Affiliation:
1Department of Cardiology, Calderdale & Huddersfield NHS Foundation Trust, HX3 0PW Halifax, UK
Affiliation:
2Ziauddin Medical College, Karachi 75600, Pakistan
ORCID: https://orcid.org/0009-0008-2594-8435
Affiliation:
1Department of Cardiology, Calderdale & Huddersfield NHS Foundation Trust, HX3 0PW Halifax, UK
Email: drazeemsheikh@hotmail.com
ORCID: https://orcid.org/0000-0001-7202-9855
Explor Cardiol. 2026;4:1012104 DOI: https://doi.org/10.37349/ec.2026.1012104
Received: February 09, 2026 Accepted: March 29, 2026 Published: April 23, 2026
Academic Editor: Geu-Ru Hong, Yonsei University College of Medicine, Korea
Implantable cardioverter defibrillators (ICDs) play a central role in the prevention of sudden cardiac death. Although implantation is generally safe, rare but clinically significant complications may occur. One such complication is inadvertent placement of a transvenous ICD lead into the left ventricle via a patent foramen ovale (PFO), which carries a risk of systemic thromboembolism and may remain clinically silent for prolonged periods. We report a case of an ICD lead malpositioned in the left ventricle through a PFO, detected 17 months after implantation during transthoracic echocardiography performed for worsening dyspnoea. Imaging demonstrated the lead traversing the interatrial septum and mitral valve into the left ventricle. The patient underwent successful transvenous extraction and reimplantation of a new right ventricular lead without complications. This case highlights the limitations of electrocardiographic clues alone and underscores the importance of multimodality imaging and meticulous implantation technique. A focused narrative review integrates current literature regarding mechanisms, diagnostic strategies, management options, and preventive considerations.
Implantable cardioverter defibrillators (ICDs) are an established therapy for the primary and secondary prevention of sudden cardiac death in patients with ischaemic and non-ischaemic cardiomyopathies [1, 2]. Standard implantation involves transvenous access with advancement of the lead through the right atrium into the right ventricle, typically positioned at the apex or septum [3]. Although ICD implantation is generally considered safe, large registry data show that procedural complications still occur and are clinically relevant, with 3.2% of implantations experiencing an in-hospital adverse event and 3.05% having any complication; in-hospital death occurred in 0.38% of cases [4]. Consistently across these studies, pocket haematoma and lead dislodgement are among the most frequent early complications; however, differences in reported rates may reflect variability in patient populations, operator experience, and device type [5–7].
Lead-related complications represent a significant subset of device-associated adverse events. Lead displacement has been reported in approximately 3.1% of device implantations, although the true incidence may be underestimated because some cases remain clinically silent [5, 6]. In contrast to earlier reports, recent studies suggest that careful intra-procedural imaging, including multi-view fluoroscopy, can reduce displacement rates, highlighting the role of technique in outcome variability.
Device-related infections occur in approximately 1–3% of patients, often requiring device revision or extraction, while procedure-related mortality remains low but not negligible in large registries [6, 7].
A particularly rare but potentially serious complication is inadvertent placement of the lead within the left ventricle. This may occur when the lead crosses an interatrial communication such as a patent foramen ovale (PFO) or atrial septal defect (ASD), subsequently passing through the mitral valve into the left ventricular cavity [7]. Building upon case series and observational studies, the reported incidence of left ventricular lead malposition is extremely low, but it carries substantial thromboembolic risk.
A malpositioned lead within the systemic circulation poses significant risks, as the lead may act as a nidus for thrombus formation and predispose to systemic embolism or stroke [8]. Additional complications include valvular injury, infection, and device malfunction [9–11]. Because many cases remain asymptomatic, diagnosis may be delayed for months or even years after implantation [12].
Given its rarity, there is currently no consensus on the optimal management of left ventricular lead malposition, and strategies are typically guided by timing, mechanism of displacement, and patient-specific risk factors [13]. This highlights a knowledge gap: while observational evidence informs anticoagulation and extraction decisions, prospective studies or standardized protocols are lacking.
Here we report a case of inadvertent ICD lead placement into the left ventricle via a PFO that was detected 17 months after implantation. The case illustrates diagnostic challenges, emphasizes the importance of careful intra-procedural imaging, and provides context for management decisions in this rare complication.
A timeline of the clinical events is shown in Figure 1.
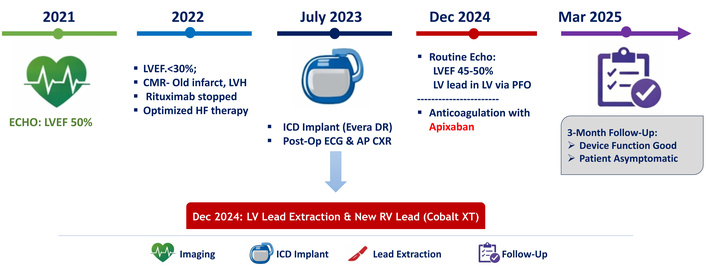
Timeline of clinical events. AP CXR: antero-posterior chest X-ray; CMR: cardiac magnetic resonance; ECG: electrocardiogram; ECHO: echocardiogram; HF: heart failure; ICD: implantable cardioverter defibrillator; LV: left ventricle; LVEF: left ventricular ejection fraction; LVH: left ventricular hypertrophy; PFO: patent foramen ovale; RV: right ventricle.
A 55-year-old woman underwent ICD implantation for primary prevention of sudden cardiac death in the context of severe left ventricular systolic dysfunction [left ventricular ejection fraction (LVEF) < 35%], narrow QRS duration (113 milliseconds), and New York Heart Association (NYHA) Functional Classification class II symptoms. Her medical history included hypertension, type 2 diabetes mellitus, chronic kidney disease stage 3, prior non-ST elevation myocardial infarction with percutaneous coronary intervention to the left anterior descending and circumflex arteries, follicular lymphoma in remission, remote ovarian cancer, and a previously known PFO.
The patient had experienced multiple hospital admissions for decompensated heart failure. Transthoracic echocardiography (TTE) in 2022 demonstrated a LVEF below 30%, compared with 50% the previous year. Cardiac magnetic resonance imaging (MRI) revealed a non-dilated left ventricle with hypertrophy, severely impaired systolic function, and non-viable myocardium within the inferior territory consistent with prior infarction. The decline in systolic function was considered multifactorial, reflecting prior myocardial infarction, progressive heart failure, and possible cardiotoxic effects of prior chemotherapy including rituximab.
Baseline ECG demonstrated sinus tachycardia, left axis deviation, left ventricular hypertrophy, and inferior Q waves (Figure 2A). Following optimisation of guideline-directed medical therapy, a dual-chamber ICD (Medtronic Evera DR) was implanted in July 2023 for primary prevention.
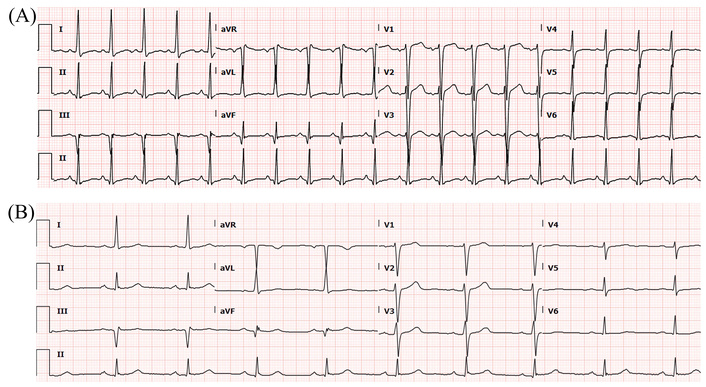
ECG before and after ICD implantation. (A) Pre-ICD implantation ECG showed sinus tachycardia, left axis deviation, left ventricular hypertrophy, and Q waves in the inferior leads. (B) Post-ICD implantation ECG showed sinus bradycardia and left axis deviation. ECG: electrocardiogram; ICD: implantable cardioverter defibrillator.
Post-procedural electrocardiogram (ECG) demonstrated sinus bradycardia with left axis deviation and no right bundle branch block (RBBB) morphology (Figure 2B).
Careful evaluation of the post-implant ECG is important for confirming the appropriate ventricular lead position. Ventricular pacing from the right ventricle typically produces a left bundle branch block-like pattern, whereas pacing originating from the left ventricle may result in an RBBB morphology, raising suspicion for inadvertent left-sided lead placement. Several reports have emphasized the diagnostic value of surface ECG in detecting abnormal lead position following cardiac device implantation and highlight that an unexpected RBBB pattern during ventricular pacing should prompt further imaging to exclude left ventricular lead malposition, including passage through a PFO [14, 15].
An antero-posterior (AP) chest radiograph (Figure 3) was reported as satisfactory; however, a lateral projection was not obtained.
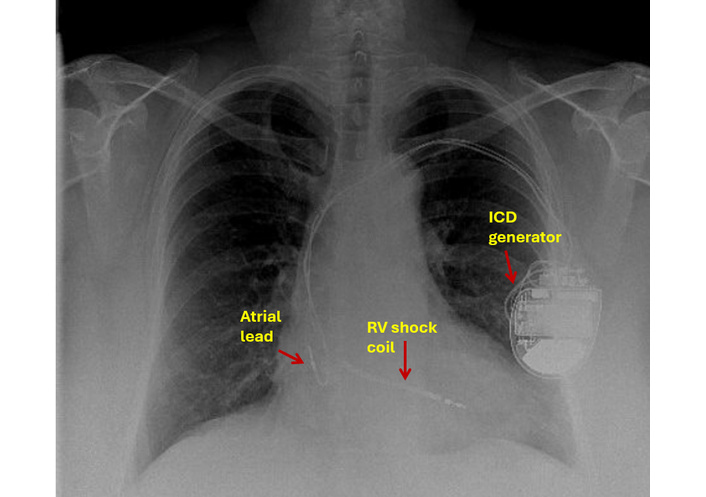
Chest X-ray: antero-posterior view showing ICD lead in an apparently good position. ICD: implantable cardioverter defibrillator; RV: right ventricle.
A review in the device clinic demonstrated high shock impedance, and the superior vena cava (SVC) coil was switched off.
Seventeen months later, in December 2024, the patient presented with worsening dyspnoea. TTE performed during evaluation demonstrated improvement in left ventricular systolic function (LVEF 45–50%), likely reflecting optimisation of guideline-directed heart failure therapy and natural variability in ejection fraction measurements rather than a direct effect of ICD implantation. The dyspnoea was attributed to fluctuations in heart failure symptoms, as there was no valvular obstruction, embolic event, or device malfunction identified. Unexpectedly, the ICD lead was visualised crossing the interatrial septum and mitral valve into the left ventricle via a PFO (Figure 4).
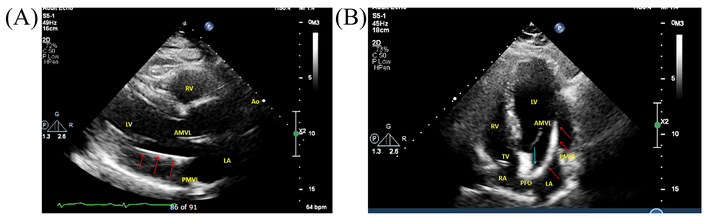
Transthoracic echocardiogram. (A) Parasternal long-axis view demonstrating the ICD lead (red arrows) traversing the mitral valve into the LV. (B) Apical four-chamber view showing the ICD lead (red arrows) traversing the PFO (blue arrow), LA, and mitral valve into the LV. AMVL: anterior mitral valve leaflet; Ao: aorta; ICD: implantable cardioverter defibrillator; LA: left atrium; LV: left ventricle; PFO: patent foramen ovale; PMVL: posterior mitral valve leaflet; RA: right atrium; RV: right ventricle; TV: tricuspid valve.
Computed tomography (CT) of the thorax confirmed the abnormal lead trajectory (Figure 5). Baseline laboratory investigations prior to the extraction, including renal function, electrolytes, and haematological parameters, are summarised in Table 1.
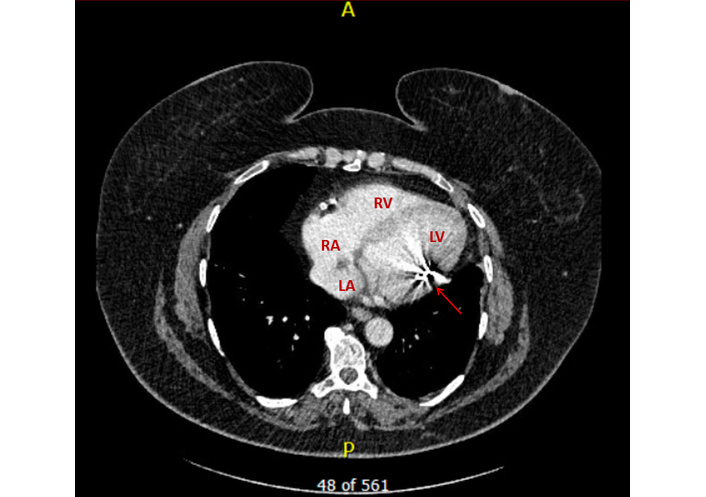
Computed tomography: crosssectional view demonstrating the ICD lead within the LV with metal artefacts (red arrow). ICD: implantable cardioverter defibrillator; LA: left atrium; LV: left ventricle; RA: right atrium; RV: right ventricle.
Laboratory investigations.
| Parameter | Result | Reference range |
|---|---|---|
| Haemoglobin (g/L) | 130 | 135–170 |
| White cell count (109/L) | 9.9 | 3.5–11 |
| Mean cell volume (fL) | 91.3 | 80–99 |
| Platelets (109/L) | 273 | 140–400 |
| Neutrophils (109/L) | 7.1 | 1.7–8 |
| Lymphocytes (109/L) | 1.5 | 1–4 |
| Sodium (mmol/L) | 142 | 133–146 |
| Potassium (mmol/L) | 4.1 | 3.5–5.3 |
| Urea (mmol/L) | 9.0 | 2.5–7.8 |
| Creatinine (µmol/L) | 137 | 48–128 |
| eGFR (L/min/1.73 m2) | 35 | ≥ 90 |
| Total bilirubin (µmol/L) | 8 | 0.0–21 |
| Alanine aminotransferase (IU/L) | 25 | 0.0–40 |
| Alkaline phosphatase (IU/L) | 60 | 30–130 |
| C-reactive protein (mg/L) | 19 | 0–10 |
eGFR: estimated glomerular filtration rate.
Given the risk of systemic embolism, oral anticoagulation with apixaban was initiated. Cardiac MRI was not performed because the device was not MR-conditional.
The patient was referred to a tertiary centre where peri-procedural transoesophageal echocardiography (TOE) demonstrated no thrombus attached to the lead or within the cardiac chambers. Transvenous extraction of the malpositioned lead was successfully performed, followed by implantation of a new right ventricular ICD lead (Medtronic Cobalt XT DR MRI) under imaging guidance.
Although the patient’s LVEF had improved to 45–50% at the time of extraction, reimplantation of the ICD was performed following multidisciplinary device team consultation. The patient had previously met clear guideline criteria for primary prevention ICD implantation (LVEF < 30% in the setting of ischaemic cardiomyopathy). Given the known variability in LVEF measurement and the persistence of an ischaemic substrate for ventricular arrhythmia, continued device therapy was considered clinically appropriate.
PFO closure was discussed in a multidisciplinary setting, but was not pursued in the absence of prior embolic events. Anticoagulation was discontinued following lead extraction.
Remote interrogation of the ICD at three months demonstrated normal device function with no recorded arrhythmic events.
At review in the haematology clinic in October 2025, the patient remained in remission and was clinically well, with no constitutional (“B”) symptoms and stable haematological parameters. At subsequent cardiology follow-up in November 2025, she reported stable symptoms corresponding to NYHA functional class II while receiving guideline-directed medical therapy for heart failure, including sacubitril/valsartan 49/51 mg twice daily, bisoprolol 10 mg once daily, and dapagliflozin 10 mg once daily. A mineralocorticoid receptor antagonist was not prescribed because of underlying renal impairment, which remained stable during follow-up.
At follow-up, the patient reported satisfaction with the care provided and improvement in her symptoms.
A targeted narrative literature review was conducted using PubMed, MEDLINE, and Google Scholar. Search terms included “ICD lead malposition,” “left ventricular lead,” “patent foramen ovale,” and “systemic embolism.” English-language case reports, case series, reviews, and guideline documents involving adult patients were included.
Consistently across recent studies, the implantation of an ICD is generally considered safe and minimally invasive; however, post-procedural complications, including lead malposition, remain clinically significant [9, 16]. During ICD implantation, particularly when imaging is limited or the technique is suboptimal, the guidewire or lead may inadvertently cross a PFO into the left atrium. In contrast to earlier reports that primarily identified malposition after clinical events, more recent series highlight the value of proactive imaging in detecting asymptomatic cases [13]. If undetected, the lead may then be advanced through the mitral valve and positioned in the left ventricle rather than in the right ventricle [9, 16].
Malposition of an ICD lead into the left ventricle via a PFO is a rare but potentially devastating complication. Although PFO is present in roughly 1 in 4 adults, clinical awareness of its implications during device implantation remains low. Many cases are only recognized after thromboembolic events, underscoring the need for vigilance.
The literature shows conflicting reports regarding the frequency and outcomes of left ventricular lead malposition. While some case series report high thromboembolic risk [8], others note long-term asymptomatic courses [17, 18], likely reflecting differences in patient selection, lead dwell time, and imaging protocols.
The clinical implications associated with this may involve thromboembolic events such as myocardial infarction and ischaemic stroke; vascular or valvular damage; and infection [9–11]. The most common pathway by which the ICD lead enters the left ventricle is via a PFO or an ASD; passage via the aortic valve through axillary or subclavian access, and ventricular septal perforations or defects account for other routes of entry [11, 13, 19]. Risk factors identified in multiple studies include congenital and acquired cardiac defects, female sex, high body mass index (BMI), and operator inexperience, although inconsistencies exist due to heterogeneous reporting [11, 13, 19–21].
The most common mechanism of inadvertent left ventricular lead placement is passage through a PFO or ASD. The foramen ovale normally closes after birth through fusion of the septum primum and septum secundum. Failure of this fusion results in a PFO, which persists in approximately 25% of adults [22].
A PFO represents a persistent communication between the right and left atria caused by incomplete fusion of the septum primum and septum secundum. Rather than a simple circular defect, a PFO typically forms a tunnel-like channel that functions as a flap valve. Under normal left-atrial pressure conditions, the septum primum remains apposed to the septum secundum, resulting in functional closure. Transient increases in right atrial pressure—such as during coughing, Valsalva manoeuvre, or instrumentation with guidewires—may separate the septal leaflets and permit passage of a catheter or pacing lead [22, 23]. During device implantation, a guidewire inadvertently entering this tunnel may traverse the left atrium and subsequently pass through the mitral valve into the left ventricle.
Malposition is more likely to occur when the procedure is performed with limited fluoroscopic guidance or when only an AP view is used. Most left ventricle lead misplacements occurred in patients with suboptimal intraoperative imaging or when TOE was not used [10]. In comparison, centres using echocardiography-guided implantation and standardized procedural protocols report lower complication rates, highlighting the importance of imagingguided technique and structured workflows in mitigating risk [24].
Besides PFOs, other ASDs (e.g., ostium secundum ASD) or iatrogenic perforations of the septum during guidewire manipulation can lead to lead migration into the left heart [25]. Variation in reporting standards and imaging protocols likely explains discrepancies in incidence rates between different case series.
Recognition of lead malposition may be challenging. Several diagnostic tools play complementary roles.
ECG can provide an important initial clue. Right ventricular pacing typically produces a left bundle branch block pattern because ventricular activation spreads from the right ventricle to the left ventricle. In contrast, pacing originating in the left ventricle produces a RBBB morphology, particularly in leads V1-V2 [9, 26]. However, inconsistencies across studies indicate that ECG alone may be insufficient, as conduction patterns vary with lead position and myocardial substrate [15]. In the present case, the expected RBBB morphology was not observed despite prolonged left ventricular lead placement, highlighting the limitations of relying solely on ECG findings.
Standard posteroanterior chest radiography may fail to detect abnormal lead positioning. Lateral projections are often more informative because a lead positioned in the left ventricle typically courses posteriorly toward the spine rather than anteriorly toward the right ventricular apex.
During implantation, the use of multiple fluoroscopic views, particularly left anterior oblique projections, may help confirm appropriate lead position. The lead’s unusual trajectory in the lateral view often provides the first visual clue [27].
Echocardiography is particularly valuable for confirming the trajectory of the lead through cardiac structures. TTE may visualise the abnormal course, while TOE provides superior visualisation of the interatrial septum and associated defects.
When echocardiography is inconclusive, cardiac CT provides high-resolution imaging with multiplanar reconstruction, enabling precise delineation of the lead trajectory and its relationship to surrounding cardiac structures.
Cardiac MRI has traditionally been contraindicated in patients with implantable cardioverter-defibrillators (ICDs); however, though modern MR-conditional devices have expanded its utility [28], MRI remains restricted in many cases and requires careful device-specific evaluation before use.
Given the potentially serious clinical consequences of lead malposition, adherence to careful procedural protocols is essential. Contemporary implantation practice should include:
Use multiplanar fluoroscopy (posteroanterior and lateral views) to confirm the appropriate lead trajectory;
Verify QRS morphology on the post-implant ECG to ensure the expected ventricular activation pattern;
Employ intraoperative echocardiography in complex or uncertain cases;
Consider screening for PFO when unusual guidewire behaviour is observed.
Although inadvertent lead malposition may remain undetected at the time of implantation, several procedural findings should raise suspicion. Unusual guidewire trajectories during advancement, unexpected resistance while positioning the lead, or atypical fluoroscopic projections may suggest entry into an abnormal pathway. If the lead appears to course posteriorly on lateral fluoroscopic views rather than anteriorly toward the right ventricular apex, this should prompt immediate reassessment of lead position.
ECG findings obtained immediately after lead placement may also provide valuable diagnostic clues. As ventricular activation during right ventricular pacing spreads from right to left, a left bundle branch block morphology is typically observed. Conversely, pacing originating from the left ventricle may produce a RBBB pattern. However, reliance on this criterion alone may be misleading, as variations in ventricular conduction patterns and lead location can produce atypical morphologies.
In addition, intra-procedural imaging plays an important role in confirming correct lead placement. The use of multiple fluoroscopic projections, particularly left anterior oblique views, can help distinguish between right ventricular and left ventricular positions. In complex cases or when an abnormal trajectory is suspected, adjunctive imaging with intraoperative echocardiography may provide additional confirmation.
One of the principal concerns associated with left ventricular lead malposition is the risk of systemic thromboembolism. The presence of an intracardiac foreign body within the systemic circulation may promote thrombus formation due to endothelial disruption and turbulent flow patterns [9]. Embolization from such thrombi may result in transient ischaemic attack, stroke, or peripheral arterial occlusion. Stroke may be the first clinical manifestation in some cases [8]. In some patients, embolic events may remain clinically silent. Silent cerebral infarction has been described in patients with intracardiac thrombotic sources and may only be detected on neuroimaging [17].
Although our patient did not report neurological symptoms, the possibility of silent embolic phenomena cannot be completely excluded in the absence of dedicated brain imaging. This represents an inherent limitation of the present case.
Observational studies suggest that systemic embolic complications may occur months or even years after device implantation if malposition remains undetected [29]. Consequently, early recognition of abnormal lead positioning is essential to mitigate long-term thromboembolic risk.
Prolonged dwell time of a malpositioned lead in the left ventricle increases the risk of infective endocarditis, especially on the mitral or aortic valves. The left ventricle is under higher pressure and flow than the right ventricle, making vegetation dislodgement more likely to result in embolism [30].
Mechanical interference with the mitral valve apparatus can occur, particularly if the lead repeatedly contacts the anterior or posterior leaflets during systole. This can cause or exacerbate mitral regurgitation, as discussed in a case by Solano et al. [31].
Improper sensing and shocking have also been described in malpositioned left ventricular leads. Left ventricular lead malposition may cause sensing abnormalities, including far-field signal oversensing, which can result in inappropriate device therapies or failure of appropriate therapy delivery during ventricular arrhythmias [32].
Various factors can impact treatment options, including the pathway underlying displacement, time since implantation, evidence of thrombus, and clinical presentation [9, 19].
Early detection typically favours prompt lead extraction because fibrotic adhesions are limited, and procedural risk is lower. In delayed presentations, extraction may carry a higher risk of thromboembolism due to thrombus formation or fibrotic attachment.
In our case, the patient had been asymptomatic, and lead misplacement had been an incidental finding, more than a year after implantation; post-procedural images up till that point had not detected any abnormalities. She underwent a successful lead extraction from the left ventricle and a new lead placement in the right ventricle. It is important to consider that possible adhesions or debris around the mispositioned lead could also lead to acute embolization during extraction.
Post-operatively, there are no specified guidelines for treatment, but it may involve percutaneous or surgical lead extraction, or a conservative approach with long-term anticoagulation [9]. There are no widely accepted definitions of what constitutes early or delayed diagnosis of lead misplacement, which further impacts management strategies. A period of < 12 months from implantation may be referred to as early, whereas > 12 months may be considered as delayed [18, 29].
There are currently no consensus guidelines regarding routine PFO closure following lead extraction, and decisions should be individualized.
During the early post-operative period, or when surgical risk is high, percutaneous lead extraction may be considered; however, lead manipulation—particularly with the use of an extraction sheath—may dislodge an existing thrombus, causing systemic embolism [9, 29]. The risk increases with the duration of lead malposition. Although extraction may eliminate the need for prolonged anticoagulation, other significant complications include vascular perforation or occlusion, and haemorrhage [18].
Percutaneous extraction with the assistance of transcatheter protection devices has been described, which mitigates the procedural risk of thromboembolism from a known thrombus or when its presence cannot be fully excluded [18]. Transcatheter cerebral embolic protection devices have been used during the extraction of malpositioned left-sided leads to help protect the cerebral circulation from embolic debris dislodged during manipulation, although the evidence remains limited and largely case-based [33]. While they do not mitigate other procedural risks such as vascular injury, perforation, or haemorrhage, their use can improve the safety profile of extraction in high-risk patients, particularly when pre-procedural imaging cannot fully exclude thrombus [34].
Early removal is associated with lower complication rates because fibrotic adhesions are less developed in recently implanted leads [35]. Extraction is typically performed under combined fluoroscopic and echocardiographic guidance to monitor for pericardial effusion or thrombus dislodgement [6]. Periprocedural heparinization is generally recommended to reduce embolic risk. In most cases, once the lead is extracted, a new ICD lead is successfully positioned in the right ventricular apex or septum [36].
Following delayed recognition (> 12 months) in patients presenting with complications of lead displacement, surgical extraction may be considered [29]. This approach is preferred when concomitant surgical correction of a cardiac defect is indicated, for example, repair of an ASD in a patient with neurological symptoms related to left ventricular lead migration, followed by implantation of a new ICD [19]. Surgical intervention is also indicated in the presence of endocarditis, significant valvular injury, or a large thrombus attached to the lead, where transvenous extraction may be insufficient or high-risk.
Conservative management is typically reserved for asymptomatic patients or those at high procedural risk, particularly when lead malposition is detected late after implantation [18]. In cases diagnosed several years post-implantation, where extraction carries substantial procedural risk, long-term systemic anticoagulation represents a pragmatic strategy [13].
The primary long-term trade-off of conservative management is persistent thromboembolic risk, as the lead remains within the systemic circulation. Although no randomized controlled trials have compared anticoagulation strategies in this setting, limited observational evidence and expert reviews suggest that chronic therapeutic anticoagulation with warfarin may reduce the risk of systemic embolic events in patients with left-sided lead malposition when lead removal is not performed [37]. Case series and registry-based observations have reported that patients maintained on long-term anticoagulation, most commonly with warfarin, experience a lower incidence of stroke and systemic embolism compared with untreated patients, although the residual risk is not eliminated [38]. These findings support anticoagulation as a reasonable strategy when lead extraction is not feasible or carries excessive risk.
If lead extraction is deferred, ICD reprogramming may be required to mitigate oversensing, inappropriate shocks, or ineffective therapy. Regular device interrogation, ECG monitoring, and periodic imaging (TTE or cardiac CT) are recommended to detect changes in lead position or the development of complications [39].
In asymptomatic patients, periodic neurological evaluation may be considered to monitor for potential thromboembolic events, including silent cerebral infarctions, which may arise from systemic embolization associated with a lead positioned within the left-sided circulation.
In our patient, erroneous lead placement into the left ventricle occurred via a PFO, without associated clinical symptoms. The patient underwent successful transvenous lead extraction followed by placement of a new right ventricular lead, without procedural complications. Currently, there are no established guidelines regarding the role of PFO closure as a precautionary measure after removal of a malpositioned left ventricular lead.
Approximately 200,000 ICD implantations are performed each year [40] globally, and this number is expected to increase as the incidence of cardiovascular disease rises and the indications for cardiac implantable electronic devices expand. As implant volumes grow, even rare complications such as inadvertent left ventricular lead placement may be encountered more frequently in absolute numbers, highlighting the need for clearer clinical recommendations. Larger studies evaluating different therapeutic strategies would help inform practice, although the rarity of this complication may limit such investigations. Long-term follow-up of affected patients would also be valuable in clarifying the short- and long-term outcomes associated with each management approach.
In the present case, despite delayed recognition, imaging demonstrated no thrombus, and the patient remained clinically stable. Transvenous extraction was therefore performed successfully without complications.
The optimal management strategy for delayed recognition of left ventricular lead malposition remains uncertain due to the absence of large prospective studies. Decisions are therefore typically individualized and guided by a multidisciplinary evaluation involving electrophysiologists, imaging specialists, and cardiac surgeons.
Several factors influence management decisions, including the duration of lead implantation, evidence of thrombus formation, patient comorbidities, and the anticipated procedural risk of extraction. In patients with long-standing lead malposition, fibrotic adhesions may increase the technical complexity of extraction and potentially elevate the risk of embolization during manipulation of the lead.
Conversely, leaving the lead in situ exposes the patient to an ongoing risk of systemic embolism. In such situations, lifelong anticoagulation may be considered as an alternative strategy, although robust evidence supporting this approach is limited. The choice between extraction and conservative management, therefore, requires careful balancing of procedural risk against long-term embolic risk.
In the present case, the absence of thrombus on TOE and the availability of an experienced extraction centre favoured transvenous lead removal. This approach allowed definitive correction of the abnormal lead position while minimising long-term thromboembolic risk.
Prevention and early recognition remain important. During device implantation, careful fluoroscopic assessment using multiple projections is essential to confirm correct right ventricular lead positioning. In particular, the left anterior oblique (LAO) view helps distinguish a right ventricular septal or apical lead from a malpositioned lead that has crossed an interatrial communication and entered the left ventricle. Failure to utilise multi-view fluoroscopy may delay recognition of abnormal lead trajectories and contribute to missed intraprocedural diagnosis.
As the global number of ICD implantations continues to increase, even rare complications may be encountered more frequently in absolute numbers. Heightened awareness and careful imaging can reduce the risk of inadvertent lead malposition.
Inadvertent ICD lead placement into the left ventricle via a PFO is an uncommon but potentially serious complication. This case highlights that reliance on ECG findings alone may be insufficient for diagnosis and that multimodality imaging—including echocardiography, fluoroscopy, and CT—plays a critical role in detection.
As the global number of device implantations continues to increase, even rare complications will be encountered more frequently in absolute terms. Meticulous implantation technique, biplanar fluoroscopic imaging, and careful post-procedural evaluation are essential preventive measures.
Future multicentre studies are required to better define optimal management strategies, particularly regarding the role of anticoagulation, indications for extraction in delayed diagnosis, and whether routine PFO closure should be considered after lead removal.
Inadvertent placement of an ICD lead in the left ventricle is a rare but potentially serious complication that may remain clinically silent for prolonged periods.
ECG findings may not always reliably identify lead malposition; therefore, multimodality imaging, including echocardiography and CT, can play a crucial role in confirming the diagnosis.
Careful procedural technique, including the use of multiple fluoroscopic views and verification of pacing morphology, is essential to prevent inadvertent lead passage through intracardiac defects such as a PFO.
AP: antero-posterior
ASD: atrial septal defect
CT: computed tomography
ECG: electrocardiogram
ICDs: implantable cardioverter defibrillators
LVEF: left ventricular ejection fraction
MRI: magnetic resonance imaging
NYHA: New York Heart Association
PFO: patent foramen ovale
RBBB: right bundle branch block
TOE: transoesophageal echocardiography
TTE: transthoracic echocardiography
We thank Muhammad Abdul Rehman Jauhar for the help with the literature search and editing of the article.
SJ: Conceptualization, Writing—original draft, Writing—review & editing. AU: Writing—review & editing. ASS: Conceptualization, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
The study was conducted in accordance with institutional guidelines and the principles of the Declaration of Helsinki. A formal ethical approval was not required for this retrospective case report.
Informed consent to participate in the study was obtained from the patient.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 456
Download: 28
Times Cited: 0
