Affiliation:
1Cardiovascular Research Center, Tabriz University of Medical Sciences, Tabriz 5166615573, Iran
ORCID: https://orcid.org/0000-0002-4492-637X
Affiliation:
2Department of Internal Medicine, Advanced Cardiac Centre, Post Graduate Institute of Medical Education & Research (PGIMER), Chandigarh 160012, Chandigarh, India
ORCID: https://orcid.org/0000-0001-5433-0433
Affiliation:
1Cardiovascular Research Center, Tabriz University of Medical Sciences, Tabriz 5166615573, Iran
ORCID: https://orcid.org/0000-0003-0109-2026
Affiliation:
3Department of Cardiology, Dayanand Medical College and Hospital (DMCH), Ludhiana 141001, Punjab, India
Email: akashbatta02@gmail.com
ORCID: https://orcid.org/0000-0002-7606-5826
Affiliation:
3Department of Cardiology, Dayanand Medical College and Hospital (DMCH), Ludhiana 141001, Punjab, India
ORCID: https://orcid.org/0000-0002-4337-3603
Explor Neuroprot Ther. 2026;6:1004148 DOI: https://doi.org/10.37349/ent.2026.1004148
Received: October 06, 2025 Accepted: March 02, 2026 Published: April 20, 2026
Academic Editor: Athanasios K. Petridis, Heinrich Heine University Duesseldorf, Germany, St. Lukes Hospital, Greece
The article belongs to the special issue Interdisciplinary Approach to Therapeutic Strategies of Neuroprotection in Present and Future
Background: Atrial fibrillation (AF) substantially increases the risk of ischemic stroke (IS), underscoring the need for effective anticoagulation strategies. Direct oral anticoagulants (DOACs) have largely supplanted vitamin K antagonists (VKAs) due to their favorable safety profile and ease of use. Factor XI (FXI) inhibitors, which target the intrinsic coagulation pathway, are emerging as potential alternatives that may offer reduced bleeding risk. This systematic review evaluates the efficacy and safety of FXI inhibitors compared with DOACs for stroke prevention in AF.
Methods: A total of 20 studies fulfilled the inclusion criteria, comprising 11 randomized controlled trials (RCTs), five systematic reviews or meta-analyses, and four narrative, cohort, or modeling studies. Eligible investigations compared FXI inhibitors with DOACs in patients diagnosed with AF. The primary outcomes assessed were stroke or systemic embolism, major bleeding, and all-cause mortality. Methodological quality was evaluated according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) framework, the revised Cochrane Risk of Bias 2 (RoB 2) tool for RCTs, and the Newcastle-Ottawa Scale (NOS).
Results: FXI inhibitors were associated with a significant reduction in major bleeding [relative risk (RR) 0.31; 95% confidence interval (CI) 0.21–0.46] and clinically relevant non-major bleeding (RR 0.66; 95% CI 0.47–0.93) compared with DOACs. Conversely, FXI inhibitors demonstrated an increased risk of stroke or systemic embolism (RR 3.17; 95% CI 2.18–4.62), as observed in the OCEANIC-AF trial [hazard ratio (HR) 3.79; 95% CI 2.46–5.83]. No significant difference was noted in all-cause mortality (RR 0.85; 95% CI 0.67–1.08). Limited evidence suggests that FXI inhibitors may also reduce bleeding-related hospitalizations.
Discussion: FXI inhibitors provide a favorable bleeding profile but are less effective than DOACs for stroke prevention in patients with AF. Further long-term RCTs are warranted to delineate their role, particularly in populations at high risk of bleeding.
Atrial fibrillation (AF), the most common sustained arrhythmia, is a major risk factor for ischemic stroke (IS), increasing the risk approximately fivefold, primarily due to thrombus formation in the left atrial appendage (LAA) [1, 2]. The prevalence of AF rises sharply with age, exceeding 10% in individuals over 80 years, according to recent studies [3]. Beyond stroke, AF is associated with heart failure (HF), cognitive decline, and increased all-cause mortality [4–8]. The risk of stroke varies from roughly 1% in younger age groups to nearly 25% in individuals aged over 85, largely determined by comorbidities such as hypertension (HTN), diabetes mellitus (DM), vascular disease (VD), and prior stroke [5, 6]. Cardioembolic events account for nearly 20–25% of all IS cases, with AF representing the most frequently documented etiology. Furthermore, individuals with AF remain at substantial risk for stroke following hospitalization, particularly those with multiple comorbid conditions [5, 9, 10].
In low- and middle-income countries (LMICs), limited access to diagnostic tools and anticoagulant therapies exacerbates the burden of untreated AF, resulting in elevated rates of preventable strokes [11]. Such disparities in healthcare access contribute to poorer outcomes in resource-limited settings [12]. The global socioeconomic impact of AF is considerable, with AF-related healthcare costs placing substantial strain on healthcare systems worldwide [13]. Epidemiological projections indicate that the prevalence of AF could double by 2050, emphasizing the urgent need for effective prevention and treatment strategies [14]. The increasing global burden of AF and associated healthcare disparities underscore the need for innovative, accessible anticoagulation therapies to improve outcomes worldwide [3, 14–16].
Anticoagulation therapy remains the cornerstone of stroke prevention in AF. Historically, vitamin K antagonists (VKAs), such as warfarin, were the primary agents; however, these have largely been supplanted by direct oral anticoagulants (DOACs), including apixaban and rivaroxaban [17]. Evidence indicates that DOACs provide superior stroke prevention in AF and confer nearly 30% lower rates of major bleeding compared with VKAs [17, 18]. Advantages of DOACs include fixed dosing and the absence of routine international normalized ratio (INR) monitoring, facilitating adherence in AF management [17, 19]. Nevertheless, DOACs are associated with persistent bleeding risk in elderly or renally impaired patients and are limited by the availability of reversal agents [17, 20]. In this context, factor XI (FXI) and factor XIa (FXIa) inhibitors, such as asundexian and abelacimab, have emerged as a novel class of anticoagulants designed to balance thromboembolism prevention with reduced bleeding risk [21, 22]. By selectively targeting the intrinsic coagulation pathway, which plays a greater role in pathological thrombosis than in normal hemostasis, these agents aim to provide effective antithrombotic activity while preserving hemostatic function [23]. Asundexian, an oral small-molecule inhibitor, achieves rapid, dose-proportional FXIa inhibition of up to 94% at a 50 mg daily dose, with a half-life of 14–17 hours, which enables once-daily dosing with predictable pharmacokinetics [24]. Abelacimab, a subcutaneous monoclonal antibody administered monthly (e.g., 90 or 150 mg), offers sustained FXI suppression due to its long half-life of approximately 25–30 days, potentially improving adherence compared to daily oral regimens [25]. These agents exhibit minimal off-target effects on platelet function and lower potential for drug–drug interactions, making them particularly suitable for patients with polypharmacy [22, 26].
Despite their favorable pharmacological profile, FXI inhibitors have demonstrated variable efficacy for stroke prevention in patients with AF when compared with DOACs. In the phase 3 Oral FXI Inhibitor Evaluation in Atrial Fibrillation (OCEANIC-AF) trial, asundexian was less effective than apixaban in preventing stroke or systemic embolism, with a hazard ratio (HR) of 3.79 [95% confidence interval (CI) 2.46–5.83]. However, asundexian was associated with a substantial reduction in major bleeding, demonstrating a 68% lower risk compared with apixaban (HR 0.32; 95% CI 0.18–0.50) [27].
Meta-analyses confirm that FXI inhibitors reduce major bleeding by approximately two-thirds compared with DOACs, offering a wider therapeutic index, particularly for high-bleeding-risk populations such as the elderly or those with chronic kidney disease (CKD) [21]. However, limitations include the lack of approved reversal agents and limited long-term safety and real-world data, necessitating further phase 3 trials to optimize dosing and identify ideal patient populations [22, 28]. Beyond AF, FXI inhibitors are under investigation for expanding indications, including venous thromboembolism (VTE) prophylaxis, secondary prevention after acute coronary syndrome (ACS), and non-cardioembolic stroke prevention, as evaluated in the Platelet Aggregation and Coagulation Inhibition in Cardiac and Stroke-Related Events-Stroke (PACIFIC-Stroke) trial [28]. Since patients with severe hepatic impairment, active bleeding, and mechanical heart valves were all excluded from the trial, recommendations on their use in these populations cannot be justified till we have more data [28]. These findings underscore the potential of FXI inhibitors to transform anticoagulation therapy for high-bleeding-risk patients while underscoring the need for additional research to establish their long-term efficacy and safety [28, 29]. Although DOACs represent the current standard of care, a residual risk of bleeding persists among high-risk patients, and direct comparisons with emerging FXI inhibitors have produced inconsistent findings regarding both efficacy and safety. Accordingly, this systematic review addresses the evidence gap by comprehensively synthesizing the available data on the comparative efficacy and safety of FXI inhibitors versus DOACs for stroke prevention in patients with AF.
A systematic literature search was conducted to identify studies published between January 2015 and August 2025. The PubMed search strategy included the following terms: (“atrial fibrillation” [MeSH Terms] OR “atrial fibrillation” OR “AF” OR “atrial flutter”) AND (“factor XI” OR “factor XIa” OR “FXI” OR “FXIa” OR “factor XI inhibitors” OR “factor XIa inhibitors” OR “asundexian” OR “abelacimab” OR “milvexian” OR “osocimab”) AND (“direct oral anticoagulants” OR “DOACs” OR “apixaban” OR “rivaroxaban” OR “dabigatran” OR “edoxaban” OR “non-vitamin K antagonist oral anticoagulants” OR “NOACs”) AND (“stroke” OR “cerebrovascular accident” OR “systemic embolism” OR “bleeding” OR “hemorrhage” OR “safety” OR “efficacy”).
Similar strategies, adapted to the syntax of each database, were applied to Scopus and the Cochrane Library. Boolean operators (AND, OR, NOT) were used to optimize the search. Web of Science and Embase were not searched due to substantial overlap with PubMed and Scopus (estimated > 90% duplication in cardiology-related topics) and institutional access limitations, which allowed feasibility without compromising comprehensiveness.
The search process was independently performed by two reviewers to enhance accuracy and minimize selection bias. Discrepancies regarding study eligibility were resolved through discussion or, when necessary, consultation with a third reviewer. In addition, reference lists of included studies, relevant systematic reviews, and clinical guidelines from the American College of Cardiology (ACC) and the European Society of Cardiology (ESC) were manually screened to identify additional studies not captured in the initial database search. This systematic review was not prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO) or any other registry. Nevertheless, the review process adhered strictly to established systematic review standards to ensure methodological rigor and transparency.
Studies were deemed eligible if they assessed the efficacy or safety of FXI inhibitors (e.g., asundexian, abelacimab) compared with DOACs, such as apixaban or rivaroxaban, in individuals with AF, for stroke prevention. Eligibility was determined based on predefined inclusion and exclusion criteria.
Inclusion criteria:
Randomized controlled trials (RCTs), prospective or retrospective cohort studies, or systematic reviews that directly compare FXI inhibitors with DOACs for stroke prevention in AF.
Studies that report at least one relevant clinical outcome, including stroke, systemic embolism, major bleeding, or all-cause mortality.
Human studies with full-text availability.
Exclusion criteria:
Case reports, case series, editorials, and conference abstracts.
Studies not reporting original data (with the exception of systematic reviews or meta-analyses providing extractable outcomes).
Publications not available in English.
The quality of eligible studies was assessed using established tools. Systematic reviews were evaluated according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guideline [30]. The initial search identified 845 records from PubMed, Scopus, and the Cochrane Library. After removing 151 duplicates, 694 unique articles remained. Screening of titles and abstracts resulted in 95 studies selected for full-text review. Of these, 75 were excluded due to irrelevant outcomes, inappropriate study populations, or inadequate methodological quality. Ultimately, 20 studies fulfilled the inclusion criteria and were incorporated into the systematic review, comprising 11 RCTs and related primary analyses, five systematic reviews or meta-analyses, and four narrative, cohort, or modeling studies.
Data from the included studies were extracted using a structured form summarizing key information, including study design, number of participants, patient characteristics, interventions, outcomes of interest, and main findings.
The quality of RCTs was evaluated using the Cochrane Risk of Bias 2 (RoB 2) tool [31], which examines methodological domains such as random sequence generation, allocation concealment, blinding, completeness of outcome data, and selective reporting. Cohort studies were appraised with the Newcastle-Ottawa Scale (NOS) [32], focusing on selection, comparability, and outcome assessment domains. Systematic reviews were assessed using the A Measurement Tool to Assess Systematic Reviews 2 (AMSTAR 2), evaluating key elements including protocol availability, adequacy of the literature search, risk of bias assessment, and management of heterogeneity.
Studies that did not satisfy established quality standards were excluded to preserve the methodological rigor and reliability of the review. An overview of the study selection process is provided in Figure 1.
PRISMA flow diagram depicting the selection process of studies included in the systematic review. * Records identified: A total of 845 records were retrieved from electronic databases (PubMed, Scopus, and the Cochrane Library). Reference lists of included studies, relevant systematic reviews, and clinical guidelines (ACC and ESC) were manually screened, as were trial registries; however, these processes did not yield any additional records. ** Records excluded: Screening was performed manually by two independent reviewers, without the use of automation tools. At the title and abstract screening stage, 599 records were excluded for failing to meet the inclusion criteria. During full-text assessment, 75 articles were excluded. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; ACC: American College of Cardiology; ESC: European Society of Cardiology. Adapted from [30]. © 2026 BMJ Publishing Group Ltd. Licensed under a CC BY 4.0.
Data were systematically extracted using a predefined form to ensure consistency across studies. Extracted variables included study design, baseline patient demographics, type of AF, anticoagulant regimens, follow-up duration, and clinical outcomes.
The primary outcomes of interest were IS or systemic embolism, major bleeding, all-cause mortality, and composite measures of net clinical benefit. Secondary outcomes, including cardiovascular death and hospitalization, were also recorded when available.
Given the expected heterogeneity in study design, patient populations, and outcome definitions, a narrative synthesis approach was employed. Quantitative pooling of results was considered only when data from high-quality RCTs were sufficiently homogeneous. Results were categorized by outcome domain to facilitate comparisons between FXI inhibitors and DOACs.
To account for methodological heterogeneity and to clearly differentiate levels of evidence, studies were grouped and analyzed according to study design: (1) primary evidence from RCTs, (2) synthesized evidence from systematic reviews and meta-analyses, and (3) supportive or exploratory evidence from narrative reviews, cohort studies, and modeling studies. This stratification was implemented to minimize overlap in data interpretation and to provide a transparent hierarchy of evidence.
A total of 20 studies were included in this systematic review. The included studies comprised 11 RCTs and related primary analyses, five systematic reviews or meta-analyses, and four narrative, cohort, or modeling studies. Collectively, these investigations evaluated the efficacy and safety of FXI inhibitors compared with DOACs for stroke prevention in patients with AF.
Due to the heterogeneity in study designs, the evidence is presented in stratified sections below to distinguish primary clinical trial data from synthesized and supportive evidence, thereby minimizing conceptual overlap.
This analysis is primarily based on data from the 11 RCTs and their primary analyses, which represent the highest level of evidence for direct comparative efficacy and safety. Phase 2 RCTs, including Assessment of FXI Antagonism for the Long-Term Evaluation of Atrial Fibrillation-Thrombolysis in Myocardial Infarction 71 (AZALEA-TIMI 71) and the Phase 2 Study of Asundexian for Stroke Prevention in Patients with Atrial Fibrillation (PACIFIC-AF), directly compared FXI inhibitors, namely asundexian and abelacimab, with DOACs, including apixaban and rivaroxaban. Primary outcomes were stroke, systemic embolism, and major bleeding, while secondary outcomes included pharmacokinetics, laboratory parameters, and patient-reported measures [23–25, 27].
The synthesized evidence from the five included systematic reviews and meta-analyses provides a quantitative summary of treatment effects observed across multiple primary trials. These reviews reported pooled estimates, indicating a reduced risk of major bleeding with FXI inhibitors [relative risk (RR) 0.30–0.39], but an increased risk of stroke or systemic embolism (RR 3.17–3.37) compared with DOACs in patients with AF. The reviews also highlighted the limited availability of long-term outcome data [26, 29, 33].
Complementary and contextual insights were obtained from four narrative reviews, cohort studies, and modeling studies. These investigations provide real-world context, mechanistic rationale, and exploratory predictions that inform the interpretation of primary clinical trial findings.
Prospective cohort studies primarily evaluated real-world outcomes with DOAC therapy in patients with AF. Reported annual stroke rates ranged from 1.0% to 1.4%, whereas major bleeding rates ranged from 2.1% to 3.0%. Treatment adherence was high, with 70–85% of patients achieving a proportion of days covered (PDC) > 80%. The majority of patients (90–93%) received anticoagulation in accordance with guideline recommendations, based on a CHA2DS2-VASc score ≥ 2, which incorporates congestive HF (CHF), HTN, age ≥ 75 years, DM, prior stroke/transient ischemic attack (TIA) or thromboembolism, VD, age 65–74 years, and sex category [34–36]. Table 1 presents the characteristics and key outcomes of the included studies, grouped broadly by study type to highlight differences across RCTs, systematic reviews/meta-analyses, and other designs.
Characteristics and key outcomes of included studies comparing FXI inhibitors and direct oral anticoagulants (DOACs) in patients with AF.
| Author (Year) | Study type/design | Population (n, characteristics) | Intervention | Comparator | Primary outcomes | Secondary outcomes | Follow-up duration |
|---|---|---|---|---|---|---|---|
| Rao et al. (2022)[37] | Multicenter, randomized, placebo-controlled phase 2 RCT | Post-MI patients, some with AF (n = 1,601; mean age 64) | Oral asundexian | Placebo | Safety: BARC 2/3/5 HR 0.98 | Efficacy: CV death, MI, stroke/stent thrombosis HR 1.05 | 1 year |
| Sharma et al. (2024)[38] | Phase 2 dose-finding RCT | 2,368 [patients with acute mild IS or high-risk transient ischemic attack (TIA); median age 70–72 years, 33–39% aged ≥ 75 years, 33–37% female, 75–79% HTN, 27–32% diabetes] | Milvexian [oral FXIa inhibitor; doses: 25 mg once daily (QD), 25 mg twice daily (BID), 50 mg BID, 100 mg BID, 200 mg BID] + dual antiplatelet therapy (APT; aspirin + clopidogrel) | Placebo + dual APT (aspirin + clopidogrel) | Composite of symptomatic IS + covert brain infarction on MRI at Day 90 (no significant dose-response; RR range 0.91–0.99 vs. placebo) | Major bleeding (BARC Type 3/5; low rates, no increase with milvexian); symptomatic IS alone (numerical reduction in most doses, RR 0.65–0.83 except 200 mg BID RR 1.40) | 90 days |
| Piccini et al. (2022)[23] | Phase 2 dose-finding RCT | AF n = 753, mean age 73.7 | Asundexian | Apixaban | Major/CRNM bleeding: asundexian 20 mg 0.50, 50 mg 0.16; pooled 0.33 | Any AE: 47% vs. 49% | ~1 year |
| Shoamanesh et al. (2022)[28] | Phase 2b RCT | Acute non-cardioembolic stroke ≥ 45 years, n = 1,808 | Asundexian | Placebo + antiplatelet | Covert brain infarcts/recurrent stroke at 26 weeks: 19–22% | Major/CRNM bleeding 4% vs. 2% | 26 weeks |
| Perera et al. (2022)[39] | Two-part, double-blind, placebo-controlled, sequential single ascending dose (SAD) and multiple ascending dose (MAD) study | 104 healthy adults (SAD panels: 48 participants in 6 panels of 8; MAD panels: 56 participants in 7 panels of 8; randomized 3:1) | Milvexian (BMS-986177/JNJ-70033093; SAD doses: 4, 20, 60, 200, 300, or 500 mg oral; MAD: once- or twice-daily for 14 days; high-fat meal effect in 200- and 500-mg panels) | Placebo | Safety/PK/PD: Favorable safety (no clinical bleeding); dose-proportional absorption (up to 200 mg); terminal t½ 8.3–13.8 h; PD correlated with aPTT | Food effect (increased bioavailability dose-dependently); renal excretion (< 20% in all panels) | SAD: up to 72 h postdose; MAD: 14 days dosing |
| Galli et al. (2023)[22] | Meta-analysis 8 RCTs | n = 9,216 | ISIS 416858, osocimab, abelacimab, milvexian, asundexian | Enoxaparin, DOACs, placebo | Vs. enoxaparin: ↓ any bleeding RR 0.49; vs. DOACs: trend ↓; vs. Placebo ↑ any bleeding RR 1.25 | Major bleeding: non-significant vs. enoxaparin/DOACs; trend ↑ vs. placebo | Short-term perioperative/6–12 months |
| de Alcântara et al. (2026)[40] | Systematic review and meta-analysis | Patients with AF pooled from 4 RCTs (n = 16,852) | FXI inhibitors (asundexian, milvexian) | DOACs (apixaban, rivaroxaban) | Major bleeding significantly lower in FXI inhibitors (OR 0.30, 0.40% vs. 1.32%; 95% CI 0.20–0.43; p < 0.001) | Stroke/systemic embolism more common in FXI inhibitors (OR 3.20; 95% CI 1.85–5.55; p = 0.004) | Median 3–48 months across trials |
| Franco-Moreno et al. (2024)[41] | Systematic review | 18 trials; AF, stroke, MI, VTE | FXI inhibitors (phase 2) | Enoxaparin, DOACs, placebo | ↓ Thrombotic complications ~50% (TKA), modest in AF/MI/stroke | ↓ Bleeding ~60% vs. enoxaparin; safety acceptable | Short-term 12 months |
| Parsa et al. (2024)[42] | Review | Synthesis of preclinical, phase I, and phase II trials focusing on patients with AF, ACS, IS, and VTE | Milvexian (oral FXIa inhibitor; doses up to 500 mg in phase I, varying in phase II; high affinity and selectivity for FXIa) | DOACs (e.g., apixaban, rivaroxaban) or vitamin K antagonists | Anticoagulant efficacy without increased bleeding risk (phase II: no dose-dependent bleeding increase; reduced thrombotic events while preserving hemostasis) | Safety/Tolerability: Mild AEs (phase I), promising risk-benefit (phase II); PK: dose-proportional (up to 200 mg), terminal t½ 8.3–13.8 h | Not applicable (review) |
| Jain et al. 2024[43] | RCT (study design paper) | Patients with AF at risk of stroke; planned n = 15,500, mean age ~70, CHA2DS2-VASc ≥ 2 | Milvexian (FXI inhibitor) | Apixaban (DOAC) | Composite of IS/systemic embolism; major bleeding | Intracranial hemorrhage, fatal bleeding, cardiovascular mortality | Planned 3 years (ongoing trial, no outcome data reported) |
| Shahid et al. (2026)[44] | Systematic review and meta-analysis | Patients with AF pooled from 3 RCTs (n = 16,845) | FXI inhibitor (asundexian, abelacimab) | DOACs (rivaroxaban, pixaban) | Stroke/systemic embolism more common in FXI inhibitors (RR 3.32; 95% CI 2.24–4.90, p < 0.00001) | Major and minor bleeding were significantly lower in FXI inhibitors (RR 0.41; 95% CI 0.33–-0.49, p < 0.00001) and (RR 0.68; 95% CI 0.49–0.93, p = 0.02), respectively | Mean 18 months in real-world cohorts |
| Markides et al. (2025)[29] | Systematic review & meta-analysis | AF; 3 RCTs (PACIFIC-AF, OCEANIC-AF, AZALEA-TIMI 71) | Asundexian, abelacimab | DOACs (apixaban, rivaroxaban) | ↓ ISTH major or CRNM bleeding OR pooled 0.39 | ↑ IS OR 3.37; ↓ all-cause mortality OR 0.82 | Short-mid-term |
| Xue et al. (2025)[33] | Systematic review 3 RCTs | Patients AF n = 16,852 | Abelacimab, asundexian | Rivaroxaban, apixaban | ↓ Major/CRNM bleeding: abelacimab 62–69%; asundexian 50–84% | OCEANIC-AF: ↑ stroke risk with asundexian 3.8×; abelacimab trend ↑ | Short-mid-term |
| Ruff et al. (2025)[25] | Phase 2 RCT (AZALEA-TIMI 71) | AF n = 1,287, median age 74 | Abelacimab 150 mg SC, 90 mg SC | Rivaroxaban 20 mg daily | Major/CRNM bleeding: 150 mg HR 0.38, 90 mg HR 0.31 vs. rivaroxaban | AE similar | Median follow-up 3 months (stopped early) |
| Raffo et al. (2025)[21] | Narrative review | AF patients from RCTs like AZALEA-TIMI 71, PACIFIC-AF, OCEANIC-AF | FXI inhibitors (abelacimab 90/150 mg monthly SC, asundexian 20/50 mg daily oral) | DOACs (rivaroxaban 20 mg daily, apixaban 5/2.5 mg BID) | Major/CRNM bleeding (1.9–2.7/100 PY abelacimab vs. 8.1/100 PY rivaroxaban; 0.4–1.2% asundexian vs. 2.4% apixaban) | IS: higher (non-significant) with abelacimab; all-cause mortality: lower (non-significant); GI bleeding: 0.1/100 PY (abelacimab) vs. 2.1/100 PY (rivaroxaban) | Median 21 months (AZALEA-TIMI 71), 12 weeks (PACIFIC-AF) |
| Piccini et al. (2025)[27] | Phase 3 RCT (OCEANIC-AF) | AF n = 14,810, mean age 73.9 | Asundexian 50 mg daily | Apixaban standard | Stroke/systemic embolism 1.3% vs. 0.4%; HR 3.79 | Major bleeding ↓ 0.2% vs. 0.7%; AE similar | Short, trial terminated early |
| Patel et al. (2025)[45] | Phase II RCT secondary analysis | 1,284 AF patients; median age 74; 44.5% women; stratified by creatinine clearance (≤ 50 vs. > 50 mL/min) | Abelacimab 150/90 mg SC | Rivaroxaban | Major/CRNM bleeding ↓ across kidney function strata | Consistent benefit | Median ~3 months |
| Zhou et al. (2026)[46] | Quantitative modeling study | Simulated AF patient population for phase III trial dose selection; based on prior trial data (n not specified, high-risk AF patients) | Milvexian (FXI inhibitor) | Apixaban | Predicted stroke/systemic embolism risk; bleeding risk (modeled outcomes) | Pharmacokinetic/pharmacodynamic profiles, dose optimization | Not applicable (modeling study, no clinical follow-up) |
| Al Said et al. (2025)[47] | Prespecified sub-analysis of phase 2 RCT (AZALEA-TIMI 71) | AF patients with concomitant APT (subset stratified at baseline; higher bleeding risk due to dual therapy) | Abelacimab 90 mg or 150 mg SC monthly | Rivaroxaban 20 mg daily (dose-adjusted for CrCl) | Major/CRNM bleeding markedly lower with abelacimab vs. rivaroxaban in patients on APT (greater absolute risk reduction in APT group: 7.1–8.1% vs. 4.6–5.0% without APT); consistent safety benefit | IS/systemic embolism (low events, numerically similar); all-cause mortality (neutral); adverse events similar | Median ~21 months (1.8 years) |
ACS: acute coronary syndrome; AF: atrial fibrillation; AF-QoL: atrial fibrillation quality of life; aPTT: activated partial thromboplastin time; AZALEA-TIMI 71: Assessment of Factor XI Antagonism for the Long-Term Evaluation of Atrial Fibrillation-Thrombolysis in Myocardial Infarction 71; BARC: bleeding academic research consortium; BMS-986177/JNJ-70033093: Bristol Myers Squibb compound number/Johnson & Johnson compound number; CHA2DS2-VASc: congestive heart failure, hypertension, age ≥ 75 years, diabetes mellitus, stroke/TIA/thromboembolism, vascular disease, age 65–74 years, sex category; CI: confidence interval; CRNM: clinically relevant non-major; FXI: factor XI; FXIa: factor XIa; GI: gastrointestinal; HR: hazard ratio; HTN: hypertension; IS: ischemic stroke; MI: myocardial infarction; MRI: magnetic resonance imaging; OCEANIC-AF: Oral Factor XI Inhibitor Evaluation in Atrial Fibrillation; OR: odds ratio; PACIFIC-AF: Phase 2 Study of Asundexian for Stroke Prevention in Patients with Atrial Fibrillation; PD: pharmacodynamics; PK: pharmacokinetics; PY: patient-years; RCT: randomized controlled trials; RR: relative risk; SC: subcutaneous; VTE: venous thromboembolism.
Evidence from RCTs indicates that FXI inhibitors substantially reduce the risk of major bleeding compared with DOACs. However, this safety benefit is accompanied by an increased risk of stroke or systemic embolism. A recent meta-analysis reported a significantly lower risk of major bleeding with FXI inhibitors (RR 0.31; 95% CI 0.21–0.46), together with a higher incidence of thromboembolic events (RR 3.17; 95% CI 2.18–4.62) compared with DOACs in patients with AF [33].
These data are consistent with another systematic review assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach, which confirmed the overall safety advantage of FXI inhibitors but underscored uncertainties regarding their efficacy for stroke prevention [26]. Furthermore, results from the phase 3 OCEANIC-AF trial, presented in 2024, demonstrated that asundexian was less effective than apixaban in preventing stroke or systemic embolism, despite being associated with a lower rate of major bleeding [27].
FXI inhibitors exhibit a superior safety profile regarding bleeding outcomes compared with DOACs. Evidence from RCTs demonstrates a significant reduction in major bleeding events (RR 0.31; 95% CI 0.21–0.46) and clinically relevant non-major bleeding (RR 0.48; 95% CI 0.36–0.64), particularly for gastrointestinal hemorrhages [26, 33].
The PACIFIC-AF trial, which included 755 patients with AF over 12 weeks, showed that asundexian (20 or 50 mg daily) reduced the incidence of major or clinically relevant non-major bleeding compared with apixaban (HR 0.33; 95% CI 0.09–0.97). No major bleeding events were reported; however, thrombotic efficacy could not be conclusively assessed due to the short follow-up period [23].
In the phase 3 OCEANIC-AF trial, which enrolled 14,810 high-risk AF patients over 155 days, asundexian was less effective than apixaban for stroke prevention (HR 3.79; 95% CI 2.46–5.83) but showed superior safety, reducing major bleeding by 68% (HR 0.32; 95% CI 0.18–0.55). The trial was terminated early due to efficacy concerns [27]. Similarly, the phase 2b AZALEA-TIMI 71 trial, which included 1,287 patients with AF and a follow-up of 1.8 years, demonstrated that abelacimab (90 or 150 mg monthly) reduced overall bleeding by 62–69% (HR 0.34) and gastrointestinal bleeding by 89% compared with rivaroxaban [25]. Figure 2 illustrates the RRs and HRs of stroke or systemic embolism and major bleeding with FXI inhibitors compared to DOACs in patients with AF.
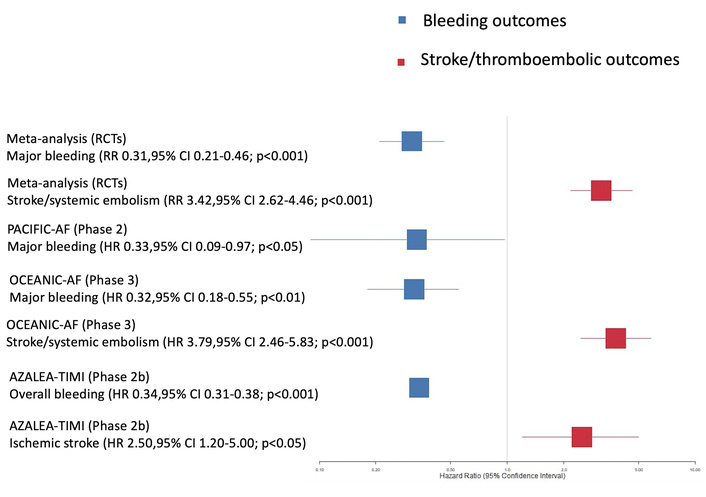
Effect estimates of stroke or systemic embolism (increased) and major bleeding (reduced) with FXI inhibitors versus DOACs in AF (pooled estimates from meta-analyses incorporating the PACIFIC-AF, OCEANIC-AF, and AZALEA-TIMI 71 trials). AF: atrial fibrillation; AZALEA-TIMI 71: Assessment of Factor XI Antagonism for the Long-Term Evaluation of Atrial Fibrillation-Thrombolysis in Myocardial Infarction 71; CI: confidence interval; DOACs: direct oral anticoagulants; FXI: factor XI; HR: hazard ratio; OCEANIC-AF: Oral Factor XI Inhibitor Evaluation in Atrial Fibrillation; PACIFIC-AF: Phase 2 Study of Asundexian for Stroke Prevention in Patients with Atrial Fibrillation; RCTs: randomized controlled trials; RR: relative risk.
FXI inhibitors appear to exert a neutral effect on mortality outcomes in patients with AF. A meta-analysis of three RCTs, including PACIFIC-AF, OCEANIC-AF, and AZALEA-TIMI 71, reported no significant difference in all-cause mortality between FXI inhibitors and DOACs (RR 0.85; 95% CI 0.67–1.08) [26]. In the PACIFIC-AF trial, which enrolled 755 patients with AF over 12 weeks, no deaths occurred in either the asundexian or apixaban arms [23]. The low event rates observed in these studies likely reflect the overall efficacy of anticoagulation therapy in patients with AF. Evidence specifically evaluating cardiovascular mortality remains limited in contemporary trials.
FXI inhibitors, including asundexian and abelacimab, demonstrate favorable pharmacokinetic profiles, characterized by predictable absorption and minimal drug–drug interactions. Early-phase trials (phase 1 and 2) have shown effective inhibition of FXIa, stable activated partial thromboplastin time (aPTT), and minimal impact on prothrombin time (PT), thereby preserving hemostasis [22]. In the phase 2 acute Myocardial Infarction (PACIFIC-AMI) trial, which included 1,601 patients post-myocardial infarction, asundexian administered at 20–50 mg daily produced dose-dependent FXIa inhibition, achieving 92–95% inhibition at the 50 mg dose [37]. Similarly, the PACIFIC-AF trial found no significant off-target effects on anti-Xa activity [23]. These findings support the specificity and favorable safety profile of FXI inhibitors as anticoagulant therapies.
Bleeding events significantly impair QoL and treatment adherence in patients with AF, often leading to therapy discontinuation [48–50]. A meta-analysis demonstrated that FXI inhibitors reduce the risk of major bleeding by 59% (RR 0.41; 95% CI 0.36–0.46) compared with DOACs [51]. Real-world data from European registries indicate high adherence to DOACs, with 70–85% of patients achieving a PDC > 80%, likely reflecting the comparatively low incidence of major bleeding [52]. The favorable bleeding profile of FXI inhibitors suggests potential improvements in patient satisfaction and persistence with therapy. However, dedicated studies utilizing standardized QoL instruments, such as the EuroQol 5-dimensions (EQ-5D) and the AF-QoL questionnaires, remain limited.
FXI inhibitors reduce major bleeding (RR 0.41; 95% CI 0.36–0.46) compared with DOACs, but are associated with an increased risk of stroke (RR 3.42; 95% CI 2.62–4.46) and systemic embolism (RR 4.26) [51]. In the phase 3 OCEANIC-AF trial, which enrolled 14,810 high-risk patients with AF and a median follow-up of 155 days, asundexian (50 mg daily) was compared with apixaban [27]. Asundexian demonstrated inferior efficacy in stroke prevention (HR 3.79; 95% CI 2.46–5.83) but achieved a 68% reduction in major bleeding (HR 0.32; 95% CI 0.18–0.55) [27]. These results underscore a critical trade-off between bleeding reduction and thrombotic risk, emphasizing the need for individualized treatment decisions, particularly in patients at high risk of either outcome.
Evidence regarding the impact of FXI inhibitors on hospitalization and LOS in AF patients is limited. In the PACIFIC-AF trial, which included 755 patients with AF over 12 weeks, no significant difference was observed in all-cause hospitalization rates between asundexian and apixaban (HR 0.91; 95% CI 0.62–1.34) [23]. Bleeding-related hospitalizations were lower with asundexian (2.1% vs. 4.8%) compared with apixaban [23]. Real-world data from European registries indicate that DOACs reduce overall hospitalization rates by 20–30% compared with VKAs (RR 0.75; 95% CI 0.68–0.83), with a mean LOS for bleeding-related admissions of 5–7 days [52]. The mean LOS for bleeding-related admissions with DOACs is 5–7 days [52]. Given the reduced risk of major bleeding observed with asundexian in the OCEANIC-AF trial (HR 0.32; 95% CI 0.18–0.55), FXI inhibitors may reduce bleeding-related hospitalizations [27].
This review evaluated the efficacy and safety of FXI inhibitors compared with DOACs in patients with AF, focusing on both primary outcomes (stroke/systemic embolism prevention and major bleeding) and secondary outcomes (all-cause and cardiovascular mortality, pharmacokinetics, patient-reported outcomes, net clinical benefit, and hospitalization/LOS). The evidence suggests that FXI inhibitors substantially reduce major bleeding (RR 0.31; 95% CI 0.21–0.46) but increase the risk of stroke or systemic embolism (RR 3.17; 95% CI 2.18–4.62) relative to DOACs. Their impact on mortality appears neutral, while early pharmacokinetic data are favorable. However, evidence on patient-reported outcomes and hospitalization remains limited.
The primary outcomes reveal a clear trade-off in FXI inhibitors' performance. A GRADE-assessed meta-analysis of RCTs confirmed a significant reduction in major bleeding compared with DOACs (RR 0.31; 95% CI 0.21–0.46), which further support the results from the PACIFIC-AF trial (HR 0.33; 95% CI 0.09–0.97 for major or clinically relevant non-major bleeding) and the AZALEA-TIMI 71 trial (HR 0.34; 95% CI 0.31–0.38 for major or clinically relevant non-major bleeding) [23, 25, 26]. Similarly, the increased risk of thromboembolic events aligns with the phase 3 OCEANIC-AF trial, where asundexian was inferior to apixaban for stroke prevention (HR 3.79; 95% CI 2.46–5.83) [26, 27].
When contextualized with established DOAC trials, important contrasts emerge. For instance, a meta-analysis by López-López et al. [16] demonstrated that apixaban reduced major bleeding versus warfarin (RR 0.77; 95% CI 0.67–0.88) while maintaining effective stroke prevention.
Furthermore, real-world data from Vinogradova et al. [34] reported annual major bleeding rates of 2.1–3.0% and stroke rates of 1.0–1.4% with DOACs. These benchmarks underscore that while FXI inhibitors may offer superior bleeding reduction, they have not yet matched DOACs in terms of long-term thromboembolic protection.
For secondary outcomes, FXI inhibitors demonstrate a neutral effect on all-cause mortality (RR 0.85; 95% CI 0.67–1.08), comparable to findings from trials with DOACs [26]. Pharmacokinetic data from early-phase trials, including PACIFIC-AMI, show dose-dependent FXIa inhibition (92–95%) and reductions in D-dimer levels, without off-target effects on anti-Xa activity [23, 24, 37]. Evidence on patient-reported outcomes remains limited; however, the bleeding reduction observed with FXI inhibitors suggests potential improvements in QoL and treatment adherence. This is consistent with findings from DOAC registries, where lower bleeding risk correlated with high adherence (PDC > 80% in 70–85% of patients) [34]. The net clinical benefit of FXI inhibitors is characterized by a marked reduction in bleeding risk but an increased risk of stroke appears less favorable compared with DOACs, as reported in recent studies and meta-analyses [27, 51]. Data on hospitalization are sparse; however, the PACIFIC-AF trial reported fewer bleeding-related admissions with asundexian compared to apixaban (2.1% vs. 4.8%), suggesting a potential to reduce hospital burden relative to DOACs in real-world practice [23].
While DOACs remain the cornerstone of stroke prevention in AF according to current international guidelines [15, 16], the distinct safety profile of FXI inhibitors has prompted consideration of their potential role in carefully selected, high-bleeding-risk populations for whom DOAC-associated hemorrhage may be unacceptable. Findings from this review suggest that subgroups potentially deriving the greatest benefit from FXI inhibitors, despite their currently demonstrated inferior efficacy, include patients with a history of major bleeding (particularly intracranial or gastrointestinal), those with severe CKD where DOAC use is contraindicated or requires complex dosing, frail elderly patients at high risk of falls, and individuals requiring concomitant antiplatelet therapy for coexisting coronary artery disease (CAD) [21, 25, 27, 45].
For example, the AZALEA-TIMI 71 trial demonstrated a consistent reduction in bleeding with abelacimab across renal function strata, highlighting a potential advantage in CKD patients [45]. A prespecified analysis of the same trial further indicated marked bleeding reduction among patients receiving concomitant antiplatelet therapy, underscoring a potential role in this challenging subgroup [47].
It is important to emphasize that the use of FXI inhibitors in these contexts remains strictly investigational. The phase 3 OCEANIC-AF trial was terminated early due to inferior efficacy, clearly indicating that FXI inhibitors are not yet suitable to replace DOACs for broad stroke prevention in AF [27]. Therefore, current clinical guidance continues to endorse DOACs as the standard therapy, given their established efficacy and manageable safety profile.
The heterogeneity of study populations, including differences in AF severity, comorbidities, and follow-up durations may limit the generalizability of findings. Reliance on phase 2 trials and interim data from phase 3 studies constrains the ability to draw firm conclusions regarding the long-term efficacy and the incidence of rare adverse events with FXI inhibitors. Furthermore, the absence of standardized assessments for patient-reported outcomes restricts a comprehensive evaluation of QoL benefits. Compared with the extensive real-world evidence available for DOACs, data on FXI inhibitors remain sparse.
Large-scale, long-term RCTs are necessary to establish the efficacy and safety of FXI inhibitors, particularly in high-risk AF populations. Direct comparative studies between different FXI inhibitors, such as asundexian and abelacimab, are warranted to identify the optimal therapeutic agent. Future studies should prioritize high-bleeding-risk subgroups to determine whether the substantial reduction in bleeding conferred by FXI inhibitors translates into a favorable net clinical benefit despite an increased thromboembolic risk, potentially informing a personalized anticoagulation strategy. Moreover, the inclusion of standardized patient-reported outcome measures, such as the EQ-5D and AF-QoL questionnaires, is recommended to assess treatment impact on QoL. Finally, real-world studies across diverse healthcare settings will be essential to validate RCT findings and support the implementation of individualized treatment strategies.
FXI inhibitors demonstrate a favorable bleeding profile compared with DOACs but confer an increased risk of thromboembolic events in patients with AF. Their neutral impact on mortality and advantageous pharmacokinetic properties suggest potential utility in patients at high risk of bleeding. Evidence on QoL and hospitalization outcomes, however, remains limited.
ACC: American College of Cardiology
AF: atrial fibrillation
AZALEA-TIMI 71: Assessment of Factor XI Antagonism for the Long-Term Evaluation of Atrial Fibrillation-Thrombolysis in Myocardial Infarction 71
CI: confidence interval
CKD: chronic kidney disease
DM: diabetes mellitus
DOACs: direct oral anticoagulants
EQ-5D: EuroQol 5-dimensions
FXI: factor XI
FXIa: factor XIa
GRADE: Grading of Recommendations Assessment, Development and Evaluation
HR: hazard ratio
HTN: hypertension
IS: ischemic stroke
LOS: length of stay
OCEANIC-AF: Oral Factor XI Inhibitor Evaluation in Atrial Fibrillation
PACIFIC-AF: Phase 2 Study of Asundexian for Stroke Prevention in Patients with Atrial Fibrillation
PDC: proportion of days covered
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
QoL: quality of life
RCTs: randomized controlled trials
RoB 2: Risk of Bias 2
RR: relative risk
VD: vascular disease
VKAs: vitamin K antagonists
RP: Conceptualization, Methodology, Investigation, Data curation, Formal analysis, Writing—original draft, Writing—review & editing. JH: Supervision, Validation, Writing—review & editing. HB: Writing—review & editing. AB: Conceptualization, Methodology, Formal analysis, Writing—original draft, Writing—review & editing. BM: Project administration, Validation, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request, or are included in the published articles referenced in this review.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Ramón Rama, Joan Ramon Torrella
