Affiliation:
1Department of Internal Medicine, Staten Island University Hospital, Northwell Health, New York, NY 10305, United States
Email: chloelahoud@gmail.com
ORCID: https://orcid.org/0000-0003-2346-8733
Affiliation:
2Division of Gastroenterology and Hepatology, NYC Health + Hospitals/South Brooklyn Health, New York, NY 11235, United States
ORCID: https://orcid.org/0009-0002-1237-5232
Affiliation:
3Division of Gastroenterology and Hepatology, Staten Island University Hospital, Northwell Health, New York, NY 10305, United States
ORCID: https://orcid.org/0000-0002-5525-4599
Affiliation:
3Division of Gastroenterology and Hepatology, Staten Island University Hospital, Northwell Health, New York, NY 10305, United States
ORCID: https://orcid.org/0009-0009-7555-1513
Affiliation:
4Department of Pathology, Staten Island University Hospital, Northwell Health, New York, NY 10305, United States
Affiliation:
3Division of Gastroenterology and Hepatology, Staten Island University Hospital, Northwell Health, New York, NY 10305, United States
ORCID: https://orcid.org/0000-0001-7915-4579
Explor Dig Dis. 2026;5:1005113 DOI: https://doi.org/10.37349/edd.2025.1005113
Received: November 16, 2025 Accepted: February 13, 2026 Published: March 05, 2026
Academic Editor: Jose C. Fernandez-Checa, Institute of Biomedical Research of Barcelona (IIBB), CSIC, Spain
Gangliocytic paraganglioma (GP) is a rare tumor that involves the gastrointestinal system and often occurs in the second portion of the duodenum. This is the case of a 73-year-old female presenting for an unintentional 10-pound weight loss over the previous year and a computed tomography (CT) scan of the abdomen and pelvis showing a mass in the second portion of the duodenum with omental nodules. She had no other symptoms. An upper endoscopy revealed a pedunculated 5 cm polypoid mass that was endoscopically resected and found to be a GP on pathology. GP is the third most frequent histopathologic type of gastrointestinal neuroendocrine tumors (NET) after gastrinomas and somatostatinomas. Clinical presentations vary from asymptomatic patients to those having gastrointestinal bleeding, melena, anemia, and abdominal pain. GP presents as a single solid well-demarcated mass that is polypoid, pedunculated, or sessile. Histological features of GP show three cell types: spindle cells, ganglion or ganglion-like cells, and epithelioid cells. Biopsy specimens might not contain all 3 characteristic cell types; thus, definitive diagnosis could be challenging. Periampullary GP should be considered as a tumor with malignant potential. The optimal treatment has not been clarified, but endoscopic resection remains the most common treatment of choice. En bloc resection with negative margins for a pedunculated lesion is possible. This is a rare case of GP in a female patient presenting with weight loss only. It highlights the importance of considering GP in the differential diagnosis for duodenal masses, while keeping a broad differential due to the low rates of diagnostic biopsies. More research is needed to establish standardized management protocols for GP and improve patient care.
Gangliocytic paraganglioma (GP) is a rare tumor that commonly involves the gastrointestinal system and often occurs in the second portion of the duodenum [1]. Aside from gastrointestinal involvement, some case reports present GP of the lungs, heart, head, and neck [2–4]. GP is characterized by its triphasic cellular differentiation: epithelioid neuroendocrine cells, spindle cells with Schwann cell differentiation, and ganglion cells [1]. Most GPs are considered benign and are amenable to excision; however, periampullary GPs should be considered as a tumor with malignant potential [1, 5].
We present the case of a 73-year-old female with a duodenal GP that was successfully treated with endoscopic resection.
The timeline is shown in Table 1.
Timeline.
| Date | Timeline event |
|---|---|
| 2014-06-01 | Past medical history: diagnosed with small bowel gastrointestinal stromal tumor (GIST) and was treated with surgery followed by adjuvant therapy with imatinib for 3 years |
| 2024-03-01 | Computed tomography (CT) scan of the abdomen and pelvis: revealing a small bowel mass in the second portion of the duodenum with omental nodules |
| 2024-04-09 | Presentation: patient presenting for an unintentional 10-pound weight loss over the previous year |
| 2024-04-11 | Esophagogastroduodenoscopy (EGD): revealing a pedunculated 5 cm polypoid mass (Figure 1A). The mass was removed in an en bloc fashion using a hot snare (Figure 1B) |
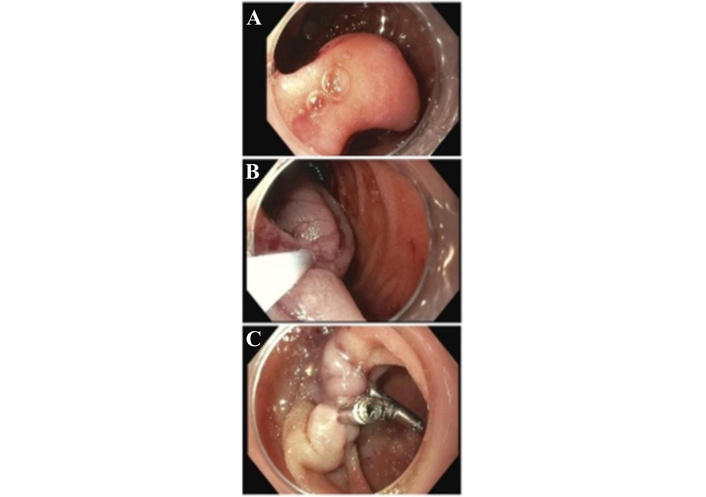
A pedunculated 5 cm polypoid mass in the second portion of the duodenum (gangliocytic paraganglioma) (A) was removed endoscopically with a hot snare (B), followed by the placement of 3 endoclips (C).
The patient is a 73-year-old female who presented for an unintentional 10-pound weight loss over the previous year. Her medical history is remarkable for a small bowel gastrointestinal stromal tumor (GIST) treated surgically in 2014, followed by adjuvant therapy with imatinib for 3 years, hypertension, coronary artery disease, and osteoarthritis. Prior to her evaluation, she had a computed tomography (CT) scan of the abdomen and pelvis, revealing a small bowel mass in the second portion of the duodenum with omental nodules. She denied any gastrointestinal symptoms, including abdominal pain, hematochezia, melena, or changes in bowel habits.
She was seen by the gastroenterology team and underwent esophagogastroduodenoscopy (EGD), revealing a pedunculated 5 cm polypoid mass (Figure 1A). A clear cap was used on the scope. The base of the mass was injected with epinephrine, and the mass was removed in an en bloc fashion using a hot snare (Figure 1B). Three endoclips were successfully placed for defect closure (Figure 1C), and the polyp was retrieved. The patient tolerated the procedure well with no complications.
Pathology showed spindle, ganglion, and epithelioid cells, confirming the diagnosis of a GP with negative tumor margins (Figures 2 and 3). Immunohistochemistry revealed a Ki-67 < 1%, was positive for synaptophysin, chromogranin, CD56 and S100, and negative for CD117, DOG1, SMA, and AE1/AE3. The patient’s diet was advanced, and she was discharged for follow-up in the gastroenterology clinic. Following her procedure, she underwent sampling of her omental nodules with a percutaneous biopsy, revealing recurrent GIST.
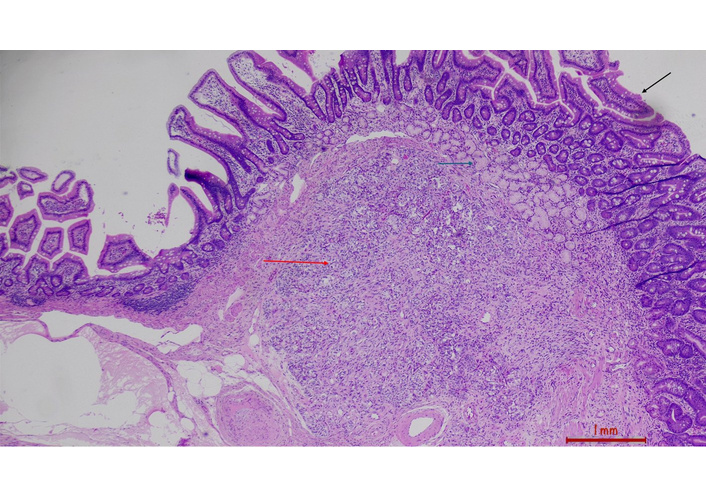
Duodenal mucosa with villi (black arrow), Brunner’s glands (green), submucosa with gangliocytic paraganglioma (red) (H&E stain, 4×).
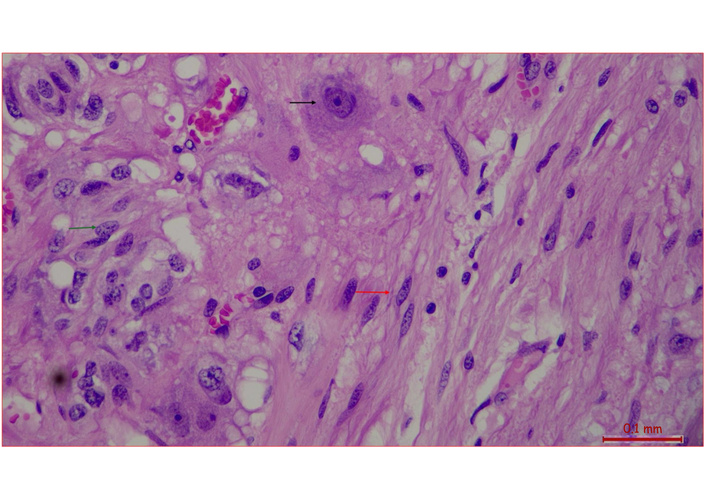
Ganglion type cell (black arrow), spindle cell (red arrow), epithelioid cell (green arrow) (H&E stain, 40×).
GP accounts for 6 to 9% of duodenal gastrointestinal neuroendocrine tumors (NET) and ranks as the third most frequent histopathologic type after gastrinomas and somatostatinomas [6, 7]. The exact embryologic origin of GPs remains unclear, but endodermal and ectodermal sources are postulated [8–10].
In a literature survey done by Okubo et al. [11], patients with GP had a mean age of 52.3 (range 15 to 84 years of age), with a slight male predominance (male to female gender ratio 114:76), and a mean tumor size of around 2.5 cm. GP is most commonly found in the duodenum (90%), and its clinical presentation varies from an asymptomatic course to one or more presenting gastrointestinal symptoms [11, 12]. In a systematic review of 263 cases with GP, the most common presentations of GP were gastrointestinal bleeding (48%) and abdominal pain (45%), followed by anemia (20%), and less commonly nausea (7%), weight loss (5.5%), and jaundice (5%) [13]. While lymph node involvement was reported in up to 12% of cases, rare complications such as intestinal obstruction, bile duct obstruction, and liver metastasis (1.1%) can also occur [13, 14].
GP tumors usually present as single solid well-demarcated lesions that could be polypoid, pedunculated, or sessile, ranging in size from 5.5 to 100 mm [11, 15]. The mass has a gray to white, yellowish appearance, commonly with an overlying mucosal ulcer with bleeding [1]. It is typically centered in the submucosal layer and infiltrates the mucosal and/or muscularis propria layer in more than 50% of cases [11]. As a result, superficial biopsies may not confirm a diagnosis, and deeper sampling is often necessary. The depth of tumor invasion beyond the sphincter of Odi or the submucosal layer has been associated with an increased risk of lymph node metastasis [13].
Histological features of GP show three cell types:
Epithelioid cells, arranged in a Zellballen (nested) pattern resembling paraganglioma.
Spindle cells, with schwann cell differentiation, are arranged in streams surrounding the epithelioid nests.
Ganglion or ganglion-like cells are scattered throughout or merged with spindle cells in fascicular clusters resembling ganglioneuroma [16, 17].
In cases with lymph node metastasis, the metastasized tumor has variable cell types, most commonly made up of epithelioid cells only (46.7%) [1]. Each of the three cell types has characteristic immunohistochemical staining, and in our case, S100 was positive in ganglion and spindle cells [1]. Okubo et al. [13] report that each of the three cell types have unique immunohistochemical profile, with the most common ones being: CD56 (100%), S-100 protein (96%), and CD56 protein (95.2%) in the epithelioid cells, spindle cells, and ganglion-like cells, respectively. Some of the other immunoreactivity markers used are synaptophysin, neuron-specific enolase, progesterone receptor, chromogranin A, pancreatic polypeptide, somatostatin, chromogranin, and cytokeratins [13]. The histological or immunohistochemical differences between tumors with or without lymph node metastasis remain unclear.
GPs are rare and difficult to differentiate from other periampullary neoplasms, including NETs. Immunohistochemical expression of progesterone and pancreatic polypeptide receptors is useful in differentiating GPs from G1 NETs [13]. Biopsy specimens might not contain all 3 characteristic cell types, and definitive diagnosis could be challenging with a low rate of diagnostic biopsies before surgical resection of around 11% [11, 13].
It is important to have a broad differential diagnosis in suspected GP cases. The differential diagnosis would include GISTs, NETs, smooth muscle tumors, adenocarcinoma, and ganglioneuromas [1, 7]. A common pitfall is misdiagnosis as NET G1 when only the epithelioid component is sampled, particularly in superficial biopsies that fail to capture the ganglion-like and spindle cell components [13]. Superficial biopsies may not confirm the diagnosis, and deeper sampling is often necessary since GP is typically centered in the submucosal layer. When tissue is limited, immunohistochemistry on the biopsy specimens is essential for definitive diagnosis, as the triphasic morphology may not be fully appreciated in small samples [13, 17].
The most critical differential diagnosis is NET G1 (carcinoid tumor), as GP is often misdiagnosed as NET G1, which carries a poorer prognosis [13]. Okubo et al. [18] report that up to 40% of duodenal lesions initially diagnosed as NET G1 may actually be GP. The key distinguishing features are that GP exhibits the characteristic triphasic morphology with ganglion-like and spindle cell components, whereas NET G1 shows a more uniform neuroendocrine cell population.
Periampullary GP should be considered as a tumor with malignant potential. Factors that could be associated with lymph node metastasis include size (> 2 cm), younger age, female gender, a high mitotic index, nuclear polymorphism, and tumors invading the submucosal layer or causing biliary obstruction [1].
The optimal treatment of periampullary GPs has not been clarified, and treatment options vary [5]. Endoscopic resection is the treatment of choice for most of the duodenal GPs. En bloc resection with negative margins for a pedunculated lesion is possible. In the published literature, endoscopic techniques successfully removing these lesions have been described. Palomino-Martínez et al. [19] report a case of GP adjacent to the ampulla of Vater that was treated with an endoscopic ampullectomy, with successful resection and confirmed negative margins on histopathology. Similarly, Manglekar et al. [20] discuss the case of a 70-year-old female who was found to have a GP in the second part of the duodenum. It was treated with endoscopic submucosal dissection (ESD), and en bloc resection was achieved. Moreover, pancreaticoduodenectomy is recommended for GPs with possible malignancy, submucosal invasion, or pancreatic GP [5]. Localized excision may be used in cases where endoscopic removal is not possible or cannot achieve negative margins [21]. Wong et al. [22] report the case of a patient who received postoperative adjuvant radiation therapy after pylorus-preserving pancreaticoduodenectomy for periampullary paraganglioma with nodal metastases. Utilization of chemotherapy for the management of this disease has not been standardized [23].
A metastatic workup is recommended at the time of diagnosis of GPs with imaging of the chest, abdomen, and pelvis.
One published case reports tumor recurrence 11 years after surgical intervention [24]. Aside from lymph node metastasis, factors affecting tumor recurrence remain unclear, and subsequently, surveillance imaging has not been standardized, but is recommended given the knowledge gap.
This case underscores the importance of including rare tumors with malignant potential, such as GP, in the differential diagnosis of duodenal masses. Histologic features, including spindle, ganglion, or epithelioid cells, combined with characteristic immunohistochemical staining, are essential for confirmation. Treatment typically involves endoscopic or surgical resection, depending on the depth of invasion. In similar cases where a duodenal GP is pedunculated, en bloc resection is possible with conventional techniques and a forward-viewing scope. Further longitudinal studies are needed to establish standardized management protocols and enhance patient care.
CT: computed tomography
EGD: esophagogastroduodenoscopy
ESD: endoscopic submucosal dissection
GIST: gastrointestinal stromal tumor
GP: gangliocytic paraganglioma
NET: neuroendocrine tumors
This case was previously presented at the American College of Gastroenterology Conference, and the abstract was published online (link: https://journals.lww.com/ajg/fulltext/2025/10002/s6118_case_of_a_gangliocytic_paraganglioma_of_the.6117.aspx). The abstract, on conference acceptance, was published in the American Journal of Gastroenterology. However, the abstract has since been modified to fit the requirements of this journal. The publication of this manuscript and the data involved in other journals is not affected by the conference organizers.
CL: Conceptualization, Investigation, Writing—original draft. MT: Writing—original draft. HK: Writing—review & editing. AE: Writing—review & editing. OC: Resources. JMC: Conceptualization, Writing—review & editing, Supervision. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
According to the policy of the Institutional Review Board (IRB) at the Staten Island University Hospital, this case report does not require ethical approval. The report was conducted in accordance with the Declaration of Helsinki.
Informed consent to participate in the study was obtained from the patient.
Informed consent to publication was obtained from relevant participants.
The datasets that support the findings of this study are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 869
Download: 8
Times Cited: 0
