Affiliation:
1Calderdale & Huddersfield NHS Foundation Trust, Halifax, HX3 0PW, UK
ORCID: https://orcid.org/0009-0006-7896-3351
Affiliation:
2Royal Preston Hospital, Preston, PR2 9HT, UK
ORCID: https://orcid.org/0009-0006-9065-2417
Affiliation:
3Lahore Grammar School JT, Lahore 54700, Pakistan
ORCID: https://orcid.org/0009-0004-2581-8498
Affiliation:
1Calderdale & Huddersfield NHS Foundation Trust, Halifax, HX3 0PW, UK
Email: drazeemsheikh@hotmail.com
ORCID: https://orcid.org/0000-0001-7202-9855
Explor Cardiol. 2026;4:101297 DOI: https://doi.org/10.37349/ec.2026.101297
Received: December 30, 2025 Accepted: February 10, 2026 Published: February 27, 2026
Academic Editor: Alexandre Abizaid, Institute Dante Pazzanese de Cardiologia, Brazil
Infective endocarditis (IE) is a rare but potentially life-threatening condition with a wide spectrum of clinical presentations, often leading to diagnostic delay. The COVID-19 pandemic has added further complexity by overlapping clinical features and imposing constraints on diagnostic pathways. We report the case of a 48-year-old male who presented with prolonged non-specific symptoms following a recent COVID-19 infection. Multiple emergency department visits resulted in an initial diagnosis of viral illness. Subsequent clinical deterioration prompted further evaluation, which revealed severe aortic regurgitation due to a large aortic valve vegetation on transthoracic echocardiography. Blood cultures grew α-haemolytic Streptococcus mitis fulfilling the modified Duke criteria for IE. The patient developed complications, including heart failure and peripheral arterial embolisation, necessitating urgent surgical aortic valve replacement. This case highlights the diagnostic challenges of IE in the context of recent COVID-19 infection, where overlapping symptoms and altered healthcare pathways may contribute to delayed recognition. Clinicians should maintain a high index of suspicion for IE in patients presenting with persistent or atypical symptoms following COVID-19 infection. Early recognition and prompt intervention are essential to prevent serious complications.
Infective endocarditis (IE), a rare but potentially life-threatening condition, is caused by bacteria, fungi, and other micro-organisms that, after entering the bloodstream, can affect heart valves, both native and prosthetic and intracardiac devices [1, 2]. Despite the advances in diagnosing and treating this condition, the mortality rate remains high, up to 18–25% in the first 3 months [3]. The epidemiology of IE has changed in the 21st century, with healthcare-associated IE accounting for > 25% of the cases, due to the increase in intravenous lines and intracardiac devices [4–6]. Despite the well-documented risk factors for IE, patients presenting with atypical signs and symptoms continue to pose a diagnostic challenge [6]. Clinical presentation in IE is diverse and can range from an acute severe infection to low-grade fever, heart failure presentation, or cerebrovascular accident. A rapid diagnosis and initiation of appropriate treatment is utmost important, as a delay in diagnosing this condition can lead to complications and poor clinical outcomes [7–9].
IE is infrequent in patients with COVID-19 compared to other cardiac conditions in this cohort. Nevertheless, a recent study [10] demonstrated a higher incidence of hospital-acquired IE during the first couple of months of the COVID-19 pandemic. In a study of 3,011 hospitalised patients over a period of three months, cardiac complications were seen in 11.6% of the cases, which included atrial fibrillation being the most common. Other complications included acute coronary syndrome, congestive cardiac failure, arrhythmias, myocarditis, IE, pulmonary embolism, and pericarditis [11].
We describe an interesting case of a patient who had an atypical presentation of IE following a COVID infection.
A timeline of clinical events is shown in Figure 1.
A 48-year-old Caucasian man with no known structural heart disease and no significant past medical history presented to the emergency department on multiple occasions over several weeks with non-specific systemic symptoms, including intermittent fever, malaise, and exertional breathlessness. He was previously fit and well, with no history of cardiac disease, intravenous drug use, or recent dental procedures. Notably, the onset of symptoms followed a recent recovery from a confirmed COVID-19 infection.
The patient was a practising Jehovah’s Witness and declined blood product transfusion under any circumstances. Initial clinical assessments during early emergency department attendances were unrevealing, and cardiovascular examination did not document a cardiac murmur at that stage.
During one presentation, iron-deficiency anaemia was identified, prompting upper gastrointestinal endoscopy and colonoscopy, both of which excluded significant pathology. Despite repeated presentations, his symptoms persisted and progressively worsened, ultimately prompting further inpatient evaluation.
Given the ongoing clinical deterioration, the patient underwent urgent surgical intervention. He subsequently had an uncomplicated postoperative recovery and remained clinically stable at both three- and six-month follow-up, with complete resolution of symptoms and no evidence of recurrent infection.
Baseline laboratory investigations demonstrated mildly elevated inflammatory markers, which were non-specific in nature (Table 1). Initial chest radiography performed during early emergency department presentations was unremarkable. However, repeat imaging later during the admission demonstrated evolving pulmonary changes consistent with acute heart failure (Figure 2).
Laboratory investigations.
| Parameter | Result | Reference range |
|---|---|---|
| Haemoglobin (g/L) | 76 | 135–170 |
| White cell count (109/L) | 8.5 | 3.5–11 |
| Mean cell volume (fL) | 79.6 | 80–99 |
| Platelets (109/L) | 148 | 140–400 |
| Neutrophils (109/L) | 7.66 | 1.7–8 |
| Lymphocytes (109/L) | 0.60 | 1–4 |
| Sodium (mmol/L) | 136 | 133–146 |
| Potassium (mmol/L) | 4.6 | 3.5–5.3 |
| Urea (mmol/L) | 6.1 | 2.5–7.8 |
| Creatinine (mmol/L) | 84 | 48–128 |
| Total bilirubin (µmol/L) | 9 | 0.0–21 |
| Alanine aminotransferase (IU/L) | 25 | 0.0–40 |
| Alkaline phosphatase (IU/L) | 159 | 30–130 |
| C-reactive protein (mg/L) | 121 | < 10 |
| Brain natriuretic peptide (BNP) (pg/mL) | 839 | < 100 |
| Erythrocyte sedimentation rate (ESR) (mm/h) | 67 | 1–10 |
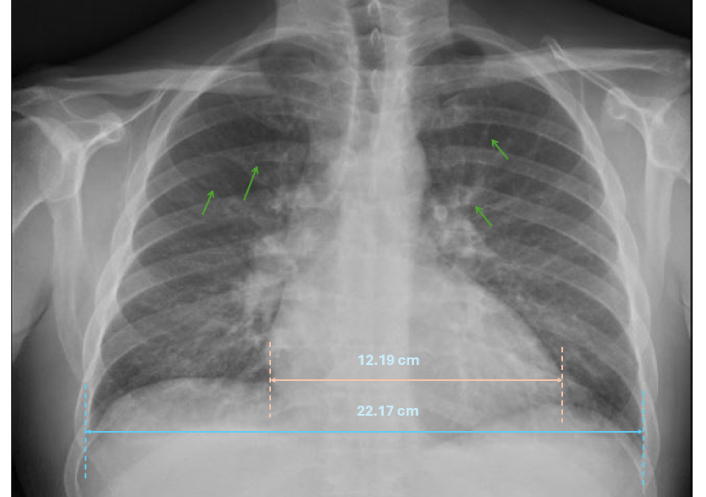
Chest X-ray demonstrating cardiomegaly with upper lobe diversion (thin green arrows).
In view of persistent symptoms and worsening clinical status, multiple sets of blood cultures were obtained prior to the initiation of antibiotic therapy, in accordance with guideline recommendations. Blood cultures subsequently grew α-haemolytic Streptococcus mitis, confirming the microbiological diagnosis of IE.
Transthoracic echocardiography (TTE) revealed a severely dilated left ventricle (LV) with mildly reduced systolic function, alongside a normal right ventricle (RV). A large vegetation measuring 2.58 cm × 1.78 cm was visualised on the flail left coronary cusp of the aortic valve (AV) (Figure 3A), resulting in torrential aortic regurgitation (AR) (Figure 3B).
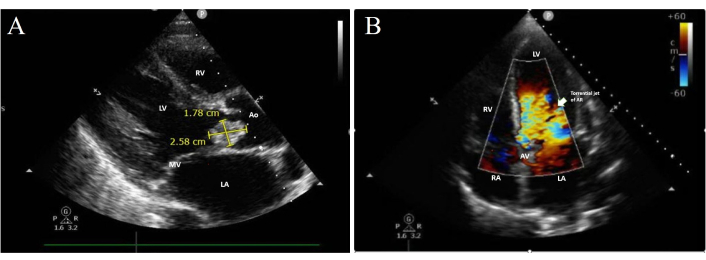
Trans-thoracic echocardiogram. (A) Parasternal view showing a large vegetation on the left coronary cusp. (B) Apical 5-chamber view showing torrential AR. LA: left atrium; MV: mitral valve; LV: left ventricle; RV: right ventricle; Ao: aorta; RA: right atrium; AV: aortic valve; AR: aortic regurgitation.
Transoesophageal echocardiography (TOE) was not performed, as the diagnosis had already been established through microbiological and transthoracic echocardiographic findings, and the patient proceeded directly to surgery due to rapid clinical deterioration.
The patient underwent urgent AV replacement with a 27-mm Edwards Inspiris Resilia bioprosthetic valve. Valve selection followed multidisciplinary heart team discussion and was guided by several specific considerations, including active infective pathology, the need for urgent surgery, the patient’s refusal of blood product transfusion as a Jehovah’s Witness, anticipated anticoagulation risks, and the desire to minimise postoperative bleeding complications.
On follow-up, the patient reported satisfaction with the care provided and a significant improvement in symptoms.
IE continues to present significant diagnostic challenges due to its heterogeneous and often non-specific clinical manifestations, which may range from acute sepsis to indolent constitutional symptoms [1–3, 12]. Atypical presentations are well recognised and frequently contribute to delayed diagnosis, particularly in patients without traditional risk factors or known structural heart disease [5, 13].
The COVID-19 pandemic has further complicated the diagnostic landscape. Systemic inflammation, endothelial dysfunction, immune dysregulation, and prothrombotic states associated with SARS-CoV-2 infection can obscure or mimic the clinical features of IE, potentially delaying appropriate investigation and treatment [9, 10, 14–17].
Several studies have demonstrated that patients with concurrent IE and COVID-19 experience significantly worse outcomes, including higher in-hospital mortality, compared with those with IE alone [9, 18].
In this case, a recent COVID-19 infection likely contributed to diagnostic delay by providing an alternative explanation for persistent fever, inflammatory markers, and cardiopulmonary symptoms. Emerging data suggest that COVID-19 may unmask previously subclinical cardiac pathology or accelerate disease progression through inflammatory and endothelial mechanisms [10, 18, 19].
In a recent systematic review, Parizad et al. [20] proposed that persistent low-grade inflammation, endothelial injury, and autonomic dysregulation represent key mechanistic contributors to long-term cardiac dysfunction. In their systematic review, they reported pooled estimates indicating a 12-month readmission rate of 28% and a mortality rate of 18%, predominantly attributable to heart failure decompensation.
However, a direct causal relationship between COVID-19 and IE cannot be definitively established, and the association in this case should be interpreted with caution.
Early and appropriate microbiological investigation remains central to the diagnosis of IE. Current guidelines emphasise obtaining multiple blood cultures before initiating antimicrobial therapy to maximise diagnostic yield and minimise the risk of culture-negative endocarditis [11, 21].
In the present case, timely blood culture collection prior to antibiotic administration was crucial in establishing the diagnosis.
Echocardiography is essential in suspected IE, with TOE preferred for its higher sensitivity, particularly in detecting small vegetations, prosthetic valves, and periannular complications. TTE may suffice in selected patients when interpreted alongside positive blood cultures and compatible clinical features [17, 21]. Differentiating vegetations from thrombi is critical: vegetations are typically irregular, mobile, and valve-attached, often with associated structural damage, whereas thrombi are more homogeneous, less mobile, and occur in low-flow regions. Clinical context and, when needed, serial or multimodality imaging improve diagnostic accuracy and inform management.
In our patient, TOE was not performed because the diagnosis was already established, and urgent surgical management was required due to rapid clinical deterioration.
Despite the patient’s relatively young age, a bioprosthetic valve was selected following multidisciplinary heart team discussion. Key factors influencing this decision included the urgency of surgical intervention, the presence of active infective pathology, the patient’s religious refusal of blood transfusion, and concerns regarding long-term anticoagulation and bleeding risk. Although mechanical valves are often preferred in younger individuals, current guidelines emphasise individualised decision-making that incorporates patient-specific clinical, surgical, and psychosocial factors [21, 22]. In this context, a bioprosthetic valve was considered the most appropriate and safest option.
This case highlights the importance of maintaining a high index of suspicion for IE in patients with persistent or unexplained symptoms following COVID-19 infection.
Clinicians should remain vigilant for overlapping clinical features and undertake timely microbiological and echocardiographic assessments to prevent delays in diagnosis and management.
Clinicians should maintain a high index of suspicion for IE in patients presenting with persistent or atypical symptoms following COVID-19 infection. Early recognition and prompt intervention are essential to prevent serious complications.
IE may present with non-specific symptoms and delayed recognition, particularly following recent COVID-19 infection.
Overlapping clinical features between COVID-19 and IE may mask classical signs and divert early clinical focus. Overlapping inflammatory and cardiopulmonary features may obscure early diagnosis.
Thorough cardiovascular examination remains essential in patients with persistent systemic symptoms.
Early multidisciplinary evaluation and timely surgical management are critical to prevent complications such as heart failure and embolisation.
Ao: aorta
AR: aortic regurgitation
AV: aortic valve
ESR: erythrocyte sedimentation rate
IE: infective endocarditis
LA: left atrium
LV: left ventricle
RA: right atrium
RV: right ventricle
TOE: transoesophageal echocardiography
TTE: transthoracic echocardiography
EA: Conceptualization, Writing—original draft, Writing—review & editing. MF: Writing—review & editing. MARJ: Writing—review & editing. ASS: Conceptualization, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
The study was conducted in accordance with institutional guidelines and the principles of the Declaration of Helsinki. A formal ethical approval was not required for this retrospective case report.
Informed consent to participate in the study was obtained from the patient.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
This research work did not receive any external funding.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1009
Download: 70
Times Cited: 0
