Affiliation:
Institute of Psychiatry, Psychology & Neuroscience, King’s College London, WC2R 2LS London, United Kingdom
Email: dr.maria.loumpourdi@gmail.com
ORCID: https://orcid.org/0000-0002-6526-0095
Explor Neurosci. 2026;5:1006128 DOI: https://doi.org/10.37349/en.2026.1006128
Received: November 29, 2025 Accepted: February 27, 2026 Published: March 17, 2026
Academic Editor: Aurel Popa-Wagner, University of Medicine and Pharmacy Craiova, Romania
Adult human hippocampal neurogenesis has been debated for decades, with methodological differences producing conflicting reports. Radiocarbon birth-dating provided population-level evidence of sustained dentate gyrus neuron turnover, while immunohistochemical studies produced variable results depending on fixation protocols. Optimized post-mortem handling has reported higher detectability of immature-neuron markers across adulthood, whereas longer post-mortem delays and prolonged fixation can reduce signal and contribute to apparent null findings; however, marker-based interpretations remain debated and require cautious, multi-marker validation. Recent single-nucleus and spatial transcriptomics further support persistent neurogenesis, identifying immature granule-cell signatures and niche programs into late life. This article critically appraises evidence from radiocarbon dating, immunohistochemistry, and transcriptomics, highlighting sources of discrepancy and convergence. Practical standards for human tissue handling, antigen retrieval, and multimarker panels are proposed to minimize methodological artefacts. Collectively, convergent evidence favors low-level, lifelong neurogenesis with potential contributions to memory precision and affective regulation, albeit at lower rates than rodents. It is concluded that integrating radiocarbon baselining, optimized immunohistochemistry, and transcriptomic validation provides a robust framework for resolving the controversy and advancing translational relevance in cognition, aging, and psychiatry.
For most of the twentieth century, the adult mammalian brain was considered a static organ, incapable of generating new neurons. This dogma, shaped by early neuroanatomists such as Santiago Ramón y Cajal [1], remained largely unchallenged for decades. In the 1960s, however, Altman and Das [2] provided autoradiographic evidence of postnatal neurogenesis in the rodent hippocampus, initiating a gradual paradigm shift. Despite this, the notion of adult neurogenesis in humans remained contentious, due in part to the difficulty of translating findings from animal models.
A turning point came with the work of Eriksson et al. [3], who administered the thymidine analog bromodeoxyuridine (BrdU) to terminally ill cancer patients and, upon postmortem examination, identified BrdU-labeled neurons co-expressing mature neuronal markers in the dentate gyrus. This landmark finding provided the first direct evidence of new neuron formation in the adult human brain and refuted the long-standing belief that neurogenesis was confined to early development. The study also raised critical questions about the rate, persistence, and functional relevance of neurogenesis throughout human adulthood, particularly given its established roles in learning and emotional regulation in animal studies.
Over the subsequent two decades, researchers employed various methodologies to characterize adult human hippocampal neurogenesis. Early immunohistochemical analyses revealed cells expressing progenitor and immature neuron markers, although many suggested a steep decline in neurogenesis with age [4]. The development of novel techniques, such as radiocarbon dating of neuronal DNA by Spalding et al. [5], added quantitative rigor to the field by enabling retrospective birth-dating of neurons and providing robust evidence of neuron turnover in adult humans.
Yet the field was soon divided by conflicting reports. Two high-profile studies published in 2018 reached diametrically opposed conclusions: Boldrini et al. [6] found abundant immature neurons in the dentate gyrus of older adults using optimized tissue handling and staining protocols, while Sorrells et al. [7] reported a near-complete absence; subsequent work and commentaries have emphasized that tissue handling, fixation duration, and marker strategy strongly influence detectability and may explain part of this divergence [6–10]. These opposing results reignited debate over the existence and significance of adult neurogenesis in humans.
Since the opposing 2018 reports, the field has increasingly converged on the view that methodological sensitivity, particularly post-mortem delay, fixation duration, antigen retrieval, and marker interpretation, can strongly determine whether immature-neuron proxies are detectable in human dentate gyrus tissue. Accordingly, recent syntheses have emphasized standardized tissue handling, multi-marker validation, and cross-modal triangulation (histology, birth-dating, and transcriptomics) to avoid false-negative or over-interpreted results. At the same time, thoughtful skepticism remains regarding the magnitude and functional relevance of any residual adult neurogenesis in humans, motivating balanced appraisal of both supportive and null findings [8, 11–15].
This review critically evaluates the principal lines of evidence supporting and opposing adult hippocampal neurogenesis (AHN), with a focus on methodological influences such as fixation protocol and antigen retrieval. It also integrates recent insights from single-cell transcriptomics and other omics technologies to offer a comprehensive synthesis of the current state of the field. We highlight areas of convergence and disagreement and propose future directions to resolve outstanding questions surrounding the biological and clinical significance of adult-born neurons in the human hippocampus.
One of the most convincing demonstrations of adult neurogenesis in humans has come from an unconventional source: Cold War-era nuclear testing. Spalding et al. [5] leveraged the sharp rise and gradual decline of atmospheric carbon-14 (14C) levels caused by mid-20th century nuclear bomb tests, the so-called “bomb pulse”, as a timestamp for cell birth. Because dividing cells integrate atmospheric 14C into their DNA, the amount of 14C in a cell’s genome reflects the atmospheric concentration at the time of the last cell division. Spalding and colleagues isolated neuronal nuclei from postmortem human hippocampi and measured 14C content to infer each neuron’s birthdate.
Strikingly, they estimated that roughly 700 new neurons are added to the adult dentate gyrus per day, amounting to ~35% turnover of dentate granule cells over the human lifespan. This directly challenged earlier assertions that primates generate essentially no new neurons after birth [16] and provided the first population-level quantitative evidence supporting AHN in humans.
The key inference highlighted by Spalding et al. [5] is summarized for the reader’s convenience in Figure 1, with interpretive notes in the caption. In brief, radiocarbon measurements showed that neuronal 14C levels were elevated relative to the atmospheric decay curve in adult samples, consistent with ongoing DNA synthesis well into adulthood. From these data, the authors modeled an annual turnover rate of approximately 1.75% of dentate gyrus neurons, implying the coexistence of a large developmentally generated population and a smaller continuously renewing pool. A major strength of radiocarbon birth-dating is that it objectively estimates cell “birth” without relying on transient immunolabels or cell-cycle markers, and it integrates neurogenesis activity over long time windows (capturing cumulative addition over the lifespan), thereby avoiding some pitfalls of snapshot histology that may miss rare or transient cell populations.
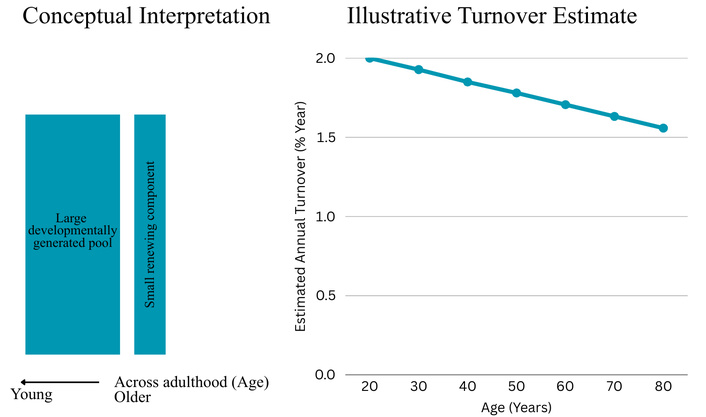
Summary of the key inference from Spalding et al. [5] radiocarbon birth-dating. This figure is a simplified representation created by the author of this review based on the data and conclusions reported in Spalding et al. [5]. It highlights the central interpretation: radiocarbon-derived neuronal “birth dates” are consistent with a largely developmentally generated dentate granule-cell population alongside a smaller renewing component persisting into adulthood. The purpose is to provide a reader-facing visual reference for the pivotal result discussed in the text; it is not a verbatim reproduction of the original figure and should be interpreted in conjunction with the methodological assumptions described by Spalding et al. [5]. Because the original figure is copyrighted, we provide an author-created summary to allow direct reader engagement with the central inference without reproducing the published artwork.
However, radiocarbon dating also comes with important caveats. It assumes 14C uptake and retention are uniform across cell types and individuals, which may not strictly hold true due to metabolic differences or DNA repair processes. There is also limited spatial resolution; the technique cannot localize new neurons to specific hippocampal subregions or distinguish neuronal subtypes. Additionally, only individuals born before or during the period of atomic bomb testing (roughly pre-1970s) have the distinctive 14C signatures needed for this analysis, restricting the applicability to older cohorts. Despite these limitations, radiocarbon birth-dating remains a cornerstone because it integrates DNA synthesis over the lifespan and provides population-level estimates that are broadly congruent with later cross-method discussions in the field [5, 8, 9]. Alternative explanations like adult DNA repair were considered but deemed insufficient: modeling shows that unrealistically extensive DNA repair would be required to account for the 14C levels observed in hippocampal neurons [8]. Thus, radiocarbon dating offers strong, population-level support for the existence of adult neurogenesis in humans. Its quantitative estimates (hundreds of new neurons per day in mid-adulthood) set a benchmark that immunohistochemical and other approaches have attempted to corroborate or refute.
While radiocarbon dating provided indirect but integrative evidence of neuron turnover, immunohistochemistry offers a more direct look at newborn cells in situ. Numerous studies have examined postmortem human brain tissue for cells expressing markers of neurogenesis, including proliferative markers [e.g., Ki-67, proliferating cell nuclear antigen (PCNA), minichromosome maintenance protein 2 (MCM2)], neural stem cell markers (e.g., SOX2, Nestin), and immature neuron markers such as doublecortin (DCX) and polysialylated neural cell adhesion molecule (PSA-NCAM). The immunohistochemical evidence has been mixed, with some reports clearly identifying putative new neurons in adult humans and others failing to detect them. Crucially, these discrepancies appear to stem largely from technical factors in tissue processing and analysis, as discussed below.
Using refined immunohistological methods, several groups have presented evidence that neurogenesis persists in the adult (and even elderly) human hippocampus. One influential study by Boldrini et al. [6] analyzed autopsy hippocampal samples from 14 neurologically healthy individuals spanning 14 to 79 years of age. Boldrini et al. [6] used rapid autopsy handling with optimized immunohistochemistry (see “Key methodological variables” below) and reported DCX/PSA-NCAM-positive immature neurons across adult ages. Within the dentate gyrus subgranular zone (SGZ) (the niche where new granule cells originate), Boldrini et al. [6] found abundant immature neurons at all adult ages.
Even in the oldest subjects (late 70s), they quantified roughly 2,000–3,000 DCX-positive young neurons per cubic millimeter of dentate gyrus, only modestly fewer (~30% less dense) than in adolescents. They also observed a small but consistent population of actively proliferating cells (around 10 Ki-67+ progenitor cells per mm2) in both young and old brains. These data indicate that cell division and neuronal maturation continue in the hippocampus even in the seventh and eighth decades of life. Notably, the densities of DCX+ cells reported by Boldrini et al. [6] align well with the independent turnover rates estimated by the 14C dating method (~1–2% of neurons renewing annually), lending cross-validation between cellular and isotopic approaches.
Boldrini et al.’s [6] findings corroborated and extended earlier partial evidence. For instance, Knoth et al. [4] had reported DCX+ cells co-expressing other immature neuron markers in adults up to 65 years old, though with an age-related decline. More recently, Tobin et al. [17] examined hippocampi from individuals with an exceptionally high mean age (~90 years) and still detected numerous neural progenitor cells (Nestin+/SOX2+) and DCX+ neuroblasts in these nonagenarians. Intriguingly, Tobin et al. [17] also noted that among their elderly cohort, those with mild cognitive impairment or Alzheimer’s pathology tended to have fewer new neurons, whereas individuals with higher counts of DCX+ cells performed better on cognitive tests. This suggests a possible link between retained neurogenesis and cognitive resilience in old age. Taken together, such studies firmly indicate that neurogenesis does not shut down completely in middle age as once feared; on the contrary, healthy older adults retain a reservoir of neural stem/progenitor cells and continue to generate new granule neurons, albeit at lower levels than in youth.
Further evidence came from Moreno-Jiménez et al. [12], who performed an incisive analysis of how tissue processing affects neurogenesis detection. By shortening the postmortem interval to under 3 hours and limiting immersion formalin fixation to ~2 hours for small hippocampal tissue blocks, they found a threefold higher yield of DCX+ cells in adult hippocampus compared to more conventionally processed tissue [11] (see “Key methodological variables” for how fixation/PMI interact with tissue block size and antigen detectability).
Importantly, these short intervals are not a universal ‘standard’ for all human hippocampal specimens; they are feasible when the hippocampus is rapidly dissected into small blocks (thin slabs), so fixative penetration is fast. Fixation time scales with tissue thickness and density because formaldehyde penetrates from the surface inward; thicker blocks generally require longer immersion to ensure uniform fixation, which can preserve morphology but may reduce immunoreactivity for sensitive epitopes such as DCX unless antigen retrieval is optimized. In practice, ‘optimal’ protocols are determined empirically for a given tissue type and block size by balancing (i) adequate fixation (morphology preservation and reduced autolysis) against (ii) antigen preservation (minimizing epitope masking), typically using pilot blocks, internal positive-control regions, and systematic testing of fixation duration and antigen-retrieval conditions [12].
In a cohort of individuals aged 40 to 85, these optimal conditions revealed 2,000–3,500 DCX-positive cells per mm3 of dentate gyrus, again indicating thousands of immature neurons persisting even in late life. Notably, Moreno-Jiménez et al. [12] also examined age-matched patients with Alzheimer’s disease (AD) and found a sharp drop (50–75% lower) in DCX+ and SOX2+/Tbr2+ progenitor cell densities relative to healthy controls. This drop in neurogenesis correlated with the severity of AD pathology, suggesting that neurodegenerative conditions can significantly impair the already modest neurogenic activity in older humans. The AD finding is important because it directly links diminished hippocampal neurogenesis to human disease: impaired generation of new neurons may contribute to cognitive deficits in AD, or conversely, AD pathology may incapacitate the neurogenic niche. In either case, it underscores that adult-born neurons, while few, could play roles in brain health that become apparent under stress or disease. In summary, multiple immunohistochemical studies employing careful protocols have converged on the conclusion that neurogenesis does persist in the adult human hippocampus throughout aging, and that these new neurons can be visualized and quantified given appropriate techniques. The typical picture is one of gradually declining but non-zero neurogenesis: an “old trickle” of new neurons that might support plasticity and repair in the aging brain.
Countering the above evidence, a few studies, most prominently Sorrells et al. [7], have reported essentially no detectable neurogenesis in adult humans beyond childhood. Sorrells et el. [7] examined 59 human hippocampal specimens ranging from neonates to 77-year-olds, using standard fixation (often several days in formalin) followed by DCX immunostaining. Their results were striking: abundant DCX+ immature neurons were seen in neonates and young children, but the numbers fell off sharply with age. By adulthood, DCX-positive cells were exceedingly rare: fewer than a dozen per mm3 in middle-aged samples, and in individuals over 50, Sorrells et al. [7] reported zero DCX+ neurons. On this basis, they concluded that neurogenesis in the human hippocampus wanes to an undetectable level in adults, essentially ceasing after early adolescence. Coming from a respected neuroanatomy lab, this publication in Nature generated enormous controversy, as it directly contradicted the multiple studies finding ongoing neurogenesis in adult humans. It also seemed inconsistent with the radiocarbon evidence that had indicated lifelong neuron addition.
In dissecting why Sorrells et al. [7] failed to see adult neurogenesis while others did, several critical methodological issues emerged. First, the tissue in that study had prolonged fixation times, often weeks in formalin, which can reduce the detectability of sensitive epitopes such as DCX and PSA-NCAM and thereby increase the risk of false-negative detection when the expected signal is sparse (as in adults), even if neonatal samples still show strong positivity [8, 9].
A potential apparent inconsistency is that the same study reported the detection of neurogenesis-associated markers in neonatal samples processed under the same general protocol. This does not, however, rule out fixation-driven false negatives in adult tissue: neonatal dentate gyrus contains a far higher density of immature neurons and typically much stronger DCX/PSA-NCAM signal, providing substantially greater ‘signal-to-noise’ and dynamic range. In contrast, any adult neurogenesis signal (if present) is expected to be sparse and lower-intensity, making it more vulnerable to epitope masking, background autofluorescence, and thresholding decisions after prolonged immersion fixation. Thus, a protocol can remain sufficient to detect robust neonatal DCX positivity while still under-detecting low-abundance adult DCX+ cells, especially when fixation is prolonged, or antigen retrieval is not optimized for sensitive epitopes [8, 9].
These fixation/PMI and antigen-retrieval dependencies, and the evidence that prolonged fixation can suppress DCX/PSA-NCAM detectability while optimized handling improves sensitivity, are synthesized in “Key methodological variables” below (including recommended controls and multipronged validation).
Secondly, over half of Sorrells et al.’s [7] adult cases had significant neurological disease (such as epilepsy, chronic seizures, or stroke). Chronic epilepsy and other conditions are known to alter hippocampal cytoarchitecture and could disrupt or deplete the neurogenic niche. It’s possible that some of Sorrells’ older “control” brains [7] were not truly representative of healthy aging. By inadvertently sampling pathological or treatment-affected hippocampi, the study may have been further biased toward finding little neurogenesis. Third, Sorrells et al. [7] relied on a single marker (DCX) at a single time-point per individual. As critics pointed out, detecting neurogenesis based on one transient marker is inherently precarious: any variability in the expression or detection of that marker can lead to false negatives. In contrast, techniques like 14C birth-dating integrate DNA synthesis over decades and are not subject to the narrow time window of marker expression. Thus, a failure to find DCX+ cells in adults does not prove neurogenesis is absent; it may simply reflect the limitations of the chosen method.
In light of these considerations, the ‘negative’ findings of Sorrells et al. [7] are often interpreted as highlighting how methodological factors (post-mortem delay, fixation, marker choice, and cohort characteristics) can drive false-negative detection in adult tissue, although debate continues regarding the magnitude and functional relevance of any residual adult neurogenesis [8–10]. The study inadvertently highlighted how not to search for new neurons in postmortem tissue: i.e., with long postmortem delays, prolonged fixation, and single-marker assays [7]. Subsequent commentary stressed that more rigorous protocols are required to capture the sparse, delicate population of adult-born neurons in humans [10]. In fact, another group led by Cipriani et al. [18] had published similar findings of very low DCX+ cell counts in adult hippocampi fixed for long periods. By contrast, studies using multiple markers and fresher tissue consistently find many immature neurons even in adults [6, 12]. In sum, the lack of DCX labeling in Sorrells’ adult samples [7] likely reflected technical shortcomings rather than a complete biological absence of neurogenesis. This does not fully resolve the debate: a few experts continue to question whether the level of neurogenesis in adult humans is functionally significant [10], but it shifts the discussion toward improving detection methods. The episode underscored that demonstrating adult neurogenesis in humans is challenging, and negative evidence must be interpreted with caution, given the sensitivity of immature neuron markers to tissue handling.
Several lines of critique caution against interpreting the existing literature as evidence for robust AHN in humans. First, some negative studies argue that true neuron birth may decline so steeply after childhood that remaining events are below reliable detection in typical postmortem datasets, especially when relying on transient protein markers [7, 16].
Second, even when DCX/PSA-NCAM-positive cells are detected, skeptics emphasize that marker expression alone does not prove recent neuron birth: these proteins can reflect immature-like phenotypes, prolonged maturation states, or plasticity-related re-expression rather than de novo neurogenesis, particularly in long-lived primate circuits [10, 16].
Third, tissue and cohort heterogeneity can bias both positive and negative results. Variations in agonal state, postmortem delay, fixation, comorbid disease, and medication exposure can suppress proliferation or reduce marker detectability, while selective inclusion of unusually “high-signal” cases could inflate estimates [7–9].
Fourth, methodological critiques apply in both directions: while prolonged fixation can reduce sensitivity (risking false negatives), extremely short fixation or aggressive antigen retrieval can raise concerns about non-specific staining or interpretation thresholds when expected signals are rare. Thus, convincing inference increasingly requires multi-marker validation, anatomical localization (SGZ), and convergence across orthogonal methods rather than reliance on any single readout [8–10].
Taken together, these arguments support a cautious interpretation: adult human hippocampal neurogenesis, if present, is likely low-level and variable across individuals, and current evidence is stronger for the existence of a small residual process than for a large, functionally dominant neuron-addition program [8, 10].
Because methodological issues (post-mortem interval, fixation, antigen retrieval, tissue quality, marker panels, and analysis thresholds) recur across both positive and negative studies, they are synthesized here as a unified framework. Subsequent sections refer back to this framework rather than repeating method details.
The stark contrast between studies like Boldrini/Moreno-Jiménez et al. (finding thousands of new neurons) [6, 12] and Sorrells et al. (finding virtually none) [7] prompted a deeper examination of methodological factors that can make or break the detection of neurogenesis markers. A consensus emerged that tissue processing protocols are a decisive factor. Kempermann et al. [8], a consortium of leading neurogenesis researchers, published a perspective synthesizing what technical lessons could be learned from the discordant studies.
They emphasized that postmortem delay and fixation duration are critical: DCX and other immature neuron markers begin to degrade within hours after death, and excessive fixation (even > 24–48 hours) can obscure these epitopes. In one analysis, up to 70% of DCX immunoreactivity was lost when tissue was fixed for weeks compared to a few hours. Kempermann et al. [8] pointed out that when human autopsy tissue is handled with rapid processing (< 12 hours postmortem, brief fixative exposure, and antigen retrieval steps), numerous DCX+ and PSA-NCAM+ cells are consistently observable in the dentate gyrus, on the order of hundreds to thousands per cubic millimeter, even in older adults.
This suggests that earlier failures were likely due to suboptimal methods rather than an absence of the cells themselves. Accordingly, the authors called for standardized protocols across laboratories: for example, limiting fixation times, using heat-mediated antigen retrieval, and including positive control tissues to ensure markers are detectable.
Another technical consideration is the use of multiple complementary markers and approaches. Because no single marker definitively proves “new neuron” identity in humans, a combination of evidence is preferable. For instance, co-labeling a cell with a proliferative marker (Ki-67 or a cell-cycle protein) and a neuronal maturation marker (DCX or NeuN) provides stronger evidence that it is a newly generated neuron. Several earlier studies that reported adult neurogenesis did so by demonstrating such co-expression or by culturing neurospheres from adult tissue, indicating the presence of mitotically active progenitors. In contrast, Sorrells et al. [7] looked at DCX alone. The Kempermann et al. [8] report and others recommend a “multipronged approach”: combine radiocarbon dating for quantitation, optimized immunohistochemistry for visualization, fluorescence-activated cell sorting (FACS) of neuronal nuclei for lineage analyses, in vitro neurosphere assays to test for proliferative potential, and even electrophysiological validation of immature neurons if possible. By converging evidence from independent methods, one can more definitively confirm the presence (or absence) of neurogenesis. This integrated strategy has not yet been fully applied in a single human study, but it represents a path forward to resolve remaining doubts.
The major markers used to infer AHN and their key interpretive caveats are summarized in Table 1.
Common adult hippocampal neurogenesis-related markers and key caveats.
| Marker | What is it used to infer | Major caveats (alternative explanations/confounds) | Best-practice interpretation/controls |
|---|---|---|---|
| DCX (doublecortin) | Immature neurons/neuronal lineage progression (late progenitor to immature neuron stage) | Epitope sensitivity to post-mortem interval and prolonged fixation; low-level expression can be missed (false negatives). Can also reflect prolonged maturation or plasticity-state changes rather than recent birth. | Use with additional markers (e.g., PROX1, NEUROD1, calretinin) and anatomical localization to SGZ/GCL. Report fixation/PMI and antigen retrieval. Validate with multiple antibodies and negative controls. |
| PSA-NCAM | Structural plasticity; often used as an ‘immature neuron’ proxy (especially with DCX) | Not specific to newborn neurons; can label plasticity-related states and some mature interneurons. Signal depends on tissue handling and antibody/thresholding. | Avoid using alone; interpret as a plasticity marker unless co-localized with neuronal lineage markers and appropriate SGZ distribution. Include isotype controls and evaluate non-SGZ staining. |
| Ki-67 | Active cell cycling (proliferation) | Marks all cycling cells (not neuron-specific); low event rate in adult human DG increases sampling error. Can be affected by agonal state, hypoxia, inflammation, and tissue quality. | Combine with progenitor markers (SOX2, Nestin) and lineage markers; quantify regionally (SGZ) and report counting frame and total cells sampled. |
| MCM2/Other MCM proteins | Licensing of DNA replication; proliferation competence | Not neuron-specific; can label cells poised for division. Like Ki-67, very sparse events in adult human tissue. | Use with cell-type markers and SGZ localization; interpret as evidence of proliferation capacity, not neuron production by itself. |
| PCNA | DNA replication/repair-associated protein; sometimes used for proliferation | Less specific than Ki-67; can be elevated during DNA repair and in non-dividing contexts, increasing false positives. | Prefer Ki-67/MCM2 for proliferation; if used, pair with additional proliferation markers and stringent controls. |
| SOX2 | Neural stem/progenitor-like state | Also expressed outside the DG niche, depending on context, it does not prove ongoing neurogenesis; affected by tissue quality. | Interpret as progenitor pool indicator; combine with proliferation markers (Ki-67/MCM2) and SGZ localization. |
| Nestin | Neural progenitor/radial glia-like morphology (in some contexts) | Not exclusive to neural stem cells; expression depends on antibody and fixation; may label reactive glia after injury. | Use with SOX2 and morphology (radial processes) plus SGZ location; consider injury/inflammation status. |
| TBR2 (EOMES) | Intermediate progenitors (transit-amplifying) | Very rare in adult human DG; detection sensitive to tissue handling and section sampling; not neuron birth by itself. | Use to support the presence of progenitor stages when co-detected with proliferation markers and SGZ confinement. |
| NEUROD1 | Neuronal differentiation program (early neuronal lineage commitment) | Can be transient and sparse; transcript/protein detection varies by assay sensitivity; interpretation depends on co-expression patterns. | Use as part of a lineage panel (with DCX/PROX1) and anatomical context; corroborate with orthogonal methods. |
| PROX1 | Dentate granule cell lineage identity (granule-cell fate) | Lineage identity marker, not an ‘age’ marker; does not distinguish newborn vs. mature granule cells on its own. | Use to confirm granule-cell identity of DCX+ cells; combine with maturity markers (calbindin) and stage markers (calretinin/DCX). |
| Calretinin | Immature to early post-mitotic neuron stage (often used with DCX) | Not exclusive to newborn granule cells; can be expressed in interneurons and varies by region; sparse signals are prone to sampling error. | Interpret as supportive when co-localized with DCX/PROX1 in SGZ/GCL; include cell-type disambiguation where needed. |
| Calbindin | Mature granule cell phenotype | Reduction can reflect dematuration or stress/injury effects; not direct evidence for or against neuron birth. | Use to define maturity gradients and to interpret ‘immature-like’ states; avoid using as a sole proxy for neurogenesis. |
| snRNA-seq ‘immature neuron’ clusters (e.g., DCX/NEUROD1/PROX1 programs) | Rare immature granule-cell states; potential ongoing neurogenesis | Dropout and sparse capture reduce sensitivity; nuclei assays under-sample cytoplasmic transcripts; clustering thresholds, doublets, and ambient RNA can blur rare states. Immature-like signatures can also reflect prolonged maturation, dematuration, or plasticity. | Require convergence: progenitor/proliferation signatures, SGZ localization (spatial), and/or independent birth-dating. Report QC, doublet filtering, batch correction, and sensitivity analyses. |
| Spatial transcriptomics neurogenic signatures | Anatomical mapping of gene programs to DG niches | Lower sensitivity on some platforms; many technologies capture multi-cell spots, causing signal dilution; deconvolution assumptions can bias rare-cell inference. | Pair with immunohistochemistry and high-resolution segmentation; interpret rare signatures conservatively; report platform resolution and detection thresholds. |
SGZ: subgranular zone; PSA-NCAM: polysialylated neural cell adhesion molecule; DG: dentate gyrus; MCM2: minichromosome maintenance protein 2; PCNA: proliferating cell nuclear antigen; snRNA-seq: single-nucleus RNA sequencing; QC: quality control.
A key interpretive caveat is that expression of DCX, PSA-NCAM, and “immature granule cell” transcriptomic signatures can, in some contexts, reflect dematuration or prolonged plasticity of existing neurons rather than de novo neuron birth. In rodents, chronic fluoxetine can shift mature dentate granule cells toward a more immature-like phenotype, including reduced expression of mature markers (e.g., calbindin) and increased expression of immature-associated features, i.e., “dematuration”. Such state changes could, in principle, increase DCX/PSA-NCAM detectability without a proportional increase in newly generated neurons. In addition, PSA-NCAM is a broader plasticity-associated molecule that can be expressed outside canonical neurogenic contexts (e.g., in subsets of mature interneurons), reinforcing that PSA-NCAM positivity alone is not definitive evidence of recent neuron birth [19–21].
Similar interpretive caution applies to transcriptomics: clusters enriched for DCX/NEUROD1/PROX1 and related programs can be consistent with ongoing neurogenesis, but they can also be influenced by (i) prolonged maturation programs (particularly in long-lived primate circuits), (ii) injury/stress-related plasticity states, and (iii) technical factors in nuclei isolation and clustering that can blur boundaries between rare immature neurons and plasticity-shifted mature populations. Accordingly, the most convincing inference is obtained when “immature” signatures converge with orthogonal indicators of recent cell birth (e.g., cell-cycle/proliferation signatures, progenitor-state markers, anatomical restriction to SGZ-associated niches, and/or independent birth-dating). In practice, this argues for reporting multi-marker panels and explicitly separating claims of ‘immature-like state’ from claims of ‘new neuron production’ when interpreting human tissue [22, 23].
In summary, a common interpretation is that methodological differences contribute substantially to discrepancies in detecting AHN, though uncertainty remains regarding the extent and functional relevance of the phenomenon in humans [8–10]. Rapid autopsy protocols, proper antigen preservation, and multi-marker confirmation have consistently unveiled populations of new neurons in adult humans. By contrast, prolonged fixation or reliance on a single marker is likely to miss these cells. Thus, to answer the question “Do new neurons grow in the adult human hippocampus?” one must ensure the search is conducted under conditions where those neurons can actually be seen. The controversy has, in effect, driven improvements in how human tissue is handled, moving the field toward a more reliable toolkit for studying neurogenesis in our own species.
Beyond traditional microscopy, recent advances in single-cell genomics and transcriptomics have opened new avenues to detect and characterize adult-born neurons in humans. These techniques can reveal molecular signatures of neurogenesis that might not be evident from protein markers alone. For example, single-nucleus RNA sequencing (snRNA-seq) can profile thousands of individual cells from postmortem brain tissue, potentially identifying rare cell types like newborn neurons by their gene expression profiles.
Limitations of single-nucleus/single-cell and spatial transcriptomic approaches should also be considered, particularly when inferring rare populations such as immature neurons. First, sparse capture leads to “dropout” (failure to detect transcripts that are present), which reduces sensitivity for lowly expressed genes and can fragment marker programs across cells; this is especially consequential when conclusions depend on a small set of transcripts (e.g., DCX/NEUROD1/PROX1). Second, nuclei-based assays (snRNA-seq) under-sample cytoplasmic transcripts and can be biased by cell-type differences in nuclei isolation, potentially distorting estimated abundances of fragile or rare states. Third, doublets, ambient RNA, batch effects, and clustering/threshold choices can blur boundaries between rare immature-neuron clusters and plasticity-shifted mature populations, motivating conservative interpretation and validation across modalities. Spatial transcriptomics partially restores anatomical context, but sensitivity can be lower than dissociation-based assays, and many platforms capture mixed-cell “spots” rather than true single cells, so rare-cell signatures may be diluted and require careful segmentation/deconvolution and histological cross-validation [24, 25].
In a landmark study, Zhou et al. [23] applied snRNA-seq to adult human hippocampal samples across a wide age range. They discovered discrete clusters of dentate gyrus cells expressing canonical immature neuron genes (such as DCX, PROX1, and NEUROD1) even in older adults. These cells’ transcriptomes resembled those of young granule neurons in rodents, confirming that a trickle of new neurons continues to be generated and matures over time in humans. Interestingly, the “immature neuron” gene signature persisted into advanced ages (the study included subjects in their 70s and 80s), although the proportion of such cells was low. The authors noted that the immature neurons in aged humans showed unique, prolonged maturation programs. For instance, continued expression of developmentally related genes well after cell birth suggests that the integration of new neurons in the human circuit might be a protracted process.
Building on these findings, Thompson et al. [26] combined single-nucleus sequencing with spatial transcriptomics to create an integrated atlas of the adult human hippocampus. Spatial transcriptomics allowed them to map where in the hippocampal tissue certain gene expression patterns were enriched. They identified localized “neurogenic niches” in the dentate gyrus, microregions enriched in transcripts associated with neural progenitors and immature neurons. These niches corresponded to the SGZ and areas around vasculature, aligning with known neurogenesis hotspots in animals. The spatial data also illustrated how microenvironmental context (e.g., proximity to blood vessels or specific glial cell types) can influence the differentiation trajectory of new neurons in humans. This kind of high-resolution mapping is helping to visualize the “hiding spots” of adult neurogenesis that are scattered in the human dentate gyrus.
Furthermore, time-course analyses are being applied to postmortem brains to infer dynamic changes. Ramnauth et al. [27] performed a spatiotemporal gene expression analysis on human dentate gyrus tissue from infancy through old age. They confirmed a dramatic drop-off in proliferative markers (like Ki-67) in early childhood, consistent with the idea that neurogenesis is highest in infancy and then declines, but importantly, they still detected persistent expression of immature neuron markers (DCX, PSA-NCAM, etc.) in adult and even elderly samples. This again supports the presence of a residual neurogenic process lasting throughout life, even if the proliferative activity becomes very low after early adulthood. The transcriptomic data also revealed age-related changes in the neurogenic niche, such as increasing expression of inflammatory and gliosis-related genes in the older dentate gyrus. Such changes hint that the aging microenvironment (e.g., more reactive glia, less growth factor support) might increasingly constrain neurogenesis with time: a hypothesis consistent with animal studies and with the reduced neurogenesis seen in neurodegenerative disease contexts.
In parallel, in vitro and ex vivo approaches continue to contribute evidence. Human hippocampal progenitor cells have been isolated from adult surgical or postmortem brain tissue and demonstrated the capacity to generate neurons in culture, indicating that neurogenic potential persists into adulthood [28]. There is also growing interest in non-invasive imaging to detect neurogenesis. While no current modality can directly visualize newborn neurons in humans, experimental MRI and magnetic resonance spectroscopy (MRS) techniques have been proposed, using cerebral blood volume or spectral metabolites as proxies for neurogenesis [29]. These techniques remain exploratory. However, the advent of PET tracers targeting neuroinflammation and synaptic density may provide indirect insights by revealing hippocampal microenvironmental changes that accompany new neuron integration [30, 31].
Overall, genomic technologies have reinforced the case that adult-born neurons exist in humans, and they are starting to characterize these cells in unprecedented detail [23, 26]. We now have molecular fingerprints of putative adult human neural progenitors and neuroblasts, which opens new possibilities for understanding how these cells are regulated.
It’s notable that even the most skeptical assessments have had to contend with this accumulating evidence. For instance, a recent commentary noted that if even a few new neurons are present, single-cell sequencing would be sensitive enough to detect them [10]. Indeed, these methods have essentially “found the needles in the haystack”, validating earlier histological observations.
As techniques continue to improve, we may detect even smaller or more transient waves of neurogenesis; for example, in response to exercise or antidepressant treatment in humans, as well documented in animal models [6, 32]. The integration of multi-modal evidence, from isotopic to histological to transcriptomic, now paints a consistent picture: the adult human hippocampus is not entirely quiescent but maintains a subdued trickle of neuron production, which modern methods can discern if we look closely enough.
If new neurons are indeed born in the adult human hippocampus, what are they good for? In rodents and other animals, AHN has been linked to specific brain functions, notably learning and memory, especially pattern separation and memory precision, as well as mood regulation under stress [33, 34]. For example, adult-born granule cells in mice have been shown to integrate into hippocampal circuits and contribute to distinguishing similar experiences and to the behavioral effects of antidepressants [35, 36]. It stands to reason that a similar process in humans, even if much smaller in scale, could influence cognition and emotion. New neurons might provide a trickle of plasticity that fine-tunes memory circuits or buffers stress and depression. However, directly testing the function of adult-born neurons in humans is challenging: we cannot experimentally ablate or label these cells in living people as is done in animal studies [8]. Thus, we must rely on correlative evidence and theoretical arguments.
One intriguing hint of functional relevance comes from the finding that older individuals with higher levels of neurogenesis markers tend to have better cognitive function. As noted, Tobin et al. [17] observed that among very old adults, those with more DCX+ cells had relatively preserved cognitive performance. Similarly, in AD patients, Moreno-Jiménez et al. [12] found that those with the greatest loss of neurogenic cells had the most severe cognitive impairments. While these studies are correlational, they suggest that adult-born neurons could play a role in maintaining certain aspects of memory or hippocampal function in the face of aging and neurodegeneration. It is conceivable that even a small number of new neurons might provide disproportionate benefits if they participate in critical circuit computations (for instance, encoding fine distinctions between memories or modulating mood-related feedback to the hypothalamus). Consistent with this, Martínez-Canabal et al. [37] recently demonstrated in a mouse model that adult-born hippocampal neurons are required for optimal cognitive flexibility. Specifically, the ability to update memories in a fear discrimination task was impaired when neurogenesis was blocked. Extrapolating cautiously, one might speculate that in humans, the few new neurons added each month contribute to our ability to adapt to new experiences, potentially affecting how we generalize or distinguish contexts in memory and how we cope with stress or emotional challenges.
Adult neurogenesis could also have implications for psychiatric and neurological diseases. Decreased hippocampal neurogenesis has been hypothesized to contribute to depression, and conversely, many antidepressant treatments and lifestyle factors like exercise increase neurogenesis in animal models [38, 39]. If adult neurogenesis exists in humans, even at low levels, it could represent a therapeutic target. Conditions such as major depression or chronic stress may suppress baseline neurogenesis, reducing hippocampal plasticity and resilience [40]. Preclinical studies in non-human primates show that stress reduces hippocampal neurogenesis [41], and some postmortem human data suggest diminished progenitor markers in depressed individuals, though this is confounded by treatment effects [6].
Conversely, interventions like exercise, environmental enrichment, or medications might stimulate proliferation or survival of new neurons in the adult human brain; a hypothesis still unproven but under active investigation [42]. The possibility that enhancing neurogenesis could aid cognition or mood in disorders such as depression, PTSD, or age-related memory decline is particularly intriguing. In AD, the presence of DCX-positive cells in aging, but their reduction in AD brains suggests retained but diminished neurogenic potential [12, 17]. This raises the question of whether bolstering endogenous neurogenesis could slow cognitive decline.
Traumatic brain injury (TBI) is a major global cause of disability across age groups and socioeconomic settings, and it provides a clinically relevant context in which endogenous neurogenesis could matter. Preclinical work consistently shows that TBI perturbs dentate gyrus neurogenesis in a complex, time-dependent manner: injury can trigger increased progenitor proliferation, yet also cause acute loss of immature neurons and/or aberrant integration (including ectopic migration and altered maturation), with outcomes influenced by injury severity and post-injury inflammation. These findings suggest that “more proliferation” after TBI is not automatically beneficial; the functional consequence depends on whether newborn neurons survive, integrate appropriately, and contribute to circuit repair rather than maladaptive plasticity [43–45].
Human evidence is limited but relevant. Postmortem analyses after TBI have reported increased proliferative/neurogenic marker expression in canonical neurogenic regions compared with non-injured controls, consistent with an injury-responsive neurogenic program that can persist beyond the acute period. While such observations do not prove that new neurons restore cognition in humans, they support the translational hypothesis that the adult neurogenic niche is injury-responsive and may be a modifiable target (e.g., via rehabilitation, exercise, enriched environments, or anti-inflammatory strategies) to improve hippocampal-dependent outcomes after TBI [46].
Skeptics argue that adult human neurogenesis may occur so infrequently as to be negligible [7, 47]. One evolutionary view posits that primates, including humans, minimized neurogenesis to preserve stable circuits for long-term learning, suggesting attempts to restore it may be biologically constrained [16]. Duque et al. [10] contend that extrapolating from rodent data has created unrealistic public expectations about neuron regeneration in humans. Even if a few neurons are added daily, their clinical relevance remains unclear unless they properly integrate and function.
Most experts now take a middle-ground view: adult human neurogenesis likely exists but plays a limited modulatory role [8]. While the absolute numbers are small, the strategic placement of these cells within hippocampal circuits may matter. They may fine-tune memory discrimination or encode temporal aspects of experience. This could be especially relevant to disorders marked by cognitive overgeneralization, such as PTSD or age-related memory interference [37]. Finally, examples from species with low neurogenesis but sophisticated cognition (e.g., dolphins, bats) suggest that neurogenesis is only one component of a complex cognitive toolkit.
In summary, while the biological role of adult neurogenesis in humans remains incompletely understood, accumulating evidence supports its persistence and potential significance. Future research will need to establish causal links between new neurons and function, ideally via noninvasive tracking or clinical trials of neurogenic enhancement strategies. Until then, it is plausible to consider adult-born neurons as subtle but meaningful contributors to human brain plasticity.
AHN remains debated, but the literature supports a more integrative interpretation: evidence is strongest for a low-level, variable process that persists beyond childhood, while uncertainty remains about its magnitude and functional impact.
Conclusion 1: Multiple lines of evidence are consistent with persistence, but not at the rodent-like scale. Radiocarbon birth-dating provides population-level estimates consistent with ongoing neuronal turnover across adulthood, while optimized immunohistochemistry has reported DCX/PSA-NCAM-positive immature neurons and progenitor-associated markers in adult and aged dentate gyrus. Transcriptomic atlases further identify rare immature granule-cell programs and niche-associated states across the lifespan. Together, these approaches support persistence, but the most conservative reading is that it is modest and heterogeneous.
Conclusion 2: Methodological sensitivity explains much of the conflict, but interpretation must be cautious in both directions. Discrepant outcomes are strongly shaped by post-mortem interval, fixation duration, tissue block size, antigen retrieval, tissue quality, marker panels, and analytic threshold; variables that can plausibly generate false negatives when signals are sparse. However, methodological concerns also apply to positive inference: rare marker-positive cells require rigorous controls, multi-marker corroboration, and transparent reporting to avoid overcalling.
Conclusion 3: “Immature” markers and transcriptomic signatures are not synonymous with new neuron birth. DCX/PSA-NCAM positivity and immature-like transcriptomic signatures can reflect neurogenesis, but credible alternative explanations remain, including prolonged maturation programs, dematuration, and plasticity-associated re-expression. The most persuasive inference, therefore, comes from convergence across orthogonal indicators of recent cell birth (e.g., proliferation/progenitor signatures, anatomically appropriate SGZ localization, and/or independent birth-dating), rather than from any single marker or modality.
Conclusion 4: The major unresolved questions are quantitative and functional, and they are testable, including in clinically important contexts such as TBI. Key open issues include (i) the true rate of AHN across individuals and contexts (healthy aging, neurodegeneration, psychiatric illness, and TBI), (ii) whether adult-born neurons reliably survive and integrate into human hippocampal circuits, and (iii) whether this process measurably contributes to cognition, affect, or recovery. Importantly, TBI is a clinically pressing setting in which hippocampal neurogenesis is consistently perturbed in preclinical models and injury-responsive neurogenic markers have been reported in humans, supporting the translational rationale for evaluating neurogenesis-targeting strategies (e.g., behavioral, rehabilitative, and anti-inflammatory approaches) as candidate modifiers of hippocampal-dependent outcomes after injury [43–46].
In sum, the field is moving beyond a binary “exists/does not exist” framing toward defining how much, under what conditions, and with what consequences. A conservative synthesis of current evidence is that AHN is real but modest, method-sensitive, and biologically variable; features that make it challenging to detect yet potentially relevant in disease- and injury-related contexts, including TBI.
14C: carbon-14
AD: Alzheimer’s disease
AHN: adult hippocampal neurogenesis
BrdU: bromodeoxyuridine
DCX: doublecortin
PSA-NCAM: polysialylated neural cell adhesion molecule
SGZ: subgranular zone
snRNA-seq: single-nucleus RNA sequencing
TBI: traumatic brain injury
ML: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. The author read and approved the submitted version.
The author declares that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 4128
Download: 30
Times Cited: 0
