Affiliation:
1Department of Adult Cardiology, National Institute of Cardiovascular Diseases (NICVD), Karachi 75510, Pakistan
Email: nylaurentian@live.co.uk
ORCID: https://orcid.org/0000-0002-7724-4017
Affiliation:
2Department of Medicine, Liaquat National Hospital and Medical College, Karachi 74800, Pakistan
ORCID: https://orcid.org/0009-0000-4942-5374
Affiliation:
1Department of Adult Cardiology, National Institute of Cardiovascular Diseases (NICVD), Karachi 75510, Pakistan
ORCID: https://orcid.org/0000-0002-4278-7919
Explor Cardiol. 2026;4:1012106 DOI: https://doi.org/10.37349/ec.2026.1012106
Received: December 18, 2025 Accepted: February 14, 2026 Published: April 29, 2026
Academic Editor: Carmine Gazzaruso, University of Milan, Italy
Diabetes mellitus significantly increases the burden of coronary artery disease and peripheral arterial disease, frequently necessitating vascular bypass surgery. Imaging plays a central role across the perioperative continuum, from preoperative risk stratification and surgical planning to intraoperative guidance and postoperative surveillance. This narrative review synthesizes current evidence on the application of multimodality imaging in diabetic patients undergoing coronary artery bypass grafting and peripheral arterial bypass procedures. A structured literature search of major databases was undertaken to identify contemporary studies evaluating echocardiography, computed tomography angiography, duplex ultrasound, magnetic resonance angiography, digital subtraction angiography, and functional nuclear imaging. Diabetic-specific factors such as diffuse multivessel disease, medial arterial calcification, renal impairment, and microvascular dysfunction substantially influence imaging performance and modality selection. While noninvasive imaging increasingly guides surgical decision-making and follow-up, the available evidence remains heterogeneous and largely observational. Advanced and hybrid imaging techniques show promise for individualized care but are limited by cost, availability, and a lack of robust outcome-driven data. Standardized imaging pathways and higher-quality comparative studies are required to optimize perioperative imaging strategies in this high-risk population.
Diabetes mellitus is a major global health challenge and a leading contributor to cardiovascular morbidity and mortality. Patients with diabetes develop accelerated and diffuse atherosclerosis, predisposing them to both coronary artery disease and peripheral arterial disease, frequently requiring surgical revascularization. Compared with non-diabetic patients, individuals with diabetes undergoing coronary artery bypass grafting or peripheral arterial bypass experience higher rates of perioperative complications, graft failure, and long-term adverse outcomes [1–4]. Imaging therefore plays a pivotal role in optimizing patient selection, procedural planning, and postoperative surveillance.
Recent advances in cardiovascular and vascular imaging have expanded the range of available noninvasive and minimally invasive techniques. However, diabetic-specific factors such as medial arterial calcification, chronic kidney disease, microvascular dysfunction, and multilevel disease complicate both image acquisition and interpretation. Despite widespread clinical use, uncertainty remains regarding the optimal sequencing, comparative effectiveness, and cost-efficiency of different imaging modalities in this population. This narrative review critically examines the current role of imaging across the perioperative course of vascular bypass surgery in diabetic patients, highlighting strengths, limitations, and gaps in the existing literature.
A narrative literature review was performed using PubMed/MEDLINE, PubMed Central, ScienceDirect, and the Cochrane Library. Searches covered the period from January 2020 to March 2025 and employed combinations of the terms “diabetes”, “coronary artery bypass”, “peripheral arterial disease”, “vascular bypass surgery” and “imaging”. English-language original studies, narrative reviews, and major guidelines addressing perioperative imaging in diabetic patients undergoing coronary or peripheral revascularization were included. Case reports, retracted articles, and studies not addressing surgical imaging were excluded. Titles and abstracts were screened followed by full-text review. Evidence was synthesized qualitatively with emphasis on diabetic-specific considerations, clinical applicability, and comparative insights across imaging modalities.
Preoperative imaging focuses on defining vascular anatomy, myocardial function, and tissue viability. Echocardiography remains investigation of choice for preoperative cardiac assessment. It has advanced techniques such as speckle-tracking enabling detection of subclinical myocardial dysfunction in diabetic patients with preserved ejection fraction (EF) as shown in Figure 1 highlighting that the 3D EF was within the normal range (52.7%) [5]. Computed tomography angiography provides high-resolution anatomical mapping of coronary and peripheral vessels, facilitating conduit selection and operative planning, although heavy calcification and contrast nephropathy limit its universal applicability in diabetics [6]. Figure 2 shows multivessel disease with significant stenosis in left anterior descending (LAD) and left circumflex coronary artery (LCX) along with reduced EF. Magnetic resonance angiography offers a radiation-free alternative in patients with renal impairment, albeit with lower spatial resolution and limited availability [7].
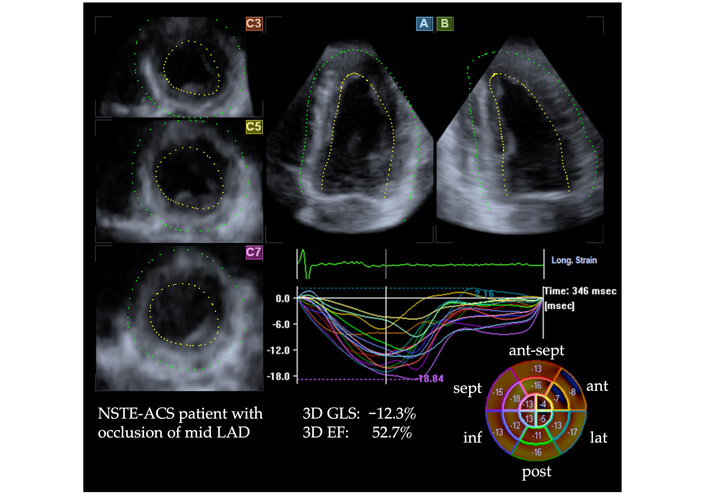
3D speckle-tracking echocardiographic strain analysis of a patient with mid-segment occlusion of the left anterior descending (LAD) coronary artery. The apical views (A and B) and corresponding short-axis sections (C3, C5, C7) are displayed, along with segmental strain curves and a bull’s-eye map. Although the 3D ejection fraction (EF) was within the normal range (52.7%), the 3D global longitudinal strain (GLS) showed a marked reduction (−12.3%). The bull’s-eye plot in the lower right clearly demonstrates substantially decreased regional longitudinal strain in the anterior segments, corresponding to the myocardial territory supplied by the LAD. NSTE-ACS: non-ST segment Elevation Acute Coronary Syndrome; ant-sept: antero-septal; lat: lateral; inf: inferior; post: posterior. Reprinted from Stokke et al. [5]. © 2025 The Author(s). Distributed under a CC BY license.
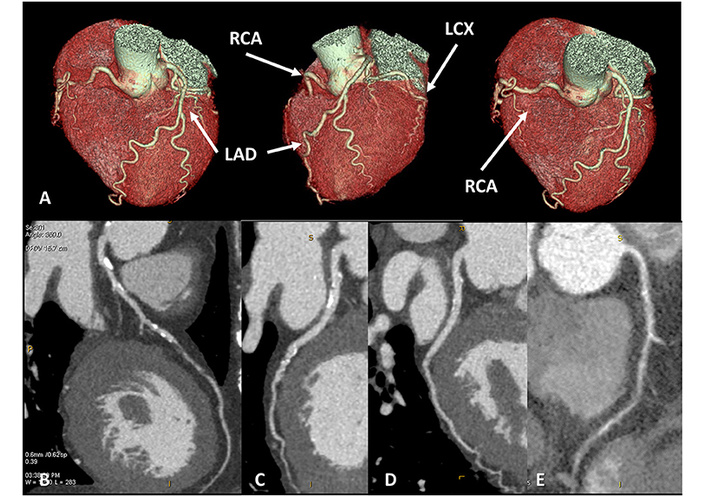
A 65-year-old female patient presented with newly diagnosed severe left ventricular systolic dysfunction (EF < 30%) and symptoms of dyspnea. Cardiac CT, performed with optimal image quality (A), revealed severe multivessel coronary artery disease. A near-occlusive lesion was identified in the mid-segment of the left anterior descending (LAD) artery (B), a significant stenosis in the left circumflex artery (C), no significant narrowing in the marginal branch, and near-occlusive disease in the right coronary artery (D, E). EF: ejection fraction; RCA: right coronary artery; LCX: left circumflex coronary artery. Reprinted from Conte et al. [6]. © 2021 The Author(s). Distributed under a CC BY license.
Intraoperatively, imaging is directed toward confirming graft quality and adequacy of revascularization. Transesophageal echocardiography enables real-time functional assessment, while Doppler-based transit-time flow measurement is widely used to verify graft patency during coronary bypass procedures [8]. Digital subtraction angiography, though invasive, remains indispensable in complex peripheral interventions requiring precise vascular detail [9], shown in Figure 3 in detail with severe stenosis of distal superficial femoral artery.
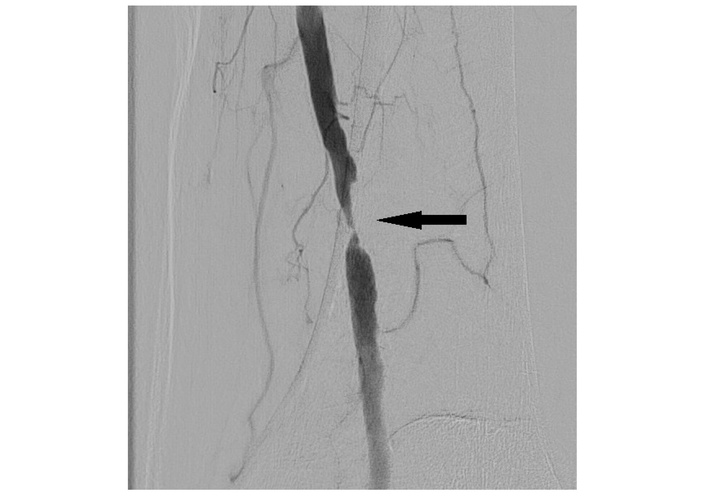
Digital subtraction angiography showing chronic severe stenosis of distal superficial femoral artery (arrow). Reprinted from Posa et al. [9]. © 2022 The Author(s). Distributed under a CC BY license.
Postoperative imaging aims to detect early graft failure, disease progression, and procedure-related complications. Duplex ultrasound is the first-line modality for routine graft surveillance owing to its safety, accessibility, and cost-effectiveness [10]. Computed tomography angiography and magnetic resonance angiography are selectively employed when ultrasound findings are equivocal or when symptoms recur. Functional imaging using positron emission tomography or single-photon emission computed tomography as shown in Figure 4 illustrates mortality risk assessment and provides incremental prognostic information by assessing myocardial or tissue perfusion and viability, particularly in patients with persistent symptoms despite patent grafts [11].
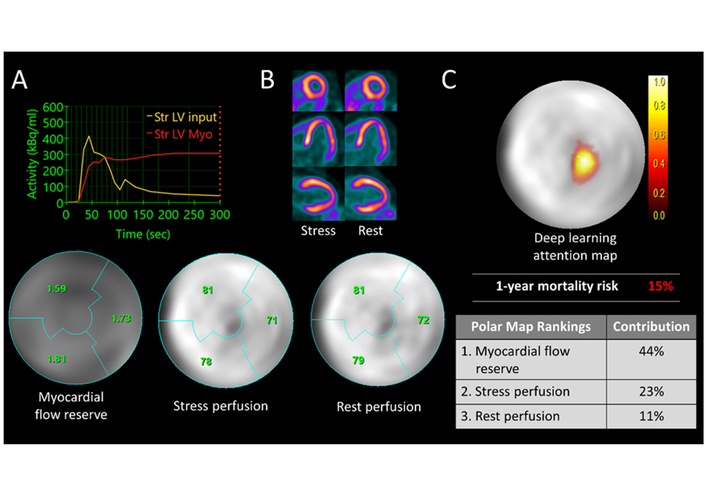
Case example illustrating mortality risk assessment using deep learning applied to multiparametric PET imaging. (A) A 72-year-old female patient demonstrated a reduced coronary flow reserve of 1.7 on the polar map. (B) Both rest and stress perfusion studies appeared normal. The patient unfortunately died three months after undergoing the PET scan. (C) The deep learning-generated attention map visualizes the patient’s individualized mortality risk. PET: positron emission tomography; LV: left ventricular; Str: strain; Myo: myocardium. Reprinted from Blach et al. [11]. © 2025 The Author(s). Distributed under a CC BY license.
Diabetic vasculopathy presents unique challenges for imaging. Medial arterial calcification reduces the diagnostic accuracy of duplex ultrasound and computed tomography angiography, while renal dysfunction restricts the use of iodinated contrast agents. Comparative evidence suggests that computed tomography angiography offers superior anatomical detail, whereas magnetic resonance angiography is preferable in patients with renal impairment [6, 7]. Functional imaging adds value in assessing myocardial viability and predicting benefit from revascularization, but its routine use is constrained by cost and limited accessibility. No single modality is sufficient in isolation; tailored multimodality strategies based on patient-specific risk profiles appear most effective.
Cost considerations are critical, particularly in low- and middle-income settings where the prevalence of diabetes is rising. Duplex ultrasound remains the most economical and widely available modality for both diagnosis and follow-up. Computed tomography angiography provides a favorable incremental cost-effectiveness ratio for operative planning but increases overall expenditure. Advanced and hybrid imaging techniques offer diagnostic advantages, but currently lack robust economic justification for routine use. A stepwise imaging approach, escalating from low-cost to advanced modalities as clinically indicated, is the most efficient strategy [11, 12].
Imaging is integral to the perioperative management of diabetic patients undergoing vascular bypass surgery. Advances in noninvasive and functional imaging have enhanced risk stratification, surgical planning, and postoperative surveillance (Table 1). Nevertheless, the available evidence remains largely observational, and diabetic-specific challenges persist. Standardized imaging pathways and high-quality comparative studies are required to define optimal imaging strategies and improve outcomes in this high-risk population.
Summary of applications, strengths and limitations of various imaging modalities.
| Imaging modality | Key applications | Strengths | Limitations | Evidence |
|---|---|---|---|---|
| Echocardiography (2D, Doppler, speckle-tracking) | Pre & postoperative LV function, GLS, diastolic recovery, intraoperative renal prognosis (RRI) | Widely available, noninvasive, sensitive for subclinical dysfunction (GLS) | Operator-dependent, limited use in obese/postoperative patients | Speckle-tracking superior to conventional LVEF for prognosis [5, 8, 13] |
| CTA | Preoperative vessel/conduit mapping, intraoperative planning, early graft failure detection, long-term patency assessment | High resolution, fast, reliable roadmap, less invasive than invasive angiography | Radiation exposure, iodinated contrast nephropathy, less accurate with heavy calcification | BYPASS-CTCA trial: reduced complications, better surgical planning [1, 6, 14] |
| DUS | Conduit mapping (saphenous/radial), intraoperative graft flow verification, postoperative surveillance (CABG & PAD) | Noninvasive, safe, real-time hemodynamic data | Operator-dependent, less accurate in calcified/distal vessels | Routine surveillance reduces graft failure, limb loss [10, 15] |
| MRA | Alternative to CTA in renal risk patients, PAD & CABG mapping, postoperative graft monitoring | No radiation, safe in CKD, high concordance with DSA | Lower spatial resolution, motion artifacts, contraindicated with implants | QISS and non-contrast MRA: > 85–90% accuracy vs. invasive angiography [7] |
| PET/SPECT | Myocardial perfusion, ischemia, viability, myocardial blood flow (PET) | Quantitative perfusion (PET), strong prognostic value, widely validated | Radiation exposure, lower spatial resolution (SPECT), cost (PET) | 2022 ASNC/AAPM/SCCT/SNMMI guideline for the use of CT in hybrid nuclear/CT cardiac imaging [16] |
GLS: global longitudinal strain; LV: left ventricular; RRI: renal resistive index; LVEF: left ventricular ejection fraction; CTA: computed tomography angiography; DUS: duplex ultrasound; CABG: coronary artery bypass graft; PAD: peripheral arterial disease; MRA: magnetic resonance angiography; CKD: chronic kidney disease; DSA: digital subtraction angiography; QISS: Quiescent-Inflow Single-Shot; PET: positron emission tomography; SPECT: single-photon emission computed tomography; 18F-FDG: 18Flourine-Fluorodeoxyglucose; MR: magnetic resonance.
EF: ejection fraction
GLS: global longitudinal strain
LAD: left anterior descending
LV: left ventricular
PET: positron emission tomography
NY: Conceptualization, Investigation, Writing—original draft, Supervision. FT: Investigation, Writing—review & editing. ABA: Validation, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 228
Download: 22
Times Cited: 0
