Affiliation:
1Department of Physiotherapy, School of Health Rehabilitation Sciences, University of Patras, 26504 Patra, Greece
2School of Health Sciences, Faculty of Environmental and Life Sciences, University of Southampton, SO17 1BJ Southampton, UK
Email: psakkatos@upatras.gr
ORCID: https://orcid.org/0000-0001-5801-9765
Explor Asthma Allergy. 2026;4:1009122 DOI: https://doi.org/10.37349/eaa.2026.1009122
Received: February 02, 2026 Accepted: March 25, 2026 Published: April 16, 2026
Academic Editor: Diego Bagnasco, University of Genoa, Italy IRCCS Policlinic San Martino, Italy
Aim: Dysfunctional breathing (DB) is a common comorbidity in asthma, yet its objective characterisation remains challenging due to reliance on subjective questionnaires. This study aimed to determine whether quantifiable breathing pattern parameters can be used to characterise asthma patients with perceived DB from those without DB.
Methods: This observational cross-sectional study involved 122 adults with physician-diagnosed asthma (GINA Steps 2–5). Participants completed the Nijmegen Questionnaire (NQ) to determine DB (NQ > 23) or non-DB (NDB). Resting breathing was recorded for 5 minutes using structured light plethysmography (SLP), a contactless optical motion-analysis system that quantifies thoracoabdominal displacement. Extracted parameters included respiratory rate (RR), inspiration time (Ti), expiration time (Te), Ti/total breath cycle duration (Ttot), and the ratio of ribcage to abdominal displacement during the inspiration phase (RCampinsp/ABampinsp). Both absolute values and within-subject variability [coefficient of variation expressed in percentage (CoV%)] were estimated. Between-group comparisons used Mann-Whitney U tests. Two binary logistic regression models evaluated the predictive value of mean parameters and their variability for characterising DB.
Results: Of the 122 participants, 38 asthmatic patients were determined with DB. The DB group showed significantly higher RR and lower Ti and Te, but no differences in Ti/Ttot or RCampinsp/ABampinsp. In contrast, within-subject variability across all parameters was significantly greater in the DB group. The regression model using absolute values showed limited predictive power (R2 = 0.251). The model incorporating variability demonstrated substantially improved predictive power (R2 = 0.540), with CoV% RR, Te, Ti/Ttot, and RCampinsp/ABampinsp emerging as significant predictors.
Conclusions: Asthma patients with DB exhibit breathing-pattern alterations, most notably increased within-subject variability of timing and TA movement parameters. Breathing pattern variability may be a promising surrogate marker to characterise DB, highlighting the value of dynamic objective physiological assessment of breathing beyond conventional symptom-based assessments of DB.
Asthma is a chronic inflammation of the airways characterised by reversible airflow limitation and bronchial hyperresponsiveness leading to recurrent episodes of wheezing, breathlessness, chest tightness, and coughing [1]. Asthma remains a significant global health concern while recent epidemiological data revealed that there were approximately 260.48 million prevalent cases and 436,190 deaths due to asthma attacks [2]. Some of the risk factors for fatal asthma attacks include poorly controlled asthma, poor medication adherence, delayed emergency care, and poor patient education for self-management [3]. Despite the typical clinical manifestations of asthma, dysfunctional breathing (DB) is a common comorbidity in asthma, particularly difficult-to-treat asthma, with nearly one-third of individuals with asthma, especially difficult-to-treat asthma, exhibiting symptoms consistent with DB [4]. Although an exact causal relationship between asthma and DB has not yet been clearly established, evidence suggests that DB is associated with poor asthma control, leading to patient overtreatment and poor quality of life [5].
DB, also currently referred to as Breathing Pattern Disorder, is a term used interchangeably in contemporary literature to describe chronic or recurrent modifications in breathing pattern associated with variable respiratory and non-respiratory symptoms [6]. Although the term Breathing Pattern Disorder has gained increasing preference in recent years to emphasize observable biomechanical breathing modifications, the term DB remains widely adopted to encompass multidimensional respiratory, biochemical, and psychophysiological components. Respiratory symptoms of DB involve dyspnea, hyperventilation, persistent coughing, excessive sighing, and a feeling of air hunger, whereas some of the non-respiratory symptoms have been reported to be fatigue, dizziness, altered vision, tingling, palpitations, and altered cognitive and emotional states. To date, the exact pathophysiological mechanism of DB remains controversial due to its multidimensional nature based on an interplay of neural, biomechanical, and psychological mechanisms [7]. Several attempts for classification of DB have been made, including hyperventilation syndrome, periodic deep sighing, thoracic dominant breathing, forced abdominal expiration, and thoraco-abdominal asynchrony [6].
To date, there is no gold standard method to objectively characterise DB in asthma patients, and several questionnaires have been developed. The most widely used questionnaire for identifying DB in asthma patients is the Nijmegen Questionnaire (NQ), initially developed to assess the presence of hyperventilation [8]. Although this questionnaire can be easily administered in clinical settings, its use has been criticised in the literature over the years. In particular, it captures a subjective dimension of DB that relies on patient recall, which may be influenced by several factors [8]. Furthermore, the use of the NQ cannot provide objective measurements of several potentially altered breathing pattern components. As a result, these components may not be sufficiently evaluated in the presence of DB in asthma patients in clinical practice.
In addition, other questionnaires, such as the Brompton Breathing Pattern Assessment Tool, provide a structured observational framework for evaluating multiple components of breathing patterns, including diaphragmatic versus thoracic contribution, respiratory rate (RR), and the presence of abnormal breath sounds during inspiration and expiration [9]. However, its application is dependent on the clinician’s expertise and subjective interpretation. Furthermore, this tool does not objectively yield quantifiable measures of breathing pattern alterations, limiting current knowledge relating to potentially altered measurements of quantifiable breathing pattern parameters in asthma patients with DB.
A breathing pattern refers to the characteristic way in which an individual inhales and exhales, encompassing multidimensional and quantifiable components such as air volume, timing indices, and chest wall movement, coordinated by the integrated function of physiological systems such as neural pathways, vital organs, and respiratory muscles [10]. In the literature, these breathing pattern components can be objectively quantified and monitored using non-invasive motion-analysis systems that track chest wall movements, allowing the objective recording of the natural behaviour of breathing over time [11]. Based on this and the current need for more objective physiological markers to objectively characterise the presence of DB among asthma patients, an exploratory secondary analysis of a previously collected doctoral research data [12] was performed to investigate the use of additional breathing pattern parameters to characterise DB among asthma patients. The current secondary analysis seeks to extract additional insights from the dataset, and it was hypothesised that potentially altered absolute mean values and within-subject variability of additional timing breathing pattern components and thoracoabdominal (TA) movement could be used as a surrogate and informative physiological marker to characterise DB in asthma patients.
An observational cross-sectional study design was used to examine whether the absolute mean values or within-subject variability [as estimated by the coefficient of variation expressed in percentage (CoV%)] of RR, inspiration time (Ti), expiration time (Te), the ratio of Ti to total breath cycle duration (Ttot) and the ratio of ribcage amplitude during inspiration phase (RCampinsp) to abdominal amplitude during inspiration phase (ABampinsp) differ between asthma patients with DB and those non-DB (NDB) in a sample with varying asthma control and severity. Furthermore, the absolute mean values and the within-subject variability of the examined parameters were incorporated into two different predictive models to assess whether altered breathing pattern components could be used to characterise DB in asthma patients. An optical motion analysis system of the TA area, known as structured light plethysmography (SLP), was used to record and quantify the examined variables through breath-by-breath analysis as described below. Breathing pattern components were recorded during resting breathing within a single recording session at the NIHR Southampton Clinical Research Facility at the University General Hospital of Southampton after ethical approval.
Adult volunteers (aged 18 years and older) with a physician-confirmed diagnosis of asthma receiving Step 2 to Step 5 asthma medication according to GINA guidelines [13] were eligible for inclusion in this study. Patients who were unable to provide written informed consent or who self-reported any acute or chronic respiratory condition (apart from asthma) were excluded. The latter criterion was applied to minimise potential confounding influences on the breathing pattern components examined for the prediction and identification of DB in asthma patients. The sample size was determined based on the number of events per predictive variable required for each predictive model [14], and thus, a pragmatic minimum sample size of 120 asthma patients was determined.
Participants’ demographic information and medication usage were collected to characterise the study population and asthma severity accordingly. Asthma control was assessed using the 7-item Asthma Control Questionnaire (ACQ-7). The NQ was used as a binary outcome measure to determine the presence or absence of DB among asthma patients and classify the total sample into two groups. In clinical practice, an NQ score greater than 23 is considered indicative of DB [15]; therefore, in the present secondary analysis, asthma patients with NQ > 23 were classified as having DB, with the rest being considered NDB.
Breathing pattern components were recorded for 5 minutes in the seated position using the SLP (Thora-3Di™, Pneumacare Ltd, Cambridge, UK). The SLP is a valid and reliable, fully contactless optical motion analysis device designed to quantify breathing pattern parameters [11]. It has been reported to minimise instrumental observer effect by avoiding contact with the individual, thereby reducing interference with natural breathing behaviour and enabling objective assessment of TA movement [16]. The system employs structured light technology to capture in real time the anterior axial displacement of the TA region during respiration [11]. A stereoscopic analysis is automatically performed through the detection of respiratory-induced distortions in a projected black-and-white grid pattern positioned on the individual’s anterior TA surface, between the clavicles and the umbilicus. These distortions are recorded by two auto-calibrated digital cameras and generate a one-dimensional displacement trace of the TA region over time.
To ensure optimal grid projection quality, participants wore a tight-fitting white T-shirt in accordance with the manufacturer’s guidelines. The participants were instructed to remain still and quiet throughout the recording period, while seated in a high-back chair. This was to minimise extraneous body movement artefacts that could otherwise bias subsequent quantification of breathing pattern parameters due to noise artefacts on the SLP’s output trace. In addition, to reduce potential participants’ behavioural modification of breathing associated with awareness of being observed, participants were deliberately given an inaccurate indication of the recording start time. Actual data collection commenced one minute after this initial notification to ensure capture of the natural resting breathing pattern.
At the time of the study, the manufacturer’s software did not support direct breath-by-breath estimation of the RCampinsp/ABampinsp during inspiration, one of the examined parameters. Therefore, an automatic peak detection algorithm, previously used by the author, was applied to the SLP output trace using MATLAB software (The MathWorks Inc, R2017b) to obtain parameterised data [mean values and standard deviations (SD)] for all examined breathing pattern components [17]. The algorithm identified local minima and maxima within each inspiratory and expiratory phase of each breath cycle reflected on the summed SLP trace, as described in prior published work [17]. The RR was defined as the number of breathing cycles per minute (bpm). The Ti was calculated as the interval between a trough and the subsequent peak on the summed trace, whereas the Te was defined as the interval between a peak and the following trough, both expressed in seconds (sec). The Ttot was calculated as the sum of Ti and Te. The ratio of RCampinsp/ABampinsp was defined as the proportion of the vertical distances between a trough and the next peak on the different SLP’s traces used to record the motion of the ribcage and abdomen separately. The within-subject variability for all examined breathing pattern components was expressed as the CoV%.
Statistical analysis was conducted using Jamovi software (version 2.7.17). Descriptive statistics were generated to summarise the demographic characteristics of the total sample as well as each group (asthma patients with DB and those with NDB). Between-group differences in the examined breathing pattern components were assessed using the Mann-Whitney U test, given that normality assumptions were not met for all examined breathing parameters within each group. A Bonferroni-adjusted significance threshold of p < 0.01 was applied. Subsequently, multiple binary logistic regression analysis was exploratory performed using the forced-entry method to characterise the presence of DB among asthma patients. Binary logistic regression is a statistical modelling technique used to estimate the probability of a binary outcome, in this case, the presence or absence of DB based on the use of absolute measures of examined breathing pattern parameters or the estimated within-subject variability of them.
Therefore, two different exploratory regression models were applied. The regression model 1 incorporated the absolute mean values of RR, Ti, Te, Ti/Ttot, and RCampinsp/ABampinsp as predictors, whereas the regression model 2 included the within-subject variability (CoV%) of these parameters. A significance level of p < 0.05 was set for both models. Furthermore, model performance was evaluated using several complementary statistical indicators. In particular, model deviance reflected the goodness-of-fit of the regression model, with lower values indicating improved model fit relative to the observed data. The Akaike information criterion (AIC) and Bayesian information criterion (BIC) showed balanced model fit and complexity, with lower values indicating better model performance.
Assessment of multicollinearity in the models was also checked and indicated elevated variance of inflation factors (VIF > 10) for absolute measures of RR (VIF: 13.09) and Te (VIF: 12.32) in the regression model 1, suggesting the presence of substantial multicollinearity between timing-related predictors. Given the known physiological interdependence between RR and Te, this finding was anticipated. Therefore, the regression coefficients in regression model 1 should be interpreted with caution due to potential instability and inflation of standard errors.
In the presence of a statistically significant predictive model, model discrimination was further explored using receiver operating characteristic (ROC) curve analysis. This aimed to illustrate the trade-off between sensitivity (the ability of the model to correctly identify individuals with DB) and specificity (the ability to correctly identify individuals with NDB). For the exploratory aim of the current secondary analysis, sensitivity and specificity were reported using the default predicted-probability threshold of 0.50.
The total sample consisted of 122 adult patients with asthma (75 females), with a mean age of 44.75 years (SD = 15.98) and a mean body mass index (BMI) of 25.75 kg/m2 (SD = 3.95). Overall, 70% of the sample had a diagnosis of atopic asthma. Participants presented with varying levels of asthma severity and control. In particular, 33 individuals had mild asthma (Step 2 on the GINA medication scheme), while the remaining participants ranged from moderate to severe asthma (Steps 3, 4, and 5 on the GINA medication scheme).
Of the 122 patients, 84 had the NQ score < 23 and were classified as having NDB, whereas the remaining 38 participants were classified as having DB. In the NDB group, there were 50 females and 34 males; in the DB group, there were 25 females and 13 males. The two groups had relatively comparable mean ages (NDB: 43.4 years, SD = 17.0; DB: 47.8 years, SD = 13.1). Within the NDB group, 58 participants had well-controlled asthma, and 26 had not-well-controlled asthma. In contrast, in the DB group, nearly all participants, except one, had poorly controlled asthma.
The Mann-Whitney U test indicated significant between-group differences in absolute timing parameters of breathing patterns such as RR, Ti, and Te; however, no significant differences were found in the absolute values of Ti/Ttot and RCampinsp/ABampinsp between the two groups (Table 1). In contrast, participants with DB exhibited significantly greater within-subject variability across all examined breathing parameters compared to those with NDB (Table 2).
The differences in the absolute values of examined breathing pattern parameters between asthma patients with DB and those without.
| Examined variable | Patients with DB (N = 38) | Patients with NDB (N = 84) | Mann-Whitney U | p-valuea | ||
|---|---|---|---|---|---|---|
| M | Min–max | M | Min–max | |||
| RR (bpm) | 17.80 | 7.40–32.00 | 15.20 | 7.09–22.00 | 1,031 | 0.002 |
| Ti (sec) | 1.37 | 0.73–3.50 | 1.54 | 1.07–3.89 | 1,078 | 0.004 |
| Te (sec) | 1.88 | 1.14–5.28 | 2.42 | 1.60–5.46 | 1,094 | 0.006 |
| Ti/Ttot | 0.40 | 0.30–0.50 | 0.39 | 0.27–0.54 | 1,504 | 0.610 |
| RCampinsp/ABampinsp | 1.32 | 0.36–12.70 | 1.29 | 0.44–4.23 | 1,447 | 0.410 |
ABampinsp: abdominal amplitude during inspiration phase; DB: dysfunctional breathing; M: median values; NDB: non-dysfunctional breathing; RCampinsp: ribcage amplitude during inspiration phase; RR: respiratory rate; Te: expiration time; Ti: inspiration time; Ttot: total breath cycle duration. a: significant result at p < 0.01 after Bonferroni correction with adjusted alpha level.
The differences in the within-subject variability of examined breathing pattern parameters between asthma patients with DB and those without.
| Examined variable | Patients with DB (N = 38) | Patients with NDB (N = 84) | Mann-Whitney U | p-valuea | ||
|---|---|---|---|---|---|---|
| M | Min–max | M | Min–max | |||
| CoV% RR | 13.80 | 3.21–28.70 | 7.42 | 0.00–20.50 | 606 | < 0.001 |
| CoV% Ti | 24.60 | 9.62–38.40 | 17.30 | 9.58–37.00 | 666 | < 0.001 |
| CoV% Te | 27.30 | 15.30–50.00 | 18.50 | 10.20–35.50 | 542 | < 0.001 |
| CoV% Ti/Ttot | 18.60 | 7.60–30.80 | 15.00 | 7.38–29.10 | 975 | < 0.001 |
| CoV% RCampinsp/ABampinsp | 26.80 | 10.40–57.80 | 16.50 | 7.48–47.30 | 572 | < 0.001 |
ABampinsp: abdominal amplitude during inspiration phase; CoV%: coefficient of variation expressed in percentage; DB: dysfunctional breathing; M: median values; NDB: non-dysfunctional breathing; RCampinsp: ribcage amplitude during inspiration phase; RR: respiratory rate; Te: expiration time; Ti: inspiration time; Ttot: total breath cycle duration. a: significant result at p < 0.01 after Bonferroni correction with adjusted alpha level.
The multiple binary logistic regression analysis using the absolute values of the examined breathing parameters did not adequately predict the presence of DB in asthma patients (deviance: 127; AIC: 139; BIC: 156; R2: 0.251). Although RR and Te emerged as significant predictors within this model, overall predictive performance remained limited (Table 3). In contrast, the regression model incorporating within-subject variability of the breathing pattern parameters demonstrated substantially improved predictive ability for characterising DB among asthma patients (deviance: 92.20; AIC: 104; BIC: 121; R2: 0.540). Specifically, within-subject variability in all timing parameters except Ti and variability in RCampinsp/ABampinsp were significant predictors of DB, indicating that greater fluctuations in breathing pattern parameters were associated with a higher probability of DB in asthma patients (Table 4).
The results of the regression model incorporating absolute values of examined breathing pattern parameters were used to characterise dysfunctional breathing among asthma patients.
| Predictors of regression model 1 | B (SE) | X2 | 95% CI for the odds ratio | Sig.a | ||
|---|---|---|---|---|---|---|
| Lower | Odds ratio | Upper | ||||
| RR (bpm) | 0.704 (0.188) | 18.83 | 1.40 | 2.02 | 2.93 | < 0.001 |
| Ti (sec) | 1.581 (1.068) | 2.01 | 0.60 | 4.86 | 39.45 | 0.139 |
| Te (sec) | 1.884 (0.779) | 5.83 | 1.43 | 6.58 | 30.28 | 0.016 |
| Ti/Ttot | –0.603 (8.966) | 0.01 | 0.00 | 0.55 | 23.40 | 0.946 |
| RCampinsp/ABampinsp | 0.212 (0.257) | 1.00 | 0.75 | 1.24 | 2.05 | 0.410 |
The Ti/Ttot variable exhibits a large SE indicating instability of the estimate due to collinearity with other timing-related parameters. Results should be interpreted with caution. ABampinsp: abdominal amplitude during inspiration phase; RCampinsp: ribcage amplitude during inspiration phase; RR: respiratory rate; Te: expiration time; Ti: inspiration time; Ttot: total breath cycle duration. a: significant result at p < 0.05.
The results of the regression model incorporating within-subject values of examined breathing pattern parameters were used to characterise dysfunctional breathing among asthma patients.
| Predictors of regression model 2 | B (SE) | X2 | 95% CI for the odds ratio | Sig.a | ||
|---|---|---|---|---|---|---|
| Lower | Odds ratio | Upper | ||||
| CoV% RR | 0.152 (0.057) | 7.69 | 1.04 | 1.16 | 1.30 | 0.008 |
| CoV% Ti | 0.065 (0.058) | 1.23 | 0.95 | 1.06 | 1.19 | 0.262 |
| CoV% Te | 0.131 (0.061) | 4.87 | 1.01 | 1.14 | 1.29 | 0.034 |
| CoV% Ti/Ttot | –0.173 (0.076) | 5.84 | 0.72 | 0.84 | 0.98 | 0.024 |
| CoV% RCampinsp/ABampinsp | 0.091 (0.031) | 10.00 | 1.03 | 1.10 | 1.16 | 0.004 |
ABampinsp: abdominal amplitude during inspiration phase; RCampinsp: ribcage amplitude during inspiration phase; CoV%: coefficient of variation expressed in percentage; RR: respiratory rate; Te: expiration time; Ti: inspiration time; Ttot: total breath cycle duration. a: significant result at p < 0.05.
The regression model 2 correctly characterised 91.7% of participants without DB (77 correctly identified as NDB, 7 misclassified as DB) and 63.2% of participants with DB (24 correctly classified as DB, 14 misclassified as NDB), using the default predicted-probability classification threshold of 0.50. The model yielded a Youden index of 0.549 and the area under the ROC curve (AUC) of 0.905, indicating excellent discriminatory ability between asthma patients with and without DB (Figure 1). Although the AUC reflects strong overall discrimination, at the selected cut-off, the model demonstrated moderate sensitivity and high specificity, indicating greater accuracy in characterising individuals without DB than in detecting all cases of DB.
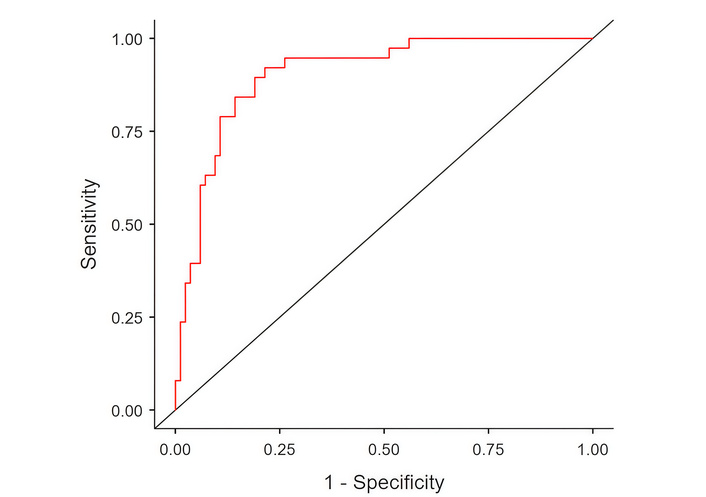
The ROC curve for the regression model involving the within-subject variability of the examined breathing pattern parameters. ROC: receiver operating characteristic.
The present exploratory secondary analysis was performed to investigate whether quantifiable breathing pattern parameters, specifically absolute mean values and within-subject variability of timing parameters and TA movement, can characterise patients with asthma who appear to have DB from those who do not. The findings provide preliminary evidence of differences in several breathing-pattern parameters between the two groups. Specifically, patients with asthma and DB demonstrated slightly higher absolute values of RR and lower Ti and Te compared with those without DB. Asthma patients with DB consistently exhibited greater within-subject variability in both timing parameters and TA movement compared with those without DB. Notably, the within-subject variability across all examined breathing pattern parameters showed better ability to characterise the presence of DB, outperforming the corresponding absolute measures.
This may be attributed to the difficulty of capturing the presence of DB using static breathing metrics of quantifiable breathing pattern parameters. Instead, DB may be more appropriately characterised by monitoring the temporal dynamics of breathing behaviour over time. Recent work in physiological signal analysis has highlighted the value of integrating linear and nonlinear features derived from multimodal physiological signals to characterise complex and dynamic biological processes [18]. Combined linear and nonlinear analyses of multimodal physiological data can capture dynamic patterns underlying physiological regulation. Such analytical frameworks further support the conceptual relevance of examining temporal variability in physiological signals, including breathing pattern dynamics, when investigating multidimensional conditions such as DB. DB has been conceptualized as a multidimensional, state-dependent phenomenon emerging from continuously interacting neural, biomechanical, and psychological processes, situated along a continuum from adaptive to maladaptive breathing control over time [7]. Consistent with this framework, quantitative indices of within-subject variability in breathing pattern parameters may be more informative for identifying asthma patients with DB than corresponding absolute values, as indicated in the present study.
To the author’s best knowledge, few quantifiable breathing-pattern parameters have been monitored to characterise the presence of DB among asthma patients. In particular, the association of higher NQ scores with greater ribcage than abdominal contribution during quiet, seated breathing has been evaluated by using the Manual Assessment of Respiratory Motion (MARM) in 84 self-referred adults with breathing concerns [19]. The results indicated a statistically significant, albeit weak correlation between absolute upper-ribcage contribution and NQ scores. In contrast, a cross-sectional, retrospective, real-world study of 209 adults with unexplained exertional dyspnea assessed proportional tidal volume variation as a simple, objective index that aligned with expert consensus for DB detection [20]. The results concluded that tidal-volume variability provides an easy, objective outcome measure to identify ventilatory irregularities relevant to DB presence.
The present exploratory secondary analysis contributes to the existing literature by demonstrating that objectively recorded breathing pattern components, specifically the within-subject variability of timing parameters and TA movement, are potentially altered in the presence of DB among individuals with asthma. Conceptually, increased within-subject variability may reflect heightened respiratory demand, occurring in parallel with changes in breathing mechanics due to different levels of asthma control. One of the proposed pathophysiological mechanisms underlying DB in asthma involves impaired coordination between ribcage and abdominal movement, resulting in excessive upper-chest breathing and suboptimal diaphragmatic function in uncontrolled asthma [6]. This interpretation aligns with descriptions of physiological mechanisms underlying breathing retraining techniques for DB in asthma management, which aim to normalise the inefficient recruitment of respiratory musculature, decrease sensations of air hunger and dyspnea [21]. Nonetheless, due to the cross-sectional design of the present secondary analysis, causal inferences regarding the relationship between increased within-subject variability and the presence of DB in asthma patients cannot be established. The current study design only permits identification of associations between the examined outcomes, but does not allow determination of directionality or causality. Therefore, it remains unclear whether increased breathing pattern variability contributes to the development of DB, represents a physiological consequence of DB, or reflects shared underlying mechanisms, such as poor asthma control or other interacting pathophysiological processes regarding asthma severity.
In addition, the findings of the regression model 2 suggest that within-subject variability may serve as a moderately useful surrogate marker for characterising DB in patients with asthma. However, the clinical applicability of the regression model 2 should be interpreted cautiously. In the present analysis, the number of DB events relative to the number of predictors resulted in an events-per-variable ratio below traditionally recommended thresholds for logistic regression modelling. This raises the possibility of model overfitting, whereby the predictive performance observed in the current dataset may be optimistically biased. Given the exploratory nature of the present analysis, the present findings are preferable to be considered hypothesis-generating, and this model should be regarded as a preliminary physiological indicator requiring further refinement and evaluation in longer independent cohorts before its consideration as a complementary marker for characterising DB.
At present, the identification of DB relies on subjective assessment tools due to the absence of robust, standardised objective diagnostic methods. As mentioned previously, symptom-based questionnaires, such as the NQ, are practical and widely implemented in clinical practice [22]. However, they predominantly capture self-reported symptom severity, often reflecting perceptual and emotional dimensions of the individuals, rather than quantifiable aspects of breathing mechanics. Research in the DB field has therefore recently focused on characterising DB via the incorporation of objective physiological assessments, including capnography (e.g., PETCO2 monitoring and hyperventilation provocation tests), TA coordination measured via optoelectronic plethysmography (OEP), and exercise-derived indices of breathing irregularity obtained by cardiopulmonary exercise testing [23–25]. These methods aim to characterise the mechanical and neurophysiological alterations associated with DB, yet substantial heterogeneity persists regarding instrumentation, the selection of breathing parameters, and studied populations, as well as proposed diagnostic thresholds. As a result, no universally accepted gold standard currently exists for the objective characterisation of DB, limiting comparability across studies and complicating clinical decision-making.
Current technological advancements in breathing pattern measurements may offer a potential future opportunity to enable objective continuous measures of breath-by-breath fluctuations of breathing patterns over time under real-life conditions, shedding more light on the characterisation of DB among asthma patients. As such, objective monitoring of within-subject variability of breathing patterns has the potential to complement traditional patient-reported outcomes by indexing real-time physiological behaviour of breathing rather than retrospective symptom appraisal. This may help circumvent limitations inherent to subjective measures, such as recall bias and susceptibility to emotional arousal, thereby contributing to a more comprehensive characterisation of DB in asthma patients.
It is acknowledged that the current secondary analysis has several limitations that may have an impact on the interpretability and generalizability of the current findings. One of the methodological limitations relates to the operational definition of DB. Classification of DB relied exclusively on the NQ, a widely used instrument to assess DB. As mentioned previously, it is commonly applied in asthma populations and has demonstrated clinical utility. However, it primarily captures patient-reported symptom perception rather than objective ventilatory mechanics. It is therefore possible that observed associations between increased breathing pattern variability and DB classification partly reflect symptom burden, perceptual sensitivity to respiratory sensations, or psychological factors rather than purely objective breathing dysfunction. Although no universally accepted gold-standard method currently exists for DB diagnosis, future studies should incorporate multimodal approaches, including clinician-based assessments and complementary objective physiological markers to enhance diagnostic robustness, allow triangulation of findings, and limit misclassification bias.
Another important consideration relates to the distribution of asthma control across the study groups. In the present sample, the majority of participants classified as having DB presented with not well-controlled asthma, whereas a substantial proportion of those without DB had well-controlled disease. This imbalance raises the possibility that increased within-subject breathing pattern variability may partly reflect physiological instability associated with poor asthma control rather than DB. Since asthma control was not incorporated as a covariate in the regression models of the current secondary analysis, the independence of breathing pattern variability as a marker of DB cannot be definitely established. However, it is important to recognize that DB has been consistently associated with poor asthma control, making differentiation between DB and physiological instability related to uncontrolled asthma inherently challenging in clinical populations [5]. Future studies should therefore employ stratified designs to better disentangle the relative contributions of asthma instability and DB to breathing pattern variability.
An additional limitation relates to the sex distribution of the studied sample. It has been reported that DB is more prevalent among females, which is reflected in the higher proportion of females in the DB group in the present study [4]. Furthermore, established physiological sex differences in breathing mechanics indicate that females typically demonstrate relatively greater thoracic contribution to ventilation compared with males [26]. Such anatomical and biomechanical differences may influence TA motion parameters and potentially confound the interpretation of no group differences found regarding the absolute measurements of TA motion. However, the significant between-group differences were primarily related to within-subject variability rather than absolute measurements of breathing pattern components, and yet no firm conclusions have been drawn regarding sex differences associated with the variability of several breathing pattern components. However, the independent contribution of sex-related physiological variation cannot be fully excluded, and future studies should consider stratified or sex-adjusted analyses to further delineate these effects.
Finally, the breathing pattern analysis was restricted to a single 5-minute resting measurement under controlled conditions, which may have an impact on the ecological validity of the breathing pattern recordings. Although this approach minimises extraneous variability, DB has been conceptualised as a dynamic, state-dependent phenomenon influenced by several factors. A brief resting recording period may not fully capture the physiological and behavioural modulation of breathing that can occur across different states. Therefore, additional studies are needed to examine prolonged or ambulatory breathing pattern measurements, to validate current results, and further evaluate their utility in characterising DB across different physiological or behavioural states.
This exploratory secondary cross-sectional analysis suggests that asthma patients classified with DB may exhibit quantifiable alterations in breathing pattern parameters compared with those without DB. In particular, increased within-subject variability of timing parameters and TA motion may be present in asthma patients with DB. These findings suggest that quantification of breathing pattern variability through innovative technological advancements capturing the physiological behaviour of individuals’ breathing may represent a potentially informative physiological marker associated with DB in asthma. However, given the exploratory and secondary analysis nature of the study, further prospective and multimodal investigations are required to validate these observations and determine their future clinical applicability.
ABampinsp: abdominal amplitude during inspiration phase
AIC: Akaike information criterion
AUC: the area under the curve
BIC: Bayesian information criterion
CoV%: coefficient of variation expressed in percentage
DB: dysfunctional breathing
NDB: non-dysfunctional breathing
NQ: Nijmegen Questionnaire
RCampinsp: ribcage amplitude during inspiration phase
ROC: receiver operating characteristic
RR: respiratory rate
SD: standard deviations
SLP: structured light plethysmography
TA: thoracoabdominal
Te: expiration time
Ti: inspiration time
Ttot: total breath cycle duration
VIF: variance of inflation factors
The author thanks the participants for their valuable contribution to this dataset. Also, the author thanks the funders for their financial support of the study, which was part of a series of clinical studies conducted for the successful completion of the author’s doctoral degree. The author thanks his supervisors, Prof. Anne Bruton (Emeritus Professor, University of Southampton) and Prof. Anna Barney (Professor of Biomedical Acoustic Engineering, Faculty of Engineering and the Environment, University of Southampton), for their meticulous guidance and contribution to the author’s PhD work that produced the dataset used for the current secondary analysis. Special thanks also go to Prof. Anna Barney for developing and providing the MATLAB algorithm used to extract the breathing pattern data during the author’s PhD work.
PS: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Resources, Visualization, Writing—original draft. The author read and approved the submitted version.
The author declares that he has no conflicts of interest.
The study had been approved by the London-Queen Square Ethics Committee (Rec no: 17/LO/1640; IRAS ID: 230295). This study complies with the 2013 Declaration of Helsinki.
All participants provided a written consent form prior to their participation in the study.
Patients’ anonymous data were agreed to be published to maintain anonymity and protect individuals’ health data.
The dataset used and analyzed during the current secondary analysis, as presented in the current manuscript, is available from the corresponding author (1st author of this manuscript) on reasonable request at psakkatos@upatras.gr.
The current study which was part of a series of clinical studies of the author’s doctoral research was funded by the British Lung Foundation and the Wessex Medical Trust [15501/01]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 584
Download: 9
Times Cited: 0
