Affiliation:
1Department of Food Science and Technology, College of Science, Kwame Nkrumah University of Science and Technology, Private Mail Bag, Kumasi, Ghana
Email: alhassanawuni@yahoo.com
ORCID: https://orcid.org/0009-0004-8784-2434
Affiliation:
1Department of Food Science and Technology, College of Science, Kwame Nkrumah University of Science and Technology, Private Mail Bag, Kumasi, Ghana
ORCID: https://orcid.org/0000-0003-0985-8727
Affiliation:
2Department of Food Science and Technology, School of Agriculture and Technology, University of Energy and Natural Resources, P.O. Box 214, Sunyani, Ghana
Email: nanafrimpong94@yahoo.com
ORCID: https://orcid.org/0000-0001-5466-4425
Affiliation:
2Department of Food Science and Technology, School of Agriculture and Technology, University of Energy and Natural Resources, P.O. Box 214, Sunyani, Ghana
ORCID: https://orcid.org/0000-0002-2703-3393
Affiliation:
3Department of Biochemistry and Biotechnology, College of Science, Kwame Nkrumah University of Science and Technology, Private Mail Bag, Kumasi, Ghana
Explor Foods Foodomics. 2026;4:1010116 DOI: https://doi.org/10.37349/eff.2026.1010116
Received: November 06, 2025 Accepted: January 14, 2026 Published: February 25, 2026
Academic Editor: Noureddine Benkeblia, University of Life Sciences, Mona Campus, Jamaica
Pearl millet (Pennisetum glaucum) is known for its agronomic, economic, and nutritional functionalities coupled with its important position as a “Smart food” in the food ecosystem. However, among agronomic products, pearl millet is now considered an “orphan crop” due to its neglect. As a result, numerous scientific methods have been investigated to clarify the antinutritional factors that prevent the bioaccessibility of minerals in pearl millet. To meet the biological needs of consumers, this review examines the various sustainable food processing techniques employed to enhance the mineral bioaccessibility of pearl millet. Additionally, the benefits of pearl millet for health are mentioned. The application of the INFOGEST digestion model as a method for comprehending mineral bioaccessibility in vitro is presented. Some well-known millet food products are reportedly being adopted to encourage and prevent the underutilization of pearl millet. In summary, the results offer optimization strategies to reduce the bioaccessibility issues associated with pearl millet minerals for human nutrition, hidden hunger, and public health mineral deficiency interventions.
Recent climate change has confronted us, devaluing the success of sustainable agriculture and increasing food insecurity. So, it makes sense to cultivate plants that are resistant to drought and high temperatures. The most suitable crop to grow in this changing environment is regarded as pearl millet since it has an intrinsic climate resilience quality [1].
Pearl millet (Pennisetum glaucum) is a vital plant for the sustenance, food security, and income generation of resource-constrained farmers in developing countries. Despite its historical importance, it has lost ground because it is not as widely grown and commercialized as other crops like wheat, rice, and maize. Farmers have been ignoring it for decades, and it has not received much research or development during the Green Revolution; it is thus regarded as an underutilized and orphan crop [2, 3]. Consequently, in recognizing the enormous potential of pearl millet and other millets, the United Nations General Assembly (UNGA) at its 75th session in March 2021 passed a resolution declaring 2023 to be the International Year of Millets (IYM), with the primary goal of raising public awareness of the nutritional and health benefits of millets as well as their ability to withstand global climate change. The IYM 2023 offered the chance to spread knowledge about the benefits of millet for better nutrition, production, sustainability of the environment, and quality of life [4].
Pearl millet is mostly cultivated in the tropical and semi-arid regions [3]. Pearl millet has high nutritional value [5]. High concentrations of minerals, including iron (Fe), zinc (Zn), magnesium (Mg), copper (Cu), manganese (Mn), potassium (K), and phosphorus (P), are found in pearl millet grain [6–8]. The total mineral content of pearl millet grain is around 2.3 g/100 g (dry weight) [7, 9]. As a “Smart Food”, pearl millet is frequently referred to as a “Nutri-Cereal” and considered beneficial because it has a higher nutritional content compared to other cereals like wheat, rice, and maize [3, 10]. Pearl millet benefits the health of both humans and animals, especially expectant mothers and their offspring [6, 11]. Recent research suggests that eating millet-based foods may be able to address several health-related problems [1, 11]. This is because millet produces nutrient-rich foods, and malnutrition in newborns, kids, and women of reproductive age is reduced when millet diets are followed. Additionally, because pearl millet has a significant amount of Zn and Fe, diets involving it raise the hemoglobin content of consumers [12]. Pearl millet’s high Ca and P content is crucial for bone development and growth as well as the production of adenosine triphosphate (ATP), the body’s energy currency. Strong phytonutrients and biologically active compounds found in millet help protect against heart disease. Patients with diabetes can benefit from pearl millet due to its low to medium glycemic index [12]. According to Babele et al. [2], eating millet products can reduce fasting glucose by 32% and eliminate insulin resistance by 43%. Due to increased dietary Mg, there is a clear reduction in severe respiratory issues and an increase in insulin activity and glucose receptors in the body [2].
Pearl millet cereals have been used to make a variety of traditional meals. The most popular traditional pearl millet meal in Ghana and many other regions of the world is porridge, both thin and thick [3, 8]. Additional millet-based dishes that are offered are roti flatbreads, alcoholic and non-alcoholic drinks (“mangisi”, “pombe”, “pito”, “boza”, “brukutu”, “jandh”, “kunu-zaki”, “bushera”, “fura”, “ben-saalga”, “mahewu”, “oskikundu”, “ugi”, “togwa”, “marewa”), and “chibuku” shake [13–15]. Indian women have long used pearl millet flour to make a wide range of foods, including cake, “wadi”, chips, “laddoo”, and bread [8, 9]. In some parts of Ghana, it is customary to produce an unfermented flour water for guests by using pearl millet flour.
Pearl millet has several anti-nutritional compounds (phytate, tannins, and polyphenols) in addition to its nutritional value and health advantages (Figure 1) [16]. These compounds cause dietary minerals to chelate in the digestive system, which lowers their bioaccessibility and bioavailability [17]. Depending on the area and genotype, pearl millet’s phytic acid content can range from 179 to 306 mg/100 g [18]. The level of phytic acid concentration of pearl millet makes it difficult to remove from processed foods and causes it to persist even after processing. Since phytic acid content is higher in pearl millet, practical home techniques to lower its levels are necessary to improve nutrient absorption [19]. For minerals to be accessible and available to consumers, good production techniques must be implemented to dramatically lower the quantities of anti-nutrients. Reducing anti-nutritional factors is required to increase the mineral content, bioaccessibility, and bioavailability of pearl millet processed products. This can be done by employing a variety of processing methods, including fermentation, soaking, dehulling, milling, malting, blanching, parboiling, acid, and heat treatments [20]. The current understanding of the antinutritional elements, bioaccessibility, and bioavailability of pearl millet and its derivatives must be expanded. These anti-nutrients interact with minerals to decrease their bioaccessibility and bioavailability, which leads to undernutrition. Thus, the main objective of this review continues to be food processing methods used to improve the mineral bioaccessibility and bioavailability of pearl millet and its products.
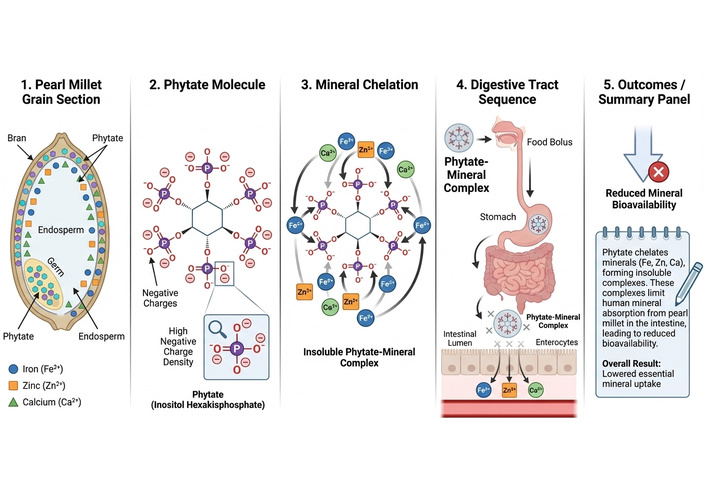
Mechanism of phytate on pearl millet minerals. The figure was created using FigureLabs (https://figurelabs.com).
Mineral concentrations in pearl millet grains generally vary based on the influence of agronomic, genetic, and class factors [21, 22]. Given this, agronomic and genetic factors may contribute to pearl millet cultivars with a broad range of mineral content [23]. According to a detailed analysis of the mineral composition of two pearl millet [Pennisetum glaucum (L.) R. Br.] cultivars, namely ICMH 1201 and ICMH 1301, Fe was above normal, but Ca and Zn were below average. The ranges were P (326–373 mg/100 g), Ca (7.2–7.4 mg/100 g), Fe (5–8 mg/100 g), and Zn (3.1–3.2 mg/100 g) [23]. Pearl millet has the highest Fe and Zn content of any millet. Its Zn and Fe levels were found to be positively correlated, although there was no discernible link with grain size. This suggests that pearl millet’s Fe concentration increases in proportion to an increase in Zn grain content [24]. Also, Satyavathi et al. [25] found that grain Fe and Zn concentrations in pearl millet were positively correlated in a study conducted in West Africa. The pearl millet grain Zn and Fe concentrations were positively impacted by the parental lines Jirani, LCIC 9702, and MORO [26]. Additionally, the study found that increasing grain Fe features naturally leads to an increase in grain Zn content. This is partly explained by the proximity of Zn and Fe in the millet grain’s scutellum [23]. Hassan et al. [27] investigated the effects of cultivating pearl millet in two different regions (South Africa and Zimbabwe) and found significant differences in the studied minerals between the two locations. They also found that major nutrients (Ca, Mg, and Na) were not as concentrated as trace minerals (Fe and Zn, for example) and that major minerals (Ca, Mg, and Na) were not as high as the trace minerals (Fe and Zn). These variations in the mineral contents of pearl millet from different locations could have been caused by the types of soil in which the grains were grown [28]. A different investigation of twenty-two cultivars of pearl millet revealed a wide range of mineral content levels, with reported values for Fe, Ca, and Zn, among others, ranging from 13.85–2,766.3, 31.77–728.7, and 43.34–809.38 mg/kg [29]. The variety and production site were identified as the causes of variations in these nutrients’ amounts [29]. Additionally, three genotypes of pearl millet were examined by Kulthe et al. [30], who also noted that the mineral contents of the grain varied from 40.07–42.67 mg/100 g of Ca, 255.67–327.82 mg/100 g of P, and 5.08–8.12 mg/100 g of Fe. The variations in mineral concentrations were caused by genetic differences [23]. Two pearl millet cultivars were treated to varying storage periods in a different study by Babiker et al. [31]. The findings indicated that the cultivars’ storage caused a progressive drop in their Ca, P, and Fe contents. Additionally, Suma and Urooj [32] examined the Fe and Zn profiles of two pearl millet cultivars, “Kalukombu (K)” and hybrid Maharashtra Rabi Bajra (MRB). The findings indicated that the total Fe content of the two cultivars, K and MRB (5 and 6.4 mg/100 g, respectively), did not differ significantly. The investigators failed to elucidate the reported results, while it is probable that the two pearl millet varieties were cultivated on soil with comparable mineral content, climate, and no genetic alteration.
According to Minnis-Ndimba et al. [23], proton-induced X-ray emission (PIXE) spectrometry has shown to be an effective method for examining the distribution of key mineral elements in millets (Table 1). The endosperm and germ are the two primary sections of pearl millet. The primary site of mineral accumulation, according to the PIXE, is the tissue of the germ (scutellum and embryo). While Ca, Mn, and Zn are concentrated in the pearl millet embryo tissues, Fe, P, and K are substantially concentrated in the scutellum. Fe is comparatively high in the scutellum and other peripheral areas, while Zn is more prominent in the embryo. In actuality, the scutellum has 15% more Fe than the embryo.
Mineral content of pearl millet.
| Minerals | Minerals content | References |
|---|---|---|
| Calcium (Ca) | 13.66–15.67 | [21] |
| 31.77–728.71 | [29] | |
| 40.07–42.67 | [30] | |
| 46 | [33] | |
| 36.42 | [34] | |
| 10–80 | [35] | |
| Copper (Cu) | 0.59–0.69 | [21] |
| 10–18 | [35] | |
| 10.6 | [36] | |
| Iron (Fe) | 4.45–4.92 | [21] |
| 13.85–2,766.31 | [29] | |
| 5.08–8.12 | [30] | |
| 121 | [34] | |
| 70–180 | [35] | |
| Magnesium (Mg) | 132.47–142.48 | [21] |
| 340.27–4,769.9 | [29] | |
| 1,028 | [34] | |
| 180–270 | [35] | |
| Manganese (Mn) | 0.54–0.92 | [21] |
| 18–23 | [35] | |
| 11.5 | [36] | |
| Phosphorus (P) | 266.03–292.66 | [21] |
| 255.67–327.82 | [30] | |
| 314 | [33] | |
| Potassium (K) | 302.52–307.06 | [21] |
| 4,500 | [34] | |
| 70–110 | [35] | |
| Sodium (Na) | 3.96–4.48 | [21] |
| 58.37–240.47 | [29] | |
| 230 | [34] | |
| 4–13 | [35] | |
| Zinc (Zn) | 1.97–2.78 | [21] |
| 43.34–809.38 | [29] | |
| 40 | [34] | |
| 53–70 | [35] |
Mawouma et al. [21], values are in mg/100 g DW (dry weight) for pearl millet flour micro-element and trace element contents. Adéoti et al. [29], values are in mg/1,000 g of micronutrients of pearl millet flour. Kulthe et al. [30], values are in mg/100 g of mineral composition of pearl millet flour. Burton et al. [33], values are mg/100 g for pearl millet minerals. Owheruo et al. [34], values are in ppm of pearl millet. Abdalla et al. [35], values are in mg/100 g and 0.001 mg/g mineral content of pearl millet. Sankara Rao and Deosthale [36] 0.001 mg/g mineral content of pearl millet.
On the other hand, the endosperm has comparatively low Zn, P, and Fe levels. It was also observed that the outer grain layers included comparatively significant concentrations of Ca and Fe. This demonstrates that the distribution patterns of these two minerals within the pearl millet grain are different [23]. Additionally, microscopic analysis showed that Zn was mostly dispersed in the pericarp, sub-aleurone, and germ regions, whereas Fe was found in the aleurone layer, scutellum region, and the tip of the germ. This demonstrates once again that the distribution patterns of Zn and Fe are different [37]. Earlier investigations reported that Zn in millet is found in the interior sections of the grain where the germ is lodged in the endosperm [38]. The Zn was absent or present in very small amounts in the peripheral tissues. In addition, Zn is equally distributed in the grain and around 8% is present in the bran of pearl millet, according to Hama et al. [38, 39]. This contrasts with Fe, which is not.
To standardize experimental conditions and enhance result comparability, an international network of excellence on the fate of food in the gastrointestinal tract (the INFOGEST network) has developed a consensus protocol for static and semi-dynamic simulated digestion, which is widely adopted in the scientific community. These protocols replicate the physiological conditions of oral, gastric, and small intestinal digestion phases, with the static method being fully controlled by the analyst, while the semi-dynamic method incorporates gradual pH variations through automated processes. It is recommended to incorporate physiologically relevant electrolytes or salts such as simulated salivary fluid (SSF), simulated gastric fluid (SGF), and simulated intestinal fluid (SIF) during the digestion phases to determine how they impact enzyme activities and digestion outcomes (Figure 2). These solutions mimic the ionic composition of digestive secretions in the various sections to support enzyme activities [40, 41]. Table 2 highlights the different types of electrolytes and their role in the phases of the digestion process. The oral phase encompasses a dilution of food with SSF, combining potential salivary amylase use and simulated mastication for solids or semisolids. This phase is essential for all simulated digestion processes, ensuring uniform dilution across food states [40]. Usually, an approximate pH of 7 at 37°C for 2 min runs if salivary amylase is used in a standard INFOGEST model [41]. The 2 min period mimics the brief period food stays in the oral cavity. In the oral phase, there is less effect on mineral solubility as most minerals are still bound to phytate and complexed with the cell wall [42]. However, other studies observed that fermented and germinated millet floursʼ soluble Fe and Zn increase after α-amylase digestion. Furthermore, oral digestion facilitates the release of minerals in the gastric phase through homogenization and exposure of mineral-binding sites, improvement [43]. It is now a mandatory phase for ensuring consistency in all food matrices before entering the gastric phase [40].
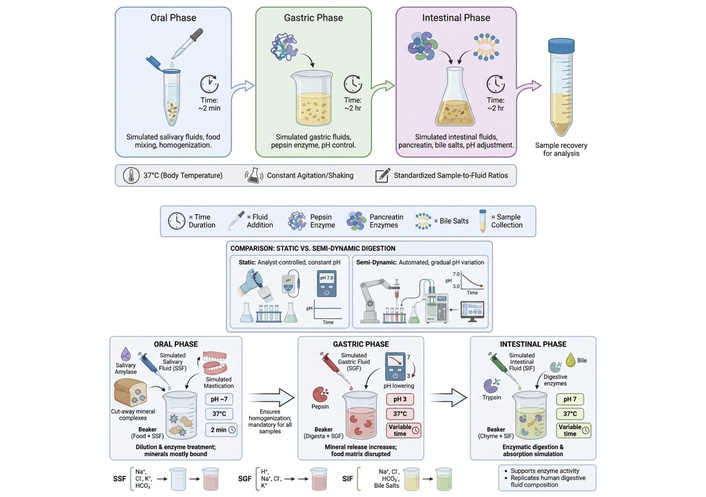
Schematic route of INFOGEST in vitro digestion models. The figure was created using FigureLabs (https://figurelabs.com).
The role of electrolytes in the INFOGEST digestion model [41].
| Electrolyte | Phase (s) used | Role |
|---|---|---|
| NaCl | Saliva, gastric, and intestinal | Preserves ionic strength, contributes to pH buffering and osmotic balance, and replicates the natural sodium levels in digestive juices. |
| KCl | All phases | Provides physiological potassium concentration; thus, important for maintaining osmotic pressure and enzyme activity. |
| KH2PO4 | Salivary, intestinal | Acts as a phosphate buffer, stabilizing pH and providing phosphate ions for buffering capacity. |
| NaHCO3 | Saliva, intestinal | Primary buffering agent that neutralizes gastric acid in the intestinal phase and maintains physiological pH (7). |
| MgCl2·6H2O | All phases | Supplies magnesium ions, essential cofactors for many digestive enzymes (e.g., amylases, proteases, lipases). |
| (NH4)2CO3 | Salivary | Mimics ammonia content in saliva, contributing to pH buffering and enzymatic activation. |
| CaCl2·2H2O | All phases | Supplies calcium ions, which promote micelle formation in lipid digestion and stabilize enzyme structures (especially amylase and lipase). |
| ZnSO4 | Intestinal | As a cofactor for certain proteolytic enzymes, thus enhancing metalloprotease activity. |
| Bile salts | Intestinal | For emulsification of fats, solubilization of lipophilic compounds for absorption, and micelle formation. |
Food from the oral phase moves to the gastric phase, which has an SGF. This phase uses the enzyme pepsin, which provides about 2,000 U mL–1 activity in acidic solution at pH between 2 and 3 at 37°C for 1.5–2 h, influencing the cleavage of protein to peptide [40, 41]. The low pH also accelerates mineral-phytate complex hydrolysis, as a result affecting solubilization of Fe and Zn [43]. The reduction of phytic acid and peptide complexes due to pre-digestion of fermented millet reportedly led to high Fe bioaccessibility [42]. Conversely, dos Santos et al. [44] reported that polyphenol-Fe complexes are precipitated when there is excessive gastric acidification.
From the gastric phase, the intestinal condition is created by mixing the chyme with SIF. Also known as the decisive step, the intestinal phase simulates biliary and pancreatic secretions under an almost neutral pH of 7 at 37°C for 2 h for bioaccessibility estimation [41]. During this stage, lipids are emulsified while peptides are hydrolyzed, resulting in the release of bound minerals into the aqueous micellar fraction. The use of ultrafiltration and centrifugation during the subsequent intestinal digestion phase was able to quantify soluble Fe and Zn [42, 43]. When enzymatic activities were increased in the intestines because of fermentation and germination, mineral bioaccessibility was increased as the phytate:mineral molar ratio was reduced [42]. Muleya et al. [43] highlighted the need for post-digestion isotopic correction as divalent cations may still be chelated by residual polyphenols.
From a nutritional point of view, bioaccessibility is the amount or degree to which micronutrients are released from the food matrix and made available for absorption after digestion in the gastrointestinal tract [45, 46]. Bioaccessibility relies on digestion, and the release of nutrients from the food matrix can be simulated using in vitro methods. It allows investigation of how factors like food processing, food matrix composition, and luminal conditions, such as pH, affect nutrient availability. Additionally, bioaccessibility is influenced by the interactions between components and physicochemical properties, including pH, temperature, and texture [47, 48]. Rodrigues et al. [49] revealed that diverse in vitro digestion (IVD) models exhibit varying levels of complexity and automation.
The percentage of a nutrient that is broken down, released from food, absorbed by intestinal cells, and made available for bodily physiological processes is known as bioavailability [47]. Following digestion, the nutrients released are taken up and absorbed throughout the intestinal tract, where bioavailable water-soluble and micellarized fat-soluble nutrients permeate the mucin layer and reach the apical membrane of enterocytes. They are then absorbed within these cells [49]. Bioavailability (in vivo) is affected by several host characteristics that in vitro techniques cannot precisely reproduce [47]. Utilizing human subjects, animal models, Caco-2 cell models, and techniques such as mineral dialysis, the bioavailability of micronutrients in crops has been evaluated [45].
The main factors affecting mineral bioaccessibility and bioavailability in pearl millet are phytic acid, phenolic compounds (polyphenols or tannins), and fibers, which are known to act as antinutritional factors due to their capacity to bind minerals such as Fe and Zn, making these minerals less bioaccessible for absorption in the human body (Figure 3) [50].
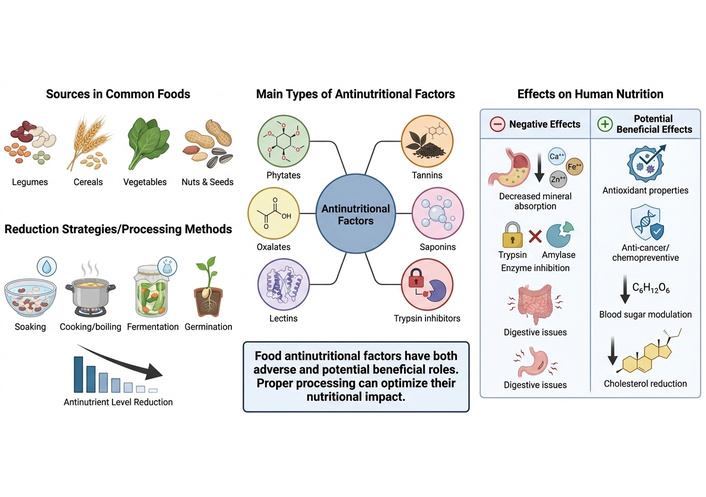
Functionalities of food antinutritional factors. The figure was created using FigureLabs (https://figurelabs.com).
According to Gibson et al. [51], phytic acid is defined as myo-inositol hexakisphosphate (IP6), which is composed of an inositol ring with six phosphate ester groups, and the salts Mg, Ca, or K phytate that relate to it. Phytates, which are mixed mineral salts of phytic acid, are a common form of phytic acid found in grains [50]. Food grains of the cereal class, oil seeds, nuts, and legumes are rich sources of phytate [52]. According to Savita et al. [52], it makes up between 3% and 5% of the dry weight of plant seeds. Phytate is mainly stored as a complex salt of minerals (Ca, Mg, K, etc.) and protein in globoids and aleurone particles of cereals and legumes. Krishnan and Meera [37] reported that the aleurone, germ, and sub-aleurone layers of pearl millet contain higher concentrations of phytic acid. Phytate is soluble in water and only slightly soluble in ethanol [53]. However, the formation of insoluble complexes decreases their solubility and hinders the absorption of bound elements and IP6 in the small intestines, thereby lowering their bioavailability [54]. While ferric phytate is insoluble at pH levels between 1.0 and 3.5, phytates become more soluble at pH values around 5.5–6.0 with Ca2+, 7.2–8.0 with Mg2+, and 4.3–4.5 with Zn2+ as the counter ion [55]. The buildup of phytic acid rises as external calcium concentration rises. According to reports, pearl millet has a phytate level of 485 mg/100 g [56]. Furthermore, phytic acid P concentration ranged between 0.198 ± 0.034 g/100 g and 0.410 ± 0.036 g/100 g, according to a thorough analysis of a panel of 145 inbred lines of pearl millet [Pennisetum glaucum (L.) R. Br.] of seed flour that came from landraces in India, West Africa, and Central America and encompassed a wide genetic variety [18]. According to a recent study, pearl millet has an extremely high phytate concentration of 878 mg/100 g [57].
A six-fold dihydrogen phosphate ester of myo-inositol, also known as cis-1,2,3,5-trans-4,6-cyclohexanehexol, is produced from the inositol ring present in the structure of phytic acid. Because the maximal amount of phosphate groups, that is, five out of six, are present in thermodynamically stabilized equatorial positions, myo-orientation is also observed in the case of phytic acid [58]. It has firmly bound mineral P in the form of a molecule that resembles snowflakes [55]. Phytate contains double-charged phosphate groups with a negative charge within typical pH ranges [59, 60]. Because of this, it is quite unstable. There are six different types of inositol phosphate, referred to as inositol monophosphate (IP1) to IP6, depending on how many phosphate groups are joined to the inositol ring. Myo-inositol phosphates are almost entirely found in the IP6 form in mature, unprocessed cereals, legumes, and oleaginous seeds that have not been preserved. This form is the most common and has the strongest mineral-binding capacity [61]. IP6 structure displays 12 dissociable protons, and its empirical formula is C6H16O24P6 [62].
Humans lack the intestinal phytase enzyme, rendering phytate in food antinutritional, as the human digestive system cannot decompose it. Phytic acid functions as a chelating agent and contains six reactive phosphate groups [50]. Its phosphate groups’ double charge makes it more likely to bind cations, mostly Fe, Zn, and Ca, and prevent their absorption [63]. Due to its negative charge at physiological pH, phytic acid has a remarkable attraction for positively charged dietary ingredients, including minerals and trace metals, giving it an exceptional chelating power [64]. According to Raes et al. [50], a cation can bridge two or more phytic acid molecules or attach to one or more of the phosphate groups on a single phytic acid molecule. Furthermore, the amount of phosphate groups and their trans and cis locations on the inositol ring determine the cation binding ability. According to research, for instance, at a pH between 5 and 7, fewer minerals were attached to inositol tetraphosphate (IP4) and inositol triphosphate (IP3), which are lower inositol phosphates, than to IP6 and inositol pentaphosphate (IP5), which are higher inositol phosphates [50]. Furthermore, phytate’s net charge of –3 and –8 at pH 1.5 and pH 7.5, respectively, results in a strong binding capacity with metals [65]. This leads to a pronounced binding capacity with counter ions or metal complexation at the near-neutral pH of the duodenum [66]. Each of the six inositol phosphate groups attached to the inositol ring can bind to any divalent mineral; generally speaking, it appears that at least five of the six sites on the inositol ring must be phosphorylated for the inositol to form a strong association with mineral ions [67]. Phytic acid and minerals interact to generate salts that are soluble at acidic pH but insoluble at physiological pH. As a result, the digestive tract absorbs these salts poorly [68]. The complexes that are produced have a lower bioavailability in the human digestive tract because they are stable, insoluble, and difficult to digest at physiological pH [64]. It should be mentioned that the phosphate esters of phytic acid are undissociated at gastric pH 2 or lower, which restricts the amount of free binding sites for minerals [60, 68]. Phytate is a strongly negatively charged ion that may form stable complexes with divalent mineral ions like Fe, Zn, Ca, and Mg by electrostatic contact, but only at pH values more than 2, which is above the pH of the stomach [69]. It should be highlighted, nonetheless, that the combination between phytic acid and minerals might primarily arise in the digestive system or in plant tissue. Essentially, there is a reduction in the bound mineral ions’ bioavailability. Since lower inositol-phosphates (like IP4 and IP3) have a lower capacity to bind minerals and typically form more soluble complexes, higher inositol phosphates (like IP5 and IP6) have a negative influence on mineral bioavailability [69].
Dietary fiber includes non-starch polysaccharides, lignin, resistant starch, non-digestible oligosaccharides, cellulose, hemicellulose, and pectin according to Krishnan and Meera [37], and pearl millet has 20.8% dietary fiber [70]. Another study has found that the total dietary fiber (TDF) content among pearl millet genotypes ranged from 7.68–16.18 g/100 g [71]. Dietary fiber is classified as either water-soluble, highly fermentable, or non-water-soluble, less fermentable based on its solubility in water and fermentation capacity. Water-soluble highly fermentable fibers include pectin, gums, inulin, galactomanans, soluble hemicelluloses (arabinoxylans or pentosans), and mucilages; non-water-soluble less fermentable fibers include cellulose, insoluble hemicelluloses, lignin, and resistant starch [72]. TDF is the sum of the soluble dietary fibers (SDF) and insoluble dietary fibers (IDF) [73]. Water-soluble non-starch polysaccharides in pearl millet account for approximately 0.66% of grain weight and water-insoluble non-starch polysaccharides for 3.88% [70]. Most of the dietary fiber present in pearl millet grains is insoluble fiber. It is composed of cellulose, hemicellulose, and lignin, whereas SDF is made up of arabinoxylan and β-glucan. From the outer to the inner layers of the grain, these five elements are dispersed in different ratios [74]. Compared with the insoluble dietary fraction, the soluble dietary fraction is often concentrated in the inner layers [74]. IDF ranging between 54 and 72 g/100 g was present in the bran-rich fraction [75]. Jha et al. [75] also reported that most of the dietary fiber in pearl millet was insoluble fiber. The endosperm fraction had an IDF content of 5.47 g/100 g, whereas the bran-rich fraction had a high insoluble fiber level of 63.32 g/100 g. Moreover, they observed that pearl millet’s coarse fraction, containing pericarp and germ, had higher IDF content, while its soluble fiber content is larger than the endosperm-rich fraction [75]. Another study conducted by Goudar et al. [57] revealed that of a TDF content of 11.76 g/100 g in pearl millet, soluble dietary and insoluble dietary fractions were 1.91 and 9.85 g/100 g, respectively. Thus, without any ambiguity, the bran of pearl millet has more IDF than the endosperm.
Fiber’s antinutritional effect results from the electrostatic interactions that hold fibers and minerals together. Fiber complexes are formed when the polysaccharide or sulphate groups deprotonate around neutral pH, forming negatively charged groups that facilitate fiber and cationic mineral binding [50]. The complexation of fiber with minerals is unclear in the literature. Other authors assert that fibers only minimally complex minerals, and much of the complexation observed in fiber-rich foods may result from other pollutants, such as phytic acid, which accumulates in the seed coat [50]. On the other hand, Jha et al. [75] found that fiber had a significant impact on the availability of minerals in pearl millet. According to their research, SDF improved Fe and Zn bioaccessibility while insoluble fiber had the opposite impact. Moreover, Krishnan et al. [76] found that a considerable decrease in the coarse fraction of pearl millet led to an increase in Fe and Zn bioaccessibility. Pectin is recognized as one of the SDFʼs with potent cation-binding properties of non-methyl esterified galacturonic acid (GalA) residues, which can ionize (based on the pH of the solution) and impart to pectin its polyanionic properties, including the ability to bind metal ions. The polyanionic nature and mineral binding capacity of pectin will be impacted by variations in its degree of methyl esterification (DM). Furthermore, according to research by Kyomugasho et al. [77] and Rousseau et al. [78], the cation binding ability of pectin is influenced by the presence of neutral sugars as well as the kind and degree of branching. According to Rousseau et al. [69, 78], lignin has a higher mineral binding capacity than cellulose and hemicelluloses due to its polyphenolic composition; however, IDF generally has a negligible mineral binding capacity.
Oxalate is a salt that is made from oxalic acid. Oxalic acid usually forms strong bonds with different minerals, like Ca, Mg, Na, and K, to form oxalate salts. The insoluble Ca oxalate tends to precipitate (or solidify) in the kidneys or in the urinary tract, resulting in the formation of kidney stones [56, 73, 79]. According to Goyal [80], oxalate is engaged in several processes, including the regulation of Ca, the management of metal toxicity, the suppression of oxidative bursts in response to pathogenic invasion, and programmed cell death. Goudar et al. [57] revealed in a recent paper that the oxalate concentration of pearl millet grain was 26.50 mg/100 g. Prior research by Suma and Urooj [32] on two types of pearl millet obtained from the Mysore, India market revealed an oxalate level of 31.6–58.4 mg/100 g for the MRB variety and 36–64.8 mg/100 g for the K variety. In addition, research by Goyal [80] stated that the oxalate level of pearl millet varied from 0.63–1.98% oxalate content. Plants contain both soluble and insoluble forms of oxalate. Soluble forms are created when oxalate combines with K, Na, and Mg (though Mg oxalate is less soluble than the K and Na salts). Insoluble forms are created when oxalate combines with Ca and Fe. Ca oxalate is not soluble at neutral or alkaline pH levels, but it readily dissolves in acid.
Strong bonds are formed between oxalic acid and Ca, Mg, Na, and K. These chemical combinations result in the formation of oxalate salts. Higher oxalate content can bind to Ca in food, making Ca unavailable for normal physiological and biochemical roles, such as the maintenance of strong bones and teeth, as a cofactor in enzymatic reactions, nerve impulse transmission, and as a clotting factor in the blood. Regular consumption of food containing excessive amounts of oxalic acid increases the risk of nutritional deficiencies. To prevent Ca, Mg, and other trace minerals like Fe from being assimilated, oxalate binds to them in the colon. When oxalate comes into contact with different minerals in the gastrointestinal tract, it forms strong chemical links with them, known as chelated bonds. Salts of Ca, Mg, Na, and K are produced because of this chemical reaction [81]. Additionally, soluble oxalate is easily absorbed from the colon into the bloodstream and stays soluble in the liquid portion of the intestinal contents when Ca2+ levels in the meal are low. When the concentration of oxalate ions in the blood that the kidneys filter rises too high, it can react with Ca2+ or Mg2+ to create insoluble oxalate crystals, which can obstruct urine flow and result in renal failure [80]. The primary antinutrient that lowers the bioavailability of Ca in both people and plants is Ca oxalate, which has the highest concentration of bound Ca and is frequently present in crystalline forms. Since Ca absorption depends on the amount of oxalic acid in foods, the Ca oxalate complex affects how well Ca is absorbed and used by the body [40, 56, 81].
An aromatic ring with one or more hydroxyl groups and a range of substitutions is what defines phenolic compounds, which are a broad class of substances [50, 70]. These are secondary metabolites found in plants. Based on their carbon structure, phenolic compounds are classified into many classes, such as stilbenes, lignans, phenolic acids, and flavonoids. According to Raes et al. [50], they can exist as monomers, polymers, or bound to conjugates. Total phenolics localization studies support the notion that polyphenols are primarily found in the pericarp, aleurone layer, and to a lesser degree in the endosperm, that is, the area directly adjacent to the epicarp of the pearl millet grain [37]. The results of the studies conducted by Krishnan and Meera [37] revealed that the content of galloyls in the pearl millet fine fraction (endosperm) and coarse fraction (bran and germ) was 0.929 and 3.317 mg/g, respectively; additionally, they reported that the content of catechol in the pearl millet fine fraction (endosperm) and coarse fraction (bran and germ) was 1.360 and 3.415 mg/g, respectively. These findings validated the notion that phenolics such as galloyls and catechol are more abundant in the bran and germ and less in the endosperm.
Fe can be complexed by ortho-dihydroxy phenolic compounds, which are molecules with galloyl or catechol groups. However, they must first be deprotonated before Fe can form a complex with galloyl or catechol groups. This can be accomplished readily at or below physiological pH [50]. Because Fe prefers octahedral geometry during complexation, it can coordinate up to three catechol or galloyl groups (catecholate or gallate groups), meaning that there may be variable coordination modes during complexation with Fe [50]. Galloyls’ structure includes a trihydroxy benzene group, whereas catechols’ structure includes a dihydroxy benzene group. As a result, galloyls bind Fe more strongly than catechols do. Because Fe may exist in both divalent and trivalent states, it forms complexes with galloyls more frequently than with catechols [37].
According to Urooj [82], food processing is the process of transforming raw materials into cooked or usable form to extend the shelf life of the product, add variety to the diet, supply nutrients that are necessary for health, and make money for the manufacturing company. Processing is divided into three categories: mechanical and bioprocesses [83], modern and traditional [84], and ambient temperature processing [82]. Cleaning, grading, size reduction, mixing, forming, concentration, and separation of food components, and radiation are all examples of ambient temperature processing. Thermal food processing involves the use of high temperatures and includes blanching, pasteurization, heat sterilization, evaporation and distillation, extrusion, dehydration, smoking, frying, and baking [82]. Value-added foods like soaked, cooked, malted, fermented, popped or puffed, extruded, and multigrain flours are made using contemporary processing techniques. The conventional techniques for processing millet encompass flaking, grinding, roasting, fermentation, malting, and decortication. Foods are processed using these methods to enhance their sensory, nutritional, and edible qualities (Figure 4) [84]. While fermentation and malting are regarded as bioprocess technologies, decortication and milling are examples of mechanical processing [83]. A few processing methods are discussed in subsequent sections.
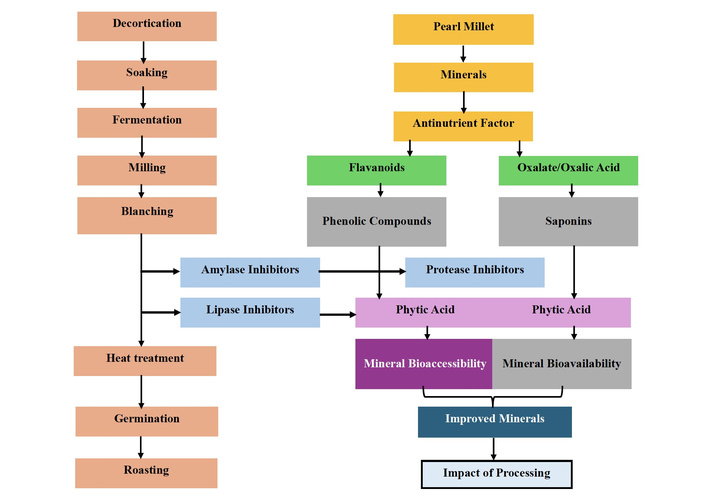
Schematic route of processing mechanisms on mineral bioaccessibility and bioavailability.
Millet and certain other coarse grains are typically dehulled and subjected to various treatments before consumption to improve their sensory and edible quality [85]. The pericarp and bran layers are mechanically removed from the pearl millet grain, reducing anti-nutritional factors such as phytate, tannins, and polyphenols, as well as some minerals and vitamins [8]. It was previously not possible to cook decorticated millet as edible, discrete grains like rice and get a soft texture [83]. Yousaf et al. [86] presented two distinct ways for decortication: a modern dehuller and a stone dehuller. Two conventional techniques previously mentioned by Saleh et al. [83] are hand-pounding and mechanical abrasive decortication.
Using this method, grains are completely submerged in liquid for a predetermined amount of time, activating endogenous enzymes such as phytases that are naturally present in grains. This can remove a significant amount of phytic acid content in grains by activating these enzymes through soaking [87]. The soaking liquid medium can be acidic, neutral, or alkaline; the pH of the soaking medium determines how much an antinutrient is broken down [75]. Five types of pearl millet varieties that were studied had a significant decrease in their phytate content, and their percentage decrease ranged from 23.6–32.4% [88]. The reason for this reduction in phytate content could be that soaking solubilizes phytic acid salts, which are stored in relatively water-soluble form (like K and Na phytate). Additionally, because phytate is present in the aleurone layer and pericarp, it could leach out into the water used for soaking [88].
In addition to increasing the availability and digestibility of nutrients (minerals), fermentation frequently improves the food’s texture, flavor, aroma, and acceptability while also decreasing or degrading certain naturally occurring toxins in the food crop [89]. Fermentation is a metabolic process that uses microorganisms to break down complex materials into simpler forms [90]. In Africa, where modern food preservation techniques are still uncommon, fermentation is commonly used due to its usefulness in food preservation [83]. There are three types of cereal fermentation: controlled, backslopping, and natural (spontaneous). Microorganisms compete during natural fermentation, with the most suited strain becoming victorious. Using a little percentage of a successful batch as starter cultures is known as backslopping. For shorter lag times, controlled fermentation employs strains. Although less efficient, natural fermentation (spontaneous) is widespread in underdeveloped nations. Additionally, solid-state or submerged/liquid fermentation is possible [89].
The main processes involved in milling are grinding, sifting, separation, and regrinding. These are repeated to extract specific parts of the grain, such as endosperm, and to obtain bran as a by-product [91]. The process involves separating endosperm, bran, and germ. To facilitate the production of fine flour, endosperm is ground into small and fine particles; hammer mills and roller mills can be used to reduce the particle size. Additionally, millet grains are typically ground by a nonmotorized grain mill that is operated by hand or another nonelectric method, particularly in rural areas for household purposes. Nevertheless, a manual grain mill that is connected to a gas or electric motor via a pulley system can also be used in this manner.
Blanching (briefly immersing food in boiling water or steam) is a thermal treatment means of processing food relative to production, harvesting, and transportation [92, 93]. One of the best pre-milling techniques for extending the shelf life of the finished product is blanching. Without significantly altering its nutritional makeup, it might slow down the enzymatic activity [8]. Concisely, blanching, which involves heating steam or hot water, helps reduce or destroy microbiological activity, removes moisture to extend shelf life, maintains food quality to promote consumer acceptability of the final product, and inactivates enzymes that could cause quality degradation.
Prior to the advent of convenience foods, heat treatment was one of the most significant pre-milling processes for pearl millet because it has a relatively short shelf life and serves as a major catalyst for increasing daily consumption [8]. Cooking is regarded as a household method of making basic, traditional staple foods [86].
It is common to use the phrases “germination”, “sprouting”, and “malting” interchangeably [86, 94]. Steeping in water, imbibition, endogenous metabolic activation, and radical emergence are all steps in the process. Hydrolytic enzymes are activated and synthesized from scratch during sprouting. Improved mineral bioaccessibility and the release of trapped minerals result from the hydrolysis of phytate by hydrolytic enzymes such as phytase activity [86]. Additionally, pearl millet’s germination lowers its phytate content. Furthermore, the decline was greater at a grain-to-water ratio of 1:5 than it was at 1:2. Thus, the phytate level was significantly decreased by a longer duration and a larger amount of soaking water [88].
To reduce the antinutritional components that impact the minerals’ bioaccessibility and bioavailability, a variety of processing techniques are commonly used. It considers where minerals and antinutritional compounds are found in plant tissues and how they interact with each other. In traditional grain processing and grain preparations, these processing methods, either separately or in combination, are frequently carried out at the home level. Additionally, several of them are used in the industries (Figure 5). Processing increases mineral availability by eliminating inhibitors and positively transforming dietary ingredients into intricate ligands for metal ions [82].
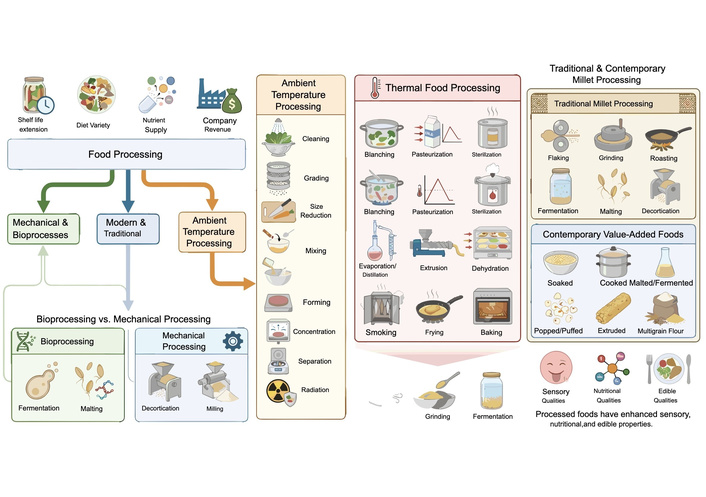
Summaries of food processing methods. The figure was created using FigureLabs (https://figurelabs.com).
Hydrolytic enzymes like phytase are activated and undergo de novo synthesis when sprouting occurs. Additionally, as germination time increases, pearl millet’s phytase activity gradually rises. After 72 h of germination, the entire pearl millet’s phytase activity rose from 0.190 μmol/g to 1.890 μmol/g in the coarse fraction and 0.461 μmol/g in the fine fraction. According to Krishnan et al. [76], the fractionated native grains’ phytase activity showed that the enzyme was primarily concentrated in the coarse fraction as opposed to the fine fraction. Enhanced mineral bioaccessibility and the release of trapped minerals resulted from increased phytase activity, which also causes phytate hydrolysis [94]. When compared to raw pearl millet flour, the Mg, Ca, Na, and Zn levels of the three-day-germinated millet flour increased [34]. Moreover, germination raised the Fe content of millet somewhat, according to Sow et al. [95]. Thorat et al. [88] found that, however, the Fe and Zn contents of germinated and malted pearl millet grains decreased when compared to the raw millet samples. In this study, the grain: water ratio during soaking resulted in the loss of minerals, with a large volume of water to grains leading to a significant loss in Zn, and vice versa in the case of Fe. They observed 8.8% to 17.5% loss upon germination, and they hinted that the low percentage of Fe could be due to the period of germination, which was short, 48 h, in contrast to other researchers who examined the effects of germination for longer periods of time, up to 96 h. After 4 days (96 h) of germination, Zanabria Eyzaguirre et al. [96] found that the pearl millet’s total Fe concentration had decreased by 50%.
In some fermented foods that are popular in India, fermentation raised the concentrations of Mg, Fe, Ca, and Zn; this rise was correlated with a decrease in phytates [97]. On the other hand, since bacteria break down protein and carbohydrates during fermentation, dry matter loss may also contribute to the rise in mineral content [98]. Food grain fermentation increases the minerals’ bioavailability [63]. Fe levels in the millet were very slightly reduced during the fermentation process, going from 7.56 mg/100 g to 7.55 mg/100 g. The short soaking time after fermentation was the cause of the minor reduction [95]. Research indicates that Hausa “koko” with bread and “akara”, a fermented porridge made from pearl millet in Ghana, has a high Fe content (15 mg/100 g) and a high Mg content (1.2 mg/100 g), but low levels of Na and Cu. The reason for the relatively high Fe content in the Hausa “koko” with bread and “akara” could be the processing method (soaking millet, milling, fermentation, and cooking), which also likely involves the processing of “akara”. The Mg content was a result of using whole grains, which are the richest sources of Mg. The low Ca content is caused by phytate’s inhibitory effect, as the phytate:Ca molar ratio (8:2) was very high above the critical level of 0.24, but it was advantageous for Zn and Fe bioavailability in the diet.
Soaking is a pretreatment that facilitates grain processing and is the most important procedure in cereal germination and fermentation processing, according to the review by Feizollahi et al. [60]. They also discussed how endogenous phytase activity and pH-dependent leaching of soluble phytate salts into the soaking medium affect the phytate level after soaking, and how discarding soak water prior to these processing steps lowers phytate, since phytate is soluble in water. Leaching of phytate is influenced by the characteristics of the phytate, which is a type of salt containing various minerals, including K, Ca, or Mg. It has been found that soaking causes the overall Fe and Zn contents to drop. It was discovered that soaking millet grains in water decreased their Fe and Zn concentrations; this could be because the minerals in the soaking water leached [88, 99]. According to other research, the Fe content of pearl millet flour decreased dramatically when the soaking duration increased from 0 to 12 h [100]. Additionally, a 24 h soaking resulted in greater Zn (18%) and Fe (48%) losses in dehulled millet grains relative to Fe loss in intact millet grains. Once more, the losses were attributed to the minerals leaching into the soaking medium because of a concentration gradient, and the variations in the percentage losses of the minerals were attributed to the locations of Fe and Zn [101]. Zn is more concentrated in the embryo, but Fe is more prevalent in the pericarp [23]. Reducing anti-nutrients through soaking has been shown to improve mineral bioavailability [102]. Jha et al. [75] investigated how soaking circumstances affected the bioaccessibility of Fe and Zn in Indian pearl millet. Apart from endosperm soaked in water, they discovered that soaking grain fractions reduced the amount of Fe. All bran fractions, except the bran soaked in water, showed a decrease in Zn content. Endosperm and bran-rich fractions showed an increase in Fe bioaccessibility because of the solubilization of insoluble fiber and a decrease in phytate content. Zn bioaccessibility, however, declined in the endosperm-rich fraction, suggesting that different inhibitory factors had different effects on mineral bioaccessibility within the grain. The effects of pre-treatments that are typically applied in homes on Fe bioavailability are helpfully understood by means of this kind of research [103]. It has been reported that soaking causes Zn and total Fe loss. Zn and Fe levels drop by roughly 20–40%, with losses increasing as soaking times are extended from 12 to 24 h and, in the case of Fe, when more water is used during soaking (grain to water ratio of 1:5) [83]. Zinc losses in the first case and Fe losses in the second case were largely caused by the type of millet and the huge volume of soaking water medium [83].
By browning the surface of the food, roasting is a processing method that uses dry heat from an oven, open flame, or other heat source to improve flavor. It has been observed that adding roasted pearl millet to beef sausages boosted the sausages’ mineral contents overall, especially those of Mg, Fe, and Ca [104]. The roasted pearl millet improved the mineral content of the sausages by adding minerals. The amount of Fe, Mg, and Ca in the sausages was considerably higher when roasted pearl millet flour was utilized.
The effects of 2, 4, and 6 min decortication on Fe-binding phenolics (galloyls and catechols) were investigated by Krishnan and Meera [37]. They found that decorticating pearl millet for 4 min enhanced the galloyls and catechols in the bran fraction while decreasing the grain fraction. However, the galloyl and catechol contents of the bran and endosperm fractions, respectively, decreased after an additional 6 min of decortication. The galloyls and catechols are more restricted to the outer regions of the grain, below the epicarp region, which explains the results that have been found [37].
According to Adebiyi et al. [105], pearl millet flours and biscuits exhibited enhanced nutritional and health-promoting properties following malting and fermentation. Except for Ca and Fe, the mineral levels in the biscuit samples were lower, indicating that the heat only marginally enhanced the minerals’ extractability. Moreover, malted and fermented flours increased the bioavailability of Fe. The Fe and Zn content of fermented pearl millet samples that underwent germination showed a notable rise with time. The biggest increases in Fe and Zn occurred during the process under ideal germination conditions. With relation to Ca, however, the pattern was different because, although there were notable fluctuations, the Ca concentrations essentially remained steady. The lowest germination conditions yielded the highest Ca amounts [106]. According to Krishnan et al. [76], the treatments reduced Fe content by 25% and 9% in the fine and coarse fractions, respectively, while decreasing Zn content by 46% in the fine fraction and 39% in the coarse fraction. The study examined the impact of germination-hydrothermal treatments followed by fractionation on Fe and Zn bioaccessibility in pearl millet fractions. The primary cause of the decline was the minerals that leached during the soaking stage of the germination process. According to Krishnan et al. [76], there was a variation in the drop between the fractions, with the coarse fraction experiencing a smaller fall than the fine fraction. The primary reason for the variation in Fe and Zn content was that these two minerals were distributed differently throughout the grain; Fe is primarily located in the scutellum, whilst Zn is primarily found in the embryo [23]. Zn bioaccessibility was only enhanced in the coarse fraction by the germinated-hydrothermal treatments, although Fe bioaccessibility was raised in both the fine and coarse fractions. This results from a general decrease in the levels of phytic acid (49%) and galloyls (48%) as well as IDF (24%) [76]. When compared to untreated pearl millet grains, the Fe and Zn contents in germinated dehulled steamed pearl millet endosperm were lower. In contrast to the untreated pearl millet, the germinated dehulled steamed endosperm had a higher bioaccessibility of Fe and Zn. Since the endosperm had a significant amount of Fe, the high amount of Fe was caused by the germs remaining in the endosperm after decortication. Moreover, the endosperm of germinated steamed grains exhibited a greater degree of phytate breakdown (80%), which influenced the bioaccessibility of Fe and Zn [75].
Mineral bioaccessibility and bioavailability significantly improve when specific chemicals are present. The bioavailability of these substances can readily be improved by quite considerable amounts even with small changes in their concentrations. For example, very few Fe and Zn promoters have been found. Depending on how well phytoferritin prevents Fe from attaching to phytates, it might be the most promising Fe absorption booster. In the small intestine, organic acids like citric and ascorbic acid convert Fe to its ferrous state and create a soluble Fe chelate that enhances absorption. It is thought that high concentrations of cysteine peptides, which can boost Fe absorption, and amino acids, which chelate Fe and are absorbed intact by amino acid carrier proteins, are responsible for the improved absorption from meat, fish, and poultry [107]. Prior research by Gibson [108] revealed that ascorbic acid increases non-heme Fe absorption by reducing Fe (III) to a more soluble Fe (II) and forming Fe ascorbate chelate. It also mitigates the negative effect of phytate.
Food-to-food fortification is a technique that increases the mineral bioaccessibility of another food by enhancing it with a local source of micronutrients. This is another way to enhance the nutrients in food. Van der Merwe et al. [109] claim that adding baobab fruit pulp to cereal porridge with a provitamin A source enhances the bioaccessibility of Fe and Zn. Baobab fruit pulp’s strong mineral bioaccessibility-enhancing content is most likely the cause of this enhancement in Fe and Zn bioaccessibility. Nonetheless, Adetola et al. [110] credited baobab fruit powder with high concentrations of ascorbic acid and citric acid, 174 mg/100 g and 4,440 mg/100 g db (dry basis), respectively, for improving Fe bioaccessibility in cereal-based porridge. It has been demonstrated that grinding cereal grains and separating the bran and germ from the endosperm during the milling process improves mineral bioaccessibility and bioavailability, even though the mineral content of milled cereals may be concurrently decreased [111]. The use of sweet potatoes as a natural source of β-Carotene in pearl millet gruel was investigated by Mawouma et al. [112]. They added mashed sweet potato to the millet gruel at levels of 20%, 30%, and 40% (v/v) to fortify it. They discovered that Fe bioaccessibility increased with incorporation level, with the largest rise occurring at the 30% level. Possible explanations for these outcomes include the activating effect of β-carotene on bioaccessibility. Typically, in the stomach, β-carotene and Fe create a soluble complex that facilitates Fe absorption even when antinutritional elements are present. The percentage bioaccessibility of Zn in the gruels enriched at 20% and 30% was considerably higher (P < 0.05) than in the control group. At 40% integration, there was a decrease in Zn bioaccessibility, nevertheless. They clarified that the rise at 20% and 30% could be attributed to the stimulatory action of β-carotene on Zn bioaccessibility, while the decrease at 40% might be the result of the compound’s extremely high antinutritional content. It has been demonstrated that sulfur-rich compound spices, such as onion, garlic, and the strong flavors of red pepper, black pepper, and ginger, improve the solubility of minerals in cereal grains.
On solid media, the degradation of phytate by yeast phytases has been investigated. According to Greppi et al. [113], Pichia kudriavzevii, which was isolated from “Gowe” and “Mawe”, affected the profuse production of phytase, which in turn caused the breakdown of phytic acid. Additionally, on both phosphate-free and high-phosphate media, the Pichia kudriavzevii TY13 strain isolated from “togwa” produces higher levels of phytase to break down [114]. The ability of the isolated yeast to break down food-grade phytin complexes in the food matrix was not tested in either study, nor were the amounts of various minerals released during the breakdown of phytate quantified. Raghavendra [115] did, however, screen for phytate-degrading lactic acid bacteria (LAB) isolated from several Indian foods in a thesis work. The investigation found that 20 isolates with the ability to break down phytate on de Man-Rogosa-Sharpe (MRS) agar containing phytate and MRS agar containing Na phytate in addition to CaCl2 were obtained from the intestines of chickens, marine fish, raw milk, and red rice. Accordingly, such cultures can break down food-grade phytin complexes, which enhances the bioaccessibility of Ca, Mg, and Zn in malted finger millet seed coat and soymilk. The levels of free Ca, Mg, and Zn increased noticeably, improving their bioaccessibility. This is because the phytin complex was destroyed by the action of phytic acid LAB, which led to the release of the bound Mg, Ca, and Zn.
The food industry has come to terms with the fact that one major indispensable tool for addressing challenges is optimization. In optimization, favorable conditions are figured out and improved for optimal outcomes. The production systems geared towards the identification of the best conditions are done systematically [116]. The old approach of using one-factor-at-a-time has been metamorphosed to response surface methodology, which is characterized by experimental modelling and variable interactions. Rheem [117] further buttressed this view by indicating that response surface methodology focuses on using statistical and practical means for experimental designs, response modelling, and factor level optimization.
Soaking and fermentation are wet processing steps adopted during the preparation of fermented foods. Optimization of these processing steps is critical for maximizing Fe, Ca, and Zn bioaccessibility, reducing antinutrients such as phytate, and retaining desirable organoleptic properties [118]. Soaking directly affects enzyme activation, phytate solubilization, and physical leaching of water-soluble nutrients and is the first major hydration step in many fermented food productions. Prolonged soaking (> 24 h) coupled with high volume of water has a tendency to increase the loss of total minerals through diffusion into the soak water, while short-to-moderate soaking (6–12 h) frequently reduces phytate and limits mineral leaching [119, 120]. Thus, the grain: water ratio is considered a significant parameter for mineral bioaccessibility outcomes. Larger water volumes, such as 1:5 (w/v) millet-water, are often characterized by high dilution, thus increasing the concentration gradient that drives soluble mineral and phytate complexed mineral to diffuse out of the grain, collectively leading to absolute mineral loss and phytate decline [121, 122]. Conversely, low water ratios such as 1:2–1:3 (w/v) limit leaching; however, reduce the effectiveness of endogenous phytase or exogenous microbial activity because of limited mobility. Thus, the optimization of soaking conditions is an essential practical means not only to balance sufficient water to permit grain hydration but also to promote enzymatic hydrolysis for antinutrient degradation while minimizing unnecessary dilution and discarding of the soak liquor. Recent studies suggest that 12 h soaking with a moderate grain:water ratio of 1:2–1:3 achieved significant phytate decline with acceptable mineral retention in millets and related cereals [119, 120]. Below the moderate grain:water ratio of 1:2, a decrease in mineral content has been reported [123] due to a decrease in mobility of endogenous and exogenous phytases from microbial activity [119, 120]. Stronger evidence from research earlier supported the preference of moderate, millet to water ratios, demonstrating a balance between a grain:water ratio of 1:3. However, it is noteworthy that the nutritional value of a processed food depends on what remains and its bioaccessibility (Figure 6). Therefore, though losses of minerals during soaking are inevitable, it does not always equate to the nutritional value of the final product [119, 120]. Feitosa et al. [124] and Luo and Xie [125] arguably reported that discarding soak water alters Fe and Zn bioaccessibility in legumes. Furthermore, it has been noted that while soaking reduces phytate levels and increases prospective mineral bioaccessibility, the simultaneous leaching of minerals into soak water may counteract these benefits, leaving the bioaccessible mineral fraction with little to no net improvement [63, 126].
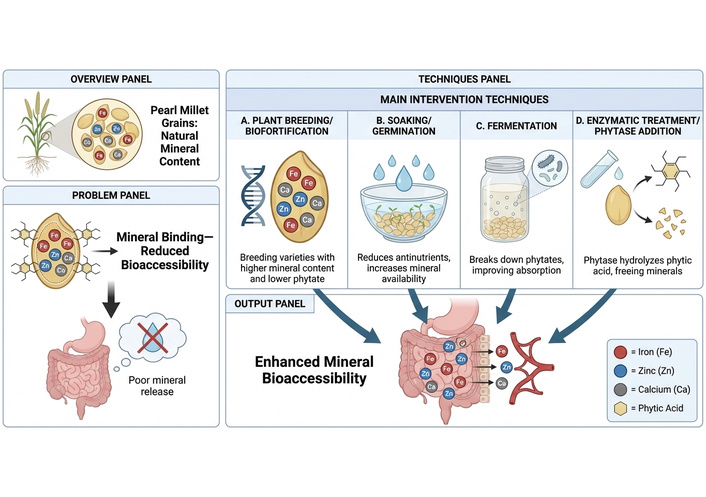
Techniques for improving pearl millet’s mineral bioaccessibility. The figure was created using FigureLabs (https://figurelabs.com).
Soaked millet undergoes modifications due to microbial acidity and enzymatic activity during fermentation, which can result in the production of a variety of food products. Low phytate content, flavor production, and partial proteolysis are characteristics of the food products produced under these conditions. This leads to improved nutrition. Porridges obtained from 24–72 h fermented cereals are known to have high reduced phytate and hence increase bioaccessibility [127, 128]. One major benefit from fermentation linked to mineral nutrition is the enhancement of microbial phytase activity and the resulting drop in pH, which increases the solubility of Ca, Fe, and Zn [129, 130]. Slurry creation resulting from adding water creates consistency during fermentation, and this influences microbial activities. Although a more diluted slurry promotes quicker acidification and enzyme diffusion, which speeds up the breakdown of phytate, it also raises the risk of mineral leaching when the fermentate is removed later in the process [122]. Thus, Atter et al. [118] recommend careful control of fermentation water and recovery of fermentation liquor where possible to avoid nutrient loss. Furthermore, Manzoor et al. [122] and Anberbir et al. [127] reported that reducing the water volume for fermentation is necessary to avoid discarding of supernatant. They added that the reduction increases acidity, taste development, and phytate hydrolysis. Additionally, they claimed that using a controlled LAB starter culture resulted in increased phytate breakdown, acidification, and high taste development between 24 and 48 h. Compared to spontaneous food fermentation, starter culture makes it easier to accelerate, break down phytate, and standardize fermentation. Thus, shorter duration and risk of over-dilution of fermentation processing for achieving enhanced mineral bioaccessibility are made possible using starter culture [128, 130]. It is therefore important to find means of balancing phytate reduction and mineral retention.
Despite being an orphan crop, millet has significant health benefits when consumed. Different processed foods made from pearl millet are found in different parts of the world, indicating the promotion of pearl millet as a smart food. Pearl millet (Pennisetum glaucum) has garnered notice for its distinct nutritional composition in the wake of efforts to mitigate food security issues, undernutrition, and malnutrition to achieve the United Nations’ 2030 sustainable development zero hunger goal. To maximize the nutritional potential of pearl millet, it is important to highlight millet food products and processing methods that work in line with mineral bioaccessibility and bioavailability.
Millet is commonly used alone or in combination with other cereals to produce a wide variety of foods and beverages. The main categories of traditional foods where pearl millet can be effectively used are fermented and unfermented flat breads, fermented and unfermented thin or thick porridge “koko” steamed and boiled products, snack foods, alcoholic and non-alcoholic beverages [3]. Pearl millets are used to make a variety of traditional foods. In sub-Saharan Africa, the most widely available traditional pearl millet food products are the fermented thin porridges, such as “ogi” and “koko”; on the other hand, there is the common unfermented thick porridge made from millet flatbreads (roti). Other foods that are sold in the market include alcoholic beverages like opaque beer or Dogon millet beer, “mbeg”, “merissa”, and non-alcoholic drinks like “pombe”, “pito”, “boza”, “kunun-zaki”, “obushera”, “mahewu”, “oskikundu”, “marewa” [104], and steamed products like “kudumu”, “fura”, and “couscous” [131]. Various nations process pearl millet to provide a wide range of edible foods. For example, millet is consumed in India in the form of bread, cake, chips, “wadi”, “idli”, chapati, and “laddoo” [86]. China, Russia, and Poland eat it as congee or porridge. Millet flour is eaten in Senegal as a porridge with meat sauce and mixed veggies. Millet is a main food consumed by the people of Namibia. In Sudan, a thin pancake prepared with pulverized millet is well-known. Food made from millet is known as chapati in Pakistan [132, 133]. According to Pswarayi and Gänzle [134], cereal-based fermented beverages are known as “mahewu” in Zimbabwe, “oshihundu” in Namibia [135], and “togwa” in Tanzania [136].
A traditional treat from Northern Ghana and Nigeria, “tuwo” is prepared from millet, maize, or sorghum, depending on the producer’s preference. It is a solid meal that's formed into balls or swallows. It can be made using whole grains or dehulled grains. Fine flour is created by milling the grains. If the flour is made from whole grains, the hulls are removed by appropriately sieving the flour. When making “tuwo or tuo-zaafi”, water is heated to boil in a saucepan, some flour is sifted into cold water to create a slurry, and the cold slurry is then added to the boiling water to ensure proper boiling. After that, the food is heated for a further 10 to 15 min, stirred, and served with any preferred soup [84]. The millet flour is sieved and then gradually added to the pot along with the boiled water and slurry. Stir until the desired thickness is reached.
“Ben-saalga”, also known as Hausa “koko”, is a fermented gruel made mostly from millet and produced in many traditional production units throughout Ghana and Burkina Faso. After washing and soaking the grain, pearl millet is ground, kneaded, sifted, settled, and cooked. In small amounts before grinding, aromatic components, including ginger, pepper, black pepper, and mint, are typically added, depending on the “ben-saalga” or “koko” producer’s custom. The supernatant from the settling process is collected and heated to almost boiling for 40 min during the final cooking stage. For consistency, the paste is then added to the supernatant and cooked for 7 min while stirring. Depending on the sourness degree of the supernatant, most processors boil either just the supernatant or a combination of fresh water and the supernatant for the manufacture of Hausa “koko”. To get the right consistency for Hausa “koko”, the heated supernatant or diluted supernatant is then gradually added to the sediment in an aluminum basin or pot while being constantly stirred.
The most popular traditional drink made in Uganda’s western highlands, where sorghum and millet are significant commercial and staple crops, is “bushera”. Both adults and small toddlers use the product. “Bushera” is prepared in a variety of ways. To manufacture flour, germinated millet or sorghum grain is mixed with boiling water and allowed to cool to room temperature. Next, incorporate the germinated millet or sorghum flour and allow the mixture to ferment at room temperature for one to six days [15].
Nigerians make “kunu”, a wholesome non-alcoholic beverage, from a variety of cereal grains, including rice, sorghum, millet, and maize. People of all age groups frequently utilize it as a dietary supplement or to relieve their thirst. “Kunu” is inexpensive and a good substitute for fizzy drink products, which offer their customers little to no nutritional value [84, 137]. To make “kunu”, the millet grain must first be cleaned and steeped in twice as much water as it has volume. Next, the grains must be washed and spiced with ginger, red pepper, cloves, and black pepper. Finally, the steeped millet grains and spices must be wet-milled in a grinding machine, sieved to remove the chaff, and the supernatant must be decanted from the slurry. Finally, the slurry should be divided into two equal parts, with one half being added to boiling water and stirred for 5 min. Lastly, the other half should be mixed with the cooled half-slurry. After adding adequate water, the mixture is stirred and given time to settle. The filtrate, which has been sweetened with granulated sugar, is then suitably mixed and sieved through a muslin cloth to produce the freshly processed millet beverage. The product is bottled in plastic bottles [84].
After being slightly moistened with water, the millet grain is processed in a locally manufactured disc attrition mill. The grain is pulverized in a hammer mill and sieved after the hull is removed, after it has dried in the sun. To make a smooth dough, mix pearl millet flour, powdered ginger, powdered black pepper, and water (95°C) in a mortar and pestle. The dough is formed by hand into balls, put in a pan of boiling water, and cooked for 30 min. While the balls are still hot, they are kneaded once more until a smooth, slightly elastic mass is achieved. After that, the dough is formed into “fura” balls [15] and floured [84].
This fermented gruel made from cereal is made and consumed in West Africa, primarily in Nigeria. According to Amadou et al. [15], “ogi” is a porridge made from fermented cake or paste derived from millet, sorghum, or maize. It is frequently used as a baby’s weaning diet, an adult’s breakfast, and a sick person’s diet [136]. The processing includes washing and steeping the grains for 12 to 72 h, decanting the fermenting water, wet-milling the grains to create a paste, wet-sieving the paste to create a filtrate, and allowing the filtrate to settle and ferment for one to three days. Warm water typically reduces the amount of time cereal needs to soak. Additionally, the producer’s preferred pH and taste determine how long “ogi” takes to ferment [136]. “Ogi” is the origin of numerous regional cuisines that go by different names. Boiling the slurry produces porridges. Other types of “ogi” include “eko” (stiff gel) and “akpan” (fluid or semi-solid cooked “ogi”) [15, 136, 138].
This is a non-alcoholic drink made by fermenting mixtures of millet, sorghum, cassava, and maize. In Tanzania, “togwa” is an indigenous fermented dish that is frequently eaten by adults and small children. Sorghum and millet, or sorghum and maize, are combined in a 1:1 (w/v) ratio to produce “togwa”. After that, water is added and combined to create a slurry in a 1:9 (w/v) ratio, which is boiled to create gruel. After cooling the gruel to around 30°C, sorghum malt and aged “togwa” are added. The mixture is allowed to ferment for one to three days. The preparation of “togwa” is akin to that of Mahewu in South Africa. The kind of inoculum utilized differs primarily in that the Mahewu recipe uses wheat flour as an inoculum [136].
“Oshikundu” is a traditional non-alcoholic fermented cereal beverage that is drunk in Namibia’s Ohangwena, Omusati, Oshana, and Oshikoto areas. When yeast is utilized for fermentation, the beverage can turn into an alcoholic one. Malted sorghum meal and pearl millet (“mahangu” meal) are used to make it. This beverage is made by women in Northern Namibia for household use, but it is also offered for sale in public markets. The beverage is consumed in a single day due to its incredibly short shelf life of less than 6 h. It has the potential to be commercialized due to its processing method, which is why Namibia’s school meal program has included it. To prepare the “mahangu” meal, boiling water is added, the mixture is allowed to cool to room temperature, and it is periodically stirred. Malted sorghum meal is then added to the mixture. In some regions, the “mahlangu” meal and malt sorghum meal are blended before the boiling water is added. The mixture is then blended and allowed to cool [132, 135]. At this stage, adding bran is optional, but regardless of the producer's strategy, it is added. Previously fermented “oshikundu” is also added. Water is added to the mixture to dilute it. The fermented “oshikundu” is ready to consume after the mixture has been allowed to ferment for several hours at room temperature.
“Mahewu” is a nonalcoholic beverage made from cereal that is produced and consumed in Zimbabwe. It is refreshing and can be used as an additional food source for young children. To make it, prepared millet or sorghum malt is combined with cooked and cooled maize porridge. The mixture is then allowed to ferment in a plastic bucket or clay pot at room temperature for 16–48 h [134].
The current study has provided insight into strategies for improving mineral bioaccessibility in millet-based diets. To improve the mineral bioaccessibility and bioavailability, processing procedures are required for the destruction of antinutritional substances. Depending on the processing criterion used, different optimized processing techniques have varying effects on pearl millet antinutrient degradation and mineral bioaccessibility. While some of these processing methods, such as prolonged soaking of pearl millet, have a deleterious effect on minerals, others have a favorable impact on mineral bioaccessibility and bioavailability. It is not possible to boost mineral bioaccessibility by concentrating on the breakdown of a single antinutritional factor. Mineral bioaccessibility is made possible by microbial starting cultures with noticeably greater extracellular enzymes, such as phytases, which reduce antinutritional elements, especially phytate, in consumer food. Additionally, adding two or more enhancers, like ascorbic acid and cysteine, during food processing is more effective at increasing mineral bioaccessibility and absorption because a treatment using both enhancers outperformed a treatment using just one of them for both Fe and Zn. Micronutrient deficiencies in developing nations can be addressed by fortifying pearl millet-based diets with a variety of food-based fortificants, such as sweet potatoes, baobab pulp, vegetables, and fruits. When used, bioenrichment with basic home processing methods could increase the nutritional bioaccessibility of foods made from pearl millet. The creation of antinutritional probiotic strains and their preservation by microencapsulation technology could aid in the bioaccessibility of minerals in the stomach as well as in the dietary matrix. The use of starter culture fermentation to lower phytate in millet offers a cheap way to address micronutrient deficiencies among marginalized communities.
IDF: insoluble dietary fibers
IP3: inositol triphosphate
IP4: inositol tetraphosphate
IP5: inositol pentaphosphate
IP6: inositol hexakisphosphate
IYM: International Year of Millets
LAB: lactic acid bacteria
MRB: Maharashtra Rabi Bajra
MRS: de Man-Rogosa-Sharpe
PIXE: proton-induced X-ray emission
SDF: soluble dietary fibers
SGF: simulated gastric fluid
SIF: simulated intestinal fluid
SSF: simulated salivary fluid
TDF: total dietary fiber
The authors thank the Kwame Nkrumah University of Science and Technology in Kumasi, Ghana, the Department of Food Science and Technology, University of Energy and Natural Resources, Sunyani, Ghana, and the Metallic Contaminants Laboratory of the Ghana Standards Authority, Accra, Ghana, for their collaborations, technical assistance, and provision of resources for a successful completion of the research. During the preparation of this work, the authors used FigureLabs for preparing the figures. After using the tool, authors reviewed and edited the content as needed and took full responsibility for the content of the publication.
AW: Conceptualization, Methodology, Software, Formal analysis, Data curation, Writing—original draft. FA: Resources, Writing—review & editing, Project administration, Supervision. EFB: Validation, Writing—review & editing, Investigation, Software, Visualization. JOK: Project administration, Writing—review & editing, Resources. FCMR: Methodology, Writing—review & editing, Investigation, Validation. All authors read and approved the submitted version.
Evans Frimpong Boateng, who is the Young Editorial Board Member of Exploration of Foods and Foodomics, had no involvement in the decision-making or the review process of this manuscript. The other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1831
Download: 101
Times Cited: 0
