Affiliation:
1Interventional Ultrasound Unit, Pineta Grande Hospital, 81030 Castel Volturno (CE), Italy
Email: giampierofrancica@gmail.com
ORCID: https://orcid.org/0000-0002-9821-7966
Affiliation:
2Department of Obesity Surgery, Pineta Grande Hospital, 81030 Castel Volturno (CE), Italy
ORCID: https://orcid.org/0000-0002-9097-0451
Explor Dig Dis. 2026;5:1005114 DOI: https://doi.org/10.37349/edd.2026.1005114
Received: November 29, 2025 Accepted: February 23, 2026 Published: March 05, 2026
Academic Editor: Nahum Méndez-Sánchez, National Autonomous University of Mexico, Mexico
The article belongs to the special issue Advances in Hepato-gastroenterology: Diagnosis, Prognostication, and Disease Stratification
In the context of the various management options available for obesity, intragastric balloons (IGBs) represent a solution for patients at high surgical risk. Swallowable IGBs are devices that can be ingested orally and then inflated within the stomach to aid in weight loss. Although generally well tolerated, these devices may migrate and cause gastrointestinal obstructive symptoms. Here we report on two cases where these obstructive complications of swallowable IGBs were promptly and non-invasively diagnosed with ultrasonography (US), which also permitted conservative management via US-guided percutaneous aspiration of the impacted balloons. These cases demonstrate that the US may provide a rapid and effective tool for managing IGBs migration, potentially reducing the need for surgical intervention.
Obesity is a growing global health concern that can be addressed using various strategies, including lifestyle modifications, medications, endoscopic procedures, and bariatric surgery [1–4]. Intragastric balloons (IGBs) have been utilized for weight loss over the past two decades; however, their use is often limited by the need for hospitalization, upper gastrointestinal endoscopy, and anesthesia [5].
The Elipse™ intragastric balloon (EIGB) is a new swallowable device that does not have these limitations and has been proven to be safe, effective for weight loss, and well tolerated [6–11]. The EIGB is compressed into a vegan capsule attached to a thin catheter. After being swallowed with water, the balloon is filled with 550 mL of liquid once it reaches the stomach. A small radiopaque ring within the EIGB allows for confirmation of its correct position through abdominal X-ray.
After approximately four months, a valve in the EIGB opens, allowing the balloon to empty and pass naturally through the digestive system without requiring endoscopic removal. The most common adverse events after EIGB placement are nausea and vomiting, occurring in 73% (35/48) and 50% (24/48) of patients, respectively. These symptoms are typically self-limited or resolve with medication within two to three days [10, 12].
While EIGB is generally well tolerated, some side effects can occur, such as intolerance requiring early endoscopic removal (about 3% of cases) [10]. The most serious complication is obstruction, which can occur at various levels, such as gastric outlet obstruction or bowel obstruction due to balloon partial deflation and migration beyond the pylorus. These severe adverse effects are rare, occurring in less than 1% of cases [10, 12].
Recently, we observed two cases of bowel obstruction caused by partial deflation and migration of the EIGB beyond the pylorus. Both cases were successfully managed with ultrasonography (US)-guided aspiration of the migrated balloons, resulting in immediate symptom relief and rapid patient discharge.
Table 1 shows the timeline of the presented cases.
Timeline.
| Case | History | Presenting symptoms | Diagnosis | Intervention | Outcome |
|---|---|---|---|---|---|
| Case 1: 42-year-old female | EIGB delivered, 4 mo. before | 23 October 2019, abdominal pain | Bowel obstruction due to deflated EIGB | US-guided percutaneous aspiration of deflated EIGB | 25 October 2019, discharged without symptoms |
| Case 2: 35-year-old male | EIGB delivered, 3 mo. before | 19 April 2024, abdominal pain | Bowel obstruction due to deflated EIGB | US-guided percutaneous aspiration of deflated EIGB | 21 April 2024, discharged without symptoms |
EIGB: Elipse™ intragastric balloon.
A 42-year-old woman was admitted to the emergency room for acute abdominal pain. Four months before, an EIGB had been delivered for weight loss. On physical examination, the patient had a distended abdomen with marked tenderness in the lower abdomen, but without rebound or guarding. Abdominal plain X-ray showed the small radiopaque marker of the EIGB in the left inferior abdominal quadrant and dilation of small bowel loops (Figure 1A). At US, the deflated EIGB was easily identified under the umbilical line just below the abdominal wall (Figure 1B).
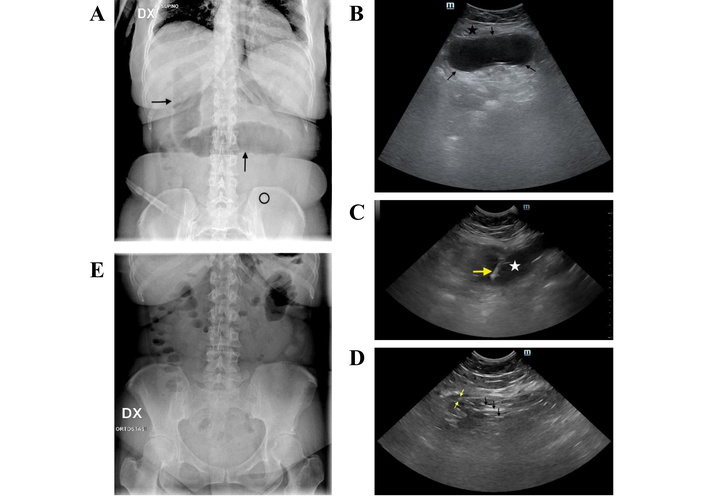
Case 1. (A) Plain X-ray of the abdomen at admission: dilated bowel loops are visible in the mid-abdomen (black arrows), and a radiopaque marker—indicating the presence of the EIGB—appears in the left lower quadrant (black circle). (B) Transverse ultrasonography (US) scan of the hypogastrium shows an oval anechoic structure with hyperechoic (bright) walls (between black arrows) just beneath the rectus abdominis muscle (black star). (C) Transverse US scan of the hypogastrium: the yellow arrow indicates the needle tip inside the nearly completely deflated balloon (white star). (D) Transverse US scan of the hypogastrium: at the end of percutaneous aspiration, only multiple hyperechoic strands (black arrows), representing overlapping walls of the balloon, are observed. The wall of a bowel loop is also seen (yellow arrows). (E) Plain abdominal X-ray taken 24 hours after admission demonstrates resolution of the mechanical ileus.
The patient was rehydrated, and a nasogastric tube was inserted. Given the absence of peritonitis and to avoid surgical intervention, a minimally invasive percutaneous approach was selected. Informed consent was obtained from the patient before the interventional procedure.
After local anesthesia with 5 mL of 2% lidocaine, an 18-gauge Chiba needle was introduced into the impacted EIGB (Figure 1C) under continuous real-time guidance; then, 250 mL of clear, viscous liquid was quickly aspirated.
During aspiration, the balloon progressively collapsed until multiple, overlapping hyperechoic linear strands signaled the complete deflation of the EIGB (Figure 1D).
No complications ensued. Six hours later, the patient reported watery diarrhea, a sign of excretion of the EIGB. Abdominal plain X-ray at 24 hours confirmed the resolution of ileus and the absence of the EIGB radiopaque marker (Figure 1E).
The patient was discharged three days after admission to the hospital.
A 35-year-old man presented to the emergency department with a four-day history of intermittent abdominal cramps and vomiting. Physical examination revealed mild dehydration and a distended abdomen with marked tenderness in the mid-abdomen, without rebound or guarding. He had swallowed an EIGB for weight control three months earlier. Computed tomography (CT) demonstrated dilated bowel loops and a partially deflated EIGB in the lower right abdominal quadrant (Figure 2A). The US displayed the balloon just beneath the abdominal wall, with no interposed bowel loops (Figure 2B).
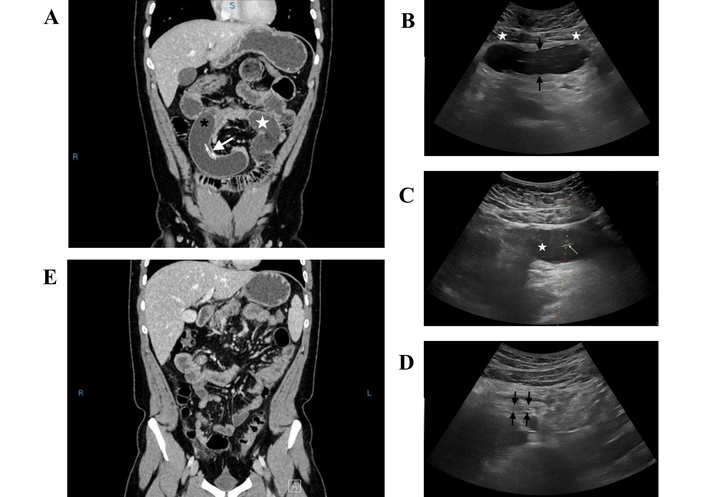
Case 2. (A) CT scan at admission shows the deflated EIGB (black asterisk) causing dilation of bowel loops (white star). The white arrow points to the radiopaque marker of the balloon. (B) Transverse US scan in the periumbilical area shows the partially deflated EIGB (between black arrows) just beneath the rectus abdominis muscles (white stars). (C) Transverse US scan shows the needle tip (white arrow) inside the deflated EIGB (white star). The dotted line on the screen indicates the chosen path for needle insertion with a lateral US-guiding device. (D) At the end of percutaneous aspiration, the balloon is entirely deflated, and the overlapping hyperechoic walls (between black arrows) are observed on US. (E) The CT scan at 24 hours shows resolution of bowel obstruction and disappearance of the EIGB. CT: computed tomography; EIGB: Elipse™ intragastric balloon; US: ultrasonography.
Following fluid resuscitation and insertion of a nasogastric tube to decompress the gastrointestinal tract and stabilize the patient, US-guided aspiration was performed using an 18-gauge Chiba needle to achieve complete collapse of the EIGB (Figures 2C and 2D). Informed consent was obtained from the patient prior to the interventional procedure.
Occlusive symptoms resolved a few hours later when watery diarrhea developed, indicating resolution of the bowel obstruction and the passage of intestinal contents. A follow-up CT scan at 24 hours showed no evidence of bowel obstruction, and the EIGB was no longer recognizable (Figure 2E). The patient was discharged the following day, 72 hours after admission.
Table 2 summarizes diagnostic tools used in these cases, the type of treatment, and outcomes.
Diagnostic tools, treatment methods, and outcomes of the two cases.
| Case | Diagnostic tools | Treatment methods | Post-treatment outcomes |
|---|---|---|---|
| Case 1 | Plain X-ray, US | Percutaneous US-guided aspiration | Relief of bowel obstruction, discharge within 72 hours |
| Case 2 | CT, US | Percutaneous US-guided aspiration | Relief of bowel obstruction, discharge within 72 hours |
CT: computed tomography; US: ultrasonography.
Both patients presented to the emergency room, deeply concerned about their abdominal pain. After their diagnosis was clarified, they consciously chose the proposed treatment: a non-invasive, US-guided intervention to deflate the obstructing EIGB. Following a quick and uneventful recovery, both patients expressed gratitude.
Our two cases confirm the diagnostic capabilities of US in detecting migrated EIGB. They also highlight the role of US-guided interventional maneuvers in treating impacted devices within a bowel loop. In both instances, the deflated and migrated EIGB was easily identified using the US. A simple, painless, and safe US-guided aspiration allowed for the excretion of the device, relieved the bowel obstruction, and prevented the need for surgical intervention.
Bibliographic research conducted on December 20, 2025, using the keywords [(Elipse intragastric balloon[Title/Abstract]) AND (ultrasonography[Title/Abstract])] retrieved no results. This suggests that our report is the first to propose US as a successful method for prompt diagnosis of EIGB-related obstructive complications and conservative management.
IGBs are commonly used as a temporary bridge therapy before surgery [13]. Most manufacturers recommend device removal after six months due to the increased risk of balloon deflation and migration. Bowel obstruction from migrated IGBs left in place longer than six months accounts for most published complications [14]. Early deflation of an EIGB (within six months), causing gastric outlet syndrome or bowel obstruction, is rare, up to 0.6% in one study [10]. This adverse event has been linked to earlier generations of the Elipse balloon, sometimes due to valve faults [8, 10]; however, our patients received the updated device. In our case, altered gastrointestinal motility and/or excessive gastric acid secretion triggered by the EIGB may have damaged the balloon valve.
The risk of balloon deflation and subsequent migration increases over time, but as seen in our cases, these complications can occur early, within the recommended six-month retrieval window.
Laparoscopic surgery is generally considered the preferred treatment for obstruction due to a partially deflated IGB. Imaging-guided percutaneous aspiration has been used rarely, in 3 out of 27 cases (11%) of bowel obstruction caused by various IGBs [11] and was not employed in two large multicenter studies on EIGB safety [8, 10]. We selected an 18 g needle over smaller gauges to ensure rapid evacuation of viscous balloon contents, balancing efficacy with safety based on our prior experience with gastrointestinal mass biopsies [15] and supporting literature [16]. Puncturing intestinal walls with an 18 g needle (even of the cutting type) has been demonstrated to be as safe as biopsies performed with smaller 20–22 g needles [15, 16].
We have previously recommended the US as the method of choice for monitoring Bioenterics Intragastric Balloon (BIB) complications and routinely use it to check intragastric devices for weight loss [17]. Our two cases suggest the potential role of the US not only as a diagnostic method for detecting migrated EIGB but also as a guidance tool for percutaneous treatment of impacted devices within a bowel loop. In both cases, the deflated and migrated EIGB was easily located with US, and a simple, painless, and safe US-guided aspiration facilitated device excretion, resolved bowel obstruction, and avoided surgery.
Although limited to only two cases, our experience suggests a potential role of US as a valuable tool for assessing migration of intragastric devices for weight loss beyond the pylorus in obese patients suitable for US exploration. Furthermore, when a safe access route is identified, US-guided needle aspiration of a partially deflated IGB causing bowel obstruction could offer a rapid and safe therapeutic option to avoid surgery. Further studies are needed to establish the role of the US in the management of bowel obstruction caused by partially deflated EIGB.
BIB: Bioenterics Intragastric Balloon
CT: computed tomography
EIGB: Elipse™ intragastric balloon
IGBs: intragastric balloons
US: ultrasonography
GF: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. CG: Conceptualization, Validation, Supervision. Both authors read and approved the submitted version.
Giampiero Francica, who is the Editorial Board Member of Exploration of Digestive Diseases, had no involvement in the decision-making or the review process of this manuscript. Cristiano Giardiello declares that he has no conflicts of interest.
Hospital Health Management of Pineta Grande Hospital has exempted this case report presented by the Interventional Ultrasound Unit and the Department of Obesity Surgery from Ethical Committee review, as all medical procedures described are consistent with standard clinical practice (document reference no. 2024/192). The study followed the Declaration of Helsinki.
Informed consent to participate in the study was obtained from all participants.
Informed consent to publication was obtained from relevant participants.
The clinical records for this study can be found in the Digital Archive of Pinetagrande Hospital, which is available for consulting only after permission from Hospital Health Management of Pineta Grande Hospital (direzionesaniataria@pinetagrande.it).
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 811
Download: 18
Times Cited: 0
Amedeo Lonardo
Amar Tebaibia ... Nadia Oumnia
Cristina Felicani ... Pietro Andreone
Rudy El Asmar ... Samer AlMasri
Ralf Weiskirchen
Vincenzo Giorgio Mirante
