Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
†These authors contributed equally to this work.
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
†These authors contributed equally to this work.
Affiliation:
2Department of Critical Care Medicine, The First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Affiliation:
1Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China
Email: cqf19760516@163.com
ORCID: https://orcid.org/0000-0003-4740-5622
Explor Endocr Metab Dis. 2026;3:101463 DOI: https://doi.org/10.37349/eemd.2026.101463
Received: September 08, 2025 Accepted: March 01, 2026 Published: March 12, 2026
Academic Editor: Charlotte Steenblock, University Clinic Carl Gustav Carus, Germany
Aim: Necrotizing fasciitis (NF) is a severe and early challenging-to-identify complication of diabetic foot (DF). This study aimed to develop and validate a novel risk assessment model for NF with DF patients utilizing conventional clinical indicators.
Methods: A retrospective analysis was conducted on 815 DF patients admitted to the First Affiliated Hospital of Chongqing Medical University between October 2018 and April 2022. Based on the presence of NF, patients were stratified into a DF group (n = 703) and a DF complicated with NF (DNF) group (n = 112). Clinically and statistically significant variables were converted into categorical form. A new risk assessment for DNF (NRADNF) nomogram was developed via multivariable stepwise logistic regression. Model performance was evaluated using the area under the receiver operating characteristic curve (AUC) for discriminative ability, the Hosmer-Lemeshow goodness-of-fit test for calibration, decision curve analysis (DCA) for clinical utility, and bootstrap resampling for stability.
Results: The final NRADNF model incorporated six indicators: age < 60 years, body temperature ≥ 38°C, foot skin necrosis, neutrophil-to-lymphocyte ratio (NLR) ≥ 8.5, hypersensitive C-reactive protein > 20 mg/L, and hemoglobin ≤ 100 g/L. The model demonstrated favorable predictive performance with an AUC of 0.815 (95% CI: 0.773, 0.857), and it was significantly superior to the RADNF model by our team (P = 0.027). Calibration curves and the Hosmer-Lemeshow test indicated good accuracy. DCA confirmed the model’s clinical net benefit, and internal validation via bootstrap resampling supported its stability.
Conclusions: Based on its favorable predictive performance and accessible indicators, the NRADNF model is suitable for preliminary screening of DNF in clinical practice.
Necrotizing fasciitis (NF) is a rare and highly destructive soft tissue infection characterized by rapid progression and severe clinical manifestations. Without timely diagnosis and treatment, it can lead to serious complications and even death [1]. Studies have indicated that the case fatality rate following delayed treatment can be as high as 100% [2]. Despite advances in medical science, early diagnosis of NF remains a significant clinical challenge [3].
Diabetes mellitus (DM) is a major risk factor for NF, and its complication, diabetic foot (DF), predisposes patients to NF if not properly treated. Furthermore, DF complicated with necrotizing fasciitis (DNF) face a higher risk of amputation [4]. Meanwhile, DNF patients are more likely to experience misdiagnosis and delays due to neurovascular disease, compromised immune function, decreased resistance to infection, and atypical signs of infection [5, 6]. Currently, the laboratory risk indicator for necrotizing fasciitis (LRINEC) scale is often used to screen for NF in the general population [7], which has demonstrated limited accuracy in predicting NF in general patients [8, 9]. Additionally, the LRINEC scale must be used with caution in DM patients, as it appears more sensitive in patients with electrolyte imbalances and kidney damage, which can confound the diagnosis [10]. Modified LRINEC (m-LRINEC) adds diabetes and kidney disease to the original LRINEC scoring system [11], but its validity should be confirmed in future larger studies. Our research group developed the first risk assessment for DNF (RADNF) scale for DNF screening, composed of temperature, hemoglobin A1c (HbA1c), and hypersensitive C-reactive protein (hs-CRP), which yielded an area under the receiver operating characteristic curve (AUC) of 0.864 (95% confidence interval [CI]: 0.785–0.943) [12]; however, due to its relatively small sample size, the scale’s validity warrants further investigation.
In order to establish a screening scale applicable to DNF and bridge the above deficiency, we retrospectively analyzed 815 patients’ data. Firstly, we analyzed the risk factors of DNF and constructed a prediction model based on these risk factors. Finally, we performed a multi-dimensional validation for the model.
A total of 1,024 patients with DF infection were initially enrolled from the First Affiliated Hospital of Chongqing Medical University between October 2018 and April 2022. Following screening, 815 patients with DF were included in the study, among whom 112 were diagnosed with complicated NF. Inclusion: the presence of a foot ulcer or gangrene in individuals with DM, Wagner classification: 2 to 5 grades. Exclusion: 1) Wagner grade 0–1 lesions (98 cases); 2) concurrent other active infectious diseases (74 cases); 3) cancer or use of immunomodulatory medications including corticosteroids and antitumor agents (28 cases); 4) hematologic disorders other than anemia (9 cases). The study protocol was reviewed and approved by the ethics committee of the First Affiliated Hospital of Chongqing Medical University (approval No. 2025-786-01), and retrospective analysis waived the patient’s informed consent form. The study complies with the Declaration of Helsinki.
We obtained data on physical examination and medical history at admission by reviewing medical records, including height, weight, body temperature, diabetes duration, diabetes complications (retinopathy, peripheral neuropathy, diabetic nephropathy), foot skin necrosis, and Wagner grade. Body mass index (BMI) was calculated as weight in kilograms divided by the square of height in meters. Blood samples were collected before antibiotic intervention. Evaluations of white blood cells, neutrophils, platelets, lymphocytes, monocytes, hemoglobin, hs‑CRP, HbA1c, albumin, sodium, creatinine, and fibrinogen followed previously described protocols [13, 14]. The neutrophil-to-lymphocyte ratio (NLR), platelet-lymphocyte ratio (PLR), and lymphocyte-monocyte ratio (LMR) were calculated.
The diagnostic criteria for DF referred to the International Working Group on the Diabetic Foot (IWGDF) guidelines [15]. Wagner grading criteria were as follows [16]: Grade 0: presence of risk factors for foot ulcers, but no ulcer is present; Grade 1: superficial foot ulcer without signs of infection; Grade 2: deep ulcers, often with soft tissue infections, but no bone lesions or deep abscesses; Grade 3: deep infection with bone tissue lesions or abscesses; Grade 4: localized gangrene (toe, heel, or dorsal foot); Grade 5: gangrene of the whole foot. The diagnosis of NF was confirmed intraoperatively based on characteristic surgical findings, including the following [12]: 1) gray necrotic fascia tissue; 2) loss of normal attachment between fascia and muscle; 3) absence of active bleeding from the tissue; 4) presence of serous pus; 5) absence of muscle involvement. Foot skin necrosis criteria referred to Karimi et al. [17]: Acute Cutaneous Necrosis: A Guide to Early Diagnosis and Treatment.
Categorical variables were described as percentages; normally distributed continuous variables were expressed as mean (standard deviation [SD]), and the t-test was used for group comparison. While variables with skewed distributions were expressed as the median (25th and 75th percentiles), the Mann-Whitney U test was used for group comparison. The statistical data were expressed as the number of cases (%), and the χ2 test was used for group comparison. Multivariable stepwise logistic regression analysis was used for the new model, and a nomogram was constructed. Odds ratio (OR) with 95% CI was reported. Receiver operating characteristic curve (ROC) analysis was performed, and the AUC, Brier score, and Hosmer-Lemeshow goodness-of-fit test were used to evaluate model performance. The bootstrap method was employed for internal validation, and the Z-test was applied to assess the difference in ROC AUC between the new model and RADNF. Statistical significance was defined as P < 0.05. All statistical analyses were conducted using SPSS version 22.0 and R version 4.3.0.
Compared with the DF group, the DNF group exhibited significantly higher body temperature, prevalence of foot skin necrosis, HbA1c, white blood cell count, neutrophil count, platelet count, monocyte count, NLR, PLR, hs-CRP, and fibrinogen levels. Conversely, age, prevalence of diabetic kidney disease, hemoglobin, albumin, lymphocyte count, and LMR levels were significantly lower in the DNF group. No significant differences in gender, BMI, diabetic duration, creatinine, and blood sodium levels between the two groups (P > 0.05, Table 1).
General clinical characteristics and baseline data of patients.
| Indicator | DF group (703) | DNF group (112) | P-value |
|---|---|---|---|
| Temperature (°C) | 36.6 ± 0.6 | 37.0 ± 0.9 | < 0.001 |
| Age (years) | 66.4 ± 12.2 | 61.4 ± 11.3 | < 0.001 |
| Male (%) | 464 (66.0) | 78 (69.6) | 0.460 |
| BMI (kg/m2) | 23.6 ± 3.5 | 22.9 ± 3.7 | 0.054 |
| Diabetes duration (years) | 10.0 (5.0, 18.0) | 10.0 (4.0, 15.0) | 0.136 |
| Diabetic peripheral neuropathy (%) | 586 (83.4) | 87 (77.7) | 0.100 |
| Diabetic retinopathy (%) | 301 (42.8) | 49 (43.8) | 0.701 |
| Diabetic nephropathy (%) | 354 (50.4) | 39 (34.8) | 0.002 |
| Foot skin necrosis (%) | 172 (24.5) | 46 (41.1) | < 0.001 |
| Wagner classification (%) | |||
| 2 | 290 (41.2) | 7 (6.2) | - |
| 3 | 243 (34.6) | 59 (52.7) | - |
| 4 | 168 (23.9) | 45 (40.2) | - |
| 5 | 2 (0.3) | 1 (0.9) | - |
| White blood cell (109/L) | 8.9 ± 4.0 | 12.9 ± 6.7 | < 0.001 |
| Neutrophil (109/L) | 6.7 ± 3.9 | 10.8 ± 6.7 | < 0.001 |
| Platelet (109/L) | 225.0 (175.0, 302.0) | 282.0 (212.5, 345.8) | < 0.001 |
| Lymphocyte (109/L) | 1.4 ± 0.6 | 1.3 ± 0.5 | 0.013 |
| Monocyte (109/L) | 0.5 (0.4, 0.7) | 0.7 (0.5, 0.9) | < 0.001 |
| NLR | 4.2 (2.9, 6.6) | 8.5 (4.3, 13.0) | < 0.001 |
| PLR | 175.2 (122.7, 243.2) | 219.4 (168.2, 329.0) | < 0.001 |
| LMR | 2.7 (1.9, 4.0) | 1.8 (1.2, 2.8) | < 0.001 |
| Hemoglobin (g/L) | 116.2 ± 20.4 | 106.5 ± 22.4 | < 0.001 |
| HbA1c (%) | 9.6 ± 2.6 | 10.6 ± 2.8 | < 0.001 |
| hs-CRP > 20 mg/L | 273 (38.8) | 87 (77.7) | < 0.001 |
| Albumin (g/L) | 35.9 ± 6.1 | 31.6 ± 6.5 | < 0.001 |
| Na+ (mmol/L) | 138.3 ± 14.1 | 138.7 ± 5.2 | 0.821 |
| Creatinine (mmol/L) | 82.0 (62.0, 112.5) | 73.0 (56.0, 109.0) | 0.142 |
| Fibrinogen (g/L) | 4.7 (3.7, 6.4) | 6.6 (4.9, 7.5) | < 0.001 |
Continuous variables are expressed as mean (standard deviation) or median (25th and 75th percentiles), and categorical variables are expressed as percentages (%). BMI: body mass index; DF: diabetic foot; DNF: DF complicated with necrotizing fasciitis; HbA1c: hemoglobin A1c; hs-CRP: hypersensitive C-reactive protein; LMR: lymphocyte-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-lymphocyte ratio.
To facilitate model comparison, temperature and HbA1c were dichotomized based on the classification criteria from RADNF [12]. Age and hemoglobin were binarized using clinically significant cut-off points of 60 years and 100 g/L, respectively. For the remaining indicators, optimal cut-off values were determined by maximizing the Youden index derived from ROC analysis. These variables were subsequently converted into binary form (Table 2).
Continuous variables are converted to categorical variables.
| Indicator | Section | Selection criteria for cut-off points |
|---|---|---|
| Age (years) | 60 | Clinical significance |
| Temperature (°C) | 38 | RADNF |
| HbA1c (%) | 11 | RADNF |
| Hemoglobin (g/L) | 100 | Clinical significance |
| White blood cell (109/L) | 9 | Youden index |
| Monocyte (109/L) | 0.5 | Youden index |
| NLR | 8.5 | Youden index |
| LMR | 2 | Youden index |
| Fibrinogen (g/L) | 6 | Youden index |
HbA1c: hemoglobin A1c; LMR: lymphocyte-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; RADNF: risk assessment for diabetic foot with necrotizing fasciitis.
DNF was set as the dependent variable. Age, temperature, foot skin necrosis, HbA1c, white blood cell, monocyte, NLR, LMR, hs-CRP, hemoglobin and fibrinogen were included as independent variables in a multivariable stepwise logistic regression analysis. A nomogram was constructed based on the regression results. The analysis revealed that age < 60 years was associated with DNF risk (Score: 86). Furthermore, temperature ≥ 38°C, hemoglobin ≤ 100 g/L, foot skin necrosis, NLR ≥ 8.5, and hs-CRP > 20 mg/L were also identified as significant factors influencing DNF development (Table 3), with corresponding scores of 100, 54, 60, 81, and 79 points, respectively (Figure 1). The optimal cutoff value determined by maximizing the Youden index was 110 points, yielding a sensitivity of 0.893 and a specificity of 0.606.
Multivariate logistic regression analysis of influencing factors of DNF.
| Indicator | β | SE | Waldχ² | P | OR | 95% CI |
|---|---|---|---|---|---|---|
| Age | –0.974 | 0.269 | 13.150 | < 0.001 | 0.378 | 0.223~0.639 |
| Temperature | 1.138 | 0.344 | 10.936 | 0.001 | 3.119 | 1.589~6.122 |
| Hemoglobin | 0.606 | 0.272 | 4.947 | 0.026 | 1.833 | 1.075~3.126 |
| Foot skin necrosis | 0.680 | 0.270 | 6.326 | 0.012 | 1.974 | 1.162~3.353 |
| NLR | 0.916 | 0.297 | 9.520 | 0.002 | 2.500 | 1.397~4.475 |
| hs-CRP | 0.898 | 0.325 | 7.652 | 0.006 | 2.455 | 1.299~4.638 |
CI: confidence interval; DNF: diabetic foot complicated with necrotizing fasciitis; hs-CRP: hypersensitive C-reactive protein; NLR: neutrophil-to-lymphocyte ratio; OR: odds ratio.
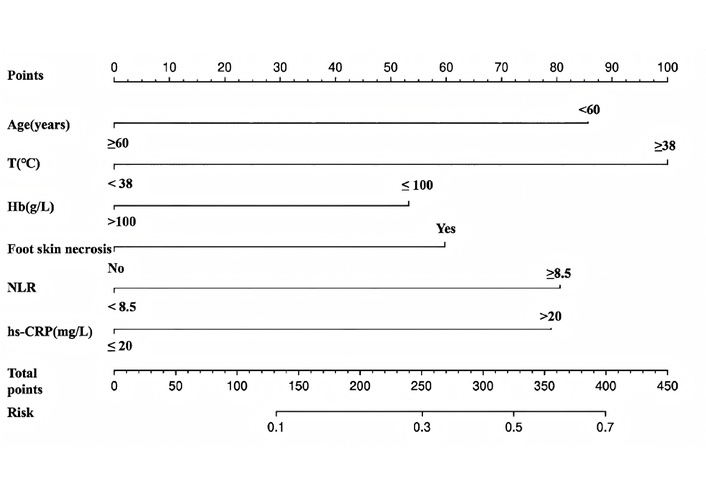
The nomogram of the new risk assessment of diabetic foot with necrotizing fasciitis. T: temperature; Hb: hemoglobin; hs-CRP: hypersensitive C-reactive protein; NLR: neutrophil-to-lymphocyte ratio.
Model differentiation: ROC analysis showed that the AUC (95% CI) of the NRADNF model was 0.815 (0.773, 0.857), whereas the AUC (95% CI) of the previously constructed RADNF model was 0.736 (0.680, 0.793) (Figure 2A). The difference in ROC AUC between the two groups was statistically significant (Z = 2.207, P = 0.027), indicating that NRADNF possesses superior diagnostic predictive efficiency.
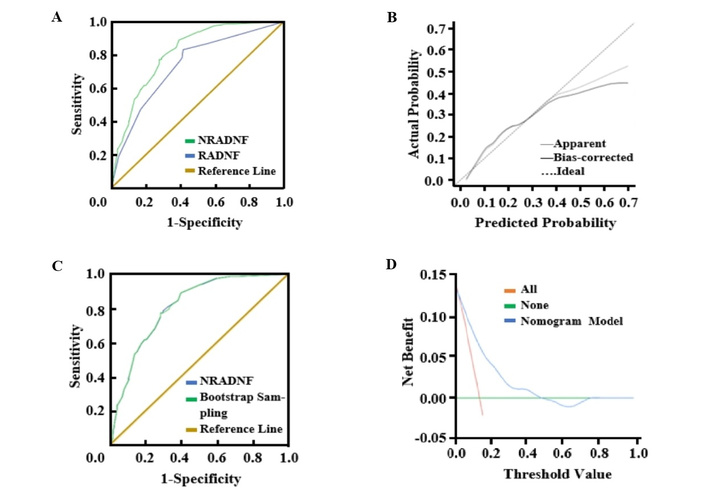
Validation and evaluation of the NRADNF prediction model. (A) Comparison of ROC between NRADNF and RADNF. (B) Calibration curve of NRADNF. (C) Comparison of ROC between NRADNF and Bootstrap repeated sampling (B = 500). (D) NRADNF prediction model decision analysis curve. NRADNF: new risk assessment for diabetic foot with necrotizing fasciitis. RADNF: risk assessment for diabetic foot with necrotizing fasciitis.
Model accuracy: Calibration curves demonstrated a strong correlation between the predicted results of the NRADNF model and the actual results (Figure 2B); the Hosmer‑Lemeshow goodness-of-fit test confirmed that the prediction model had good accuracy (χ2 = 8.736, P = 0.272).
Model stability: Bootstrap validation with 500 repetitions yielded an ROC AUC (95% CI) of 0.815 (0.773, 0.857) (Figure 2C), indicating that the NRADNF model has excellent stability. The Brier Score is 0.096, approaching 0, which further suggests good overall model performance.
Model validity: Clinical decision curve analysis suggested a net benefit value interval of 0.048–0.478 (Figure 2D).
NF is a dangerous and insidious infectious disease that can progress rapidly and cause deterioration, potentially endangering the patient’s life. In the meta-analysis of 674 NF patients by Ahn et al. [18], found that 57.4% had concurrent diabetes. Patients with DF had a higher risk of NF due to peripheral vascular and/or peripheral neuropathy, poor resistance, delayed wound healing, and persistent infection [4]. Additionally, rapid inflammatory development causes local tissue swelling that compresses nerves and blood vessels, aggravating nerve damage and ischemia, creating a vicious cycle. If this circle is not broken, lesions progress, potentially leading to adverse events such as amputation and death. Therefore, establishing a risk assessment model for early detection is essential for improving patient prognosis.
Currently, the LRINEC scale is often used to assess NF risk in clinical practice. This scale was developed by Wong et al. [7] based on 89 patients with NF and 225 patients with cellulitis or soft tissue abscesses, and includes CRP, white blood cell count, hemoglobin, creatinine, sodium, and blood glucose with a total score of 0 to 13. A score of 8 or higher warrants suspicion of NF and immediate performance of the “finger test” (finger dissection)-making a 2 cm incision under local anesthesia to probe between the skin and fascia [1, 19]. If the test is positive, requiring further surgical incision and drainage. However, recent studies have noted that the LRINEC score lacks sensitivity for the early diagnosis of NF [9]. Furthermore, when DNF occurs, the application value of some indicators on the scale is limited. First, renal impairment in DF patients can alter electrolyte and creatinine levels, affecting diagnostic accuracy. Second, blood glucose has limited value in DNF because DF patients often have poor glycemic control [20], making it difficult to distinguish infection-induced hyperglycemia from baseline stress. Literature on NF prognosis also suggests that blood glucose values, without adjustment for baseline levels, do not correlate with prognosis [21]. Third, due to compromised immunity and peripheral neuropathy in diabetic patients, lower extremity infections may not trigger a systemic response as robustly as in healthy individuals; consequently, CRP may not rise to high levels, limiting its applicability in the LRINEC scale for DNF [12]. Compared to the RADNF model (16 DNF, 93 DF), the NRADNF model has a larger sample size and yields a significantly higher ROC value.
In this study, we performed univariate analysis on significant indicators and combined them based on clinical significance and availability for multivariate stepwise logistic regression to build the NRADNF model. The model includes six conventional indicators: age, body temperature, foot skin necrosis, hemoglobin, NLR, and hs-CRP. While age is generally a risk factor for DF, due to functional decline over time, our logistic regression showed that patients < 60 years were more likely to develop NF. This seemingly contradictory finding may be explained by the fact that NF affects skin and subcutaneous tissue, which is abundant in adipose tissue. Aging is associated with body fat redistribution, characterized by increased truncal fat and decreased subcutaneous fat in the limbs [22], which may be less conducive to the development of NF in the lower extremities of older patients. Pathophysiologically, NF involves the formation of bacterial emboli in local microvessels, blocking blood flow [23], and presents as a necrotic eschar, a key identifying feature [24]. Studies have also linked NF amputation risk to soft tissue and skin necrosis, etc. [25]. Because NF progresses rapidly, systemic manifestations are more pronounced than in simple DF, reflected by significant increases in temperature, white blood cell count, NLR, and hs‑CRP. Hs-CRP is more sensitive than standard CRP and can detect inflammation at lower concentrations [26], making it suitable for early DNF screening. NLR, an emerging inflammatory marker, is widely used in infection, cardiovascular disease, tumors, etc. Neutrophils are the first responders to infection, triggering an inflammatory cascade [27]. Excessive inflammation can deplete lymphocytes, leading to immunosuppression and further inflammatory expansion [28], a conclusion supported by our data. Kim et al. [29] also identified NLR as a classification predictor for DNF in a study of 1,581 DF ulcer patients. Additionally, anemia is a common complication of infection (inflammatory anemia). One study showed that over 20% of sepsis patients had hemoglobin levels < 100 g/L at admission [30].
The NRADNF model was visualized using a nomogram to reflect the weight of each indicator. Validation confirmed the model's predictive efficacy, stability, and clinical benefit. A total score exceeding 110 warrants high suspicion of DNF. The high risk of DNF patients should be comprehensively re-evaluated, and a finger test should be performed immediately; if positive, surgical intervention is required. Several limitations must also be acknowledged. First, as a single-center study conducted at a tertiary hospital, our cohort may be subject to selection bias due to the overrepresentation of severe cases, which limits the generalizability of our findings. Second, although this study lacks external validation, it may limit the generalizability of its findings. Third, to mitigate the well-documented limitations of stepwise regression, such as overfitting and biased parameter estimation, we supplemented it with clinical expertise and bootstrap resampling to ensure the robustness of variable selection. We look forward to further validation and refinement in clinical practice.
This study developed the NRADNF model based on common clinical indicators, which demonstrates substantial clinical utility and can be employed for the preliminary screening of DNF.
AUC: area under the receiver operating characteristic curve
BMI: body mass index
CI: confidence interval
DF: diabetic foot
DM: diabetes mellitus
DNF: diabetic foot complicated with necrotizing fasciitis
HbA1c: hemoglobin A1c
hs-CRP: hypersensitive C-reactive protein
LMR: lymphocyte-monocyte ratio
LRINEC: laboratory risk indicator for necrotizing fasciitis
NF: necrotizing fasciitis
NLR: neutrophil-to-lymphocyte ratio
NRADNF: new risk assessment for diabetic foot with necrotizing fasciitis
PLR: platelet-lymphocyte ratio
RADNF: risk assessment for diabetic foot with necrotizing fasciitis
ROC: receiver operating characteristic curve
XL: Writing—original draft, Formal analysis, Visualization. XZ: Writing—original draft. CC: Conceptualization, Investigation. XN: Conceptualization, Investigation. QH: Conceptualization, Investigation. ZT: Conceptualization, Writing—review & editing. WZ: Conceptualization, Writing—review & editing. ZD: Conceptualization, Writing—review & editing. QZ: Validation, Supervision. QC: Validation, Supervision. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
This study was approved by the ethics committee of the First Affiliated Hospital of Chongqing Medical University (approval No. 2025-786-01). The study complies with the Declaration of Helsinki.
The retrospective analysis waived the patient's informed consent form.
Not applicable.
The dataset for this study can be obtained from the corresponding author.
This work was supported by the Chongqing Diabetic Foot Disease Clinical Treatment Center project [WSJKW2022-162]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 734
Download: 33
Times Cited: 0
