Affiliation:
1Department of Pharmacy Practice, School of Pharmacy, Massachusetts College of Pharmacy and Health Sciences, Boston, MA 02115, United States
Affiliation:
1Department of Pharmacy Practice, School of Pharmacy, Massachusetts College of Pharmacy and Health Sciences, Boston, MA 02115, United States
2Well Life Medical, Peabody, MA 01960, United States
Email: jennifer.goldman@mcphs.edu
ORCID: https://orcid.org/0000-0002-9617-3998
Explor Endocr Metab Dis. 2026;3:101465 DOI: https://doi.org/10.37349/eemd.2026.101465
Received: January 25, 2026 Accepted: March 26, 2026 Published: April 02, 2026
Academic Editor: Marijn Speeckaert, Universitair Ziekenhuis Ghent, Belgium
Epiploic appendagitis is a rare, self-limiting inflammatory condition of the colonic epiploic appendages. It commonly presents with acute focal lower quadrant abdominal pain and may mimic appendicitis, colitis, or diverticulitis. Rapid weight loss has been proposed as a potential predisposing factor, although clinical evidence remains limited. This report describes a case of epiploic appendagitis occurring in temporal association with substantial and relatively rapid weight reduction during treatment with tirzepatide, a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. A 45-year-old individual with a history of obesity, prediabetes, hypertension, hypothyroidism, and inflammatory bowel syndrome presented to primary care with right lower quadrant abdominal pain. Initial evaluation attributed symptoms to constipation; however, pain progressed despite management. Computed tomography (CT) of the abdomen and pelvis with contrast was obtained due to concern for appendicitis and demonstrated imaging findings consistent with epiploic appendagitis localized to the cecum. The patient had experienced clinically significant and relatively rapid weight loss during tirzepatide dose escalation prior to presentation. Management was conservative with nonsteroidal anti-inflammatory drugs and supportive care, without surgical intervention. Symptoms resolved completely within two weeks, and tirzepatide therapy was continued without recurrence. Although epiploic appendagitis has not been identified as a safety signal in randomized trials, systematic reviews, or pharmacovigilance analyses of GLP-1/GIP receptor agonists, this case highlights epiploic appendagitis as an important diagnostic consideration in patients presenting with focal abdominal pain during periods of substantial weight loss. Awareness of this uncommon condition may help prevent misdiagnosis and unnecessary interventions as pharmacologic weight loss therapies become more widely used.
This case describes epiploic appendagitis occurring in temporal association with substantial and relatively rapid weight loss during tirzepatide therapy. Epiploic appendagitis is an uncommon, self-limiting inflammatory condition of the colonic epiploic appendages that frequently mimics more common causes of acute abdominal pain, including appendicitis, colitis, and diverticulitis. To our knowledge, published reports of epiploic appendagitis occurring during glucagon-like peptide-1 (GLP-1)/glucose-dependent insulinotropic polypeptide (GIP) receptor agonists associated with weight loss are limited. This case represents among the first reported instances highlighting epiploic appendagitis in the context of modern incretin-based therapies, as distinct from prior reports with bariatric surgery or spontaneous weight loss. Although causality cannot be inferred with a single case, the timing of symptom onset in the context of significant weight reduction highlights the importance of considering epiploic appendagitis in the differential diagnosis of focal abdominal pain in patients undergoing pharmacologically induced weight loss. For the purposes of this report, the pace of weight loss is contextualized using both absolute and percent weight loss rates and compared with expected trajectories reported in clinical trials of tirzepatide. The patient's rate of weight loss is further contextualized against the guideline-recommended target of 10% body weight reduction within 6 months [1].
GIP and GLP-1 receptor agonists are increasingly prescribed for the management of type 2 diabetes and obesity due to their demonstrated efficacy in achieving glycemic control and clinically meaningful weight loss. Dual GIP/GLP-1 receptor agonists, however, remain relatively new in clinical practice. As their use expands, substantial weight reduction, previously uncommon outside of surgical interventions, is now frequently observed. While weight loss is associated with important improvements in cardiometabolic comorbidities such as hypertension and type 2 diabetes, rapid weight reduction may also introduce clinical considerations that are not yet fully characterized. Because medication-associated rapid weight loss is a relatively recent and widespread phenomenon, its potential complications remain understudied, highlighting the need for continued clinical awareness and careful patient monitoring.
Key events are summarized in the timeline of the patient’s clinical event (Table 1).
The timeline of the patient’s clinical event.
| Date | Clinical event | Weight (kg) | Weight (lb) | Percent of change from prior weight (%) |
|---|---|---|---|---|
| Jun 2022 | Baseline weight | 117.72 | 259.00 | - |
| Nov 16, 2022 | Tirzepatide increased to 5 mg weekly | 121.09 | 266.40 | +2.86% |
| Dec 28, 2022 | Dose increased to 7.5 mg weekly | - | - | - |
| Feb 7, 2023 | Dose increased to 10 mg weekly | 109.59 | 241.10 | –9.50% |
| Mar 27, 2023 | Dose increased to 12.5 mg weekly | 101.36 | 223.00 | –7.51% |
| Mar 31, 2023 | Acute right lower quadrant abdominal pain. CT abdomen/pelvis showed epiploic appendagitis at the level of the cecum | 101.36 | 223.00 | 0.00% |
| Apr 2023 | Managed conservatively with NSAIDs and supportive care. Symptoms resolved. Tirzepatide was restarted at 10 mg with gradual titration | 101.36 | 223.00 | 0.00% |
| Oct 2025 follow-up | Patient on tirzepatide 15 mg weekly with no recurrence | 78.18 | 172.00 | −22.87% |
CT: computed tomography; NSAIDs: nonsteroidal anti-inflammatory drugs.
A 45-year-old non-binary Caucasian individual, assigned female at birth, presented to primary care with right lower quadrant abdominal pain. The patient’s gender identity has no known relevance to disease pathophysiology in this case. The individual is referred to hereafter as “the patient”. At the time of encounter, the patient had a past medical history of class II obesity and a body mass index (BMI) of 37.68 kg/m2, prediabetes, hypertension, hypothyroidism, and inflammatory bowel syndrome. The patient’s medication list at the time included tirzepatide 12.5 mg once weekly, metformin extended release (ER) 500 mg two tablets twice daily, amlodipine 5 mg once daily, losartan 100 mg once daily, vortioxetine 20 mg once daily, and levothyroxine 112 μg once daily. The patient also uses a continuous positive airway pressure (CPAP) machine at night.
At the most recent follow-up in October 2025, the patient weighed 78.18 kg (172 lb), representing a total weight reduction of 39.54 kg (87 lb), corresponding to a 33.6% decrease from baseline body weight. At baseline, the patient had class III obesity with a weight of 117.72 kg (259 lb) and was initiated on tirzepatide 2.5 mg once weekly in June 2022 to assist with weight loss. The patient experienced a slight weight increase during the 2.5 mg initiation phase, which is expected given that this dose is intended for gastrointestinal tolerability rather than therapeutic weight loss. The dose was later escalated to 5 mg once weekly in November 2022 and gradually increased to 12.5 mg by March 2023. On March 31, 2023, the patient presented to their primary care with right lower quadrant abdominal pain. The tirzepatide dose had been escalated from 7.5 mg to 10 mg in February 2023 and further increased to 12.5 mg on March 27, 2023, four days prior to the onset of acute abdominal pain.
By March 2023, the patient’s weight had decreased from 117.72 kg (259 lb) in June 2022 to 101.36 kg (223 lb), representing a reduction of 16.36 kg (36 lb), corresponding to a 13.9% decrease in baseline body weight. Most of the weight reduction occurred following dose escalation to 5 mg or greater. In randomized clinical trials, tirzepatide has demonstrated mean weight reductions ranging from approximately 15% at the 5 mg maintenance dose to 20.9% at the 15 mg dose over 52 weeks [2]. General lifestyle-based weight management guidelines typically recommend a target reduction of 5 to 10% of baseline body weight over approximately six months. The magnitude and pace of weight reduction observed in this patient occurred during active pharmacologic dose escalation and exceeded the minimum guideline-recommended threshold for clinically meaningful weight loss.
Physical examination demonstrated focal right lower quadrant tenderness. The patient denied fever or dietary changes prior to the start of the abdominal pain. Initial clinical suspicion included constipation and appendicitis. A bowel regimen was initiated but did not improve symptoms, and the patient reported progressive abdominal pain exacerbated by movement, coughing, and car travel. Due to persistent symptoms and concern for appendicitis, a computed tomography (CT) of the abdomen and pelvis with contrast was obtained (Figure 1). The images from the CT scan confirmed epiploic appendagitis at the level of the cecum.
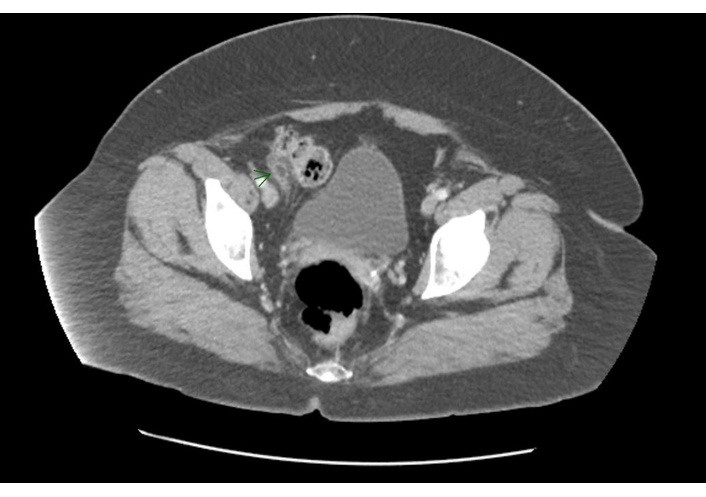
Contrast-enhanced computed tomography (CT) of the abdomen/pelvis. Contrast-enhanced CT of the abdomen/pelvis demonstrating an ovoid fat-density lesion adjacent to the cecum with surrounding inflammatory change, consistent with epiploic appendagitis. The normal caliber of the appendix and absence of appendiceal enlargement, and periappendiceal fluid reduce suspicion for appendicitis and support epiploic appendagitis as the primary diagnosis.
The CT demonstrated an ovoid fat-density lesion lateral to the cecum with surrounding focal fat stranding and a hyperattenuating ring, consistent with epiploic appendagitis. The appendix was visualized and appeared normal. No ascites, peritoneal fluid collections, intraperitoneal air, abdominal wall hernia, colonic wall thickening, pericolonic inflammatory changes, or findings suggestive of active colitis or terminal ileitis were noted. Lesion size was not documented in the imaging report. The radiologist’s impression was “Epiploic appendagitis at the level of the cecum. Normal appendix”.
Appendicitis was the primary diagnostic concern given the patient’s right lower quadrant pain and progressive symptoms. Other potential etiologies included constipation, diverticulitis, inflammatory bowel syndrome flare, and epiploic appendagitis. CT imaging demonstrated a focal fat density lesion with a hyperattenuating rim adjacent to the cecum and a normal appearing appendix, findings consistent with epiploic appendagitis and excluding appendicitis.
Vital signs and anthropometric measurements at presentation are summarized in Table 2. No laboratory tests were conducted at the time of the encounter, as appendicitis was suspected, and a CT scan was required for a diagnosis.
Vital signs and anthropometrics at presentation.
| Test | Result | Reference range | Interpretation |
|---|---|---|---|
| Blood pressure | 138/78 mmHg | 90/60–120/80 mmHg | Elevated |
| Heart rate | 100 BPM | 60–100 BPM | High-normal |
| Temperature | 97.4°F | 97.0–99.0°F | Normal |
| SpO2 | 99% | 95–100% | Normal |
| Actual weight | 101.36 kg (223 lb) | N/A | N/A |
| Height | 1.638 m (5 ft 4.5 in) | N/A | N/A |
| BMI (kg/m2) | 37.68 | 18.5–24.9 | Obese (class II) |
BMI: body mass index; SpO2: percutaneous oxygen saturation.
The patient was initially treated with a bowel regimen without symptom improvement. Following review of the CT findings confirming epiploic appendagitis, conservative management was recommended. The patient was advised to take over-the-counter nonsteroidal anti-inflammatory drugs as needed for pain control and to follow up in two weeks if symptoms persisted, and to seek urgent medical evaluation if symptoms worsened. No surgical intervention was required.
The patient reported using ibuprofen 200 mg twice daily for the first two days following diagnosis, followed by an increase to four doses daily for approximately nine days. During the initial eight days, the patient described being largely bed-bound due to severe pain and limited mobility. Symptoms gradually improved and fully resolved after approximately two weeks with rest and as-needed analgesia.
The patient noted that lying in the prone position provided the greatest pain relief from abdominal pain. During the acute phase, the patient also reported that consuming pureed, lightly seasoned foods and clear liquids was more tolerable than solid foods. The patient further reported minimal weight loss while receiving tirzepatide 2.5 mg weekly, with more rapid weight reduction observed following escalation to higher doses. The patient recalled a history of being overweight during adolescence.
Epiploic appendagitis is a rare, self-limiting inflammatory condition of the colonic epiploic appendages that frequently mimics more common causes of acute abdominal pain, such as appendicitis or diverticulitis. Early recognition is clinically important because accurate diagnosis can prevent unnecessary antibiotic therapy, invasive procedures, or surgical consultation. In this case, epiploic appendagitis occurred in temporal association with substantial weight reduction during tirzepatide therapy. To our knowledge, there are limited published reports describing epiploic appendagitis occurring in the context of GLP-1/GIP receptor agonist-induced pharmacologic weight loss. This case adds to the emerging literature and represents one of the first reported instances in the context of modern incretin-based therapies, distinct from bariatric surgery or spontaneous weight loss. Epiploic appendagitis is most commonly caused by torsion or thrombosis of an epiploic appendage, leading to ischemia, edema, aseptic inflammation, and eventual resorption by the peritoneal cavity [3, 4]. Although epiploic appendagitis has not been identified as an adverse event or safety signal in randomized trials, systematic reviews, or pharmacovigilance analyses of GLP-1/GIP receptor agonists, it is biologically plausible that rapid weight loss could increase susceptibility to this condition. The epiploic appendages are droplet-shaped fatty projections attached to the taeniae on the surface of the colon, most commonly along the taenia libera and taenia omentalis of the cecum and sigmoid colon [3]. Approximately 50 to 100 epiploic appendages form two rows along the surface of the colon [3, 4]. These structures are supplied by one or two small arteries and a single draining vein, rendering them vulnerable to changes in perfusion [3, 4]. The epiploic appendages develop during adulthood and tend to be more prominent in individuals with obesity [3]. Although their precise function is not fully understood, they are thought to provide mechanical cushioning during peristalsis and compression of colonic vessels, or may serve as reservoirs for blood supply [3]. As pharmacologic weight-loss therapies become increasingly common in clinical practice, awareness of uncommon causes of focal abdominal pain may help clinicians avoid misdiagnosis and unnecessary interventions in patients undergoing substantial weight loss.
Several risk factors for epiploic appendagitis have been described, including male sex, age between 40 and 50, obesity, hypertension, diabetes, increased fat around the abdominal organs, visceral adiposity, intensive strenuous exercise, and the presence of abdominal hernias [3, 4]. In a retrospective cohort study, patients with epiploic appendagitis were found to have 60% greater abdominal adipose volume and a 117% increase in visceral adipose area compared with controls [3]. Given these associations, abrupt changes in visceral fat volume or perfusion may increase the likelihood of torsion or thrombotic ischemia in susceptible individuals.
Tirzepatide has been shown to significantly reduce visceral and abdominal tissue [5]. Weight loss has been associated with decreased perfusion of abdominal subcutaneous and visceral adipose tissue compartments [6]. Each epiploic appendage is perfused by a limited vascular supply, which makes these structures particularly sensitive to changes in blood flow. In a randomized trial, patients receiving tirzepatide achieved mean weight reductions of 15% to 20.9% after 52 weeks, compared to a 3.1% reduction with placebo [2]. In the present case, epiploic appendagitis occurred in temporal association with substantial and relatively rapid weight loss during tirzepatide dose escalation, consistent with the hypothesis that physiologic changes accompanying weight reduction may have influenced susceptibility, although causality cannot be inferred. Epiploic appendagitis most commonly affects appendages along the descending colon (24.5%), sigmoid (22.6%), cecum (14.5%), or the ascending colon (14.5%) [4].
Another possible mechanism relates to anatomic and vascular changes accompanying a substantial reduction in visceral adiposity. Epiploic appendages are pedunculated fat-containing structures supplied by a limited vascular pedicle, making them susceptible to torsion and venous outflow obstruction. During periods of rapid visceral fat loss, decreased surrounding fat volume may alter the relative mobility of the appendages along the colonic surface. This increase in mobility may theoretically predispose a vulnerable appendage to torsion. Torsion typically results in a narrow vascular stalk, leading to ischemia and localized aseptic inflammation [3, 4]. In parallel with weight loss changes in adipose tissue perfusion, this theoretically may further influence susceptibility in these structures with limited blood supply. In this patient, the transition from class III obesity with presumed high visceral adiposity to a state of rapid fat reduction during dose escalation may have theoretically created the anatomical conditions necessary for torsion. This remains speculative, and causality cannot be inferred from a single case.
Diagnosis is typically established with imaging, with CT considered the diagnostic modality of choice [3, 4]. Characteristic CT findings include an ovoid fat-density lesion measuring between 1.5 and 3.5 cm in diameter with a surrounding hyperdense ring corresponding to inflamed serosa [3, 4]. Ultrasound at the site of maximal tenderness may reveal a hyperechoic, noncompressible mass with absent central blood flow on Doppler imaging and can be useful in select populations, such as pregnant patients or those with contraindications to radiation exposure [3]. Magnetic resonance imaging is used less frequently but may offer advantages in pediatric or pregnant females due to the absence of ionizing radiation [3]. Laboratory findings are often normal, with mild leukocytosis representing the most commonly reported abnormality [3, 4]. Symptoms of epiploic appendagitis consist of: pain (100%), left abdomen (53%), right abdomen (42%), tenderness (59.5%), rebound tenderness (27.4%), nausea (20.2%), fever (13.4%), vomiting (11.5%), bowel changes (6.4%) [3, 4].
Management of epiploic appendagitis is typically conservative, with or without the use of nonsteroidal anti-inflammatory drugs [3, 4]. Treatment may occur in either inpatient or outpatient settings, depending on symptom severity and diagnostic uncertainty. Surgical intervention, consisting of laparoscopic excision of the affected appendage, is reserved for select cases [3, 4]. While some studies report higher recurrence rates with conservative management compared to surgical treatment, other data suggest no significant difference in recurrence [3, 4]. Symptoms generally resolve in one to two weeks, consistent with the clinical course observed in this case.
This report is subject to several limitations, including its single-case design, incomplete control of potential risk factors, missing information, and the presence of clinical confounders such as baseline obesity and inflammatory bowel syndrome. As such, causal inference cannot be drawn, and the findings should be interpreted as hypothesis-generating. Despite these limitations, this case underscores the importance of considering epiploic appendagitis in the differential diagnosis of acute focal abdominal pain, particularly in patients undergoing substantial weight loss. Early recognition and confirmation with CT may help avoid misdiagnosis, unnecessary interventions, and support appropriate conservative management as pharmacologic weight loss therapies become more widely used.
Several factors could have influenced abdominal pain presentation or the patient’s susceptibility to epiploic appendagitis independent of tirzepatide exposure. These include baseline obesity and presumed visceral adiposity, gastrointestinal comorbidities such as inflammatory bowel syndrome, constipation, and bowel habit changes, reduced oral intake, and nausea during dose escalation. Activity changes and other triggers reported in the literature, such as strenuous exercise or hernia-related effects, could not be fully assessed from available clinical documentation. Accordingly, this report should be interpreted as a temporal association and hypothesis-generating observation rather than evidence of causality.
This case describes epiploic appendagitis occurring in temporal association with relatively substantial and rapid weight loss during tirzepatide therapy. Although epiploic appendagitis has not been identified as an adverse event signal in randomized trials, systematic reviews, or pharmacovigilance reports of GLP-1/GIP receptor agonists, this observation highlights a biologically plausible temporal relationship that may be related to abrupt changes in visceral adipose tissue and regional perfusion. Clinicians across specialties should consider epiploic appendagitis in the differential diagnosis of focal abdominal pain in patients experiencing rapid weight loss, particularly when imaging findings are consistent and systemic signs are limited. Increased awareness of this uncommon condition may help prevent misdiagnosis, avoid unnecessary interventions, and support appropriate conservative management as pharmacologic weight loss therapies continue to expand in clinical practice.
Epiploic appendagitis may be considered in the differential diagnosis of focal abdominal pain in patients undergoing substantial pharmacologic weight loss when vital signs are stable, and laboratory abnormalities are minimal. CT confirmation can prevent unnecessary antibiotics or surgical workup. When imaging supports epiploic appendagitis, treatment is generally conservative and typically managed with nonsteroidal anti-inflammatory drugs (NSAIDs) and supportive care, with resolution in 1–2 weeks. The patient reported symptomatic relief with prone positioning and improved tolerance of lightly seasoned, pureed foods and clear liquids during the acute phase. Tirzepatide continued during recovery and was only lowered afterward, and was later titrated up to 15 mg without recurrence. However, evidence is insufficient to estimate recurrence risk, and decisions regarding continuation or re-initiation of therapy should be individualized.
BMI: body mass index
CT: computed tomography
GIP: glucose-dependent insulinotropic polypeptide
GLP-1: glucagon-like peptide-1
BMA: Investigation, Data curation, Writing—original draft. JDG: Conceptualization, Supervision, Writing—review & editing, Formal analysis, Visualization. Both authors read and approved the submitted version.
Jennifer D. Goldman reports receiving honoraria for educational speaking engagements and advisory activities from Novo Nordisk and Lilly. These relationships are outside the scope of the present manuscript and unrelated to the clinical topic discussed. The companies had no role in the conception, preparation, review, or submission of this manuscript. The other author declares that there are no conflicts of interest.
Not required.
Written informed consent to participation in the study was obtained from the participant.
Written informed consent for publication of the de-identified clinical details in this case report was obtained from the patient.
The datasets that support the findings of this study are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 962
Download: 35
Times Cited: 0
