Affiliation:
1Endocrinology and Metabolism Division, Department of Medicine, The University of British Columbia, Vancouver, BC V5Z 1M9, Canada
2The Centre for Menstrual Cycle and Ovulation Research, Division of Endocrinology, The University of British Columbia, Vancouver, BC V5Z 1M9, Canada
3Women’s Health Research Institute, University of British Columbia, Vancouver, BC V6H 3N1, Canada
4School of Population and Public Health, University of British Columbia, Vancouver, BC V6T 1Z3, Canada
Email: Jerilynn.prior@ubc.ca
ORCID: https://orcid.org/0000-0003-3232-0597
Affiliation:
2The Centre for Menstrual Cycle and Ovulation Research, Division of Endocrinology, The University of British Columbia, Vancouver, BC V5Z 1M9, Canada
5Honourary Professor, Division of Endocrinology and Metabolism, Department of Medicine, University of British Columbia, Vancouver, BC V5Z 1M9, Canada
6Professor Emeritus, Department of Anthropology, Indiana University Bloomington, Bloomington, IN 47405-7100, USA
ORCID: https://orcid.org/0000-0002-6765-4557
Explor Endocr Metab Dis. 2026;3:101460 DOI: https://doi.org/10.37349/eemd.2026.101460
Received: November 15, 2025 Accepted: January 20, 2026 Published: February 25, 2026
Academic Editor: Marijn Speeckaert, Universitair Ziekenhuis Ghent, Belgium
The article belongs to the special issue The Fountain of Youth: Decoding the Hormonal Regulation of Aging
The dominant paradigm for healthy ageing in women+ (all genders) focuses on estrogen and sees the menopause, per se, as a major health problem (with low estrogen and progesterone levels). In reality, the risks for diseases that increase at older ages originate during the menstruating years. Rarely discussed evidence supports the central role of progesterone and normally ovulatory menstrual cycles in preventing early cardiovascular disease, fragility fractures, dementia, and cancers. Menstrual cycles with normal and predictable lengths but disturbed ovulation, including short luteal phases with lower progesterone production as well as anovulation without progesterone, likely occur in over 25% of all such cycles. These Subclinical Ovulatory Disturbances are usually an adaptive and protective response to physiological, sociocultural, or emotional stressors. Ovulatory disturbances and risks for health issues during ageing are intrinsically related to the social determinants of health—wholesome food, plentiful physical activity, strong communities, and access to timely and appropriate medical care. This review discusses the empirical evidence that normal ovulation and progesterone production during the premenopausal years lead to the prevention of early heart attacks and fragility fractures. Few studies document the effects of prevalent Subclinical Ovulatory Disturbances on brain issues (sleep, night sweats, ischemic strokes, pain, and addictions) and cancer risks. Serious gaps in women+’s fundamental reproductive physiology must be addressed with unbiased (population-based), rigorously collected longitudinal physiological, hormonal, and sociocultural data. Progesterone therapy during perimenopause and menopause also indirectly leads to healthy ageing through effective treatment of night sweats, hot flushes, and disturbed sleep, which are associated with cardiovascular problems and osteoporosis. Not only is progesterone effective for vasomotor symptoms in menopause, but also effective in perimenopause, a time of high and chaotic estrogen levels. In sum, strong summarized evidence suggests that progesterone and ovulation need further exploration for their important roles in promoting healthy ageing for women+.
The quest for eternal youth, like the search for the holy grail, is a fool’s errand. On the other hand, many people would likely choose to be healthy for more of the years of their lives (healthy ageing) than just to be alive for a greater number of years (longevity). Some suggest that for women+ (of all genders) to achieve healthy ageing, they need to somehow delay menopause (defined as one year without menstrual flow), thereby putting off reproductive ageing. That is clearly important, and women+ might well prefer a later age at menopause, but only provided that delay did not extend the hormonal and metabolic chaos of perimenopause.
Currently, any role progesterone—specifically and only referring to the ovarian hormone, not synthetic progestins, but not applying to the chemically identical therapy, oral micronized progesterone (OMP)—may play is hidden because ovulation is silent and unrecognized by a woman+ or those close to her [1]. In addition, gynecology persistently asserts (medical students are taught; all textbooks state) that normal-length, predictable menstrual cycles are ovulatory. Ovulation is also not, as presently taught, solely a present/absent function. Rather, ovulatory function is a graded, carefully tuned, and variable characteristic, with the practical integrating factor reflecting progesterone production being the luteal phase length (LPL) [2]. Thus, decades-long exposure to cyclic progesterone or, conversely, to silent or Subclinical Ovulatory Disturbances (SOD) is unrecognized. Also, if a published, scientific report states that the women studied were ovulatory without specifically stating the method used to determine if ovulation has occurred, there is a high likelihood that this assertion is based on inaccurate or insufficient testing. Ovulation and LPLs are key features of healthy menstrual cycling and yet are often ignored in attempts to understand women+’s reproductive system and the role of progesterone in healthy ageing. Fundamental aspects of this review are based on research that both co-authors have done to accurately document ovulation both cross-sectionally and prospectively within women+ over long durations and in different sociocultural and geographical environments. As active investigators for many decades, we are well qualified to discuss evidence that progesterone is a linchpin in healthy ageing. Over time, we have each documented the hormonal patterns and other features of ovulatory and anovulatory cycles in several different premenopausal cohorts [1, 3–5]; we have also both documented ovulation in population-based samples [6, 7].
The dominant, near-total focus on estrogen (E2) as women+’s key to eternal youth and sexual allure is deeply flawed. This “E2-model” is a culturally and economically driven preference rather than a scientifically well-grounded fact. There are still those, including the USA’s Food and Drug Administration (FDA), who argue that estrogen prevents menopausal women+’s dementia and heart disease despite strong scientific evidence to the contrary.
Some support for estrogen’s seemingly magical role likely arises because of misinterpretation of large convenience sample studies that showed decreased heart attacks and lower mortality in healthy, economically secure, usually White, and educated women who were able to afford, and sought menopausal hormone therapy (MHT). Although MHT was often a first treatment for vasomotor symptoms (VMS), sometimes MHT was prescribed because women+ believed that it would prevent the diseases of ageing [8].
The prevention-oriented USA Women’s Health Initiative (WHI)’s, two large, hormone randomized controlled trials (RCTs), proved that oral, horse-derived estrogen (alone or with daily progestin) did not prevent heart disease, dementia, or breast cancer. Study treatments did temporarily prevent fractures in asymptomatic menopausal women+. However, for two reasons, it is erroneous to credit estrogen for the healthier, longer lives in these community women+ taking estrogen-dominant MHT. The first is that, especially the early analyses, even of population-based cohorts such as the Framingham Heart Study, did not control for all important confounders. In this selected study sample, higher socioeconomic status, better education, and resulting positive social determinants of health (SoDH), themselves, are associated with lower mortality, increased longevity, and a longer reproductive life span [9]. The second reason is that treatment of VMS alone likely secondarily decreases the risk for heart attacks and hip fractures associated with night sweats and hot flushes that are left untreated. Thus, before we have valid scientific data on whether or not MHT is related to healthier ageing, we will need trials comparing samples of women taking and not taking MHT to be subdivided into those who seek treatment for VMS and sleep problems and those without these symptoms who nonetheless want MHT for non-medical reasons (e.g., youthful appearance). All analyses must be adjusted for confounders (education, ethnicity, anthropometric and lifestyle factors, as well as SoDH). The evidence for these complex but practical ideas will be presented in the respective heart and bone ageing sections of this review.
Estrogen (and testosterone for men) are the hormones usually associated with healthy reproductive lives, and thus healthy ageing [10].
This review of healthy ageing in women+ is based on what may be seen as a new but evidence-based understanding that progesterone is an essential partner of estrogen in the menstrual cycle system [11]. This concept is supported by early work, including multi-species analysis of reproductive tissues in response to estradiol and progesterone. The data showed that estrogen consistently acted to increase cell proliferation or growth [12]. By contrast, progesterone’s actions counteracted estrogen-stimulated proliferation while also increasing cell differentiation and maturation [12]. This innovative concept of estrogen-progesterone partnership means that thinking of estrogen as women+’s elixir of youth is fundamentally flawed—one must understand estrogen and progesterone in context, and together.
Progesterone levels during the post-ovulatory (LP) phase of the menstrual cycle are the only time (other than during pregnancy) when levels are greater than the universal-baseline human level (in men, menopause, and children). Although the LPL is widely assumed to be “fixed” at 14 days, it may last from 4 to 16 days. The best data suggest that a healthy LPL is 10 to 16 days (as assessed by the Quantitative Basal Temperature© [QBT©] method of Vollman [13] and Prior et al. [14]). Note that the normal LPL would become 13–19 days if using the urine luteinizing hormone (LH) surge in the so-called “ovulation test”. (The reason for the three-day longer LPL with the urine LH surge is that the QBT© requires a clear elevation of the basal temperature; it takes roughly a day after the LH peak before ovulation occurs, and a further couple of days after ovulation before progesterone importantly increases the first morning temperature).
Vollman [13] made the observation from which we infer that the normal LPL of 10 days is the essential minimum in QBT© analysis for balanced estradiol-progesterone. He noted that those with consistently shorter LPLs did not become or sustain pregnancies. This observation is supported by temporarily decreased fecundity in women with short LPLs [15], but awaits validation in a prospective, population-based sample of non-contracepting women+. This normal LPL, however, also has criterion validity because community-based prospective evidence documented that those with a mean shorter LPL during a one-year prospective study were losing significantly more cancellous spinal bone mineral density (BMD) than those with longer LPLs, and the mean LPL accounted for 23% of the total change in BMD variance [4]. There is also construct validity from an international meta-analysis supporting this observation [16].
The premise that progesterone exposure is a key component of women+’s healthy ageing, however, is strongly suggested by snippets of primary data on ovulation (present or absent) during the 25–50 years of menstruating life. It is also supported, as mentioned above, by the shorter LPL association with bone loss. However, there are exceedingly few studies of ovulation in population-based data. One important early study was in a nearly complete cohort of non-contracepting Bolivian women+ living in a high-altitude agricultural community [3, 17]. Another is a point prevalence serum progesterone-level-confirmed investigation in over 3,000 Norwegian women of whom ~1,500 were tested during the potential LP [7]. Results, as shown in Figure 1, were shocking—between approximately a quarter and a third of women did not have a serum progesterone ovulatory threshold value [7]. That means a serum progesterone level above 9.5 nmol/L.
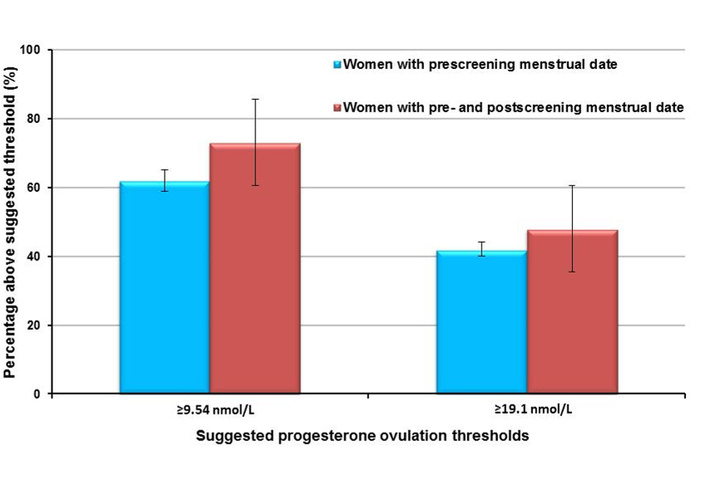
Percent of the premenstrual population with documented ovulation by two different ovulation threshold values. These results, from a population-based, regularly cycling (21–35 days cycle length) cohort of 1,545 women ages 20–49.9 in Nord Trondelag, Norway, show the percentage of cycle-timed (cycle day 14 and following until three days before the expected or known next flow) serum progesterone values above the 9.5 nmol/L and 19.1 nmol/L serum ovulatory threshold values. The blue bars indicate those providing only the date the last menstrual flow started, and the red bars indicate those who also provided the start date of the next flow [7]. Adapted from [7]. © 2015 Prior et al. Licensed under a CC BY.
The ovulatory threshold was proposed by the majority of experts in an informal survey. A progesterone level was assessed between cycle days 14 and 3 days before each woman+’s predicted cycle length (CL). As Figure 1 shows, a slightly higher percentage of women were ovulatory if they also had provided (as only about 10% did) the date their next flow started [7]. These data were difficult to publish [18].
Much more data in randomly sampled cohorts that have used valid methods to document ovulation and LPLs are necessary to scientifically confirm the progesterone-healthy-ageing hypothesis presented here.
Ovulation and normal cyclic endogenous progesterone levels, as already indicated, are not inevitable despite normal-length (21–35 days) and predictable menstrual cycles. An important aspect of women+’s reproductive physiology is that ovulation is adaptive [6, 17, 19, 20]; this is a protective function because it prevents fertility when it would be imprudent (for health or social reasons). Given the common association of relative energy insufficiency with hypothalamic reproductive suppression, and since progesterone requires additional energy to raise the core temperature, lower levels resulted in protective decreased energy requirements [21].
Menstrual cycles are variable in total CL, follicular phase (FP), and LPL during the menstruating years [22]. How much of this variability relates to the inverse association of LPL with an individual’s psychosocial and physiological stressors is currently unknown from unbiased, ideally population-based data. It is important to realize that when a woman is under threat, although progesterone levels and LPLs decline, serum estrogen levels appear to be protected—they remained at ~80% of ovulatory levels in women whose cycles did not achieve ovulatory status in the Norwegian population-based data from the study of Prior et al. [7].
The primary changes in menstrual cycles in those under duress, be it physiological (as in illness or over-exercise), nutritional (insufficient calories to meet energy expenditure needs), or social-emotional in characteristics, involve ovulation and progesterone, not CL and estrogen. Psychosocial stress, at least in the Western urban studies so far published, appears to be the most prevalent of “threats” that are related to adaptive suppression of ovulation. Evidence says that there is a suite of hypothalamic adaptive, reversible, and protective responses that are similar for all of these stresses—the most common change is shortening of the LPL [20]. Note that all these adaptive changes in ovulation will be invisible unless the clinician or scientist is specifically checking for such changes.
Prior [23] first learned the importance of social stressors related to negative cardiovascular effects from studies of subordinate female monkeys who had similar CL as their dominant female neighbors, and were not different in weight. The subordinate, stressed female monkeys, however, had significant ovulatory disturbances. Pathological analysis of the coronary arteries of all the monkeys after three years of a high-fat diet showed that the subordinate and dominant females had different coronary artery findings [23]. The dominant monkeys had clean arteries while the subordinate monkeys had lipid plaques and coronary artery atherosclerosis, not different from the vasectomized male monkey with whom they had shared a high-fat Western-type diet [23]. Progesterone “deficiency” in response to stress is a frequent and unrecognized risk for accelerated cardiovascular disease (CVD) and for early mortality.
Progesterone also appears to secondarily improve cardiovascular, bone, and probably malignancy-related health. This evidence rests on effective treatment of the most bothersome symptoms in perimenopause and menopause (VMS), meaning both night sweats and daytime hot flushes. OMP RCT data over three months showed it to be effective for VMS treatment in both perimenopause [24] and menopause [25].
In addition, OMP has a unique pharmacological “adverse effect”: it significantly improves deep sleep and overall sleep quality [26]. Sufficient sleep plays an important role in preserving healthy lives and functions. We will discuss progesterone’s therapeutic roles on VMS and sleep in the subsequent brain section.
Finally, progesterone may play currently unrecognized roles in the brain. Most brain research in women has focused on estradiol and, until recently, has primarily considered progesterone to cause adverse symptoms. It appears that progesterone and estrogen have opposite effects on the central interpretation of pain. Estradiol is associated with increased pain syndromes and central sensitization. Progesterone may play an opposing role in decreasing the central amplification of pain signals. This could aid in promoting healthy ageing.
In summary, estrogen is almost universally touted as promoting women’s healthy ageing, while its unique ovarian partner hormone, progesterone, is either not studied, ignored, or blamed for adverse effects [27]. Our purpose in this review is to cast some scientific light on progesterone’s unique roles in women+’s vitality, reproductive life span, and unique variables related to healthy ageing.
In addition, we will also outline basic, clinical, and epidemiological research that is still needed (as in the BOX—research needed to fill fundamental research gaps). This is an exploration of progesterone’s roles in the adaptive, protective reproductive system, and evidence that Cyclic Progesterone Therapy during the premenopausal years and daily progesterone treatment in symptomatic perimenopause and menopause could be considered as a clinical therapeutic tool in extending women+’s vibrant and productive lives.
Before we can discuss the increasing evidence that progesterone plays an important role in preventing heart attacks and maintaining a healthy cardiovascular system, we must deal with current concepts about women’s heart disease and estrogen. These have just been tweaked in what most scientists consider a non-scientific direction by the USA’s FDA. This agency just removed the “black box” warning about vaginal estrogen/estradiol (which is justified based on epidemiological data) and about the safety of systemic estrogen treatment (which is not).
Considerable press currently touts estrogen as preventing heart disease in women+ during menopause (starting after one-year without flow and normally in those ≥ 45 years old). This widely held belief is based on decades of pharmaceutical marketing from the 1940s until the mid-1970s that has persuaded healthcare providers and the public that menopause is an “estrogen deficiency” state rather than a normal life phase. Commercial hype further asserted that estrogen-only therapy would prevent heart disease caused by a lack of estrogen. One outcome of this campaign is that “hormone replacement therapy” became a popular and widely used term and is still so today, in both scientific publications [28] and common language.
For many decades, almost half of menopausal women in North America were prescribed the candy-coated red Premarin® pills (0.625 mg dose), a complex form of estrogen derived from pregnant mare’s urine. The notion that estrogen prevents everything bad about ageing, and a detailed treatment recipe, was promoted by zealots such as Dr. Robert Wilson, author of Feminine Forever, a book that was widely read starting in the 1960s. Although he always denied it, there is now firm evidence, as reported by his son Roland in the New York Times in 2002, that his book and outreach work were funded by the estrogen-producing Wyeth pharmaceutical firm.
Eventually, in 1975, epidemiological data showed marked increases in endometrial cancer (EC) in those women who used Premarin® as a sole menopausal treatment [29] for “estrogen deficiency”. This striking evidence of a serious cancer as an adverse effect of estrogen-only menopausal therapy was initially strongly contested [30]. Epidemiological experts doubted that estrogen could do such a thing; interestingly, one was Professor Alvin Feinstein, a physician, whose effort to make clinical diagnosis scientific was an inspiration to Prior as a medical student [31].
Although the FDA had approved Premarin® in 1941, no scientific evidence from RCT data was then available. USA and international feminists and scientists began demanding unbiased research investigations of the basic question. Does estrogen alone, or estrogen with progestin, prevent heart disease or osteoporosis, or cause breast or EC? (After the uterine cancer scare of 1975, progestin to prevent EC became required with estrogen in menopausal women who had not had a hysterectomy) [32].
The large, placebo-controlled USA prevention trials (the WHI) were designed and began to recruit in 1992, and eventually their protocol was published [33]. The WHI studies enrolled women from 40 USA centers, none of whom were currently taking hormone therapy (although close to 50% previously had), and all of whom lacked important hot flushes/flashes, night sweats, or other symptoms requiring treatment. In the WHI’s randomized controlled hormone therapy trials, the WHI gave estrogen (Premarin® 0.625 mg/day) treatment alone in ~11,000 women with hysterectomy/with or without ovariectomy studied over 6.8 years [34], and estrogen-progestin (Premarin® 0.625 mg plus Provera® 2.5 mg daily) treatment in ~17,000 women without hysterectomy treated with drug or placebo over 5.2 years [35]. These treatments were prescribed to the population of women currently receiving this therapy in the USA, including those many years into menopause. The mean age of participants was 63.3 years (SD 7.1), which was more than 10 years after the average age of menopause.
In July 2002, the WHI’s estrogen-progestin RCT arm was stopped early because results showed that it caused breast cancer [35], increased blood clotting, and other adverse effects, and did not prevent heart disease. What was not clear to the women+ or physicians reading about these results was that, by design, all of the menopausal women in these prevention trials were asymptomatic (not seeking treatment for VMS, sleep problems, or other menopause symptoms). Evidence of harm is a game-stopper for prevention trials [36].
It may surprise many readers that WHI was intended for prevention. In the last 20+ years of heated discussions about those huge WHI RCTs, rarely has mention been made that the results of these studies are virtually irrelevant for MHT as a treatment. The calculation of risk-benefit fundamentally changes when women+’s lives are disrupted by night sweats waking women+ twice or more times a week. WHI hormone RCTs were not treatment-of-symptom-nor-disease studies.
What is important are the most fundamental of WHI trial results—effects on mortality. These huge RCTs documented that neither the estrogen-alone nor the estrogen-progestin prevention RCTs improved mortality [34, 35]. That conclusion was reconfirmed after 18 years of follow-up of the entire ~28,000 women cohort—MHT caused no improvement in mortality versus placebo [37].
One of the many explanations for why WHI hormone trials were not successful was the “timing hypothesis”. This meant that the therapy had been started too late following menopause to be of its expected benefit. Despite the popularity of the “timing hypothesis”, careful analysis has also shown no significant decreased mortality in WHI participants who were younger than 60 years old, or less than 10 years into menopause [38, 39].
Many also argue that the estrogen (made from horses) and the progestin (rather than progesterone) in the WHI are the reasons it did not produce the expected cardiovascular prevention results. However, the Kronos Early Estrogen Prevention Study (KEEPS) was also a prevention RCT and tested physiological types of estrogen (transdermal estradiol 50 µg/day) and progesterone as cyclic OMP 200 mg at bedtime for 12 days/month compared with oral Premarin® (0.45 mg/day) and the same cyclic progesterone schedule each administered over 48 months. The KEEPS trials, however, had non-clinical outcomes such as changes in carotid intimal media thickness and the development of risk factors like high blood pressure and lipids rather than heart attacks or blood clots [40]. The KEEPS results showed, however, that the same increase in carotid intimal media thickness occurred on placebo as on those taking “bioidentical” hormone therapy (estradiol-progesterone). Thus, KEEPS provided further evidence that estrogen does not primarily prevent ageing of cardiovascular tissues [40].
Since these large RCTs in the USA women without symptoms did not show decreased heart disease nor improved mortality, how do we explain the beneficial effects reported in the many large convenience sample MHT treatment studies [41, 42]? For a start, most who ask for and achieve a prescription for MHT have a personal physician whom they can afford to pay. Plus, they are usually well-educated and often in upper socioeconomic strata. These women are also less likely to be obese (body mass index [BMI] ≥ 30), and given their positive SoDH, were already at decreased risk for CVD. Thus, they were initially at lower mortality because of non-medical variables and SoDH [43].
Importantly, evidence from the Study of Women Across the Nation (SWAN) showed that those with the most intense and persistent VMS had a higher rate of CVD than did those with mild or no symptoms [44]. Since there is strong RCT meta-analysis data that estrogen or estrogen-progestin therapies effectively treat hot flushes [45], that is another explanation for the decreased CVD in those in the convenience sample treated with estrogen or estrogen-progestin treatment. An analysis of the placebo-arm participants of the WHI also showed that the participants with the most intense VMS at baseline had an almost two-fold increased hip fracture risk [46]. Both arms of the WHI hormone RCTs showed that estrogen or estrogen-progestin prevented fractures [34, 35]. Although neither WHI hormone RCT arm showed cardiovascular protection [34, 35], the thought that it did has persisted. Thus, MHT was understood to prevent fracture and also heart disease in convenience samples, leading to the notion that estrogen or estrogen-progestin also caused healthy ageing. In summary, given that intense and untreated VMS are associated with both heart disease and osteoporotic fracture, and MHT is or is thought to be effective for both, it is not surprising that there is a general belief that estrogen prevents the major diseases of ageing.
With estrogen-cardiovascular system confusions now discussed, we can finally come to the issue that is of primary importance in a consideration of heart disease—i.e., the effects of progesterone that could improve healthy ageing. Before describing the large cohort study evidence that low endogenous progesterone is related to incident heart attacks, we will first discuss progesterone’s association with cardiovascular physiological changes—endothelial function, the fundamental control of blood flow through endogenous nitric oxide-release, as well as the length of the electrical QT interval on the electrocardiogram (ECG).
Endothelial function is of basic importance to the entire heart and blood vessel system [47]. Key to the concept that estrogen prevents heart disease is the evidence that estrogen improves endothelial function. But what does endothelial function mean? That endogenous nitric oxide from the vascular endothelium is released following constricted blood flow; there is greater release and improved flow in those at the least risk for CVD. Nitric oxide causes increased blood flow by increasing blood vessel diameter, which is called “flow-mediated dilatation (FMD)”. FMD was documented in a random sample from a large, prospective USA cohort, the Multi-Ethnic Study of Atherosclerosis (MESA). It included Chinese, Black, and Hispanic as well as White participants and had 50.2% women+ [48]. FMD significantly predicted cardiovascular mortality in both the unadjusted and multivariable-adjusted models [48]. This is the justification for various methods for testing endothelial function by venous occlusion plethysmography in the forearm or brachial ultrasound assessments of FMD.
In a random-ordered, within-person assessment of FMD by forearm venous occlusion plethysmography in healthy early menopausal women, Dr. Tom Elliott and others studied participants [49] after intra-brachial artery infusion of vehicle, physiological E2, physiological progesterone (P4), or both E2 and P4 levels. Results showed that progesterone was the only intervention that was significantly different from vehicle/placebo [49]. We had great difficulty getting that published (hence the strange title), because experts assumed that, by definition, the “estrogen deficiency of menopause” would cause abnormal FMD. But these healthy, early-in-menopause participants had normal baseline FMD values [49].
Progesterone and endothelial function, as assessed by FMD, are clearly controversial. Both studies in menopausal women [50] and in premenopausal women with endogenous hormones suppressed by gonadotropin-releasing hormone antagonists [51] show conflicting results with our above study [49]. With progesterone, they either show no change [50] or a negative FMD change [51]. However, most studies of progesterone have methodological problems, in particular, that they often investigate progestins (synthetic “knock-offs”) rather than progesterone. The study of Honisett et al. [50] in menopausal women used a lower-than-LP equivalent dose of progesterone (100 mg/day). The premenopausal study appeared to be of excellent design, but failed to recognize that gonadotropin-releasing hormone agonist treatment, itself, produced an unphysiological state. They did not assess nor take into account the likelihood that the higher cortisol and catecholamine levels, because of the acute onset of “medical menopause” with its rapid drop in endogenous estradiol levels, altered FMD. That is likely since higher cortisol levels, for example, in those with Cushing syndrome, showed significantly lower FMD than in matched controls [52]. That study assessed the chronic cortisol excess in both middle-aged women (n = 12) and men (n = 10) and compared FMD versus appropriate controls. Despite that, cortisol levels did not correlate with FMD [52].
Another cardiovascular influence of progesterone and estradiol is on the electrical system in the heart. The ECG records the rhythm and rate of the heart’s conduction. It also documents the height and duration of the QRS complex, the sinus node’s P wave, and the repolarization T wave. The interval from the Q to the T on the ECG, if too long, is called the “long QT syndrome”. It is known to be more common in women than men [53]. The long QT syndrome can lead to a chaotic and ineffective heartbeat (arrhythmia) and sometimes sudden death.
Although it is well known that congenital syndromes are associated with the long QT syndrome, as well as are a number of different drugs, few cardiologists or physicians in general know that estrogen lengthens the QT interval. The strongest evidence that gonadal steroids alter the QT interval comes from an experimental study in a strain of rabbits with a congenital long QT syndrome. These female or male animals were randomized to treatment with vehicle/placebo, E2, progesterone (P4), or dihydrotestosterone [54]. Over eight weeks of observation, of the seven E2-treated rabbits, five had serious arrhythmias, and two died a sudden cardiac death; of the nine P4-treated rabbits, none had a serious arrhythmia or died [54]. Note, however, that the dose of progesterone was likely quite high since it is described as “pregnancy levels”. The six male rabbits given dihydrotestosterone (the skin-active form of testosterone) also had no arrhythmias nor sudden deaths [54].
Despite these animal data and the sex differences in long QT syndrome in humans, few studies have examined the rate-corrected QT interval (QTc) for heart rate during the menstrual cycle. Studies would need to document the within-woman QTc during the mid-FP when only E2 is present at a moderate level, plus during the LP when P4 levels are high, and E2 levels continue to be moderate. Ideally, the QTc would be assessed without knowledge of cycle phase or ovulatory status. We conducted such a study with 62 women during a single cycle [55]. Because it was just following the major lockdowns during the SARS-CoV-2 pandemic, a larger-than-expected proportion of normal-length cycles were without ovulation. Also, the small number of normally ovulatory cycles required us to combine normally ovulatory and short LP cycles, thus decreasing any potential effect of progesterone on the QTc. The results showed only a small, non-significant decrease in QTc during the LP versus FP of ovulatory cycles [55]. It is of interest that, for the first time, QTc was also documented during the premenstrual phases of anovulatory cycles that have only ~20% lower E2 levels [7] and are lacking P4. Results showed that the anovulatory QTc tended to be longer than during the usual FP one [55].
In a three-month RCT of progesterone for hot flushes in menopausal women who were without baseline heart disease risks, progesterone caused no cardiovascular risk-related changes in weight, waist circumference, blood pressure, lipids, blood glucose, or risk for clotting [56].
Research studies of cardiovascular risks in a primate colony at Bowman Gray University in Georgia investigated several family-like groups of cynomolgus monkeys (one male with a vasectomy and five females) following three years of a Western-type high cholesterol and fat diet, with the primary outcome of coronary artery blood vessel pathology [57]. In the process of these experiments, investigators observed two social categories of female monkeys: those who were dominant and those who were subordinate. The dominant females got more grooming, were first to the food, and won any within-female arguments. The investigators put all the females daily through a cage-like device that allowed taking of basal temperature, a vaginal swab (to determine when the equivalent of a period started), and sometimes a blood test (always followed by a treat)! The subordinate females, although having similar cycle intervals and body weights as dominant females, were significantly less likely to have normal progesterone levels [23]. At autopsy, they also had the much larger adrenal glands (as evidence of higher cortisol levels and stress) [58]. The final results of these investigations showed that dominant females had maintained normal coronary artery vascular anatomy, but the subordinate females with more frequent ovulatory disturbances (based on lower progesterone levels) and higher cortisol (based on larger adrenals) had similar fatty streaks and thickening of the coronary artery vascular intima as the males [59]. A later study found that the subordinate female monkey’s coronary artery atherosclerosis still occurred on a low-fat diet [60]. Thus, in female cynomolgus monkeys, social stress and resulting lower progesterone (plus likely also higher cortisol) levels were related to coronary artery vascular disease.
Few studies have assessed premenopausal women’s risks for heart disease related to reproduction, ovulation, or progesterone. However, several epidemiological studies show that those with frequent first-trimester (< 12-week) miscarriages and stillbirths have a higher risk of coronary artery disease [61]. It is known that early pregnancy loss is associated with lower body weight (BMI < 20), lower serum progesterone levels (< 38.16 nmol/L), as well as increased age and higher perceived stress [62]. Thus, especially in the first trimester, recurrent miscarriages are likely related to inadequate ovulation and lower corpus luteum progesterone production.
However, the most important study addressing premenopausal hormone levels and subsequent cardiovascular risk was a prospective study. Netherlands cohort study that arose following its recruitment after the introduction of mammographic screening in women in their 40s [63]. Over 11,000 women ages 40–49 participated by collecting a first morning urine on cycle day 22 of three cycles in a row. Over the next 5–9 years, 45 women in catchment hospitals had acute myocardial infarctions or coronary artery surgeries [63]. Those with CVD (called cases) were matched three-to-one with others recruited around the same time in the cohort. The investigators found no difference between “cases” and controls in age at menopause. All case and control urinary hormones were analyzed. Estrogen and testosterone urine levels (corrected for creatinine) did not differ between the two groups. As shown in Figure 2, the women+ with early heart attacks versus controls had similar urine pregnanediol per creatinine levels [63]. However, those women with heart attacks had a significantly higher percentage of anovulatory menstrual cycles than did controls (29% versus 14%, P = 0.02) [63].
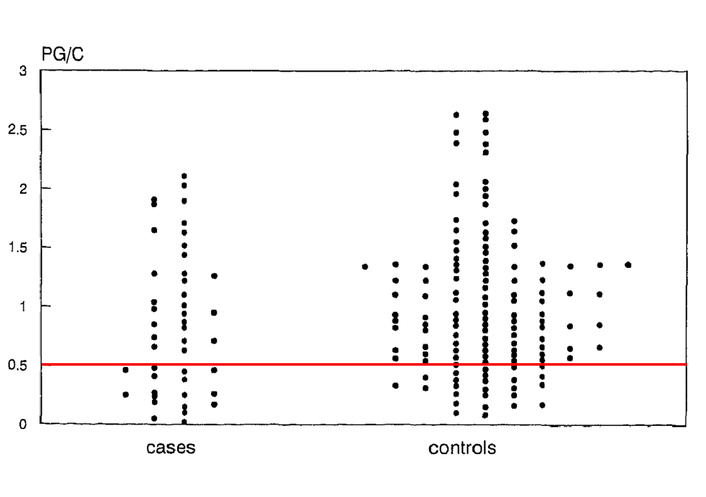
Assessing progesterone in cases with heart attacks versus controls. The first-morning urines collected on cycle day 22 in three successive cycles and assessed for pregnanediol glucuronide (PG)/creatinine (C) in those with heart attacks on the left (cases, n = 45) and controls (n = 135) on the right. This figure is modified by the horizontal red line indicating the authors’ designation of an ovulatory threshold [63]. Adapted with permission from [63]. © 1997 Published by Elsevier Inc.
The above cardiovascular results suggest that progesterone, as well as estradiol, sometimes work in parallel (on endothelial function) and sometimes with contrasting actions (on QTc), in their influences on the cardiovascular system. Evidence from cynomolgus monkey studies also suggests that the lower progesterone levels arise from social stress and directly influence atherosclerosis development in the coronary arteries. In summary, it is likely that premenopausal women who develop normally ovulatory cycles earlier and maintain them longer through the premenopausal years and into perimenopause would have a lower risk of heart attacks than women with more frequent SOD. Cardiovascular risks related to SOD are a major burden of disease in women related to early mortality and symptomatic or unhealthy aging [64].
Bone health during women’s reproductive adulthood requires both estrogen and progesterone. Maximum BMD is reached during the mid to late teens (for BMD at most measurement sites except spine, this is before the age of 20) [65]. This peak BMD is a measure of all the “construction material” that the woman+’s adult “bone remodeling system” has to work with for the remainder of her life.
The majority of physicians and the public assume that “estrogen deficiency” in menopause causes women to have more fractures than men. In fact, the reality of bone health is much more complex than that simplistic and inaccurate belief. Rather than focusing on a single hormone, we need to consider the whole of the bone biology system underlying the normal changes in bone throughout life and the several important variables that influence this complex bone remodeling system.
Estrogen has long been wrongly thought to “build bone”, but it importantly only decreases bone resorption or loss. Thus, anti-resorptive bone therapies such as bisphosphonates (such as risedronate, alendronate, or zoledronic acid) or denosumab treatment (an antibody to the Receptor Activator of Nuclear Factor kappa-B [RANK]-ligand that acts to cause bone resorption), have been the primary treatments for osteoporosis and widely used, especially in treating osteoporosis and fragility fractures in menopausal women+ [66].
Fundamental to bone biology and the bone remodeling system is understanding that its two main phases, resorption and formation, are tightly linked [67]. Resorption means the process of getting rid of old or diseased bone. It is also essential in providing necessary serum calcium (for example, during lactation). Bone formation means the process of laying down osteoid (the protein matrix of bone), which is subsequently calcified (or more accurately, mineralized). The coupling of resorption and formation means that when bone resorption decreases, bone formation is also decreased. This is a concept that few physicians or experts currently consider.
The result of this fundamental resorption-formation linkage in the two main bone remodeling processes is that a menopausal woman with fractures who has a yearly intravenous infusion of the bisphosphonate, zoledronic acid, will gain effective control of her likely rapid bone loss (a good thing) and show increased BMD (as resorption pits are filled and mineralized in an ongoing and passive process). But she will also have a decrease in new bone formation (not good). Since the dual energy X-ray absorptiometry (DXA) method of assessing BMD will show an increase, everyone is happy. But bone biopsy studies will not show an increased mineralization rate. Further, if she continues to have this bisphosphonate therapy for five or ten years, she is likely at increased risk of a new and different kind of rare fracture (called “atypical femoral fracture”). We will come back to what may cause such an odd fracture after we discuss further the role of progesterone in stimulating bone formation.
Estrogen is essential for healthy bones in both men and women [68, 69]. But in order to understand the relative and collaborative roles of both estradiol and progesterone, we must appreciate three things about bone physiology in women+ [69]: 1) estradiol and progesterone are part of a carefully controlled, adaptive bone remodeling system; 2) adult bone is maintained, lost or gained by the tightly linked processes of resorption of old bone and formation of new bone; 3) the timing of resorption and formation are imbalanced making bone resorption the most rapid and the dominant change. Bone resorption by osteoclast bone cells, especially in cancellous (honeycomb-like) bone present in the vertebrae, ribs, and at the ends of long bones, is a rapid process occurring over about three weeks [67]. This resorption takes place within multiple small bone multicellular units, the within-bone entity through which all bone remodeling takes place. By contrast, bone formation by osteoblasts is very slow. Thus, osteoblastic bone formation requires about 3–4 months for completion [67]; plus, over the years and decades, that new bone continues to gain an increase in its mineral content [69].
Estradiol (the dominant premenopausal form of estrogen) prevents bone resorption through complex actions of the RANK-system as well as by increasing gut calcium absorption. Estradiol’s main action (when it is at steady or increasing levels) is to decrease bone resorption. But paradoxically, if estrogen levels decrease, especially at a rapid rate, bone resorption increases. Prior first learned of this through a pivotal early study by Robert Lindsay [70] in which he studied the metacarpal BMD in 43 women who had undergone ovariectomy and hysterectomy. Estrogen-alone therapy prevented bone loss and increased BMD over four years. But when stopped and studied over a further four years without therapy, they lost all the bone they had gained during estrogen therapy [70]. Therefore, steady estrogen levels during the menstruating years should prevent bone resorption and loss. The issue occurs when estrogen levels rapidly drop, as in perimenopause, a paradoxical situation in which the average estrogen level is increased (over premenopausal levels) but bone loss is more rapid than in the first years of menopause [71].
In its most subtle form, because menstrual cycle estrogen levels decrease from the midcycle estrogen peak to the low during the next flow, there is a small increase in bone resorption each cycle [72]. This down-swinging estrogen and bone loss become a major issue in perimenopause when estradiol levels average higher [71] but are swinging wildly [73].
Progesterone, by contrast to estradiol, is the hormone of bone formation [74]. It directly stimulates osteoblasts that create the osteoid (protein foundation) or “matrix” of bone before it becomes mineralized (or calcified). Osteoblasts are long-lived, in contrast to the osteoclasts that disappear within weeks. Osteoblasts transform themselves to play three roles in bone—as the bone-forming cells already discussed, as the “lining cells” that are flatter osteoblasts in a tight row that protect the surfaces of bone within cancellous bone structures, and also get buried deep within bone as “osteocytes”. Osteocytes are former osteoblasts deep in bone that modulate bone remodeling by sensing mechanical and gravitational stresses on bone. Osteocytes at least partially do this by making the hormone sclerostin, which decreases bone formation. A student-led investigation of Prior’s discovered, however, that progesterone does not appear to interact directly with sclerostin levels in menopausal women [75].
That progesterone, as well as estradiol, was important in bone health was discovered by Prior and colleagues during a one-year, prospective observational study in 66 healthy women+. These were healthy, community women ages 21–42 who were screened to be of normal weight (BMI 18.5–24.9), non-smokers, and also required to have two consecutive normal-length (21–36 days) [76], and normally ovulatory (LPL ≥ 10 days) cycles confirmed before entry into the study [4] (Normal serum estradiol and progesterone levels across an ovulatory cycle have been documented in > 60 women by Landgren [77]). Notably, during this study, CL remained within the normal range in 97% of all cycles [4]. Despite maintenance of normal-length cycles, and although 19 of the 66 women trained for and ran a marathon during that study [78], yet were not different than the rest of the women+ in cycles or bone change, as a whole, these healthy young women+ were losing cancellous spinal BMD [4]. Also, surprisingly, given their careful screening, over a quarter of all cycles were without normal ovulation (mostly short LP [SLP] lengths, < 10 days, with a few anovulatory cycles) [4].
The whole-year ovulatory characteristic categories of these 66 women—e.g., the 13 with consistently normally ovulatory cycles of ≥ 10 days LPL, to any anovulatory cycles plus also SLP ones—are strongly related to the rate of cancellous spinal BMD loss as shown in Figure 3 [4].
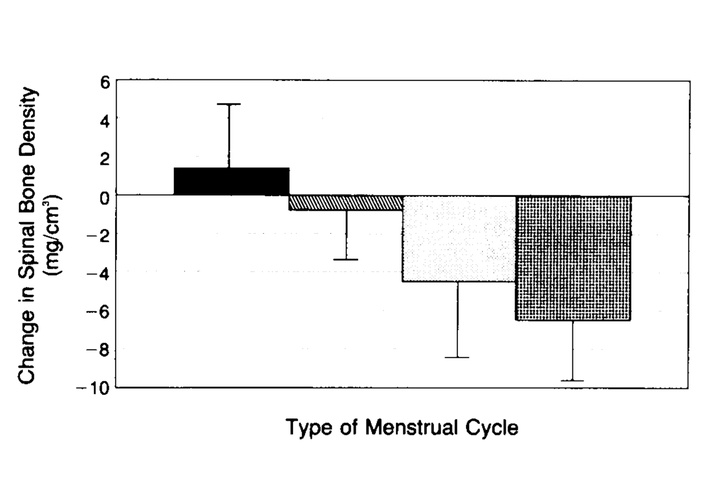
The one-year change in cancellous spinal bone mineral density was predicted by the whole-year ovulatory characteristics within normal-length cycles. BMD was assessed by quantitative computed tomography (Y axis). On the X axis are ovulatory characteristics data from the 66 healthy, initially normally ovulatory women within 97% normal-length menstrual cycles. Those 13 participants in the black bar had entirely normally ovulatory cycles (LPL ≥ 10 days); those 12 participants in the slant-hatched bar experienced one short luteal phase (< 10 days); those 28 in the white/lightly stippled bar had more than one short luteal phase cycle but no anovulatory cycles; and those 13 in the cross-hatched bar experienced at least one anovulatory cycle plus short LPL and normally ovulatory cycles (T = 5.37, P < 0.0001) [4]. LPL: luteal phase length. Reprinted with permission from [4]. © 1990 Massachusetts Medical Society.
These data clearly showed that both normal production of progesterone (as reflected by a ≥ 10-day LPL [2]) as well as usual estradiol levels (indicated by normal CL [77]) were needed to prevent cancellous spinal bone loss. Bone density in this investigation was measured by a highly reproducible (SD 1%) quantitative computed tomography (QCT) method in which a 3-dimensional ball in the center of cancellous bone was assessed within three lower spine vertebrae. At that time, the technology for today’s “bone density scan” had not yet been developed. In the analysis of these data, 23% of the variance in change in spinal BMD was accounted for by the proportion of the CL made up of the LPL as documented by QBT©. In a subsequent analysis, mean LPL alone accounted for 20% of BMD change [79]. Taking luteal length into account allowed analysis of the exercise characteristic of being a runner or not; these data showed a positive ovulation and exercise relationship to gain in QCT cancellous spinal bone over one-year [78]. Previously, exercise was thought to only cause increases in cortical BMD, the outer compact tissue of long bones.
That decreases in LPL were prevalent in healthy women was supported by a subsequent study in younger women led by nutrition and exercise scientist, Dr. Susan Barr [5]. These data integrated the stressful feelings that may be related to food choices (called cognitive dietary restraint) with ovulatory disturbances, both being related to loss of BMD by DXA, the current clinical “bone scan”. This was a two-year prospective observational study with over 100 university-aged women, which confirmed that SOD was related to spinal bone loss but also explained eight percent of cortical BMD loss in the total hip [5].
However, these observational studies can only show associations. Do we know from RCT that progesterone causes increased BMD? The answer is yes. Two studies, as explained below, provide these data. One was a one-year randomized placebo-controlled treatment trial (of the progestin, medroxyprogesterone acetate [MPA], that acts through the osteoblast progesterone receptor [79]) in normal-weight premenopausal women with disturbances of both cycles and ovulation [80]. The second was a meta-analysis of all available RCTs in menopausal women that had been randomized to estrogen alone or estrogen plus progesterone/MPA [81].
The premenopausal one-year RCT had a two-by-two factorial design with the primary outcome being one-year change in spinal BMD by DXA [80]. Since it was conducted before OMP became available in Canada, we used MPA instead, since it was known to act through the progesterone osteoblast receptor [74, 79]. We gave MPA cyclically (to “replace” the universally missing or inadequate LPs in the whole suite of adaptive protective hypothalamic reproductive suppressive changes [20]). We also gave an additional 1,000 mg of calcium (as calcium carbonate). MPA was administered as a 10 mg pill daily for 10 days at the end of the month or cycle. That study stratified 61 normal-weight (with a BMI of 18.5–24.9) premenopausal women 20–40 years old by their documented menstrual/ovulatory disturbances into four groups: hypothalamic amenorrhea (no period for six months), oligomenorrhea (cycles longer than 36 days but less than for six months), normal-length cycles with anovulation, normal cycles with SLP [80]. Participants were randomly assigned to four therapy groups: cyclic MPA with calcium; cyclic MPA with calcium placebo; placebo MPA with active calcium; and both placebos. Results showed a borderline positive effect of calcium (P = 0.07) but a highly significant BMD increase related to cyclic MPA (P < 0.0001). Interestingly, many of the women with ovulatory disturbances had (re)gained normally ovulatory cycles (as tested in the cycle after stopping experimental therapy) [80]. However, those with amenorrhea had the most positive BMD responses to cyclic MPA (because, during at least six months without any cyclic estradiol, they had developed a low rate of bone resorption). The practical result was that those on active MPA gained 1.7% of spinal BMD, calcium prevented significant spinal bone loss, while those on both placebos lost 2% of spinal BMD by DXA during that year [80].
Why did we not give estradiol as well as cyclic MPA to the women with amenorrhea and oligomenorrhea? Because clinical and basic science evidence says that estradiol (especially in higher doses such as in the birth control pill [combined hormonal contraceptives (CHC)]) impedes or prevents recovery of hypothalamic disturbances of cycles and ovulation [82]. Thus, in premenopausal women, there is strong evidence that progesterone (without estrogen) causes a gain in spinal BMD by increasing bone formation [80].
The evidence that progesterone causes bone formation in menopausal women+ is shown in a meta-analysis of RCTs that randomized them to daily estrogen alone or estrogen with cyclic MPA or cyclic OMP [81]. The primary outcome, again, was one-year change in spinal BMD by DXA. We found eight trials in 1,058 women+. As expected, estrogen alone increased BMD [81]. However, results showed that in the estrogen plus MPA/progesterone arm, there was a further and significantly greater gain (P = 0.00001) in spinal BMD than from estrogen-alone [81]. There were insufficient data to assess the hip BMD that had been measured in only a few of these trials. Thus, there is evidence that progesterone causes bone formation in estrogen-treated menopausal women.
Since normal estradiol levels are necessary (during the FP and the midcycle estrogen peak) before ovulation can occur, and, as a consequence of ovulation, progesterone is produced in the LP, it is further evidence that both estradiol and progesterone during the years from age 20 until menopause are needed to prevent fractures of osteoporosis during older ages. Recent prospective data using spine QCT also suggested that those perimenopausal women with higher or more frequent progesterone levels had less bone loss during perimenopause [83].
A meta-analysis of premenopausal one-year studies of cycles and ovulation has also documented that the net spinal BMD by DXA loss/year in those with more prevalent SOD was 0.86% yearly or almost one percent loss per premenopausal year [16]! Evidence in most of these studies was that SOD was related to social/emotional or cognitive dietary restraint stressors [16]. It is known that stressors increase cortisol and that treatment with medications that act similarly to cortisol, such as prednisone are related to bone loss [84] and fractures [85]. It turns out that cortisol (or a similar compound in experimental rodents) both increases bone resorption that causes BMD loss, as well as interferes with osteoblastic bone formation. Cortisol competes with progesterone for the progesterone osteoblast receptor [86].
In premenopausal women with hypothalamic disturbances of cycles and ovulation, does BMD loss occur (in those with amenorrhea/oligomenorrhea) because of a lack of normal estrogen levels? Or is it related to ovulatory disturbances (with losses of normal levels and durations of progesterone) that are always present in someone with cycle disturbances? Alternatively, could it be primarily because of the higher cortisol, which is also present in and related to the hypothalamic cycle-length and ovulatory disturbances that cause the BMD loss? It is likely a combination of all of these processes. Without sufficient estrogen, more cortisol will be in the protein-unbound and thus active state, and thus more effectively acting. We know that progesterone displaces cortisol from the progesterone osteoblast receptor [86]. Thus, if progesterone levels are lower, higher cortisol will be more likely to paralyze osteoblastic new bone formation.
Prospective studies show that very rapid spinal bone is gained around the time of puberty [87]. However, it is clinically common for adolescent women to be “treated” with “the Pill” or CHC for problems such as acne, irregular periods, heavy flow, or premenstrual moodiness [88, 89]. What, if any, is the evidence that CHC use is related to adolescent peak bone mass? CHC conventionally includes ethinyl estradiol, a synthetic and long-lasting type of estrogen that is about four times as powerful as physiological 17-beta estradiol. CHC also always includes one of many different progestins. During investigations of the relationships between BMD and oral contraception in the Canadian Multicentre Osteoporosis Study, Prior discovered that those who had ever used CHC (for three months or longer) had slightly but significantly lower BMD values [90]. With the later recruitment of 16–24-year-old women, she examined the rate of BMD change over two years by use of not of CHC [91]. There was a trend toward less positive bone gain that led to an exploration of the literature for other similar prospective studies. It turned out that there were four other studies examining the two-year change in BMD by CHC use or not in 885 young women+ [92]. As shown in Figure 4, in a meta-analysis after systematic review of five international studies, two-year results documented that teen-aged women+ who took CHC experienced significantly less bone gain than did those who did not (P = 0.0006) [92].
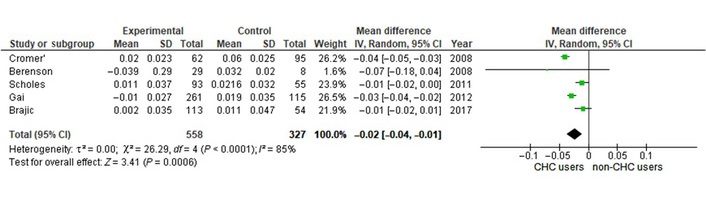
This forest plot shows that those adolescents using CHC have a 2-year weighted mean loss of spinal BMD compared to adolescents not taking CHC. These random effects meta-analysis of five international observational studies tracked spinal BMD changes over two years in adolescents by those who took CHC (called “experimental”, n = 558) or who did not (controls, n = 327) [92]. Note that there is considerable heterogeneity as expressed by the I2 value of 85%. BMD: bone mineral density; CHC: combined hormonal contraceptives. Reprinted with permission from [92]. © 2019 The Authors.
These were astonishing results! Why? It is clear that CHC (including oral, vaginal ring, and patch formulations) contains high levels of estrogen; even in the lowest doses available, these are equivalent to about four times physiological levels. CHC also contains likely luteal-phase-similar levels of progestins. How, then, could CHC be related to significantly negative adolescent spinal BMD changes [92]? To answer, we first have to understand what is happening in adolescent bone.
Rather than the adult bone remodeling system, teenagers are gaining bone density as well as bone length (called “modeling”). It is during this period that they reach their peak bone mass as documented prospectively in population-based data [65]. The best explanation is that CHC’s relatively high dose of estrogen prevents the within-bone activities of resorption and formation that are necessary for bone modeling. Said another way, in the presence of CHC, adolescent bone is not “free” to gain its normal dramatic increases [87].
It is also of importance that several studies suggest that adolescence is a time of increased “sensitivity” and thus risk for depression in the teens who are prescribed CHC for various reasons [93–95].
Although most believe that bone loss is rapid because of “estrogen-deficiency” in menopause (when women have been without a period for a year), the reality is that spinal BMD loss by DXA is greater during later perimenopause (when women have still menstruated within the year), and estradiol levels are significantly higher than in premenopausal women [71]. Also, in a meta-analysis of spinal BMD change in the last three years of perimenopause compared with the first three in menopause, there was a significantly greater rate of later perimenopausal than early menopause bone loss (–1.8%/year versus –1.2%/year) [71]. Estradiol levels are not only higher on average in perimenopause [71, 96] but much more variable [73]. As we learned earlier, rapid loss or dropping levels of estrogen produced dramatic increases in bone resorption and hence bone loss. Despite this, early evidence has suggested that the level of spine BMD that a woman has before the rapid bone loss in the last years of perimenopause is related to her subsequent risk for fracture [97].
In addition, perimenopause is a time when progesterone levels (even in ovulatory cycles) are decreasing [98]. Recent evidence has suggested that preservation of endogenous progesterone longer during perimenopause was associated with less bone loss [83].
Thus, across the life cycle, women+’s bone health requires both estradiol and progesterone to gain peak bone mass, preserve BMD during the menstruating years, protect as much as possible of bone during perimenopause, and thus prevent the fractures and osteoporosis that increase with older ages and with frailty [99].
When discussing the brain and healthy ageing, dementia and strokes are the first things that come to mind. We will start by outlining what little is known about each of these major diseases for older women+ before going on to talk about some key brain physiology in which progesterone plays a central role. These physiological functions involve core temperature, respiration, sleep, and VMS, as well as pain and addictions.
It has long been believed that estrogen prevents women’s dementia. However, there are few data on the effects of progesterone on cognitive abilities. What is known is that, in a randomized, placebo-controlled trial, estrogen alone (oral conjugated equine estrogen [CEE], 0.625 mg/day) or estrogen with MPA (2.5 mg/day) in the WHI memory study failed to improve dementia risks [100]. Even more troubling, these experimental treatments worsened cognitive function compared with the placebo [100]. Nonetheless, it is rightly asserted that this risk should not prevent women with premature menopause (before age 40) or early menopause (before age 45) from receiving MHT as a true replacement for their early end to ovarian production of estradiol and progesterone. Since the WHI results, two population-based studies using health administration data have confirmed more prevalent dementia in those who are using or have used MHT [101, 102].
There is little scientific evidence that progesterone either impairs or improves cognitive function. One random-ordered, double-blind study gave 400 mg of OMP or placebo to premenopausal women during the early FP [103]. This study used magnetic resonance imaging and noted that women+ performed less well on cognitive tests, especially memory for faces, when they had taken active OMP. These results were ascribed to adverse cognitive effects because of the increase in the progesterone metabolite, allopregnanolone [103]. Rather, poor test results are likely because this high dose of progesterone during the daytime caused major drowsiness and decreased alertness secondary to progesterone’s known beneficial effects on deep sleep (which we will discuss in detail later in this section).
By contrast to that inappropriate randomized cross-over placebo-controlled study of a single daytime high OMP dose followed immediately by cognitive testing, an appropriate study of bedtime OMP (300 mg daily) and cognition was performed in menopausal women by Schüssler et al. [26]. After the evidence that sleep was improved by the progesterone therapy, this morning wide battery of cognitive tests on day 21 of therapy showed that the within-woman there were also no negative cognitive changes following OMP therapy compared with placebo [26].
Is progesterone related to the risk of ischemic stroke? This is a question that has not been tested in humans but has been well-studied in experimental animals. A hypothesis article in Neuroscientist in 2009 outlined the potential experimentally known ways in which progesterone may be beneficial in the brain [104]. These included mechanisms to decrease edema, oxidative stress, inflammation, and excitotoxicity while increasing cellular repair. Further studies included individual animal data but showed some indication of increased death in the progesterone arm [105]. A subsequent min-review of progesterone for ischemic stroke following the failure of progesterone therapy in two multicentre RCTs to treat human traumatic brain injury, however, cast light on the yet-unresolved translational problems of progesterone for ischemic stroke, including interactions of progesterone with the tissue plasminogen activator treatment, sex-specific effects, need for multicentre pre-clinical trials, and matching designs for pre-clinical and clinical studies. All of these still need solving before progesterone could be tried for its potential neuroprotection following strokes in humans [106].
A USA feminist psychologist was the first to report, in 1934, vaginal changes in association with the core basal temperature increases during the menstrual cycle, as had been documented earlier in Germany [107]. Dr. Seward [108] noted that vaginal smear changes typical of what we now know as the LP occurred when the temperature was higher. Early on, it was correctly assumed that this temperature-increasing effect during the cycle occurred through interactions between women’s ovarian function and the brain [109]. Only later, with identification and chemical characterization of ovarian steroids, were the post-ovulatory LP and progesterone levels associated with this small but consistent rectal first-morning (so-called “basal”) temperature increase (0.2–0.3°C) [13].
Estradiol may at least partially block progesterone’s temperature-increasing activity [110], consistent with the menstrual cycle system’s counterbalancing effects of estradiol and progesterone. This estrogen effect may cause the basal temperature nadir around the time of peak menstrual cycle estradiol. Thus, progesterone’s action on the pre-optic area of the hypothalamus increases core temperature. However, this progesterone action effect likely has no direct effect on healthy aging. An indirect effect of this temperature increase related to higher progesterone levels is the requirement for an additional 300 kCal/day in energy intake to maintain a stable weight [21]. It could be speculated that this physiological increased energy requirement may be helpful in preventing the development of overweight and obesity.
The specificity of progesterone’s core temperature-raising action has historically led to many attempts to detect ovulation and control fertility using temperature-monitoring methods (which must be done upon awakening, before activity or eating, and only in someone who is not ill or having a fever). For many decades, those seeking to follow Catholic birth control and fertility instructions used a standardized education system related to oral temperatures and observations of cervical mucus that was initially called the “Billings Method” (in recognition of early champions of the method). It is now one of the methods under the umbrella of Natural Family Planning. Basal body temperature, however assessed (e.g., finger ring, vaginal, and other sensor-facilitated methods), needs to be biologically and statistically assessed (i.e., detection of ovulation) with a method that is valid and reliable. Unfortunately, multiple strategies use various analytical methods (most of which are a “black box”); few, if any, have been validated by the gold standard for ovulation since there is no such requirement. Ovulation documentation requires ultrasound identification of the day of dominant follicle collapse and hence ovum release. At-home, non-invasive, community women-appropriate, and quantitative methods to document the LPL as well as the presence or absence of ovulation, were first proposed by Rudolph Vollman [111], a Swiss ob-gyn physician. He later published his mean temperature quantitative method and the most extensive longitudinal documentation of menstrual cycles and ovulation (although requiring daily rectal temperature measurements) [13].
From this historically important work, Prior and colleagues [14] were able to develop a new quantitative method for assessment of first morning temperature data, the Least Squares Method©. Prior validated the new QBT© method, and also Vollman’s Mean Temperature Method© versus blindly assessed daily measurement of the serum LH peak [14]. Although these QBT© methods have not yet been validated by ovarian ultrasound, they, along with the daily Menstrual Cycle Diary©, have provided data that predict BMD loss and reliably indicate changes in caloric intakes and cardiac repolarization (QTc), suggesting context validation. In short, QBT© has made it feasible to assess the importance of ovulation, LPL, and progesterone levels for women+’s health, although it also requires considerable investment by the individual woman+.
Sleep is a normal function with restorative properties for humans. Normal sleep patterns and durations have important physiological and psychological benefits for human beings. However, at present, there are no strong data linking healthy aging to restorative/restful sleep that is sufficiently long (> 6 h) and not too long (> 9 h) [112]. An epidemiological study from the 1980s related sleep disturbances to a marked increased risk for depression and other emotion-related issues [113]. In the one-year follow-up, those with initially documented as having “insomnia” had a 40% (95% confidence interval [CI] 19.8–80) higher likelihood of developing new, major depression [113]. In addition, a normal 8 to 9 h of sleep in young nulliparous women during and after pregnancy was necessary to prevent the development of insulin resistance, abnormal lipids, obesity, and other unhealthy cardiovascular risks called the “metabolic syndrome” [114].
OMP, as an exogenous therapy in a dose of 300 mg at bedtime daily, decreases sleep latency, improves deep sleep (slow wave), and prevents sleep disturbances (waking after sleep onset) as has repeatedly been shown in randomized placebo-controlled trials. The first of these trials, surprisingly, was a polysomnographic study in healthy young men [115]. This investigation showed that “deep sleep” (with slow wave characteristics) was significantly improved by progesterone and associated with the gamma-aminobutyric acid (GABA)-A system in the brain and increased levels of allopregnanolone, a metabolite of progesterone [115].
The bulk of RCT evidence for improved sleep related to progesterone therapy comes from menopausal women [26, 116]. The study of Schüssler et al. [26] demonstrated typical progesterone-related increases in deep sleep and also countered the criticism that progesterone therapy causes morning drowsiness and decreased cognition (as previously discussed). Further studies showed decreased sleep disturbances when women were taking progesterone rather than a placebo, again in a polysomnographic sleep study [116].
We will shortly discuss improvement in sleep in healthy menopausal women who were randomized to progesterone or placebo in the treatment of hot flushes and night sweats [25]. What had previously been undocumented was the benefit of progesterone for deep sleep in perimenopausal women. Prior and colleagues [24] showed this, for the first time, in their 2023 RCT of daily progesterone (300 mg at bedtime) for three months in 189 perimenopausal women being treated for night sweats, waking them at least twice a week, and hot flushes. Figure 5 shows the sleep quality results in this RCT in perimenopausal women with problematic night sweats.
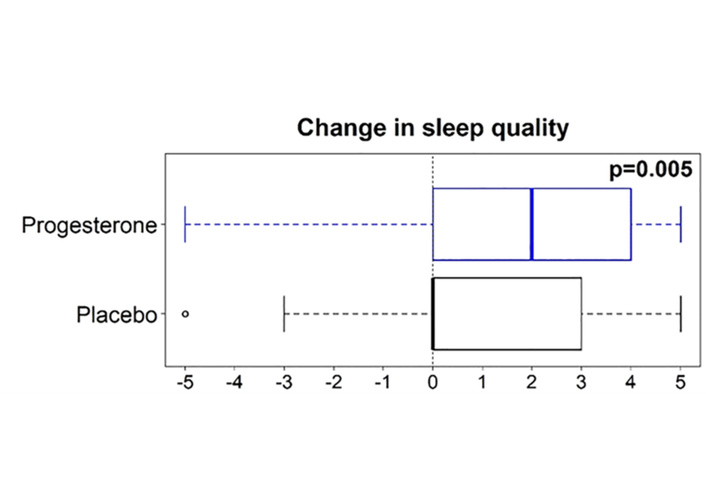
Progesterone therapy improves sleep quality in perimenopausal women+ with VMS. At the end of a three-month RCT of oral micronized progesterone (300 mg at bedtime daily) or an identical placebo for perimenopausal VMS, those on progesterone (blue, n = 90) versus those on placebo (black, n = 86) reported a significant improvement in sleep quality. Participants scored the question about a 3-month change in sleep quality on a –5 to +5 scale, with the median represented by the solid vertical line and data analyzed by Wilcoxon rank sum testing [24]. RCT: randomized controlled trial; VMS: vasomotor symptoms. Reprinted from [24]. © 2023 The Author(s). Licensed under a CC BY.
Those two studies will be discussed in more detail with VMS below.
Although oral progesterone treatment improves deep sleep, the mechanism is unclear. Why? Because RCT evidence shows that vaginal, transdermal, or buccal progesterone administrations do not alter sleep. Some recent evidence surprisingly suggests that this sleep-improving effect of oral progesterone is facilitated by changes within the gut microbiome [117].
It is currently unknown whether endogenous progesterone improves deep sleep during pregnancy or the menstrual cycle’s LP. New data show that the wearable sensor, the Oura Ring®, has had its sleep function-related results validated by the sleep gold standard, polysomnography. The Oura ring shows sleep-wake accuracy of 96% and also the ability to document the four sleep stages [118]. This study, which included evaluation of 106 people from adolescence to midlife, included 65 women+, tested over 3,000 h, versus polysomnography. The Oura ring detected deep sleep, although with more data scatter than other sleep phases; the accuracy was 94%, sensitivity 75%, and specificity 95% [118]. The Oura ring has recently been used to longitudinally track sleep during pregnancy in 17 community-dwelling women in the USA of Hispanic and/or White ethnicity [119]. Results showed that sleep was increasingly disturbed the closer a woman was to childbirth. In addition, there was a significant decrease in deep sleep as the pregnancy progressed, and especially in the third trimester [119]. That doesn’t fit with a positive role for progesterone since progesterone levels, although high throughout pregnancy, are highest in the third trimester. We cannot rule out that progesterone could be preventing even worse sleep disturbances, but this is impossible to scientifically test since pregnancy maintenance requires high progesterone levels.
Although there have been some small studies of menstrual cycle phases and sleep [120, 121], to our knowledge, none have shown sleep cycle phase-specific changes. All studies have problems, including documentation of ovulation and especially having insufficient power since studies included only 13 and six participants, respectively [120, 121]. Centre for Menstrual Cycle and Ovulation Research (CeMCOR) scientists are currently studying perceived sleep quality longitudinally in premenopausal women. This research is examining daily “sleep disturbance” scores in almost 700 Menstrual Cycle Diaries© in 53 nominally healthy and initially normally cycling and ovulatory premenopausal community-dwelling women studied over a mean of 13 cycles each.
Thus, in summary, restorative sleep of appropriate duration is important for general health as well as for psychological well-being, but its association with healthy ageing is currently unclear. OMP therapy has pharmacological effects to decrease sleep latency and sleep disturbances while increasing deep sleep. This has now been shown in RCT data in men, menopausal, and perimenopausal women. It is unclear if physiological, endogenous progesterone, during the LP and pregnancy, has sleep-promoting effects. It remains unknown, largely because, until recently, objective sleep stage measures were not applicable to community-dwelling women. Now, with Oura Ring® technology, accurate sleep documentation is possible, and thus, random-sample, prospective studies are potentially possible to answer these unanswered questions.
Respiratory function is also largely controlled by the brain through sensing carbon dioxide and oxygen levels. It is well documented that progesterone increases respiration through many mechanisms, most clearly shown in studies in pregnancy [122]. Disordered sleep breathing and sleep apnea, however, are beyond the scope of this review.
Currently, the major indication for MHT should be problematic hot flushes/flashes and night sweats. Although these often-dramatic temperature-metabolic and adrenergic experience changes are documented worldwide in midlife and older women [123], they also may occur in men. In men, they are usually due to dramatic decreases in testosterone levels, interpreted in the brain through lower catechol estrogen levels. This usually occurs because of orchidectomy for testicular or prostate cancer or androgen ablation treatment in the latter situation. Night sweats, although similar to daytime hot flushes, have recently been found to be more physiology-based and less influenced by the environment and other variables than are daytime hot flushes [124]. VMS are clearly disturbing for some and a nuisance for many more, but their etiology remains quite mysterious.
Although VMS are largely framed as “estrogen deficiency symptoms”, an early (1989) estrogen implant therapy clinical observational study was revealing [125]. A prominent group of UK gynecologists treated 12 menopausal women+ with estradiol implants (50 mg) with the expectation that these treatments would provide sufficiently high estradiol levels that they would be without VMS for at least six months. Instead, these women+ returned within a mean of 8 weeks with “estrogen deficiency symptoms (flushes, sweats, mood swings, and irritability)” [125]. These puzzled clinicians therefore measured serum estradiol when each symptomatic woman returned; results showed a mean estradiol level of 2,153 pmol/L (where the usual midcycle estradiol peak was 1,200 pmol/L) [125]. The authors ascribed these women’s symptoms to estrogen “tachyphylaxis”, that is, a phenomenon associated with addiction. This link between VMS and addiction may be apt, as discussed later.
However, the best evidence suggests the cause for VMS is not “estrogen deficiency”, nor static and low levels of estradiol, but rather dynamic estradiol downswings from levels to which the brain had become accustomed [126]. In a similar phenomenon as the brain response to dropping glucose levels, it “sees” estradiol downswings as an emergency. Multiple studies show dramatic release of myriad hormones and neurotransmitters, especially sympathetic discharges (central norepinephrine) that coincide with the galvanic skin response and women’s subjective experience of a hot flush [127, 128]. Casper and Yen [129] hypothesized that ovarian steroid withdrawal (likely estradiol, not progesterone) triggers opioidergic release, setting off a chain reaction with both hormonal and physiological responses associated with women’s VMS experience changes. VMS understanding, from a decade of human experiments, is best summarized in a review by Dr. Robert Freedman [130].
The strong association of daytime hot flushes with situational and environmental stress is an important aspect of VMS physiology that is only now beginning to be clinically recognized. Studies in perimenopause from the USA SWAN showed that those with the worst VMS were those who reported “difficulty paying for the basics” of housing and food [131]. One counselling psychologist went so far as to state in her book, Menopause and Emotions [132], that all of the symptoms of menopause were caused by elevated stress hormone levels.
The Freedman body of research and 2014 review emphasize that the down-swing in estradiol leads to increased central norepinephrine release [130]. That is an explanation for the important role of situational stress in triggering hot flushes. This association of environmental stress with daytime hot flushes was confirmed by an elegant experiment [133]. In this study, 21 community-dwelling menopausal women reporting more than eight hot flushes during the daytime were randomized to two randomly ordered study sessions. They had a button to push when they felt a hot flush starting (subjective VMS), and were monitored continuously for galvanic skin responses (objective VMS) during each four-hour session. In one session, women were invited to a large, quiet, and homey room with low lighting, which made it easy to chat and enjoy the company; but in the second session, which was a loud environment with bright lights, they were forced to watch a violent video. Results showed no report bias, but women had more VMS during the stressful session [133]. The importance of psychosocial stress related to daytime hot flushes may explain the continuation of daytime VMS (predominantly) into menopause when estradiol levels are no longer wildly down-swinging [134].
Because VMS, especially night sweats [135], are such a major, although brief, destabilizing brain event leading to dramatic release of multiple neurotransmittors and downstream stress hormones (such as norepinephrine and cortisol), it is not surprising, as mentioned earlier, that those with the most intense and persistent VMS are more likely to have subsequent CVD events [136]. In over 3,000 USA women ages 45–52, in the SWAN, most in perimenopause, and including Chinese, Japanese, Black, and White ethnicities who underwent 16 follow-up visits over 22 years, VMS were assessed by the number of days a week on which they occurred. Those with VMS on more than six days/week were compared with those with fewer days, and the incidence of CVD events (myocardial infarction, stroke, revascularization, and heart failure) was examined. Results showed that the hazard ratio (HR) for CVD was 1.51 (1.05–2.17), which just achieved significance (P = 0.03) [136]. However, if the persistence of VMS (occurring on more than a third of all follow-up evaluations versus fewer) was examined, VMS had a strong association with subsequent CVD events, HR [95% CI], 1.77 [1.33–2.35], P < 0.0001. These data are more remarkable because they are after adjustment for medications (including MHT), demographics, and traditional CVD risk factors [136].
Likewise, given that estradiol downswings are associated with surges in RANK-ligand, which increase bone resorption, VMS are also associated with bone loss [46]. In an eight-year prospective examination of hip fracture risk, the menopausal women (ages 50–79) in the WHI clinical trials (over 23,000), who were not treated with hormone therapy, were examined, comparing those with baseline moderate/severe VMS versus those with none. Hip fractures were increased (HR 1.78 (1.20–2.64) P < 0.01) [46]. Those who had BMD measured also had significantly lower final values at both the total hip and the spine [46]. Thus, more intense and longer-lasting VMS are associated with both a 50–80% increased risk for CVD in older age and an almost two-fold increased likelihood of a hip fracture. Both hip fracture and CVD are associated with decreased life expectancy and/or inhibit healthy aging.
In this context, effective and safe treatment of problematic VMS in menopausal and perimenopausal women is essential for general health and likely healthy ageing. That is especially true of treatment of night sweats that are associated with elevations in blood pressure [135], disturbed sleep [137], and higher levels of central norepinephrine. There is no doubt that estrogen therapy improves VMS, with estrogen-progestin having a significantly greater benefit than estrogen alone [45] because MPA is effective VMS treatment in RCT data [138]. There is also no question that stopping estrogen-dominant MHT is associated with a rebound increase in VMS [139] that is clinically important for at least a third of treated women. That experience is entirely congruent with the dropping-estrogen neurophysiology previously described.
That progestins, especially MPA, are effective for the treatment of VMS has been known for decades [138]. However, the only RCT of which we are aware, of OMP (300 mg at bedtime for 12 weeks after a 4-week untreated run-in) as sole therapy (i.e., without estrogen) for problematic VMS in menopausal women+ is one by Hitchcock and Prior [25]. This research was funded by donations to the Centre for Menstrual Cycle and Ovulation Research with arms-length provision of progesterone (OMP) and identical placebo capsules by their original maker, Besins Healthcare International. This trial enrolled 133 healthy local community-dwelling women+ ages 44–62 years and within 10 years of menopause whose VMS were problematic. That meant symptoms ranging from waking twice a week with night sweats to “severe VMS” as defined by the USA’s FDA, meaning ≥ 50 VMS of moderate-severe-intensity/week [25]. This trial was also pre-planned to test changes in cardiovascular risks; all 133 menopausal participants had low heart attack risks at baseline. All women kept Daily Menopause Diary© records throughout the run-in and the 12-week blinded trial while taking active progesterone or an identical placebo. Daytime hot flushes and night sweats were recorded daily as the absolute number of episodes and their average subjective intensity on a 0–4 scale [25].
Results of the RCT of progesterone for VMS in menopausal women+ showed a significant reduction in the intensity and frequency of both day (hot flushes) and night (sweats) VMS, although their decrease on placebo was also significant [25]. There were no serious adverse events, and progesterone was well tolerated. Importantly, “sleep disturbances” were also significantly improved on progesterone compared with placebo [25].
In post-hoc analyses in the same menopause VMS progesterone trial, we asked two further questions: 1) Did “severe VMS” respond the same or differently as average VMS to progesterone treatment? 2) Did suddenly stopping progesterone cause a rebound increase in VMS? The sub-study in the 46 women+ with Severe VMS intensity separately analyzed the number and intensity of VMS change versus placebo. The results showed even greater reduction in VMS compared with the main trial in those 46 participants with “severe VMS” [140].
To answer the second question, we invited participants at the end of the trial to continue Daily Menopause Diary© recording for a further month. Investigators were blinded to randomization but participants could have known what they had been taking (because at trial end a therapy-information letter was sent to her by the pharmacy managing the trial). The 34 women+ who agreed to this sub-study were similar to all in the RCT. Results showed that suddenly stopping progesterone caused no rebound increase in VMS [140]. The 17 who recorded Daily Menopause Diary® data during the month off, who had stopped placebo, had a minimal increase in VMS that became non-significantly different from run-in within a week. By contrast, although VMS gradually increased in those who discontinued progesterone, night sweats and hot flushes were still about twenty percent less than at the beginning of the study despite being off treatment for four weeks [140]. Thus, progesterone markedly differs from estradiol; progesterone withdrawal does not provoke a rebound increase in VMS.
Another progesterone-only treatment RCT in women with problematic VMS was designed to study perimenopausal women+. We knew from previous observational work that night sweats were common in perimenopause and often were cyclic around flow in midlife women whose menstruation remained regular [141]. No previous perimenopause-only trial had been conducted, but because it was known that VMS were more variable than in menopausal women, the trial was planned to include about 20% more participants. This trial was funded by a peer-reviewed grant from the Canadian Institutes for Health Research, with progesterone and identical placebo capsules again provided (arms-length) by Besins Healthcare International [24].
This RCT eventually enrolled 189 participants, the majority of whom participated remotely (facilitated by secure, signed courier management of experimental therapy). Again, the primary outcome was VMS Score from the Daily Perimenopause Diary©, which integrated the sum of each night’s sweat and hot flush mean number X intensity in the final month versus the run-in month. The primary outcome did not reach statistical significance, but a VMS score of 3 was a pre-stated, clinically important change. The 95% CI included 4, thus this trial could not exclude a clinically important benefit [24]. This RCT had important secondary outcomes assessed by a questionnaire asking about trial-related perceived changes in hot flushes, night sweats, sleep quality, and flow. This used a Likert scale in which participants could score change as none, worse (–5), or better (+5) (Figure 5). These results showed only significantly improved daytime VMS intensity, but significant overall night sweat improvement (intensity and frequency) versus placebo. In addition, sleep quality was markedly improved, and women perceived no change in flow [24]. In addition, we were requested by the Data Safety Monitoring Committee to screen all women with the PHQ-9 clinical test of depression [142]. There was no statistical difference in change in PHQ-9 across the study by therapy, thus progesterone did not increase depression, even in perimenopausal women+ who have increased susceptibility to this major mood issue [143].
Thus, in summary, evidence indicates that progesterone was significantly more effective for hot flushes and night sweats in menopause than was a placebo [25]. Progesterone was well-tolerated, safe, and with no serious adverse events. Furthermore, progesterone’s effectiveness increased the more intense the VMS [140]. Finally, there was no withdrawal rebound increase in VMS when progesterone was abruptly discontinued [140], a marked contrast to the evidence with menopausal estrogen therapy withdrawal [139]. In addition, progesterone was effective and well tolerated by perimenopausal women in the first and only RCT for VMS for this life phase [24]. Despite the high variability of VMS in perimenopause and recruiting insufficient numbers for statistical power by the primary outcome, this trial showed significantly improved night sweats and sleep without perceived changes in menstrual flow. In effectively treating VMS and sleep problems, and decreasing the catecholamine and glucocorticoid excesses, secondary positive outcomes for subsequent CVD and osteoporosis are likely (based on the literature we have previously reviewed).
Chronic pain shows sex differences because it is more prevalent in women than in men, in addition to the above brain function discussions [144, 145]. Some evidence suggests that estrogen enhances pain and that progesterone, and normal ovulation, may counterbalance that and/or prevent central sensitization [146] and the progression to a chronic pain syndrome. Most of all, few studies of pain and its pathophysiology have been done; much more research is needed.
Addiction is another important brain-related function. We made reference earlier to the rapid increase in neurotransmitters, stress hormones, and cytokines following a drop in estradiol levels. The clinical correlate of this is an estrogen withdrawal rebound increase in VMS, often to worse symptoms than previously experienced [139]. This resembles the responses in addiction, suggesting that VMS are an estrogen-addiction withdrawal symptom [147]. This withdrawal phenomenon is mediated through the brain opiomelanocortin system. Withdrawal from full-dose progesterone, however, does not produce a rebound increase in VMS but rather a slow and longer-than-4-week gradual increase [140]. Some evidence, which we can only briefly summarize here, suggests that progesterone decreases the addiction response and may act as an inhibitor of the central opioid responses leading to this addiction-related [148–150].
Brain function is a frontier for the investigation of the counterbalancing and cooperative roles of progesterone and estradiol within women’s reproductive system [11].
Estradiol (the premenopausal form of estrogen) and progesterone together form the women+’s reproductive system as mentioned previously [11]. Each of these two unique ovarian hormones interacts with the other in the production of healthy, life cycle-specific menstrual cycles over three to five decades of women+’s lives. Estradiol is a powerful growth stimulator that requires progesterone to counterbalance it while also facilitating cell and tissue maturation [12]. Taken at face value, proliferation leads to potential cell-division-related DNA errors and increased malignancy risk. In the early days of discovery about causes for heart attack (1950–1970s), the assumption was that women had fewer and later ages at heart attacks than men. Based on that information, the Coronary Drug Project was designed and conducted; it was an RCT to determine whether estrogen therapy (as CEE in high doses of 2.5 and 5.0 mg versus 0.625 mg for women) prevented men’s increased heart attack risk [151]. It was a totally unsuccessful study with major and serious adverse effects [151]. Although there were fewer smokers in the 2.5 mg estrogen group during the trial, their rate of lung cancer was observed to be increased. The trial was ended with a non-significant malignancy risk-related results, and researchers were committed by the funder to evaluate the risk of cancer after some years of follow-up. A publication as a letter in 1978, almost a decade after that poorly conceived study was discontinued, reported increased rates of all types of malignancies in men who had taken 2.5 mg of CEE for 56 months versus those treated with a placebo [152]. At 84 months post-study end, the cancer rate was over 5.2% in those who formerly took CEE versus 3.2% in those who were on placebo [152]. These data illustrate the potential malignancy risk related to estrogen’s cell-proliferative actions.
In this section, we will briefly cover what is known about women+’s most common gynecological cancer, that of the endometrium, before lightly skimming over breast and other cancers related to women+’s complex reproductive system and with implications for healthy ageing.
EC, cancer of the uterine lining, affects 3% of women+ and is the only gynecological cancer whose incidence is increasing worldwide [153, 154]. That EC was increased by estrogen-only treatment in “intact” (i.e., without a hysterectomy) menopausal women was a starkly jarring revelation in 1975 [29]. It was discovered that EC had a high prevalence in menopausal women who were treated with CEE as a sole therapy. That association, despite being contested, led to guidance that all MHT in those with an intact uterus must include progesterone or progestin. Caution suggests that the dose of progesterone match the estrogen dose, and that the duration of cyclic progesterone be longer than 10 days [155, 156].
The majority of EC are hormonally responsive (endometrioid classification) and effectively “cured” by hysterectomy if they are discovered early and before they have spread beyond the uterus. However, few community women and front-line healthcare workers currently understand the risk factors for EC [157]. In addition, because the only EC “screening” tool is an endometrial biopsy, which is expensive, invasive, uncomfortable, or very painful, there are few systematic, prospective investigations of EC risk factors. To make risk evaluation more difficult, the endogenous menstrual cycle equivalent in premenopausal women+ is anovulation in normal-length, predictable menstrual cycles. But ovulation is silent. Over one year of prospective monitoring, 26% of highly screened healthy, normal-weight premenopausal women at low risk for ovulatory disturbances maintained regular cycles but had fairly frequent SLP, and some even experienced anovulation [4]. Thus, EC’s likely primary premenopausal risk factor, SOD, is invisible because, by definition, such “progesterone-deficient” cycles are normal-length, and predictable (aka “regular”).
EC is specifically and markedly increased in polycystic ovary syndrome (PCOS), a premenopausal condition that usually presents in adolescence, and is characterized by irregular and far-apart cycles, ovulatory disturbances, and androgen excess [158]. PCOS is a risk factor for insulin resistance and type 2 diabetes mellitus (T2DM), which are EC risk factors, as is obesity. EC is increased by 5 to 17-fold in those with PCOS [159, 160], in a systematic review and in a population-based study in Taiwan (China), respectively. Recently, the Azziz laboratory estimated that the annual costs for treating EC in PCOS were almost half a million dollars a year in the USA [161].
What makes detection more difficult is that most EC risks are non-specific in nature, such as increased insulin resistance and obesity. The main pre- and perimenopausal indicator of risk for EC is “abnormal uterine bleeding” (AUB), meaning changes in the duration, timing, and/or amount of menstrual flow. AUB is universal in perimenopause, a time when increased EC is likely, given the much higher estradiol and the declining progesterone levels. Furthermore, 25% of perimenopausal women in a population-based Canadian study who sought healthcare had heavy flow and cramping as the reason [162].
Risks for EC are surprisingly also non-specific in menopausal women. Asymptomatic menopausal bleeding (silent uterine bleeding in women > 40 years of age who have been without flow for at least a year) is a non-specific sign, and indicates EC in a small percentage of women tested by endometrial biopsy, and in only 4.3% of hysterectomy specimens at pathology [163]. So, again, there is a natural reluctance in women+ to have such an invasive test as an endometrial biopsy performed, especially if the healthcare provider shares with her an accurate likelihood of the diagnosis of EC.
To make matters worse, about 10% of menopausal women will normally have a further flow as documented in a prospective cohort [164] and confirmed in a population-based investigation [165]. In Prior’s clinical experience, EC can be excluded if menopausal women+ with such further flow volunteer (or respond to an open-ended question) that they felt different before it started, typically with bloating, breast tenderness, or “mood swings”. Thus, this “postmenopausal bleeding” is not truly asymptomatic, is often a one-time occurrence, and usually occurs within three years of the final menstrual flow.
EC incidence is related to higher BMI, which may explain its increasing prevalence. Estimates from a huge UK family practice database (population-based) suggested that 41% of EC was related to obesity [166].
Progesterone or progestins alone have been used to treat early-stage endometrioid EC [156]. However, better yet would be a reliable, inexpensive, and simple way to detect premenopausal ovulation to determine whether the LPL and progesterone production are sufficient to support pregnancy and to prevent EC in women with regular menstrual cycles.
The cause for ovarian cancer, this difficult-to-diagnose and deadly women+’s malignancy, used to be considered “incessant ovulation”. However, as mentioned, ovulation is silent within normal-length cycles [1], so how would authors of these studies know if ovulation (rather than simply menstrual cycling) was “incessant”? Also, making that hypothesis improbable is the long-term follow-up of the WHI RCT data. In 2024 follow-up of CEE-alone versus placebo showed a significantly increased risk for ovarian cancer (HR 2.04) and mortality (HR 2.79) [167]. However, there was no increased risk for ovarian cancer in the WHI arm treated with CEE plus MPA [167].
There is finally some hope for ovarian cancer prevention, given the strong pathophysiologic evidence that ovarian cancer originates in the fimbria of the fallopian tubes [168]. Evidence is beginning to suggest a benefit from “opportunistic salpingectomy”, i.e., surgically removing rather than relying on clamping or other methods of tubal sterilization, plus if a woman’s family is complete, removing the tubes when abdominal surgery is required for appendectomy or most other reasons [169].
As the most prevalent of women’s cancers (although lung cancer is more deadly), it is of grave concern that an increasing percentage of women are being diagnosed with breast cancer. The risks in women with benign breast disease include greater breast density [170], older age at diagnosis, nulliparity, and a family history of breast cancer [171].
Given the much greater risk for breast cancer in women than men, what about the roles of the ovarian hormones, progesterone and estradiol? The breast is a more complex tissue than the endometrium. One example is that breast tissue itself makes estrogen. Many assert that estrogen is not related to breast cancer risk, yet all of the major breast cancer treatments include methods to decrease estradiol: by aromatase inhibitors, suppression of the reproductive system through treatment with gonadotropin-releasing hormone agonists or antagonists, or bilateral ovariectomy, to give a few examples. The literature is also confused because rodent models poorly reflect human physiology, and further because, even in scientific publications, synthetic progestins, that may increase breast cancer as does MPA are commonly mistaken for progesterone and vice versa [12]!
The WHI CEE + MPA arm showed an increased rate of breast cancer [35] versus a nonsignificant result in the WHI estrogen-only trial [34]. Some experts, based on the follow-up data, even tout that CEE alone “prevents breast cancer”. On examining the WHI hormonal and observational study protocols [33], it turns out that, to have 75% statistical power to show a breast cancer effect, the CEE-only WHI arm would have needed to study 25,000 women; they only enrolled about 11,000 [34]. It has also been learned that MPA in breast tissues does not act through the progesterone receptor, as it does in bone, but rather has its breast cancer effects through the proliferation-inducing glucocorticoid receptor [172].
Despite the persistent, insistent defenses of estrogen related to breast cancer [173] and sometimes blaming of progesterone [174], evidence continues to indicate that progesterone (but not the various synthetic progestins) may be preventative. These data include the basic science investigation of breast cancer tissue showing that, in the presence of progesterone, the estrogen receptor alpha (ERα) no longer has proliferative effects [175]. The strongest evidence was from the large eight-year prospective French teacher’s insurance investigation, Étude Épidémiologique auprès de femmes de la Mutuelle Générale de l'Éducation Nationale, called “E3N” for short, in menopausal women+ and their risk for breast cancer related to MHT use and type or not [176]. This study, in more than 80,000 menopausal women+, examined the breast cancer risk of those who used hormone therapies of different sorts versus those who were untreated, who served as controls. Results showed that estradiol treatment alone resulted in a significant 1.29 increase in the Relative Risk (RR) for breast cancer; those using estradiol with various progestins had a 1.69 RR for increased breast cancer risk [176]. However, in those who took estradiol with progesterone, the RR was 1.00 (95% CI 0.83, 1.22), suggesting that the estradiol risk was prevented by progesterone treatment [176].
Despite these data, and protests against blaming progesterone for breast cancer [177], no published study to our knowledge has yet tested progesterone for prevention in women+ at high breast cancer risk, nor as an early treatment. However, a recent pragmatic, short-term trial was performed with diagnostic tissue in women with early estrogen-receptor positive invasive breast cancer prior to definitive surgery or other therapy [178]. The primary outcome was the proliferation marker Ki67 in this short, less than two-week trial, which showed that megestrol, a progestin that acts through the breast progesterone receptor, significantly added to the effect of an aromatase inhibitor to decrease breast cell proliferation [178].
It is clear that progesterone is likely preventative for endometrial and perhaps also ovarian and breast cancers. But much more basic science, physiological, clinical, and RCT clinical trial evidence is needed in the area of malignancy, progesterone, and healthy ageing.
The questions these reviewed and diverse data about the effects of progesterone on healthy cardiovascular, skeletal, brain, and malignancy-related ageing will never be conclusively answered without tools and data from these three fundamental trials.
There are several potential chemicals specifically stimulated by the elevated progesterone levels created following ovulation, and that have no other major influences, yet have a duration of action, meaning they may be measurable in the next FP. Since the average/usual gynecologically healthy community woman not seeking pregnancy is aware of her menstrual cycle primarily during flow, the test must be conducted during the first week following the start of menstruation. This test, which we will call the “Test of Normal Ovulation” (ToNO), will have a significantly lower level in a short or insufficient length from a normal-length LP, and an absent level in those with anovulation. It must be validated against the ovulation gold standard, collapse of the dominant follicle, and compared with serum progesterone a week later, in a sufficiently powered trial that uses the duration from follicle collapse to flow to indicate the LPL.
This second fundamental research study is a prospective, population-based investigation of reproductive maturation starting with 1,000 peri-pubertal girls ages 9–10 and following them until age 20, documenting markers of maturation (i.e., breast and areolar sizes) and menarche, CL, and the onset of ovulation and its development to normal LPLs using ToNO. This cohort could then be followed using health administrative data in the Province of British Columbia, given that every person has a unique personal health number. These data, for example, could better describe the origins or pathophysiology of menstrual cramps, PCOS, and endometriosis.
This third and final study will be of randomly sampled adult premenopausal women+ (of all genders), ages 20–45, in 5-year age cohorts from four communities, each with 1,500 women. A comprehensive baseline questionnaire will document all appropriate ethnicity/race, medical, family, and reproductive histories as well as physical activity, diet and SoDH. A baseline physical examination will measure height, weight, waist circumference, blood pressure, and, in a nested random cohort, endothelial function. These women will document menstrual cycles with the Menstrual Cycle Diary© and perform ToNO testing monthly, plus answer annual postage-paid or online questionnaires (about new health issues, pregnancies, lactation, hospitalization, etc.), forms to complete for another year. All will be followed with administrative data by personal health number until death (with documented, adjudicated cause) or age 90.
Consistent observational prospective and point prevalence epidemiological data suggest that ovulation is not inevitable despite menstrual cycles that are regular (i.e., normal-length and predictable). Ovulation and progesterone production are the “undiscovered continent” in the exploration of women’s health related to successful ageing. Therefore, the fundamental research outlined in the BOX is essential. In the meantime, use of the easier methods to document basal temperature, such as the Oura Ring®, could have data analyzed with QBT© despite the need for this method to be further validated versus the collapse of the dominant follicle.
Sufficient evidence now suggests that Cyclic Progesterone Therapy (300 mg at bedtime cycle days 14–27, or for 14 days on and 14 off for those with cycle disturbances) without further needed trials could become the “go-to” therapy for adolescents struggling with problematic menstrual cramps, heavy flow, irregular periods, and acne. Progesterone is likely much safer and more physiological than “non-contraceptive” use of CHC. Why? Because Cyclic Progesterone is a physiological therapy, but CHC has been shown in meta-analysis to be harmful for adolescent peak bone mass, plus related to the risk for serious mood disturbances (major depression) when taken during the teen years, as discussed earlier in the bone section.
RCT evidence shows that Cyclic Progesterone Therapy (based on the MPA surrogate) will be positive for bone in those with the suite of hypothalamic disturbances of menstrual cycles and ovulation. Again, as described earlier in the bone section, OMP given in a “LP replacement” way (300 mg at bedtime cycle days 14–27) is more likely to be effective at improving BMD and also safer than the alternative, CHC, or high-dose estrogen for hypothalamic amenorrhea.
PCOS is a condition one of whose likely causes is a hormonal imbalance in the brain: overly rapid LH pulsing leading to androgen excess and anovulation as discussing the malignancy section. Cyclic Progesterone Therapy, both based on physiology and in feasibility evidence, seems to “work” in PCOS. New Phase II 6-month evidence in androgenic PCOS, so far reported only in abstract [179], showed that cyclic progesterone with spironolactone (an anti-androgen) is feasible based on a six-month open-label trial with highly positive PCOS-specific quality of life benefits. Results documented that participants were happy taking it, would like to continue it, and that it had no serious side effects. Cyclic Progesterone Therapy for PCOS, despite the current lack of placebo-controlled or CHC-comparative blinded trial data, can currently be trialed on a “study of one” basis for three to six months within a patient-physician partnership in deciding its person-specific effectiveness or not.
OMP (in a dose of 300 mg at bedtime daily) would ideally be the first menopausal VMS treatment if employing criteria for both effectiveness and safety. In placebo-controlled, three-month RCT data, daily bedtime progesterone is effective for VMS in menopause and significantly improves sleep.
Importantly, in the often-difficult-to-treat life phase of perimenopause, RCT data showed that progesterone effectively treated night sweats, markedly improved disturbed sleep, and also allowed participants randomized to it to experience significantly decreased “perimenopausal interference with activities of daily life”. Further, RCT data showed that progesterone did not cause an increase in perimenopausal depression, which is at higher risk during this time.
In either perimenopause or menopause, if vaginal dryness remains problematic, vaginal estrogen or dehydroepiandrosterone (DHEA) is the advocated and safe treatment. Systemic estrogen therapy will almost always be unnecessary.
Would cyclic progesterone—in women with PCOS, endometriosis, or other ovulation or cycle disturbances—help prevent CVD? Likely, based on the large prospective cohort study showing the association of low and anovulatory urinary progesterone levels with heart attacks in women in their 50s. This remains to be proved in RCT data. In addition, progesterone therapy had no negative effects on cardiovascular risk factors in a 3-month RCT in healthy menopausal women+. That information, combined with evidence that progesterone improved endothelial function, suggests it would decrease CVDs that are such an important part of symptomatic, diseased ageing.
Cyclic progesterone for those with disturbed cycles during the premenopausal years and into perimenopause would also likely prevent EC, and possibly ovarian and breast cancers. Much more focused, yet multidimensional research is needed.
In addition, evidence reviewed in the cardiovascular section showed that Silent or SOD within clinically normal menstrual cycles is related to cardiovascular issues, including early-in-menopause heart attacks. A woman in her 30s with a family history and/or personal risk factors for CVD should likely also be screened for ovulatory disturbances. The simplest way for this in a clinical setting is ordering a serum progesterone level on cycle day 22 in two cycles. If one or both progesterone levels are not ≥ 9.5 nmol/L (or ≥ 3 ng/dL), she should be treated with Cyclic Progesterone Therapy while she and her clinician work to decrease the potential causes (most commonly social stressors) of SOD. The same is likely true for women with regular cycles who have been assessed to have Unexplained Infertility, recurrent early pregnancy loss, or any stillbirths, all of which appear to have their origins in SOD.
Most importantly, women and clinicians need to recognize that untreated symptomatic perimenopause and menopause appear to carry increased risks for heart attacks and hip fractures. Current evidence suggests that these risks will be decreased or eliminated with effective vasomotor symptom and sleep disturbance treatment. The safest effective VMS and sleep therapy is with daily OMP (300 mg at bedtime), which improves deep sleep, prevents sleep disruptions, and treats night sweats in both menopausal and perimenopausal women+.
Related to bone health, not only does Cyclic Progesterone Therapy (through progesterone-receptor-acting MPA) significantly increase BMD in premenopausal women+ with hypothalamic suppression of reproduction (from amenorrhea to regular cycles with SLP), it likely adds to the fracture-preventing benefit of standard, anti-resorptive osteoporosis therapy in menopausal women+.
Menopausal women+ at increased fracture risk (as documented by a nationally appropriate fracture risk assessment tool (FRAX)) are typically first given an anti-resorptive therapy (such as a bisphosphonate or denosumab) as recommended by osteoporosis guidelines. However, as previously discussed in the bone section, since resorption and formation are tightly linked, suppressing resorption will also suppress new bone formation. Many of these menopausal women+ are highly symptomatic with both night sweats and sleep disturbances, as well as having high life stressors and other fracture risks. The combination of an anti-resorptive medication with daily OMP will likely increase BMD (and also likely decrease fracture risk) more than the anti-resorptive osteoporosis drug alone, while further treating important, risk-disturbing symptoms (VMS and sleep).
This review also showed that symptomatic menopause and perimenopause likely put women+ at increased risk for rapid bone loss/hip fractures and CVDs. Thus, there is not only a symptom indication to treat with OMP (300 mg at bedtime daily) but an additional rationale for also potentially improving bone and cardiovascular health. This hypothesis still needs testing in an RCT, although such a placebo-controlled trial may not have equipoise, or be able to effectively recruit, knowing what we now know. Since progesterone increases bone formation through the osteoblast progesterone receptor, this additional therapy will likely cause a greater improvement in BMD as previously shown in the meta-analysis of RCTs of menopausal women+ randomized to estrogen alone or estrogen with cyclic progesterone/cyclic MPA.
In short, both women+ and clinicians need to become aware that estrogen, although a powerful growth factor and absolutely necessary during the premenopausal years, requires the complementary and counterbalancing effects of normal LPLs with healthy cyclic progesterone levels to cause reproductive vitality and promote healthy ageing.
AUB: abnormal uterine bleeding
BMD: bone mineral density
BMI: body mass index
CEE: conjugated equine estrogen
CHC: combined hormonal contraceptives
CI: confidence interval
CL: cycle length
CVD: cardiovascular disease
DXA: dual energy X-ray absorptiometry
EC: endometrial cancer
ECG: electrocardiogram
FDA: Food and Drug Administration
FMD: flow-mediated dilatation
FP: follicular phase
KEEPS: Kronos Early Estrogen Prevention Study
LH: luteinizing hormone
LPL: luteal phase length
MHT: menopausal hormone therapy
MPA: medroxyprogesterone acetate
OMP: oral micronized progesterone
PCOS: polycystic ovary syndrome
QBT: Quantitative Basal Temperature
QCT: quantitative computed tomography
QTc: rate-corrected QT interval
RCTs: randomized controlled trials
SLP: short luteal phase
SOD: Subclinical Ovulatory Disturbances
SoDH: social determinants of health
SWAN: Study of Women Across the Nation
ToNO: Test of Normal Ovulation
VMS: vasomotor symptoms
WHI: Women’s Health Initiative
We appreciate the value of Dharani Kalidasan in assisting with reference finding and obtaining permissions for and refining the Figures in this review. We thank Yvette M. Vigna for her patient and careful management of Prior’s EndNote programme.
JCP: Conceptualization, Writing—original draft, Writing—review & editing. VJV: Writing—review & editing. Both authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Support for this work is provided by donations through the Endocrinology Division of the University of British Columbia (UBC) to the Centre for Menstrual Cycle and Ovulation Research. Additional support for the work of Dharani Kalidasan, MSc, was provided by the UBC Department of Medicine. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 4910
Download: 128
Times Cited: 0
Amar Mann ... Sudarshan Ramachandran
Pravinath Ramachandran ... Geoffrey Hackett
Eugenie Macfarlane ... Markus Joachim Seibel
Carter Coggins ... Vikrant Rai
