Affiliation:
1Institute of Allergology, Charité – Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany
2Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Immunology and Allergology, Berlin, Germany
ORCID: https://orcid.org/0009-0000-0999-2788
Affiliation:
1Institute of Allergology, Charité – Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany
2Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Immunology and Allergology, Berlin, Germany
ORCID: https://orcid.org/0000-0002-4500-7615
Affiliation:
1Institute of Allergology, Charité – Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany
2Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Immunology and Allergology, Berlin, Germany
ORCID: https://orcid.org/0000-0002-1466-8875
Affiliation:
1Institute of Allergology, Charité – Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany
2Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Immunology and Allergology, Berlin, Germany
Email: katarina.stevanovic@charite.de
ORCID: https://orcid.org/0000-0003-3930-9761
Explor Asthma Allergy. 2026;4:1009118 DOI: https://doi.org/10.37349/eaa.2026.1009118
Received: November 19, 2025 Accepted: February 24, 2026 Published: March 22, 2026
Academic Editor: Mario Di Gioacchino, Italian Society of Allergy and Clinical Immunology, G. d’Annunzio University, Italy
The article belongs to the special issue Bridging Experimental and Translational Allergology
The composition and biophysical characteristics of the plasma membrane are pivotal in regulating mast cell immune functions by influencing receptor distribution, activation, and intracellular signaling pathways. This article highlights the impact of plasma membrane components, such as cholesterol and lipid rafts, on the function of the Mas-related G protein-coupled receptor X2 (MRGPRX2), a key mediator of IgE-independent mast cell activation and pseudoallergic reactions. We discuss how variations in membrane fluidity, lipid composition, and microdomain organization influence MRGPRX2 conformational dynamics, ligand accessibility, and downstream signaling efficiency. These membrane-driven effects may help explain the heterogeneity of mast cell responsiveness across tissues and disease states. Integrating insights from structural biology, biophysics, and clinical immunology emphasizes that plasma membrane composition and dynamics regulate MRGPRX2-mediated signaling, positioning the membrane environment as a promising therapeutic target for modulating mast-cell hyperreactivity. By outlining this conceptual framework, we introduce a unifying hypothesis that membrane-driven regulation is a critical, yet underrecognized, determinant of MRGPRX2 responsiveness in different tissues and disease states.
Skin mast cells are mainly situated in the upper dermis, where they play a central role in host defense. Upon activation, they can rapidly release histamine and a wide range of inflammatory mediators, leading to clinical manifestations such as skin inflammation, itch, wheals, and swelling. Although mast cells can be activated by several surface receptors, only FceRI, Mas-related G protein-coupled receptor X2 (MRGPRX2), and the complement receptors can directly trigger degranulation [1].
The cell surface is composed of the plasma membrane, a dynamic mosaic of phospholipids, cholesterol, and sphingolipids that form the bilayer matrix. Embedded within this matrix are membrane proteins, while carbohydrate chains extend outward to mediate cell recognition and signaling. Membrane fluidity and organization of lipid microdomains regulate receptor positioning, clustering, and accessibility, ultimately shaping how signaling pathways are initiated and propagated [2, 3]. Among the various cell surface receptors on mast cells are the G protein-coupled receptors (GPCRs), the largest known group of integral membrane receptor proteins, which play important roles in cellular activation [4]. The influence of plasma membrane fluidity on GPCRs translocation and conformational changes has been described previously [5, 6].
Within the GPCR family is the MRGPRX2, a central mediator of non-IgE-mediated mast cell activation, especially in skin inflammatory diseases such as atopic dermatitis and urticaria [7, 8]. Activation of MRGPRX2 is mainly associated with pain, itch, inflammation, and pseudo-allergic reactions [1, 9, 10]. Its narrow expression profile, being almost exclusively present in tryptase-chymase-positive mast cells (MCTC), to which skin mast cells belong, makes MRGPRX2 particularly noteworthy [1]. This receptor responds to a diverse range of agonists, including neuropeptides, host-defense peptides, and multiple small-molecule drugs, all of which typically contain protonated amine groups and large hydrophobic domains [11]. Recent studies have suggested that plasma membrane composition also influences MRGPRX2 function by determining which receptor conformations are stabilized and thereby modulating ligand-induced responses [12]. Evidence from MRGPRX2 knock-in and Mrgprb2-deficient mice demonstrates that variations in MRGPRX2 surface expression or membrane environment are associated with differences in ligand responsiveness, degranulation capacity, and the cycle of neuropeptide-induced inflammation and vascular hyperpermeability [13].
This perspective advances the hypothesis that MRGPRX2 is a prototypical “membrane-sensitive” receptor whose ligand bias, signaling strength, and disease contributions are tightly coupled to local membrane architecture, including cholesterol content, raft organization, and phospholipid remodeling. We propose that membrane dynamics are not passive modulators but active determinants of MRGPRX2 behavior, offering a conceptual framework that may explain the marked variability of MRGPRX2-driven responses across cells, tissues, and disease states. In this context, this article focuses on MRGPRX2 and its membrane environment, particularly the role of plasma membrane fluidity, addressing the current gaps in both basic and translational immunology.
Cholesterol and the formation of lipid rafts are important for shaping the plasma membrane and regulating its fluidity, flexibility, and stability. These microdomains, enriched in cholesterol and sphingolipids, create well-organized yet dynamic regions that promote the gathering of signaling proteins and support targeted communication between receptors and effectors (Figure 1) [14, 15].
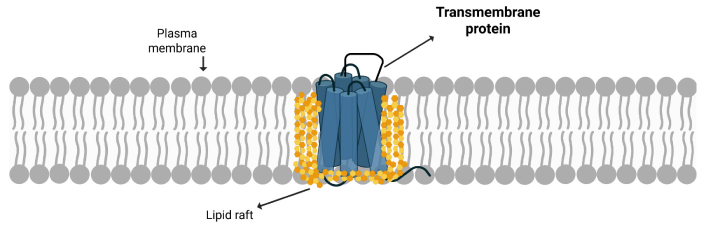
An illustration of a transmembrane protein localized within a lipid raft in the plasma membrane. Created in BioRender. https://BioRender.com/v45p4j7.
MRGPRX2 often resides in these cholesterol-dense regions, where raft association stabilizes the receptor and enhances signal transmission efficiency [6, 16]. The receptor contains specific cholesterol-binding motifs, known as the cholesterol-recognition amino acid consensus (CRAC), that promote direct cholesterol interaction, thereby encouraging MRGPRX2 to localize within these ordered domains [16, 17].
The physical state of the lipid bilayer, defined by its fluidity, plays a dual role in regulating the activity of these membrane bound receptors. Cholesterol limits phospholipid motion, which decreases local fluidity, but also fluctuations in cholesterol packing and raft composition create localized gradients of fluidity that act as dynamic regulators of receptor flexibility [18, 19]. These subtle variations in fluidity, combined with the receptor’s raft association, may fine-tune the conformational balance between active and inactive MRGPRX2 states [15, 20].
Importantly, the relationship between cholesterol, membrane fluidity, and receptor function is reciprocal. On the one hand, cholesterol stabilizes receptor conformation and membrane order. In contrast, the activity and clustering of receptors, such as MRGPRX2, can modify local lipid organization, thereby altering fluidity. Studies have shown that cholesterol extraction by methyl-β-cyclodextrin increases membrane fluidity and reduces GPCRs signaling efficiency [5, 16]. Given that MRGPRX2 contains the CRAC motif, it is possible that lipid rafts also contribute to MRGPRX2 activation and G protein coupling [16]. Conversely, excessive cholesterol accumulation can render the membrane overly rigid, thereby restricting receptor mobility and impairing efficient oligomerization of MRGPRX2s [18]. The bidirectional interplay between fluidity and cholesterol content thus establishes a finely balanced system critical for maintaining the switch-like responsiveness of MRGPRX2 to its broad spectrum of ligands.
Beyond conformational modulation, membrane rigidity also governs the lateral mobility and spatial redistribution of GPCRs across the plasma membrane [14, 19]. In general, increased cholesterol content and reduced fluidity can restrict receptor diffusion within the bilayer, thus limiting clustering of GPCRs, including MRGPRX2, into specialized membrane regions that facilitate interactions with G proteins and downstream effectors [14, 16, 20–22]. Conversely, more fluid membranes may facilitate rapid MRGPRX2 translocation and promote efficient coupling to intracellular signaling complexes [16, 21].
This dynamic equilibrium may in part explain why MRGPRX2 reactivity varies across cell types, tissue environments, and disease conditions. Membrane remodeling events, such as those arising from metabolic stress, inflammatory signaling, or pharmacological interventions, can alter the cholesterol-to-phospholipid ratio and change bilayer viscosity, thereby directly influencing receptor localization and responsiveness [6, 20, 23]. For example, the hepatocyte membrane phospholipid composition is dynamically remodeled in response to metabolic stress (such as obesity and high-fat diet), inflammatory signaling, and pharmacological activation of liver X receptors (LXRs), leading to changes propagated by a stressed endoplasmic reticulum [23]. Understanding these biophysical dependencies is crucial for deciphering how MRGPRX2 contributes to mast cell-mediated hypersensitivity and for developing strategies that target membrane lipids as potential modulators of receptor activity.
Recent advances in structural biology offer fresh insight into why MRGPRX2 is responsive to its membrane surroundings. Cryo-electron microscopy and molecular simulations have revealed that the receptor has a wide, surface-exposed ligand-binding pocket positioned close to the membrane interface, rather than tucked away deep inside the transmembrane region as in most class A GPCRs [24, 25]. This unique orientation indicates that parts of the binding cavity are in direct contact with lipid headgroups, so even subtle shifts in membrane order or fluidity can shape how ligands access and activate the receptor. A key structural feature contributing to this property is the configuration of the extracellular loop region 2 (ECL2). MRGPRX2 lacks the canonical disulfide bridge between ECL2 and transmembrane helix 3, a stabilizing bond between the two specific cysteine residues that enclose the binding site in other GPCRs; therefore, in its absence, ECL2 remains short, flexible, and partially open toward the lipid interface, as shown in Figure 2 [24–26].
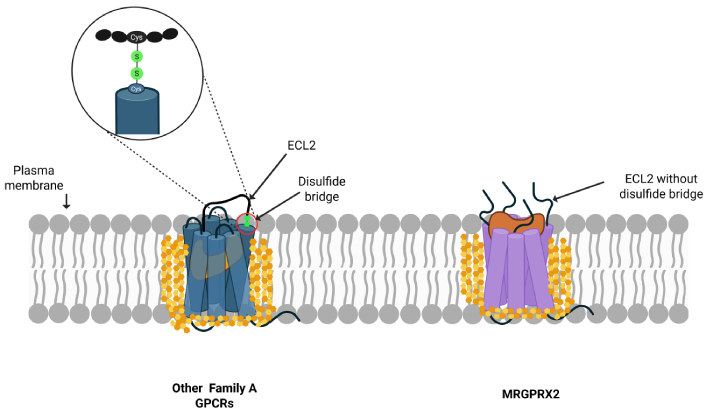
Comparison of MRGPRX2 with other GPCRs’ structures. Typical GPCRs have a conserved ECL2 disulfide bridge that stabilizes ligand binding, whereas MRGPRX2 lacks this feature, contributing to its unique ligand recognition and signaling properties. ECL2: extracellular loop region 2; GPCRs: G protein-coupled receptors; MRGPRX2: Mas-related G protein-coupled receptor X2. Created in BioRender. https://BioRender.com/v45p4j7.
This unique structural feature suggests MRGPRX2 may be more responsive to membrane lipid environment changes than receptors with constrained ECL2 domains [25, 27].
When the membrane becomes more fluid, increased molecular motion within the bilayer may permit greater flexibility of ECL2 and transmembrane helices, facilitating ligand entry and conformational rearrangements. It’s been demonstrated that mechanical disturbance of the plasma membrane can trigger ligand-independent conformational transitions in GPCRs [28]. Time-resolved fluorescence microscopy and GPCR conformation-sensitive fluorescence resonance energy transfer (FRET) revealed that subjecting endothelial cells to fluid shear stress, hypotonic environments, or membrane-fluidizing compounds, such as benzyl alcohol, significantly increased the activity of bradykinin B2 GPCR, highlighting the influence of changes in the lipid bilayer environment on GPCR conformational dynamics [28].
Computational modeling shows that MRGPRX2’s main binding pocket is put together from parts of transmembrane helices 3 to 6, with clusters of acidic and hydrophobic residues right near the lipid headgroup surface [11, 24, 25]. This constitution allows both ligands and the surrounding lipids to interact with the receptor. Even small changes in membrane fluidity, whether from cholesterol, fatty acid saturation, or temperature, can subtly shift how these helices are packed, changing the shape of the binding cavity and influencing how long ligands stay bound and how easily the receptor is activated.
In the past decade, the MRGPRX2 receptor has gotten a lot of attention due to its role in inflammatory skin diseases and as mast cell activator, alternative to IgE receptor signaling [7]. In chronic spontaneous urticaria (CSU) where mast cell hyperreactivity can occur without allergen-specific IgE, skin biopsies from CSU patients consistently reveal an increased number of MRGPRX2-positive mast cells and elevated levels of its endogenous agonists, such as substance P and vasoactive intestinal peptide (VIP), compared with healthy skin, allowing for higher mast cell degranulation and histamine release [2, 29]. It has also been shown that CSU patients often exhibit systemic lipid imbalance or oxidative stress, may experience variable sensitivity to neuropeptides and cationic drugs [2]. Therefore, disease conditions or medications that change lipid raft stability can potentially alternate MRGPRX2-mediated mast cell signaling [21].
From a therapeutic perspective, maintaining balanced membrane order emerges as an attractive target to control MRGPRX2-driven inflammation. Compounds that restore physiological cholesterol-to-phospholipid ratios or stabilize raft architecture could downregulate receptor hyperactivity without directly blocking ligand binding. Meanwhile, selective MRGPRX2 antagonists, including several new small molecules now in clinical testing, have demonstrated potent inhibition of neuropeptide-induced mast cell degranulation, supporting their use in diseases such as CSU and drug hypersensitivity [7, 30]. Combining membrane modulators with receptor-specific agents could thus represent a dual-level therapeutic approach to prevent untoward mast cell activation.
Emerging biochemical evidence indicates that plasma membrane fluidity plays a critical role in regulating GPCR function, including MRGPRX2. Given its importance in inflammatory skin diseases, disruptions in membrane organization may heighten mast-cell excitability and increase hypersensitivity risk. MRGPRX2 thus exemplifies how the membrane context of mast cells can be leveraged for therapeutic intervention, uniting principles from immunology, nanomedicine, and receptor pharmacology. Key outstanding questions include whether distinct lipidomic signatures predict MRGPRX2 hyperreactivity, how disease-associated variants interact with specific membrane environments, and whether vesicle- or nanoparticle-based strategies could be used to locally normalize mast-cell membranes. Addressing these points will deepen our understanding of MRGPRX2-mediated hypersensitivity and may ultimately support the development of therapies that modulate receptor activity by targeting its membrane environment.
CRAC: cholesterol-recognition amino acid consensus
CSU: chronic spontaneous urticaria
ECL2: extracellular loop region 2
GPCRs: G protein-coupled receptors
MRGPRX2: Mas-related G protein-coupled receptor X2
LL: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. MB: Writing—original draft, Writing—review & editing. TZ: Writing—original draft, Writing—review & editing. KS: Validation, Writing—review & editing, Supervision. All authors read and approved the submitted version.
Torsten Zuberbier, who is the Associate Editor and Guest Editor of Exploration of Asthma & Allergy; Katarina Stevanovic, who is the Guest Editor of Exploration of Asthma & Allergy, had no involvement in the decision-making or the review process of this manuscript. TZ has received institutional funding for research and/or honoraria for lectures and/or consulting from Amgen, AstraZeneca, AbbVie, ALK, Almirall, Astellas, Bayer Health Care, Bencard, Berlin Chemie, FAES, HAL, Henkel, Kryolan, Leti, L’Oréal, Meda, Menarini, Merck MSD, Novartis, Pfizer, Sanofi, Stallergenes, Takeda, Teva and UCB, Uriach; in addition, he is a member of ARIA, DGAKI, ECARF, GA2LEN, and WAO. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 689
Download: 30
Times Cited: 0
Vesna Trajkova ... Kristina Trpcheva Stojkov
