Affiliation:
1Department of Cardiology, Hitit University Çorum Erol Olçok Education and Research Hospital, Çorum 19040, Turkey
ORCID: https://orcid.org/0009-0005-9790-6435
Affiliation:
2Department of Cardiology, Faculty of Medicine, Hitit University, Çorum 19040, Turkey
Email: macitkalcik@yahoo.com
ORCID: https://orcid.org/0000-0002-8791-4475
Affiliation:
3Department of Cardiology, Gediz State Hospital, Kütahya 43600, Turkey
ORCID: https://orcid.org/0009-0001-8505-9460
Affiliation:
1Department of Cardiology, Hitit University Çorum Erol Olçok Education and Research Hospital, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0002-8604-0972
Affiliation:
1Department of Cardiology, Hitit University Çorum Erol Olçok Education and Research Hospital, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0002-2412-0985
Affiliation:
2Department of Cardiology, Faculty of Medicine, Hitit University, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0002-2444-7523
Affiliation:
1Department of Cardiology, Hitit University Çorum Erol Olçok Education and Research Hospital, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0002-6496-7849
Affiliation:
2Department of Cardiology, Faculty of Medicine, Hitit University, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0002-3920-1382
Affiliation:
4Department of Biochemistry, Hitit University Çorum Erol Olçok Education and Research Hospital, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0001-9557-678X
Affiliation:
2Department of Cardiology, Faculty of Medicine, Hitit University, Çorum 19040, Turkey
ORCID: https://orcid.org/0000-0002-2544-1975
Explor Cardiol. 2026;4:1012105 DOI: https://doi.org/10.37349/ec.2026.1012105
Received: March 13, 2026 Accepted: April 08, 2026 Published: April 24, 2026
Academic Editor: Chiara Caselli, Consiglio National Research Council (CNR), Italy
High-sensitivity cardiac biomarkers have transformed modern cardiology by enabling earlier diagnosis and refined risk stratification in acute coronary syndromes and heart failure. However, increasing analytical sensitivity has also amplified the clinical impact of immunoassay interferences, particularly those caused by heterophile antibodies. These endogenous antibodies can interact with assay antibodies and generate false-positive or false-negative results, most notably in sandwich immunoassays used for cardiac troponins and natriuretic peptides. Persistent, clinically implausible biomarker elevations related to heterophile antibody interference have led to unnecessary invasive procedures, inappropriate pharmacologic treatment, repeated hospital admissions, and prolonged diagnostic uncertainty. Conversely, false-negative results may delay diagnosis, result in inappropriate discharge, and contribute to adverse clinical outcomes in high-risk patients. This review summarizes the immunological basis and analytical mechanisms of heterophile antibody interference, highlights cardiac biomarkers most commonly affected, and outlines key clinical red flags that should prompt suspicion. Practical laboratory strategies include polyethylene glycol precipitation as an initial approach to detect antibody-mediated interference, followed by dilution studies, heterophile blocking reagents, and cautiously interpreted alternative platform testing within assay-specific reference frameworks, together with close clinician-laboratory collaboration. Greater awareness of analytical interference is critical as cardiology becomes increasingly biomarker-driven. Integrating clinical judgment with structured laboratory verification can substantially reduce diagnostic error, improve patient safety, and prevent avoidable healthcare utilization in contemporary cardiovascular practice.
Over the last two decades, cardiology practice has become increasingly biomarker-driven, particularly in suspected acute coronary syndrome (ACS), high-sensitivity cardiac troponin (hs-cTn) assays, and in heart failure evaluation, natriuretic peptides have become central components of diagnostic algorithms. This transformation has also brought an evolution from classical enzyme biomarkers to the current generation of hs-cTn assays, enabling earlier diagnosis, improved risk stratification, and faster therapeutic decision-making [1]. However, as sensitivity increases, interpretation of low-level biomarker changes has become more critical, and the impact of erroneous results on patient management has grown [2].
As immunoassay-based measurements gain importance in clinical decision-making, analytical accuracy and management of interference-related errors have become an essential part of patient safety in cardiology. Immunoassay interference may lead to serious consequences such as unnecessary invasive procedures, incorrect labeling of myocardial injury, inappropriate antithrombotic or anti-ischemic treatments, and avoidable hospitalizations [3]. Therefore, when laboratory results are inconsistent with the clinical presentation, the possibility of analytical interference should always be considered [3].
Among immunoassay interferences, heterophile antibodies remain a recognized but often underappreciated source of analytical interference, even in modern assays that are considered highly specific. Heterophile antibodies can interact with animal-derived antibodies used in assays with weak or multispecific binding and, particularly in sandwich immunometric assay designs, can generate false analytical signals, resulting in either false-positive or false-negative results [4]. In cardiac troponin measurements, clinically implausible and persistent elevations related to heterophile antibody interference have been reported, clearly demonstrating the risk of inappropriate ACS management [5, 6]. A recent case showing a 4-year-long elevation of hs-troponin due to heterophile antibodies further highlights the ongoing clinical relevance of this issue [7].
Although heterophile antibody interference is generally considered uncommon, its reported frequency varies across studies depending on assay architecture, detection strategies, and patient populations. Estimates from laboratory-based investigations suggest interference rates ranging from approximately 0.05% to 3% in immunoassay measurements, although the true prevalence is likely underestimated because many cases remain clinically unrecognized [8–13]. Importantly, even low-frequency interference may have disproportionate clinical consequences when high-stakes biomarkers such as cardiac troponins or natriuretic peptides are involved. A summary of representative frequency estimates reported in the literature is presented in Table 1.
Reported frequency of heterophile antibody interference across different immunoassay settings.
| Study | Assay | Population | Frequency |
|---|---|---|---|
| Kricka 1999 [8] | General immunoassays | Review of clinical immunoassay interference literature | Approximately 0.05–3% (assay-dependent estimates) |
| Levinson and Miller 2002 [9] | Modern sandwich immunoassays | Review of heterophile antibody interference in clinical samples | Variable prevalence; platform-dependent and often underrecognized |
| Tate and Ward 2004 [10] | Blocked immunometric assays | Clinical laboratory immunoassay review | As low as ~0.03% with blocking strategies |
| Martins et al. 2004 [11] | Multiplex cytokine immunoassay | Human serum samples | Interference detected but frequency not precisely quantified |
| Bjerner et al. 2005 [12] | Clinical immunoassays | Clinical laboratory setting (review) | Persistent but uncommon interference despite assay improvements |
| Ismail 2006 [13] | Immunoassay confirmatory testing (dilution/blocking strategies) | Clinical laboratory interpretation framework | Frequency difficult to quantify; interference remains clinically significant and often misinterpreted |
This narrative review was conducted through a comprehensive search of PubMed, Scopus, and Web of Science databases using combinations of keywords including heterophile antibodies, immunoassay interference, cardiac troponin, natriuretic peptides, and analytical error. Original research articles, case reports, systematic reviews, and relevant laboratory medicine guidelines published in English were screened for clinical relevance. References of selected papers were additionally hand-searched to identify further pertinent studies. Emphasis was placed on reports demonstrating clinical consequences, diagnostic challenges, and practical confirmation strategies related to heterophile antibody interference in cardiac biomarker testing. The literature search covered studies published up to January 2026. Priority was given to clinically relevant original studies, case series, systematic reviews, and guideline-based laboratory medicine recommendations focusing on cardiac biomarker testing and immunoassay interference. Publications were selected based on their relevance to mechanisms of interference, diagnostic implications, and practical confirmation strategies in cardiovascular clinical settings.
The aim of this review is to systematically summarize the mechanisms by which heterophile antibodies cause interference in immunoassays, the diagnostic pitfalls they create in cardiac biomarkers (particularly troponin and natriuretic peptides), and their impact on clinical decision-making. Additionally, practical suspicion and verification strategies that strengthen clinician-laboratory collaboration will be discussed.
Heterophile antibodies are endogenous immunoglobulins capable of binding to animal-derived antibodies used in immunoassays, leading to potential assay interference. These antibodies may occur naturally or may develop following exposure to animal antigens through environmental contact, therapeutic monoclonal antibody treatments, or diagnostic procedures. In addition, the likelihood of antibody-mediated interference may be increased in patients with autoimmune diseases, recent or recurrent viral infections, or immune responses related to certain medications, including over-the-counter preparations [9, 10, 14]. Their presence can cause non-specific binding within immunoassays and may result in either falsely elevated or falsely decreased analyte measurements [14].
The main mechanism of heterophile antibody interference involves their ability to cross-link capture and detection antibodies in sandwich immunoassay formats. In this situation, heterophile antibodies create an artificial bridge between assay antibodies even when the target analyte is absent, leading to false signal generation and falsely elevated results [9]. Conversely, in some cases, heterophile antibodies may block the interaction between the analyte and assay antibodies, producing falsely low measurements. The magnitude of interference depends on antibody affinity, concentration, and assay design characteristics [14, 15].
Human anti-animal antibodies (HAAAs), including human anti-mouse antibodies (HAMAs), represent a clinically important subgroup of heterophile antibodies. HAMA-related interference is particularly relevant because many monoclonal antibodies used in immunoassays are derived from murine hybridoma technology, which increases the likelihood of interaction with circulating anti-mouse antibodies [16]. When assay antibodies originate from other species, the probability and pattern of interference may differ depending on the antibody source and assay design characteristics [9]. These antibodies usually develop after therapeutic or diagnostic exposure to animal-derived immunoglobulins and are well known to cause clinically significant immunoassay interference [17]. This is particularly relevant in cardiac biomarker assays such as troponin measurements, where analytical accuracy is critical for diagnosis and treatment decisions [18].
Although heterophile antibodies and HAAAs are closely related, they are not identical entities. Heterophile antibodies are typically naturally occurring, low-affinity, multispecific antibodies that can bind assay antibodies from different animal species, whereas HAAAs usually develop after exposure to therapeutic or diagnostic animal-derived immunoglobulins and tend to show greater specificity [4]. In addition, other causes of analytical interference, such as macrotroponin formation, represent a different mechanism involving circulating macromolecular complexes rather than antibody-mediated assay bridging [9, 17]. Distinguishing between these mechanisms is important because their diagnostic confirmation strategies and clinical implications may differ.
Several strategies have been developed to reduce heterophile antibody interference. These include the use of heterophile blocking reagents, assay redesign using antibody fragments or modified antibody structures, and confirmation of suspicious results using alternative assay platforms or methodologies [18]. Despite these preventive measures, heterophile antibody interference continues to represent an important analytical challenge in clinical immunoassay testing, requiring close collaboration between clinicians and laboratory specialists [8, 9, 14–18].
Heterophile antibodies interfere with immunoassays through several biochemical and structural mechanisms, most prominently by disrupting antigen-antibody binding dynamics in sandwich assay configurations. The most widely recognized mechanism is the bridging of capture and detection antibodies, where heterophile antibodies bind simultaneously to both assay antibodies, generating a signal in the absence of the target analyte. This leads to falsely elevated biomarker concentrations and may mimic clinically significant disease states [8, 17]. The major biochemical and structural pathways through which heterophile antibodies disrupt immunoassay performance are summarized in Table 2.
Mechanisms of heterophile antibody-mediated immunoassay interference.
| Mechanism | Underlying analytical interaction | Assay type most affected | Result pattern | Clinical risk |
|---|---|---|---|---|
| Antibody bridging | Cross-linking of capture and detection antibodies independent of the analyte | Sandwich immunoassays | False-positive elevation | Overtreatment |
| Blocking of analyte binding | Steric hindrance prevents analyte-antibody interaction | Sandwich immunoassays | False-negative result | Missed diagnosis |
| High-affinity nonspecific immune complex formation | Stable circulating antibody-mediated macrocomplex formation | High-sensitivity platforms | Persistent abnormal values | Chronic mislabeling |
| Platform-specific reagent interaction | Interaction with assay-specific blocking agents or antibody sources | Manufacturer-dependent | Inter-assay discordance | Diagnostic confusion |
In contrast, heterophile antibodies may also cause false-negative results. This occurs when these antibodies block antigen-binding sites or sterically hinder analyte interaction with assay antibodies, preventing proper immune complex formation. The direction and magnitude of interference depend on multiple factors, including antibody specificity, epitope accessibility, and assay reagent composition [8, 18].
Assay design plays a critical role in susceptibility to heterophile antibody interference. Sandwich immunoassays are generally more vulnerable compared with competitive immunoassays because they rely on simultaneous binding of two antibodies to generate measurable signals. High-sensitivity assays, which use lower detection thresholds and signal amplification strategies, may be particularly prone to detecting low-level nonspecific interactions caused by heterophile antibodies [10, 17].
Another key determinant is antibody affinity and concentration. High-affinity heterophile antibodies can create stable non-specific immune complexes, increasing the likelihood of persistent assay interference. Similarly, higher circulating concentrations of heterophile antibodies increase the probability of cross-reactivity with assay reagents. Patient-specific immunological variability, therefore, plays an important role in determining interference patterns and reproducibility [8, 12].
Platform-dependent effects are also clinically relevant. Different manufacturers use varying antibody sources, blocking reagents, and assay architectures, which may result in discordant results across platforms. When heterophile antibody interference is suspected, initial confirmatory approaches typically include polyethylene glycol (PEG) precipitation and the use of heterophile blocking reagents to remove or neutralize interfering antibodies before repeat measurement. Alternative assay platform testing may provide additional supportive information; however, inter-platform differences should be interpreted cautiously because assay-specific characteristics and reference intervals may vary. Lack of concordance between platforms can represent a useful supportive clue for analytical interference rather than true biomarker elevation when interpreted together with targeted laboratory confirmation strategies [10, 15, 17, 18].
Overall, heterophile antibody-mediated assay interference represents a complex interaction between patient immunology and assay engineering. Understanding these mechanisms is essential for recognizing unexpected laboratory results and preventing diagnostic errors, particularly in high-stakes clinical settings such as emergency cardiology and ACS evaluation [8, 10].
Heterophile antibody interference has been described across a range of cardiac and cardiovascular-relevant immunoassays, but the clinical impact is greatest when biomarkers directly trigger high-stakes decisions in emergency cardiology or long-term disease management. Among these, cardiac troponins and natriuretic peptides are particularly vulnerable because they are often interpreted as objective evidence of myocardial injury or hemodynamic stress, respectively, and are frequently acted upon without delay [19–21].
Cardiac troponins are the most consequential targets of heterophile antibody interference, given their central role in diagnosing myocardial infarction and guiding invasive management. Multiple reports illustrate that heterophile antibodies can produce persistently elevated troponin concentrations despite the absence of clinical, electrocardiographic, imaging, or angiographic evidence of ongoing myocardial injury [19–21].
A typical clinical pattern is a “discordant troponin phenotype,” characterized by persistent elevation with little or no dynamic rise-and-fall, minimal correlation with symptom timing, and repeated negative ischemia workups. In one Journal of the American College of Cardiology: Case Reports publication, extensive evaluation ultimately concluded that circulating heterophile antibodies were responsible for false-positive troponin elevation, reframing the differential away from structural myocardial disease and ACS [19]. Similar pitfalls have been highlighted in case reports demonstrating that heterophile antibody-related false-positive troponin results can lead to diagnostic confusion and the potential for unnecessary management of suspected ACS [20]. More recently, a case series highlighted that heterophile antibody-mediated troponin elevation remains underrecognized and can drive repeated investigations unless interference is systematically considered and confirmed [21].
From a practical standpoint, troponin interference may trigger unnecessary diagnostic and therapeutic pathways, particularly when analytical artifacts are not recognized early. Importantly, platform dependence is common; repeating troponin on an alternative platform can reveal marked discordance, supporting analytical rather than biological causes [21, 22].
Natriuretic peptide assays, including B-type natriuretic peptide (BNP) and N-terminal pro-BNP (NT-proBNP), are also susceptible to heterophile antibody effects, which may lead to heart failure misclassification, inappropriate initiation or escalation of therapy, and distorted prognostic assessment. A notable example is a case series describing patients without heart failure who nonetheless showed apparently high BNP concentrations on one assay platform, while alternative platforms produced values within reference limits, strongly suggesting immunoassay-related false-positivity [23].
Although BNP and NT-proBNP belong to the same biological pathway and are frequently discussed together in clinical practice, their analytical characteristics differ across assay platforms because of differences in antibody targets, molecular structure, and circulating peptide forms. BNP is a biologically active peptide consisting of 32 amino acids and is more susceptible to proteolytic degradation by circulating and tissue peptidases, whereas NT-proBNP is a longer and more stable 108-amino-acid fragment with different clearance characteristics. These differences may influence assay performance, circulating fragment composition, and susceptibility to analytical variability [23–25]. Therefore, suspected immunoassay interference may affect BNP and NT-proBNP differently depending on assay architecture and platform-specific design features, and results from one peptide should not automatically be assumed to mirror the behavior of the other.
In clinical practice, false elevation of natriuretic peptides may prompt unnecessary echocardiography, diuretic intensification, or labeling of chronic heart failure in otherwise stable patients. Conversely, interference can theoretically mask true elevation (false-negatives), risking delayed diagnosis and undertreatment, especially when natriuretic peptides are used as “rule-out” tools in dyspnea evaluation [23].
Although the clinical impact of heterophile antibody interference is most prominent in cardiac troponins and natriuretic peptides, interference has also been reported in selected cardiovascular-related assays such as digoxin and D-dimer. Digoxin provides a representative example, with case reports describing markedly false elevations caused by human anti-mouse antibodies that could have resulted in inappropriate antidote administration or unnecessary discontinuation of therapy if unrecognized [26]. Similar observations have shown that interference affecting digoxin immunoassays may be mitigated using heterophile blocking reagents, highlighting the role of assay architecture in determining susceptibility [27].
D-dimer assays, widely used in thromboembolic risk assessment, have likewise been affected by heterophile antibody interference. Reported cases describe persistently elevated D-dimer concentrations that are discordant with imaging and clinical findings. Confirmation may be supported by heterophile blocking reagents, dilution studies, PEG precipitation, or alternative assay platforms; however, careful interpretation of these follow-up tests is essential because apparent correction after intervention does not always exclude residual analytical interference and may itself be misleading if interpreted without clinical and assay-specific context [28–30]. Although such interference is uncommon, it remains an important analytical consideration when laboratory results are incompatible with the clinical picture [31].
In particular, dilution testing, blocking strategies, and platform comparison require structured interpretation, as emphasized in previous methodological studies addressing common pitfalls in the evaluation of endogenous antibody interference in immunoassays [13, 32, 33].
Taken together, these observations indicate that heterophile antibody interference is not restricted to a single biomarker but represents an additional analytical consideration in cardiovascular laboratory interpretation beyond the core cardiac biomarkers, particularly cardiac troponins and natriuretic peptides (Table 3).
Cardiac biomarkers affected by heterophile antibody interference.
| Biomarker | Direction of interference | Typical clinical consequence | Key diagnostic clue |
|---|---|---|---|
| Cardiac troponins | False elevation (most common) | Unnecessary ACS treatment, angiography | Persistent elevation without expected rise-fall kinetics and discordance with clinical presentation |
| BNP | False elevation | Misdiagnosis of HF | Elevated BNP concentration inconsistent with imaging findings or clinical evidence of HF |
| NT-proBNP | False elevation or suppression | Inappropriate HF risk stratification | Marked inter-platform discordance or unexpected values inconsistent with clinical status |
| Digoxin | Markedly false elevation | Unnecessary antidote or therapy cessation | Markedly elevated digoxin level without compatible clinical toxicity findings |
| D-dimer | False elevation | Unnecessary thromboembolism workup | Persistently elevated D-dimer despite negative imaging for thromboembolism |
ACS: acute coronary syndrome; BNP: B-type natriuretic peptide; NT-proBNP: N-terminal pro-B-type natriuretic peptide; HF: heart failure.
Recognizing heterophile antibody interference requires a high index of suspicion, especially when biomarker results are discordant with the overall clinical assessment. A major red flag is an apparently abnormal cardiac biomarker in a patient whose symptoms, electrocardiogram, imaging, and overall clinical trajectory do not support the laboratory finding. In the era of high-sensitivity troponin assays, small numerical elevations can occur for many reasons, but a result that is persistently “abnormal” without a coherent clinical explanation should prompt consideration of analytical interference [34, 35].
Another important warning sign is the absence of expected biomarker kinetics. In true acute myocardial infarction, troponin typically demonstrates a rise and/or fall pattern over serial sampling, and clinical decision pathways rely heavily on these temporal changes. When serial measurements remain stable at an unusual “plateau” level, particularly if the timing of symptoms does not match the biomarker pattern, interference should be considered alongside alternative biological explanations [34, 36].
Certain patient-related factors may increase the likelihood of heterophile antibody interference. These include exposure to therapeutic monoclonal antibodies, autoimmune diseases, recent or chronic viral infections, repeated diagnostic procedures involving animal-derived antibodies, and occupational or environmental animal antigen exposure [4, 17]. In addition, assay-related characteristics such as sandwich immunoassay architecture, antibody source variability, and platform-specific blocking strategies may influence susceptibility to interference [10, 32]. Awareness of these clinical and analytical risk factors may facilitate earlier recognition of unexpected laboratory results.
Inter-assay or inter-platform discordance is a particularly valuable clue. If repeated testing using a different manufacturer’s platform yields markedly different values, this strongly supports an analytical artifact rather than true myocardial injury. A recent PubMed-indexed case report highlighted that discrepant troponin results across assays were the key trigger for investigating heterophile antibody interference and avoiding misdiagnosis [37].
Additional red flags include persistently abnormal results despite resolution of symptoms or normalization of other clinical parameters, particularly when repeated investigations remain inconclusive. Such scenarios increase the risk of unnecessary invasive testing, inappropriate antithrombotic therapy, and avoidable hospitalizations, underscoring the need to integrate clinical judgment with laboratory interpretation [34, 37].
Although heterophile antibody interference is generally considered uncommon, reported prevalence varies substantially across assay platforms, analytical designs, and study populations. Available laboratory-based estimates suggest frequencies ranging from approximately 0.05% to 3%, but the true prevalence is likely underestimated because many cases remain clinically unrecognized or are not systematically investigated [8]. Importantly, even low-frequency interference may have disproportionate clinical consequences when high-stakes biomarkers such as cardiac troponins or natriuretic peptides are involved. Therefore, clinicians should interpret unexpected or discordant biomarker results cautiously and consider analytical interference even when the estimated probability appears low.
Finally, clinicians should remember that heterophile antibodies can affect other cardiovascular-relevant immunoassays as well; therefore, a broader “interference mindset” is warranted when laboratory findings repeatedly conflict with the clinical picture [38]. An integrated overview of suspected interference patterns and confirmation strategies is presented in Figure 1.
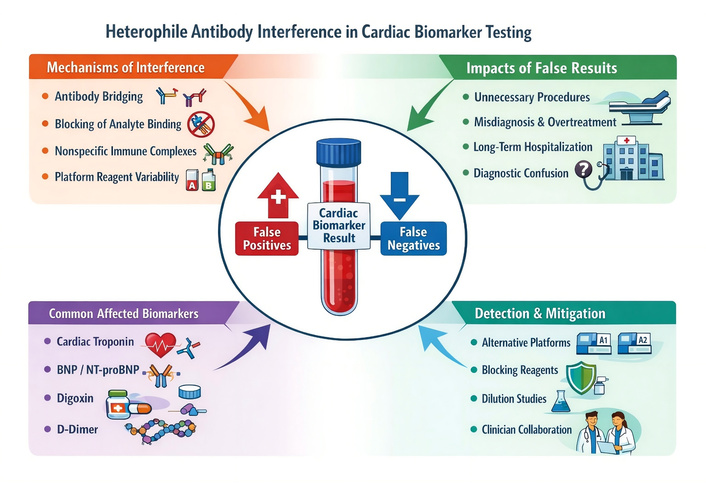
Schematic overview of heterophile antibody interference in cardiac biomarker immunoassays. The figure illustrates key mechanisms of interference (antibody bridging, analyte-binding blockage, immune complex formation, and platform-related effects), commonly affected biomarkers (cardiac troponins, natriuretic peptides, digoxin, and D-dimer), major clinical consequences, and practical laboratory strategies for confirmation. (This figure was generated with the assistance of an AI-based image-generation tool (ChatGPT/OpenAI). The figure was conceptually designed and verified by the authors, and no copyrighted material was used). AI: artificial intelligence; BNP: B-type natriuretic peptide; NT-proBNP: N-terminal pro-B-type natriuretic peptide.
When heterophile antibody interference is suspected, confirmation requires a structured laboratory approach combined with close communication between clinicians and laboratory professionals. Because no single test definitively proves interference, a stepwise diagnostic strategy is recommended, integrating clinical suspicion with targeted analytical verification methods [39, 40].
PEG precipitation represents one of the practical early confirmatory techniques, particularly when macromolecular complexes are suspected. PEG precipitation has also been incorporated into interference investigations involving high-sensitivity troponin assays. In a PubMed-indexed report describing heterophile antibody interference affecting hs-cTnT measurements, pretreatment strategies led to a marked reduction in the apparent troponin concentration, supporting analytical rather than biological elevation [41]. A decrease in measured biomarker concentration exceeding approximately 20% after PEG precipitation supports the presence of antibody-mediated interference and suggests that the original result may be analytically unreliable [15, 17].
Another important strategy involves the use of heterophile antibody blocking reagents (e.g., heterophile blocking reagent/tubes). These reagents are designed to neutralize heterophile antibody activity and thereby reduce non-specific antibody binding within immunoassays. A substantial reduction in the reported analyte concentration after pretreatment supports the presence of heterophile-mediated interference and may be decisive when laboratory findings are inconsistent with the clinical presentation [40, 41].
Serial dilution testing is also widely used in interference investigations. In true analyte measurement, serial dilution typically produces approximately linear results after adjustment for the dilution factor. In contrast, nonlinearity following dilution suggests the presence of heterophile antibodies or related endogenous interfering factors. Dilution studies are particularly useful when blocking reagents are unavailable or when additional analytical confirmation is required to support suspected interference [40].
Repeating the measurement using an alternative assay platform represents another practical component of the verification strategy. Different manufacturers employ distinct antibody sources, assay architectures, and blocking approaches. Therefore, clinically relevant discrepancies between platforms strongly suggest analytical interference rather than true biomarker elevation. Cross-platform comparison is especially valuable in suspected cases of troponin interference, where rapid clarification may help prevent unnecessary invasive investigations [39, 40].
In practical clinical settings, a stepwise workflow may improve recognition and confirmation of suspected interference. First, clinicians should reassess clinical-biochemical concordance and evaluate whether observed biomarker kinetics are physiologically plausible. Second, laboratory-based confirmation may begin with PEG precipitation when antibody-mediated interference is suspected. Third, heterophile antibody blocking reagent treatment can help demonstrate signal suppression attributable to interfering antibodies. Fourth, serial dilution testing may be performed to assess analytical linearity. Finally, repeat measurement using an alternative assay platform or consultation with laboratory specialists may be required in unresolved cases [15, 17, 39–41]. A structured workflow integrating these steps may help avoid unnecessary invasive procedures and reduce diagnostic uncertainty.
Overall, confirming heterophile antibody interference requires integration of clinical reasoning with method-specific analytical verification tools. Early recognition and appropriate laboratory confirmation can prevent repeated emergency presentations, unnecessary invasive procedures, inappropriate pharmacologic therapy, and long-term diagnostic labeling driven by spurious biomarker results [39, 42]. A structured approach to suspicion and laboratory confirmation of heterophile antibody interference, including PEG precipitation, blocking reagents, dilution-based techniques, and platform comparison, is outlined in Table 4.
Stepwise practical strategies to suspect and confirm heterophile antibody interference.
| Strategy | Principle | Expected finding | Clinical utility |
|---|---|---|---|
| PEG precipitation | Remove macrocomplexes | ≥ 20% signal reduction | Practical initial screening |
| Heterophile blocking reagents | Neutralize interfering antibodies | Significant result reduction | High specificity |
| Serial dilution | Assess assay linearity | Nonlinear response | Simple screening |
| Alternative platform testing | Different antibody architecture | Markedly discordant values | Supportive confirmation |
| Clinician-laboratory collaboration | Integrated interpretation | Early recognition | Prevents harm |
PEG: polyethylene glycol.
Heterophile antibody-mediated assay interference can directly compromise patient safety because cardiac biomarkers frequently trigger time-sensitive, high-stakes decisions. When falsely elevated results are interpreted as true myocardial injury or decompensated cardiovascular disease, patients may be exposed to unnecessary diagnostic procedures, inappropriate treatments, and avoidable hospitalization [43–45].
Unnecessary invasive investigations represent one of the most serious consequences. A case report described a patient who underwent coronary angiography solely due to false-positive troponin I results, illustrating how analytical artifacts can precipitate invasive testing with associated procedural risks and costs [43]. Similarly, a published case report described repeated clinical concern and diagnostic investigations driven by heterophile antibody-related false-positive troponin elevation, highlighting that unrecognized assay interference can result in recurrent presentations and repeated escalation of care [44].
Overtreatment is another major patient-safety issue. False-positive troponin values may trigger ACS pathways, including antiplatelet and anticoagulant therapy, which carry bleeding risks and may complicate future procedures. Even when clinicians ultimately determine that angiography is normal, the initial laboratory signal can still drive prolonged observation, serial testing, and exposure to medications that confer harm without benefit [44, 45].
Beyond immediate clinical harm, heterophile interference creates a psychological and economic burden. Patients may experience significant anxiety from being labeled with “myocardial infarction” or “chronic myocardial injury,” and may undergo repeated follow-up visits and testing. Health systems may incur avoidable expenditures related to admissions, imaging, and invasive procedures performed in response to spurious laboratory signals [45].
At the healthcare-system level, heterophile antibody interference may also increase diagnostic workload, prolong emergency department observation times, trigger avoidable imaging procedures, and generate unnecessary invasive testing such as coronary angiography. These effects contribute to increased healthcare costs and inefficient resource utilization, particularly in high-volume emergency cardiology settings where biomarker-driven decision pathways are routinely applied [43–45].
Finally, persistent assay interference can create long-term diagnostic confusion. Reports involving interference-related discrepancies (including macrocomplex-related troponin artifacts) show that patients may undergo prolonged or repeated investigations unless interference is systematically considered and resolved through clinician-laboratory collaboration [41]. Collectively, these impacts highlight why analytical interference should be treated as a patient-safety issue rather than a purely laboratory concern [43–46].
Current cardiology and laboratory medicine guidance increasingly acknowledges the importance of analytical quality in cardiac biomarker interpretation, yet heterophile antibody interference is still not systematically embedded in most routine clinical pathways. Consensus documents emphasize that biomarkers, particularly cardiac troponins, must be interpreted in a clinical context, and that analytical causes of unexpected results should be considered when laboratory data are inconsistent with the patient’s presentation [47].
The Fourth Universal Definition of Myocardial Infarction underscores that a diagnosis of myocardial infarction requires evidence of myocardial injury in a compatible clinical setting and highlights the need to exclude alternative explanations when biomarker findings are incongruent. While heterophile antibodies are not a core focus of the document, it supports the broader principle that analytical false-positives must be considered before confirming myocardial injury in low-probability scenarios [47].
From the laboratory medicine perspective, guidance on immunoassays stresses that analytical errors and endogenous interferences can have clinically significant downstream consequences and therefore require proactive risk mitigation. Practical reviews outline how immunoassays remain vulnerable to occasional analytical errors and propose structured thinking to minimize risk, particularly when results are clinically implausible [48]. Additional perspective articles emphasize that interferences may represent rare but high-impact “black swan” events that can cause misdiagnosis or inappropriate treatment unless laboratories and clinicians actively consider and investigate them [49].
Despite this awareness, major gaps remain: There is no universally adopted, cardiology-specific algorithm for systematic detection and management of heterophile antibody interference in troponin or natriuretic peptide testing. As a result, recognition often depends on individual clinician suspicion and local laboratory expertise, which can delay resolution in acute care settings [48, 49].
Future progress in preventing heterophile antibody interference will likely require parallel advances in assay engineering, laboratory workflows, and data-driven surveillance. From an assay-design perspective, manufacturers are increasingly focused on reducing susceptibility through improved antibody selection, optimized blocking formulations, and refined architectures that minimize non-specific bridging. Because interference remains platform-dependent, broader transparency around assay antibody composition and blocking strategies could facilitate more predictable troubleshooting and cross-platform verification in suspected cases [50].
Laboratory practice is also evolving toward more standardized reflex pathways. When biomarker results are clinically discordant, structured workups, such as immediate retesting on an alternative platform, targeted pretreatment with blocking reagents, and dilution-based linearity checks, should become more consistently embedded into local protocols. Wider availability of validated interference investigation kits and clearer manufacturer guidance on their limitations could shorten time-to-resolution and reduce unnecessary downstream testing [50].
A major emerging direction is the integration of artificial intelligence (AI) into clinical laboratory medicine. AI-assisted systems may support anomaly detection by identifying result patterns that deviate from expected biological behavior and flagging outliers that could warrant interference workup, particularly in high-volume settings where rare events are easily missed [51]. In addition, machine learning approaches are currently being explored as potential tools to detect laboratory errors across the testing process, which may help distinguish physiological variation from analytical artifacts and prioritize cases for expert review [52].
Future research should also focus on establishing standardized diagnostic algorithms for suspected immunoassay interference in cardiac biomarker testing and improving collaboration between cardiology services and clinical laboratories. Prospective studies evaluating the real-world frequency and clinical consequences of heterophile antibody interference across different assay platforms would further help define its true impact and guide the development of practical clinical pathways [15, 17, 48, 49].
More broadly, recent advances in AI-supported cardiovascular prognostic modeling, personalized metabolic risk stratification, and circadian rhythm-informed prevention strategies illustrate how data-driven approaches are increasingly shaping precision cardiovascular medicine. Although these developments are not specific to immunoassay interference, they highlight the growing potential of integrative analytical frameworks to improve the interpretation of complex biomarker signals and reduce diagnostic uncertainty in future cardiology practice [53–55].
Overall, combining improved assay robustness with standardized investigative pathways and AI-enabled surveillance offers a realistic roadmap to reduce heterophile antibody-related diagnostic error in cardiology [50–52].
Heterophile antibody interference remains an important yet frequently underrecognized source of analytical error in cardiac biomarker testing. As cardiology becomes increasingly biomarker-driven, the clinical consequences of false-positive or false-negative laboratory results become more significant. hs-cTn assays and natriuretic peptide measurements, which play central roles in acute and chronic cardiovascular disease management, are particularly vulnerable to immunoassay interference.
Recognition of heterophile antibody interference requires integration of laboratory data with clinical context. Discordance between biomarker results and the patient’s symptoms, imaging findings, or expected biomarker kinetics should prompt consideration of analytical interference. Early suspicion and timely collaboration between clinicians and laboratory specialists are essential to avoid unnecessary invasive procedures, inappropriate pharmacologic therapy, and prolonged diagnostic uncertainty.
Future improvements in assay design, laboratory diagnostic pathways, and advanced data analytics may help reduce the impact of immunoassay interference. However, clinician awareness will remain a cornerstone of safe biomarker interpretation. Ultimately, strengthening clinic-laboratory communication and maintaining a structured approach to unexpected laboratory results will be critical to improving patient safety and diagnostic accuracy in modern cardiology practice.
ACS: acute coronary syndrome
AI: artificial intelligence
BNP: B-type natriuretic peptide
HAAAs: human anti-animal antibodies
HAMAs: human anti-mouse antibodies
hs-cTn: high-sensitivity cardiac troponin
NT-proBNP: N-terminal pro-B-type natriuretic peptide
PEG: polyethylene glycol
The authors confirm that no AI or AI-assisted tools were used for interpreting the referenced article or for generating scientific content. Limited assistance was obtained solely for language editing, grammatical refinement, and preparation of Figure 1, without any involvement in data interpretation, analysis, or conceptual input.
ÖBÇ: Conceptualization, Writing—original draft. MK: Conceptualization, Writing—review & editing. AS: Conceptualization, Writing—original draft. MMY: Conceptualization, Investigation, Writing—original draft. OK: Conceptualization, Investigation, Writing—original draft. MY: Writing—review & editing. MCÇ: Conceptualization, Investigation, Writing—original draft. LB: Supervision, Validation. MY: Supervision, Validation. YK: Supervision, Validation. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 895
Download: 39
Times Cited: 0
