University Mahabad Branch (IR.IAU.MAHABAD.REC.1404.012).
Informed consent to participate in the study was obtained from all participants.
Not applicable.
The datasets that support the findings of this study are available from the corresponding author upon reasonable request.
The research is funded by the TCUE 2024-2027 Plan, approved by agreement on September 16, 2024, and has been selected within the framework of an operational program co-financed by the European Regional Development Fund (ERDF) and the Regional Government of Castile and León (067/230003 POC). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1333
Download: 32
Times Cited: 0
Butheinah A. Al-Sharafi, Samih A. Odhaib
Nagamani Gumpeny ... Sridhar R. Gumpeny
Bruno Vecchiatto ... Fabiana S. Evangelista
