Affiliation:
1Fondazione Paolo Procacci, 00193 Roma, Italy
2College of Medicine, University of Baghdad, Baghdad 10071, Iraq
Email: giuvarr@gmail.com
ORCID: https://orcid.org/0000-0002-3822-2923
Affiliation:
3Department of Internal Diseases Propaedeutics and Emergency Medicine, Faculty of Public Health in Bytom, Medical University of Silesia, 41-902 Bytom, Poland
ORCID: https://orcid.org/0000-0003-4754-3988
Affiliation:
4Department of Medical and Surgical Sciences and Translational Medicine, Sapienza University of Rome, 00189 Rome, Italy
ORCID: https://orcid.org/0000-0001-5228-3733
Explor Immunol. 2026;6:1003240 DOI: https://doi.org/10.37349/ei.2026.1003240
Received: January 06, 2026 Accepted: March 06, 2026 Published: March 15, 2026
Academic Editor: Shubhada Chiplunkar, Homi Bhabha National Institute (Deemed University), India
Pain is among the most prevalent and debilitating symptoms experienced by patients with cancer, significantly impairing functional status and overall quality of life. Its origin is multifactorial, involving direct tumor infiltration, treatment-induced tissue and neural injury, and immune-mediated inflammatory processes. This section examines the epidemiology of cancer-related pain, delineates its complex pathophysiology with particular focus on neuroimmune interactions, and reviews current pharmacological and interventional approaches for managing both acute and chronic pain in oncological settings. Advancing knowledge of immune-driven pain mechanisms holds promise for the development of more targeted, effective, and personalized analgesic strategies in cancer care.
Pain remains one of the most prevalent and burdensome problems encountered in oncology, affecting approximately 55% of patients undergoing active antineoplastic therapy, 66% of individuals with advanced-stage malignancies, and up to 80% of patients receiving palliative care [1]. Beyond the active treatment phase, approximately one-third of cancer survivors experience persistent, often chronic, and neuropathic pain long after treatment completion [2]. The etiology of cancer-related pain is multifactorial, arising from direct tumor invasion, nerve compression or infiltration, treatment-induced injury (e.g., surgical trauma, chemotherapy-induced neuropathy), and systemic inflammation [3]. Despite advances in cancer therapies and pain management protocols, adequate analgesic control is not achieved in 30–50% of patients [4]. Undertreated cancer pain is associated with functional impairment, sleep disturbances, emotional distress, and reduced adherence to oncologic treatment regimens, ultimately worsening clinical outcomes and quality of life [5].
Comprehensive pain management requires more than pharmacological optimization. Effective interventions should incorporate personalized, interdisciplinary models that integrate immunological, psychosocial, and disease-specific factors [6]. From a pathophysiological perspective, cancer pain is a complex and heterogeneous entity, comprising nociceptive, neuropathic, and inflammatory components that frequently coexist in the so-called mixed pain. Nociceptive pain typically results from tissue injury or mechanical compression by the tumor mass, while neuropathic pain commonly arises from direct neural infiltration or injury to somatosensory pathways secondary to cancer progression or its treatment [5]. Tumor cells release various algogenic mediators, protons (H+), adenosine triphosphate (ATP), bradykinin, prostaglandins, and endothelins, which activate or sensitize peripheral nociceptors through ion channels and G-protein-coupled receptors, such as TRPV1 (transient receptor potential vanilloid 1), ASIC3 (acid-sensing ion channel 3), and P2X receptors, on sensory neurons [7, 8]. Concurrently, the tumor microenvironment (TME) sustains a chronic inflammatory state enriched with tumor-associated macrophages (TAMs), neutrophils, and T lymphocytes, which secrete pro-inflammatory cytokines (e.g., IL-1β, IL-6, TNF-α) and chemokines (e.g., CCL2, CXCL1), further enhancing nociceptive signaling and contributing to peripheral sensitization [9]. Chemotherapy-induced peripheral neuropathy (CIPN) represents an example of a major iatrogenic cause of cancer-related neuropathic pain. Neurotoxic agents such as paclitaxel disrupt axonal transport, impair mitochondrial function, and induce neuroimmune activation within the dorsal root ganglia (DRG) and spinal cord [10, 11]. Sustained nociceptive input and neuroinflammation promote central sensitization through microglial and astrocytic activation, resulting in hyperalgesia, allodynia, and pain chronification. The immune system plays an integral role in this pathophysiology, mediating neuroimmune crosstalk that drives persistent cancer pain [12]. This narrative review highlights the epidemiology, pathophysiology, and pharmacological management of cancer pain, with an emphasis on immune mechanisms contributing to its development and chronification.
This narrative review was conducted following the SANRA criteria for narrative review articles [13]. A literature search was conducted to identify relevant studies examining the epidemiology, pathophysiology, and management of cancer pain, with particular emphasis on neuroimmune interactions and immunomodulatory mechanisms. A systematic search was performed in PubMed/MEDLINE, Scopus, and Web of Science databases up to December 2025. The search strategy employed combinations of Medical Subject Headings (MeSH) terms and free-text keywords including: (“cancer pain” OR “oncologic pain” OR “malignant pain” OR “tumor pain”) AND (“neuropathic pain” OR “nociceptive pain” OR “inflammatory pain” OR “mixed pain” OR “nociplastic pain”) AND (“immunity” OR “immune system” OR “inflammation” OR “cytokines” OR “chemokines” OR “neuroinflammation” OR “neuroimmune” OR “immune cells” OR “macrophages” OR “T cells”) AND (“pathophysiology” OR “mechanisms” OR “sensitization” OR “nociceptors” OR “tumor microenvironment”) AND (“pharmacological treatment” OR “analgesics” OR “opioids” OR “adjuvant therapy” OR “immunomodulation” OR “pain management”). Additional searches were conducted focusing on specific therapeutic interventions and emerging approaches: (“chemotherapy-induced peripheral neuropathy” OR “CIPN”), (“immune checkpoint inhibitors” OR “immunotherapy”), (“central sensitization” OR “peripheral sensitization”), and (“interventional analgesia” OR “neuromodulation”). Reference lists of identified articles, recent systematic reviews, and clinical practice guidelines were manually screened to identify additional relevant publications.
Given the narrative nature of this review and its aim to provide a comprehensive overview of current knowledge regarding cancer pain mechanisms and management, we included multiple study types to capture the available evidence. Eligible publications comprised: original research articles including randomized controlled trials, observational studies, cohort studies and case-control studies examining cancer pain epidemiology, pathophysiology, or treatment outcomes; systematic reviews and meta-analyses synthesizing evidence on cancer pain mechanisms or therapeutic interventions; mechanistic studies including laboratory investigations, animal models, and translational research elucidating molecular and cellular pathways in cancer pain; clinical practice guidelines and consensus statements from major professional organizations; and narrative reviews providing expert synthesis of specific cancer pain topics. Priority was given to peer-reviewed publications in English from high-impact journals, with emphasis on recent evidence published within the last 5 years (2020–2025) to reflect contemporary understanding and practice. However, seminal earlier publications establishing foundational concepts in pain neurobiology, neuroimmunology, and cancer pain mechanisms were also included when they provided essential background or remained the primary reference for specific concepts. Studies were excluded if they focused exclusively on pediatric populations, non-malignant chronic pain without a cancer context, or were case reports with fewer than three patients. Conference abstracts, dissertations, and non-peer-reviewed sources were generally excluded unless they reported unique data not available in peer-reviewed publications.
Given the narrative approach, formal quality assessment tools for systematic reviews were not employed. Findings were synthesized narratively and organized thematically according to the review’s main objectives: epidemiology, pathophysiological mechanisms, neuroimmune interactions, and pharmacological management strategies. During the evaluation of the articles, we found a substantial heterogeneity across studies regarding cancer types, disease stages, patient populations, pain assessment instruments, treatment regimens, and outcome measures. In fact, cancer pain encompasses diverse clinical entities spanning multiple tumor types, anatomical locations, and disease trajectories, which inherently limits the ability to draw uniform conclusions. This narrative approach permits comprehensive coverage of a complex, multifaceted topic but lacks the systematic reproducibility and quantitative synthesis of formal systematic reviews. The inclusion of diverse study types precludes any statistical meta-analysis. Furthermore, the predominance of studies from high-income countries may limit generalizability to resource-limited settings where cancer pain management resources and practices differ substantially.
Pain remains one of the most prevalent symptoms in oncology [1]. Among cancer survivors, nearly one-third report ongoing, often neuropathic, pain months or years after treatment completion [14, 15]. The multifactorial etiology includes tumor invasion, nerve injury, treatment-related neurotoxicity, and systemic immune activation [3]. Despite the evolution of clinical guidelines and multimodal analgesic strategies, pain remains inadequately controlled in 30–50% of patients, particularly those with neuropathic or mixed pain syndromes and in low-resource healthcare settings [4]. Undertreated cancer pain contributes to physical disability, disturbed sleep, psychological distress, and reduced treatment compliance, all of which negatively impact clinical outcomes and quality of life [5]. Addressing these challenges requires optimized pharmacotherapy and broader implementation of interdisciplinary, mechanism-based pain management models that integrate immunological and psychosocial considerations [16].
Pain associated with cancer can be broadly categorized into four principal types: nociceptive, neuropathic, inflammatory, and mixed pain. Each type is characterized by distinct pathophysiological mechanisms, receptor involvement, and anatomical sites of origin [17].
Nociceptive pain arises from the activation of nociceptors, specialized peripheral sensory neurons that detect actual or potential tissue damage. These high-threshold sensory neurons express various receptors that respond to mechanical, thermal, and chemical stimuli [18]. In the cancer context, nociceptive pain typically results from direct tumor invasion, causing tissue destruction, mechanical compression of adjacent structures, or stretching of visceral organs [19]. Key receptors involved in nociceptive pain include TRPV1, which responds to heat and acidic conditions; ASICs, particularly ASIC3, activated by tissue acidosis; and purinergic receptors (P2X2/3 and P2Y receptors) that detect ATP released from damaged cells [20]. Additional nociceptive receptors include bradykinin receptors (B1 and B2), prostaglandin receptors (EP and IP receptors), and mechanoreceptors such as Piezo2 channels [21]. Nociceptive pain occurs predominantly in somatic tissues, including bone (especially common in metastatic disease), muscles, skin, and connective tissues, as well as in visceral organs such as the liver, pancreas, and gastrointestinal tract [22].
Neuropathic pain develops as a direct consequence of injury or dysfunction affecting the somatosensory nervous system itself, rather than from nociceptor activation [23]. In oncology, neuropathic pain commonly results from direct tumor infiltration or compression of peripheral nerves, nerve roots, or the spinal cord, as well as from treatment-related nerve damage, including CIPN, radiation-induced plexopathy, and surgical nerve injury [24]. The pathophysiology involves aberrant neuronal excitability, ectopic discharge generation, altered ion channel expression, and central sensitization mechanisms [25]. Neuropathic pain is mediated by changes in voltage-gated sodium channels (particularly Nav1.7, Nav1.8, and Nav1.9), which become upregulated in injured neurons and contribute to spontaneous firing and mechanical allodynia [26]. Voltage-gated calcium channels, especially the N-type (Cav2.2) and T-type channels, play crucial roles in neurotransmitter release and pain signal transmission [27]. Additionally, dysregulation of potassium channels reduces the neuronal threshold for action potential generation [28].
Inflammatory pain represents a protective response to tissue injury and inflammation, mediated by complex interactions between the immune system and sensory neurons [19]. In the TME, inflammatory pain is driven by immune cell infiltration, including TAMs, neutrophils, mast cells, and lymphocytes, which release numerous pro-inflammatory mediators [29]. These mediators sensitize and activate nociceptors through specific receptors and signaling pathways [30]. Primary receptors involved in inflammatory pain include Toll-like receptors (TLRs), particularly TLR4, expressed on both immune cells and sensory neurons, which recognize damage-associated molecular patterns [31]. Cytokine receptors such as IL-1R, TNFR, and IL-6R mediate the pronociceptive effects of inflammatory cytokines [32]. Chemokine receptors, including CCR2 (responding to CCL2/MCP-1) and CXCR2 (activated by CXCL1 and CXCL8), contribute to immune cell recruitment and nociceptor sensitization. Nerve growth factor (NGF) receptor (TrkA) and its co-receptor p75NTR mediate the sensitizing effects of NGF, which is abundantly released in inflamed tissues [33]. Inflammatory pain occurs at sites of active tumor growth where immune infiltration is prominent, in areas affected by cancer-related inflammation, such as bone marrow in leukemia, in tissues responding to cancer treatment (radiation-induced mucositis, surgical wounds), and systemically in paraneoplastic inflammatory syndromes [34].
Mixed pain represents the simultaneous presence of two or more pain mechanisms, most commonly a combination of nociceptive and neuropathic components, with inflammatory processes frequently contributing to both [35]. This phenotype is particularly prevalent in cancer patients, where a single tumor may cause direct tissue damage (nociceptive), infiltrate neural structures (neuropathic), and provoke substantial immune responses (inflammatory) concurrently [36]. Mixed pain syndromes involve the complex interplay of multiple receptor systems and anatomical sites described above. The receptors engaged in mixed pain encompass the full spectrum of nociceptive receptors (TRPV1, ASICs, P2X receptors), neuropathic pain-related ion channels (voltage-gated sodium, calcium, and potassium channels), and inflammatory mediator receptors (TLRs, cytokine receptors, chemokine receptors, TrkA) [37]. This results in overlapping sensitization mechanisms affecting both peripheral nociceptors and central pain pathways. Mixed pain typically manifests in anatomically complex cancer presentations such as vertebral metastases causing both bone pain and nerve root compression, head and neck cancers involving tissue destruction with cranial nerve involvement, and post-treatment pain syndromes combining surgical trauma, nerve damage, and ongoing inflammation [38]. Understanding these distinct pain categories and their underlying receptor mechanisms is essential for implementing targeted, mechanism-based therapeutic approaches in cancer pain management [39].
Understanding pain pathophysiology requires knowledge of the fundamental biological processes that modulate nociceptive signaling: nociceptor activation, sensitization, desensitization, and central sensitization. These mechanisms represent distinct cellular and molecular events that determine the intensity, duration, and quality of pain perception.
Peripheral sensitization describes the increased responsiveness and reduced threshold of nociceptive neurons in the peripheral nervous system [40]. This phenomenon occurs through multiple molecular mechanisms operating at the level of the nociceptor terminal and cell body. Inflammatory mediators, including prostaglandin E2, bradykinin, serotonin, and NGF, activate intracellular signaling cascades involving protein kinase A (PKA), PKC, and mitogen-activated protein kinases (MAPKs) [41]. These kinases phosphorylate ion channels and receptors, altering their biophysical properties to enhance excitability [42]. For instance, phosphorylation of TRPV1 channels reduces their activation threshold, causing them to open at lower temperatures and in response to milder acidic conditions [43]. Similarly, voltage-gated sodium channels undergo trafficking changes, with increased insertion into the neuronal membrane and altered gating kinetics that promote repetitive firing [44]. Peripheral sensitization also involves transcriptional changes in the DRG, where increased expression of pronociceptive ion channels, receptors, and signaling molecules occurs in response to inflammatory signals [45]. The biological consequence is that previously innocuous stimuli can now trigger pain (allodynia), and noxious stimuli produce exaggerated pain responses (hyperalgesia) [46]. In cancer patients, the TME continuously releases sensitizing mediators, maintaining nociceptors in a hyperexcitable state that contributes to ongoing pain and tactile allodynia in tumor-bearing tissues [29].
Desensitization represents the biological process by which repeated or prolonged stimulation leads to a progressive reduction in response magnitude [47]. At the receptor level, this involves conformational changes that reduce responsiveness despite continued ligand presence. Mechanistically, desensitization is mediated by receptor phosphorylation through G-protein-coupled receptor kinases and arrestin binding, which uncouples receptors from intracellular signaling, as well as receptor internalization via endocytosis [48]. For ion channels such as TRPV1, calcium-dependent desensitization occurs through calmodulin-mediated channel inactivation following calcium influx [49]. This process serves a protective role by limiting excessive neuronal excitation and contributes to the tolerance observed with some analgesic therapies [50]. However, in chronic pain states, including cancer pain, persistent inflammatory signaling and activation of multiple receptor systems frequently overcome desensitization, resulting in sustained sensitization rather than adaptation [51].
Central sensitization refers to a state of hyperexcitability within central pain pathways, particularly in the spinal dorsal horn and higher brain centers [37]. Unlike peripheral sensitization, it involves functional and structural changes in second-order neurons and their synaptic connections. Key mechanisms include enhanced synaptic transmission driven by increased neurotransmitter release and heightened postsynaptic responsiveness. NMDA (N-methyl-D-aspartate) receptor activation plays a central role, as prolonged nociceptive input removes the magnesium block, allowing calcium influx that activates intracellular signaling pathways such as PKC and CaMKII, ultimately strengthening synaptic transmission through phosphorylation of AMPA receptors and related proteins [52]. Glial activation, particularly of microglia and astrocytes, contributes through the release of pro-inflammatory cytokines and chemokines that enhance neuronal excitability and maintain neuroinflammation [53]. Clinically, these processes produce expansion of receptive fields, reduced pain thresholds, amplified responses to stimulation, and persistent neuronal activity after stimulus cessation. In cancer patients, central sensitization contributes to referred pain, allodynia beyond tumor boundaries, and persistent pain despite tumor reduction, and is particularly relevant in CIPN [54]. The process may also spread anatomically to supraspinal structures, including the thalamus, anterior cingulate cortex, and insula, contributing to affective and cognitive dimensions of cancer pain [55]. In chronic cancer pain, persistent peripheral input sustains both peripheral and central sensitization in a self-perpetuating cycle that complicates analgesic control [19].
Neuropathic pain in cancer patients arises through multiple distinct pathogenic mechanisms, each contributing to the complex neuropathic pain phenotype observed clinically. Direct tumor-related neuropathic pain commonly results from mechanical nerve compression or infiltration by expanding tumor masses, which may involve peripheral nerves, plexuses, or spinal nerve roots [56]. Vertebral metastases can produce radiculopathy through epidural extension and vertebral collapse, while retroperitoneal and head-and-neck tumors frequently affect major plexuses or cranial nerves. Mechanical compression leads to ischemic injury, impaired axonal transport, and, in severe cases, Wallerian degeneration, all of which promote neuropathic pain [57]. Tumor infiltration of neural tissue represents another key mechanism, particularly in cancers characterized by perineural invasion, such as pancreatic, prostate, and head and neck malignancies [58]. Tumor cells migrate along nerve sheaths and release neurotrophic factors that sensitize nociceptors and promote ectopic neural activity, while interactions among cancer cells, Schwann cells, and immune cells generate sustained neuroinflammation [59]. Neuroinflammation is a central mechanism across cancer-related neuropathic pain etiologies. Immune cell infiltration within the TME leads to the release of pro-inflammatory cytokines and chemokines that directly sensitize nociceptors and recruit additional inflammatory cells, sustaining neuronal hyperexcitability [60]. Reactive satellite glial cells in the DRG further contribute to this process by releasing inflammatory mediators that enhance neuronal firing [61]. Iatrogenic mechanisms also play an important role. Radiation therapy may induce neuropathy through direct neural injury, microvascular damage, and progressive fibrosis, often manifesting months or years after treatment and associated with persistent neuroinflammation [62]. Surgical interventions can similarly produce neuropathic pain through nerve transection, stretch injury, or ischemia, leading to Wallerian degeneration, neuroma formation, and aberrant regeneration [63]. Microglia, the resident immune cells of the central nervous system, play a central role in cancer-related pain by shifting from a surveillant state to an activated phenotype that promotes neuroinflammation and central sensitization [53]. In cancer pain, spinal microglia are activated by peripheral nerve injury from tumor compression or infiltration, chemotherapy-induced neurotoxicity, and danger-associated molecular patterns released from damaged tissue [64]. Once activated, microglia undergo morphological and functional changes and release pro-inflammatory cytokines and chemokines that enhance neuronal excitability and sustain neuroinflammatory signaling within the dorsal horn [65]. Purinergic signaling, particularly ATP-mediated activation of P2X receptors, represents a key pathway linking peripheral tissue damage to spinal microglial activation and neuropathic pain [28]. Microglia further contribute to pain amplification by disrupting inhibitory neurotransmission through downregulation of potassium-chloride cotransporter 2 (KCC2), leading to disinhibition of pain pathways, and by releasing brain-derived neurotrophic factor (BDNF), which strengthens synaptic transmission and promotes long-term potentiation of nociceptive signaling [66]. In cancer-specific contexts, tumor-derived factors can activate spinal microglia even without direct central nervous system invasion, establishing tumor-to-spinal cord signaling that contributes to persistent pain [29]. Moreover, immune cells populate the TME. TAMs, regulatory T cells (Tregs), and myeloid-derived suppressor cells (MDSCs) promote immune evasion by releasing IL-10 and TGF-β (transforming growth factor-beta) and suppressing cytotoxic T cell responses [67, 68]. These immune mechanisms simultaneously contribute to neuroinflammation and heightened nociceptive transmission [68]. Persistent, uncontrolled cancer pain activates the hypothalamic-pituitary–adrenal (HPA) axis and sympathetic nervous system, elevating glucocorticoids and catecholamines that impair T cell function and natural killer (NK) cell cytotoxicity, potentially weakening antitumor immunity [69]. Chronic pain also disrupts dendritic cell maturation and enhances Treg activity, reducing the efficacy of immunotherapies [70, 71]. Neurotoxic chemotherapeutic agents further exacerbate neuropathy through mitochondrial damage, oxidative stress, and glial activation [10, 11, 72]. These drugs additionally modulate immunity, causing immunogenic cell death or lymphocyte depletion, which can alter antitumor immune responses [73, 74]. Immune checkpoint inhibitors (ICIs) such as anti-PD-1/PD-L1 and anti-CTLA-4 therapies enhance T cell-mediated antitumor responses but can cause immune-related adverse events (irAEs), including peripheral neuropathies, myositis, and arthralgia [75]. These irAEs result from autoimmune nerve damage, cytokine-mediated inflammation, and complement activation, often necessitating corticosteroid treatment that may attenuate antitumor efficacy [76].
Pharmacological management remains largely based on the World Health Organization (WHO) three-step analgesic ladder, which recommends a graded approach depending on pain severity: non-opioid analgesics [e.g., paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs)], weak opioids (e.g., codeine, tramadol), and strong opioids (e.g., morphine, fentanyl, oxycodone) for moderate-to-severe pain. Contemporary practice increasingly incorporates adjuvant analgesics and personalized regimens based on pain mechanisms and individual patient factors [77]. Acute nociceptive pain often responds to NSAIDs or corticosteroids, particularly in cases of bone metastases or inflammation-driven pain [78]. Opioids remain essential for moderate-to-severe pain but are limited by adverse effects such as tolerance, constipation, cognitive impairment, and opioid-induced hyperalgesia [79]. Optimized dosing strategies, opioid rotation, and multimodal analgesia help balance analgesic efficacy with safety. Moreover, beyond their well-recognized adverse effects, opioids exert important immunomodulatory actions that are particularly relevant in cancer patients, where immune function influences tumor progression and treatment outcomes [80]. Opioid receptors, especially the mu-opioid receptor, are expressed on immune cells, including lymphocytes, NK cells, macrophages, and dendritic cells, enabling direct modulation of immune responses [81]. Preclinical studies have consistently shown that chronic opioid exposure suppresses both innate and adaptive immunity, notably by reducing NK cell cytotoxicity and impairing T-cell proliferation and cytokine production, thereby weakening antitumor immune surveillance [81, 82]. Opioids also influence innate immunity by altering macrophage polarization toward an immunosuppressive phenotype and impairing dendritic cell maturation and antigen presentation, potentially facilitating tumor progression [83]. Clinical evidence regarding opioid-induced immunosuppression and cancer outcomes remains inconclusive. Observational studies suggest possible associations between opioid use and cancer recurrence or survival, although confounding factors limit causal interpretation, and prospective data remain limited [84]. Importantly, immunomodulatory effects vary among opioids, with agents such as fentanyl and buprenorphine demonstrating less immunosuppression than morphine in experimental models, highlighting potential differences related to receptor pharmacology and systemic exposure [85]. The route and duration of opioid administration may also influence immune effects, with lower systemic exposure potentially reducing immunosuppressive consequences [86]. In the context of cancer immunotherapy, opioid-induced immune modulation may theoretically affect treatment efficacy, although current clinical evidence does not support withholding necessary opioid analgesia [87]. Overall, while opioids may influence immune function, effective pain control remains essential, as uncontrolled pain itself can suppress immunity and negatively affect clinical outcomes. Therefore, balanced pain management strategies, including multimodal analgesia, opioid-sparing approaches, and individualized opioid selection, may help optimize analgesia while minimizing potential immunological effects.
Neuropathic and mixed pain syndromes often necessitate co-analgesics. Antidepressants such as duloxetine and nortriptyline and anticonvulsants including pregabalin and gabapentin effectively reduce neuropathic pain, including CIPN, by modulating neuronal excitability via serotonin-norepinephrine reuptake inhibition or calcium channel modulation [88]. For refractory pain or intolerable opioid-related side effects, alternative pharmacologic strategies are under investigation. These include NMDA receptor antagonists (e.g., ketamine), cannabinoid-based therapies, and anti-NGF monoclonal antibodies. Among NMDA receptor antagonists, esketamine, the S-enantiomer of ketamine, shows particular promise for short-term analgesic benefit in patients with refractory chronic pain and could represent a potential candidate for investigation in cancer pain management, due to its greater analgesic potency and potentially more favorable side-effect profile compared with racemic ketamine [89]. Esketamine exhibits approximately twice the affinity for NMDA receptors as R-ketamine, allowing for lower effective doses and reduced psychomimetic effects while maintaining analgesic efficacy. Clinical studies have demonstrated esketamine’s effectiveness in reducing opioid-refractory cancer pain, particularly in patients with neuropathic components and central sensitization [90]. The intranasal formulation of esketamine, originally developed for treatment-resistant depression, offers a non-invasive administration route that may enhance accessibility for outpatient cancer pain management, though evidence specific to cancer populations remains limited and warrants further investigation. Dextromethorphan, another NMDA receptor antagonist traditionally used as an antitussive agent, has emerged as a potential adjuvant analgesic for cancer-related neuropathic pain [91]. Its mechanism involves not only NMDA receptor antagonism but also sigma-1 receptor agonism, serotonin-norepinephrine reuptake inhibition, and nicotinic receptor antagonism, creating a multimodal analgesic effect. Clinical trials have shown that dextromethorphan, particularly in combination formulations with quinidine (which inhibits dextromethorphan’s rapid first-pass metabolism and increases bioavailability), can reduce neuropathic pain [92] and could be a promising tool for cancer patients, including those with CIPN. The favorable safety profile, lack of addiction potential, and oral bioavailability make dextromethorphan an attractive option for long-term management of cancer-related neuropathic pain, though optimal dosing strategies and patient selection criteria require further clarification through larger randomized controlled trials.
Cannabinoid-based therapies have emerged as potential adjuncts in cancer pain management due to their analgesic and immunomodulatory properties mediated through the endocannabinoid system, including CB1 receptors in central pain pathways and CB2 receptors on immune cells [93]. Cannabinoids exert analgesic effects through multiple mechanisms, including reduction of peripheral nociceptor sensitization, inhibition of inflammatory mediator release, modulation of ion channel activity, and suppression of central pain transmission and neuroinflammation [94]. Clinical evidence remains limited but suggests modest analgesic benefit, particularly with standardized preparations such as nabiximols, in patients with cancer pain inadequately controlled by opioids, although results are inconsistent and adverse effects such as sedation and cognitive impairment may occur [95]. Cannabinoids also exert immunomodulatory effects, primarily through CB2 receptor activation, which suppresses inflammatory responses and immune cell activity [96]. Preclinical studies indicate that cannabinoids may possess antitumor properties through mechanisms including apoptosis induction, inhibition of angiogenesis, and modulation of tumor cell proliferation, although these findings have not been definitively confirmed in clinical settings [97]. Conversely, concerns remain regarding potential protumorigenic effects under certain conditions, and interactions with cancer immunotherapy are not fully understood, though current evidence does not support withholding cannabinoids when clinically indicated for symptom management. From a clinical perspective, cannabinoids may be considered as adjunctive therapy in selected cancer patients with refractory pain, particularly those with neuropathic components or opioid-related adverse effects, using standardized formulations and careful dose titration [98]. Overall, cannabinoids represent a promising but still evolving component of multimodal cancer pain management, and further high-quality clinical trials are needed to clarify their long-term efficacy, safety, and impact on oncologic outcomes.
Given the immunological mechanisms underlying cancer pain, immunomodulatory approaches are being explored. These include cytokine inhibitors (e.g., anakinra, tocilizumab), CCR2 antagonists, TLR4 blockers, and glial cell modulators (e.g., minocycline), which target neuroimmune mechanisms contributing to nociception and central sensitization [99]. Effective pharmacologic management thus requires a multimodal, mechanism-based approach tailored to the patient’s disease stage, pain phenotype, and comorbidities, ideally delivered within an interdisciplinary pain management framework. The complex interplay between tumor biology, immune activation, neural sensitization, and emerging precision therapeutic strategies is summarized schematically in Figure 1.
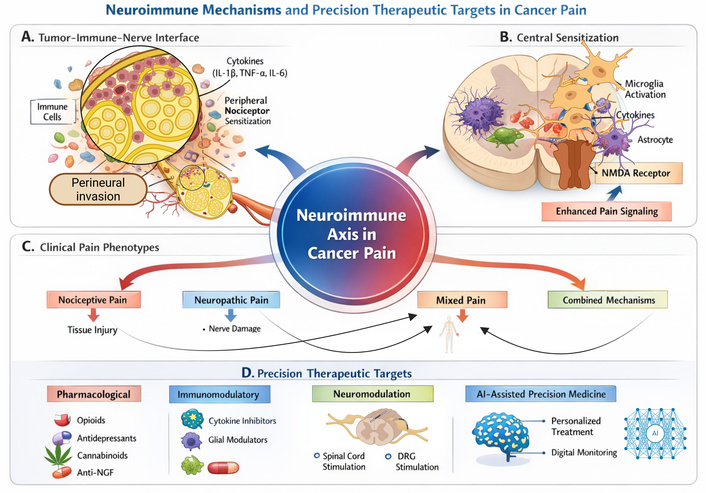
Neuroimmune mechanisms and precision therapeutic targets in cancer-related pain. This schematic illustrates the neuroimmune mechanisms underlying cancer pain and their therapeutic implications. Peripheral tumor-immune interactions and central sensitization drive nociceptive signaling, resulting in nociceptive, neuropathic, and mixed pain phenotypes. Corresponding precision treatments include pharmacological, immunomodulatory, neuromodulatory, and AI-assisted approaches. NMDA: N-methyl-D-aspartate; NGF: nerve growth factor; DRG: dorsal root ganglia. Created in BioRender. Varrassi, G. (2026) https://BioRender.com/vvr5kzf.
Effective pharmacologic management thus requires a multimodal, mechanism-based approach tailored to the patient’s disease stage, pain phenotype, and comorbidities, ideally delivered within an interdisciplinary pain management framework (Table 1).
Summary of pharmacological approaches for cancer pain management.
| Class | Examples | Primary use | Key points |
|---|---|---|---|
| Non-opioids | NSAIDs, steroids | Inflammatory/Bone pain | Short-term side effects limit use |
| Opioids | Morphine, fentanyl, oxycodone | Moderate-severe pain | Mainstay therapy: tolerance, immunosuppression |
| Co-analgesics | Duloxetine, pregabalin | Neuropathic pain, CIPN | First-line for neuropathic components |
| NMDA antagonists | Ketamine, esketamine | Refractory, central sensitization | Esketamine: better profile, intranasal |
| Cannabinoids | THC/CBD formulations | Adjunctive, refractory pain | Limited evidence; immunomodulatory |
| Immunomodulatory | Anti-NGF, cytokine inhibitors, glial modulators | Bone pain, inflammatory pain | Emerging; target neuroimmune mechanisms |
NSAIDs: non-steroidal anti-inflammatory drugs; NMDA: N-methyl-D-aspartate; CBD: cannabidiol; THC: delta-9-tetrahydrocannabinol; NGF: nerve growth factor; CIPN: chemotherapy-induced peripheral neuropathy.
Despite advances in oncological care, cancer pain remains a significant clinical challenge. Epidemiological evidence indicates that 55% of patients undergoing active treatment, 66% with advanced disease, and 80% receiving palliative care experience substantial pain [1]. Chronic pain persists in many survivors, often neuropathic in nature [12, 14]. The multifaceted etiology of cancer pain necessitates individualized, mechanism-oriented treatment approaches, where emerging tools such as artificial intelligence (AI) may optimize pain assessment and therapy. The pathophysiology involves complex nociceptive, neuropathic, and inflammatory interactions. Tumor-secreted algogenic substances directly activate nociceptors, while the inflammatory TME, enriched with immune cells and cytokines, drives sensitization and neuroplastic changes. The intricate neuro-immune interactions underlying cancer pain have been comprehensively examined in recent detailed reviews that provide extensive mechanistic analyses complementing this narrative overview [100, 101]. Wang et al. [100] presented an exhaustive examination of molecular mechanisms driving cancer pain, including a detailed discussion of ion channel modulation, neurotransmitter systems, and intracellular signaling cascades at the molecular level, alongside comprehensive treatment strategies spanning pharmacological, interventional, and emerging targeted approaches. Amit et al. [101] provided an in-depth analysis of bidirectional neuro-immune cross-talk in cancer, elucidating how nerve fibers infiltrating peripheral tumors release neuromodulatory factors that shape immune cell behavior and tumor progression, while tumor-infiltrating immune cells reciprocally modify neuronal activity including nociceptive signaling. These extensive reviews collectively emphasize that cancer pain represents a complex interplay of neuronal, immune, and tumor cell interactions, requiring an integrated understanding of neuro-immune axis mechanisms to develop novel therapeutic approaches that strengthen antitumor immunity while providing effective analgesia. This narrative review synthesizes key advances in this evolving field, highlighting their clinical relevance to cancer pain management. In particular, emerging immunomodulatory, neuromodulatory, and AI-assisted approaches are broadening therapeutic options and enabling more mechanism-based, personalized care [102]. Among immunomodulatory strategies, monoclonal antibodies targeting NGF, such as tanezumab, have demonstrated significant analgesic efficacy in cancer-related bone pain by reducing peripheral sensitization, although safety concerns have limited regulatory approval [103]. Other approaches include IL-1 receptor antagonists and chemokine receptor inhibitors, which aim to reduce neuroinflammation and nociceptive signaling, while glial modulators such as minocycline and propentofylline show promise in reducing neuropathic pain through inhibition of microglial activation [104]. Neuromodulatory interventions provide mechanism-based alternatives for refractory cancer pain. Spinal cord stimulation (SCS), including high-frequency and burst paradigms, has demonstrated sustained pain reduction and improved functional outcomes, particularly in patients with neuropathic pain or post-treatment syndromes [105]. DRG stimulation and peripheral nerve stimulation offer more targeted analgesia for focal cancer-related pain syndromes. AI is also transforming cancer pain assessment and management. Machine learning and natural language processing can identify patients at risk for severe pain, predict treatment response, and support personalized analgesic selection based on clinical, genetic, and treatment-related factors [106]. Additionally, wearable sensors, digital monitoring platforms, and AI-assisted symptom management tools enable continuous pain assessment, early intervention, and improved clinical outcomes [107]. Although these emerging strategies require further validation and integration into clinical practice, they represent a shift toward precision, mechanism-based cancer pain management tailored to individual patient characteristics.
Limitations: This review is narrative in nature and thus limited by heterogeneity among included studies, variable clinical settings, and evolving therapeutic standards. Translating mechanistic insights into routine clinical practice remains constrained by a paucity of high-quality, randomized trials investigating targeted immunomodulatory or neuromodulatory interventions. Future research should prioritize mechanistically stratified clinical trials and multidisciplinary implementation strategies to close these gaps.
Cancer pain is a prevalent, multifactorial condition driven by nociceptive, neuropathic, nociplastic, and immune-mediated mechanisms, often exacerbated by treatment-induced neurotoxicity. Despite therapeutic advances, optimal control remains challenging. Emerging immunomodulatory, neuromodulatory, and AI-assisted approaches hold promise for precision diagnostics and personalized management in oncology pain care.
AI: artificial intelligence
ASIC3: acid-sensing ion channel 3
ATP: adenosine triphosphate
CIPN: chemotherapy-induced peripheral neuropathy
DRG: dorsal root ganglia
irAEs: immune-related adverse events
NGF: nerve growth factor
NK: natural killer
NMDA: N-methyl-D-aspartate
NSAIDs: non-steroidal anti-inflammatory drugs
PKA: protein kinase A
TAMs: tumor-associated macrophages
TLRs: Toll-like receptors
TME: tumor microenvironment
Tregs: regulatory T cells
TRPV1: transient receptor potential vanilloid 1
We extend our sincere gratitude to the Paolo Procacci Foundation for its invaluable support and assistance throughout the publication process. In particular, its assistance was indispensable in researching bibliographic sources and preparing the illustrations.
GV: Conceptualization, Methodology, Visualization, Writing—original draft, Writing—review & editing. DM: Formal analysis, Writing—review & editing. MLGL: Formal analysis, Methodology, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
Giustino Varrassi and Matteo Luigi Giuseppe Leoni, who are the Guest Editors of Exploration of Immunology, had no involvement in the decision-making or review process of this manuscript. The other author declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
This research received no external funding.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1302
Download: 23
Times Cited: 0
