Affiliation:
Institute of Family Medicine, University of Szeged, 6725 Szeged, Hungary
Email: albert.varga@med.u-szeged.hu
ORCID: https://orcid.org/0000-0002-8879-3927
Affiliation:
Institute of Family Medicine, University of Szeged, 6725 Szeged, Hungary
ORCID: https://orcid.org/0000-0002-8513-5750
Explor Cardiol. 2026;4:1012103 DOI: https://doi.org/10.37349/ec.2026.1012103
Received: June 15, 2025 Accepted: March 26, 2026 Published: April 19, 2026
Academic Editor: Erkin Mirrakhimov, Internal Medicine of Kyrgyz State Medical Academy, Kyrgyzstan
Coronary vasospasm, affecting both epicardial arteries and the coronary microcirculation, is a significant yet frequently underdiagnosed and undertreated cause of coronary syndromes. When promptly identified, it carries a relatively benign prognosis. Recognition can be straightforward in non-cardiology settings when triggered by known spasmogenic agents such as misoprostol (obstetrics) or 5-fluorouracil (oncology). Vasospasm may also be incidentally revealed during noninvasive functional testing, typically presenting as ST-segment elevation during early recovery phases, or after administration of agents like aminophylline following dipyridamole or β-blockers following dobutamine. In patients with high clinical suspicion but negative Holter or stress test findings, targeted provocation with ergonovine or hyperventilation protocols can safely induce vasospasm and unmask regional wall motion abnormalities, indicating epicardial involvement. Hyperventilation-Doppler echocardiography enables the detection of microvascular dysfunction through reductions in coronary flow velocity in the mid-distal left anterior descending coronary artery. A multi-stress, multi-marker functional testing approach offers a noninvasive, safe, and effective diagnostic strategy. Inducible wall motion abnormalities are specific for epicardial spasm, while Doppler-detected flow reduction is more sensitive for microvascular dysfunction. Early diagnosis is essential, as coronary vasospasm, though potentially life-threatening, is highly manageable with appropriate therapy. Management of patients with proven epicardial coronary artery or microvascular vasospasm involves starting therapy with calcium channel blockers and nitrates, and avoiding β-blockers, as they can worsen vasospasm by blocking β2-mediated vasodilation and leaving α1-mediated vasoconstriction unopposed.
Coronary artery spasm (CAS) is a well-established mechanism contributing to a wide spectrum of acute and chronic coronary syndromes, ranging from angina pectoris to myocardial infarction and sudden cardiac death [1]. Although CAS is a known cause of coronary syndromes, its clinical identification is challenging. Guideline-recommended invasive tests are complex and risky, involving catheterization, radiation exposure, nephrotoxic contrast agents, and procedural risks [2–4]. Consequently, CAS is often underdiagnosed and undertreated [5], which may lead to inappropriate interventions. For example, coronary stenting in the context of CAS is generally ineffective and can even be harmful [6], while β-blockers may exacerbate CAS and worsen symptoms [7]. In this review, we examine the recent literature and propose a practical, stepwise diagnostic approach—beginning with clinical history, progressing through standard functional testing, and culminating in targeted provocation testing—to improve the recognition and management of CAS.
Epidemiologic data show wide differences in the prevalence of CAS in different countries. The frequency of CAS seems to be greater in Asian than in Western populations, in females compared to males, and the prevalence tends to decrease after the age of 70 years. However, it is important to emphasize that CAS is found everywhere, at all ages, and in all populations, when it is appropriately looked for [1].
A patient’s clinical history can be highly suggestive of CAS when angina presents with four key features: (1) occurrence at rest, particularly at night; (2) a diurnal pattern, often more pronounced in the early morning hours; (3) worsening or lack of control with β-blocker therapy; and (4) precipitation by hyperventilation. Another important clue is the presence of angina with non-obstructive coronary arteries (ANOCA), although CAS can occur at any level of underlying organic coronary artery disease (CAD) [8].
A powerful indicator is the recurrence of angina following successful coronary stenting, especially when angiography confirms patency of the stented segment. CAS should also be considered in cases of syncope or cardiac arrest, particularly when preceded by chest pain and in the absence of obstructive CAD [9].
Although the mechanisms are complex, heterogeneous, and partially unknown, the clinical recognition of drug-induced CAS is easy, and the therapy consists of the immediate withdrawal of the suspected drug and the start of a cardioprotective treatment with calcium-channel blockers and nitrates, avoiding β-blockers that may exacerbate spasm through blockade of β2-vasodilation, leaving unopposed the α1 vasoconstriction. Particular attention should be given to the occurrence of CAS in clinical settings far removed from traditional cardiology practice. In these contexts, CAS may be triggered by drugs whose rare but potentially life-threatening adverse effects include CAS (Table 1). Such reactions have been reported with medications commonly used in various specialties:
Drugs with vasospastic potential.
| Drug | Disease | Clinical specialty | Receptors or mediators |
|---|---|---|---|
| Amoxicillin, amphetamine | Infection, doping | GP, ER | 5-HT1 receptors, α1-adrenoreceptors |
| Aspirin high-dose | Pain, fever | GP, rheumatologist | Thromboxane A2 vasoconstriction |
| BB, non-selective* | Anxiety, glaucoma | Psychiatry, ophthalmology | β2-adrenergic receptor blockade |
| Bromocriptin | Hyperprolactinemia | Endocrinology | Dopamine D2, α1-agonist |
| Capecitabine, 5-FU | Chemotherapy | Oncology | Increases endothelin-1, decreases NO |
| Cisplatin | Chemotherapy | Oncology | Endothelial dysfunction |
| Cocaine | Addiction | ER | α1-adrenoreceptors |
| Cyclosporine | Transplant | Nephrology, transplant unit | Endothelial dysfunction |
| Ergot alkaloids** | Bleeding, migraine | Obstetrics, neurology | 5-HT1 and α-adrenergic receptor |
| Marijuana | Addiction | ER | CB-1 and CB-2 |
| Methacholine | Bronchial reactivity | Pneumology | Muscarinic receptor agonist |
| Misoprostol | Hysteroscopy | Gynecology | Prostaglandin E receptor subtype EP3 |
| Pseudo-ephedrine | Nasal decongestion | GP | α1-adrenoreceptors |
| Triptans*** | Headache | Neurology, GP | 5-HT1 B/1D receptor agonist |
*: e.g., propranolol; **: e.g., ergotamine, dihydroergotamine; ***: e.g., sumatriptan, zolmitriptan. BB: β-blocker; 5-FU: 5-fluorouracil; CB: cannabinoid receptors; ER: emergency room; GP: general practitioner.
Gynecology and obstetrics, such as ergot-derivatives for uterine bleeding [10] or misoprostol to facilitate hysteroscopy [11];
Neurology, such as sumatriptan for migraine treatment [12];
Ophthalmology, such as carteolol eye drops for glaucoma [13];
Pneumology, with methacholine for bronchial reactivity testing [14];
Oncology, during treatment with chemotherapeutic agents such as 5-fluorouracil or the pro-drug capecitabine [15];
Recreational drug use [16–18] and sports, involving performance-enhancing stimulants [19].
The broad range of triggers reflects the complexity of coronary tone regulation, primarily involving α1-adrenergic vasoconstrictive receptors, and to a lesser extent, histamine H1 receptors.
Drug-induced CAS can occur through four main mechanisms affecting endothelial cells, vascular smooth muscle cells, autonomic neurons, and inflammatory cells:
Endothelial cells: Endothelial dysfunction reduces nitric oxide-mediated vasodilation and enhances vasoconstriction.
Vascular smooth muscle cells: Hyperreactivity, often mediated by the Rho-kinase pathway, increases contractile sensitivity and CAS risk. In CAS, the affected vascular smooth muscle cells exhibit hypercontractility and an exaggerated response to vasoconstrictive signals. This excessive reactivity can be stimulated by various vasoconstrictors, including endothelin-1, serotonin, and histamine, which induce severe abnormal contraction of the muscle layer [2]. A primary molecular mechanism underlying this response is the activation of the RhoA/Rho-kinase pathway. This pathway increases smooth muscle contractility by inhibiting myosin light chain phosphatase, thereby promoting phosphorylation of myosin light chains. Enhanced Rho-kinase activity is strongly linked to the development of CAS. Clinically, inhibiting Rho-kinase pharmacologically has proven highly effective in relieving CAS. For instance, intracoronary administration of the Rho-kinase inhibitor fasudil rapidly reverses provoked CAS in patients with vasospastic angina, underscoring the pivotal role of this signaling cascade. Collectively, experimental and clinical evidence suggests that a fundamental hypercontractile state of the vascular smooth muscle—partly driven by Rho-kinase-mediated increases in calcium sensitivity—plays a crucial role in the pathophysiology of CAS [20].
Autonomic neurons: Excessive sympathetic activation or parasympathetic withdrawal leads to an imbalance favoring vasoconstriction.
Neutrophils and inflammatory cells: Inflammatory responses and production of reactive oxygen species can impair endothelial function and promote CAS [20]. In particular, systemic and local inflammation and the associated increased oxidative stress aggravate endothelial dysfunction and vascular smooth muscle hyperactivity, as also shown by the association between high levels of circulating interleukin-6 and CAS clinically. The increased inflammatory state can also be an actionable therapeutic target in the future, with targeted therapy reducing the levels of interleukin-6, such as tocilizumab [20].
Despite the complex and often heterogeneous underlying mechanisms, clinical recognition is typically straightforward if one thinks of it. Management involves stopping the suspected drug and starting therapy with calcium channel blockers and nitrates. Avoid β-blockers, as they can worsen CAS by blocking β2-mediated vasodilation and leaving α1-mediated vasoconstriction unopposed.
CAS can be revealed during functional testing in two distinct scenarios: either as an incidental finding during routine ischemia testing for CAD, or as the result of targeted provocation testing when CAS is clinically suspected.
In some cases, CAS is unexpectedly uncovered at the end of a negative maximal exercise stress test. Despite the absence of obstructive CAD, patients may develop ST-segment elevation, a hallmark of transmural ischemia, accompanied by severe inducible regional wall motion abnormality (RWMA) on echocardiography. These changes typically resolve either spontaneously or following administration of intravenous or sublingual nitrates or sublingual nifedipine [21].
A similar vasospastic response has been reported following vasodilator stress testing with dipyridamole or adenosine, particularly after aminophylline administration. Aminophylline antagonizes adenosine A2 receptors (vasodilatory) and simultaneously stimulates α1-adrenergic vasoconstriction, thereby triggering CAS [22, 23].
CAS is even more frequently observed during dobutamine stress echocardiography, particularly at high doses. While low-dose dobutamine predominantly stimulates β2-adrenergic (vasodilatory) receptors, high-dose infusion activates α1-adrenergic (vasoconstrictive) receptors. The vasospastic response is intensified by co-administration of atropine and further exacerbated by β-blockers, which blunt β2-mediated vasodilation and leave α1-mediated vasoconstriction unopposed [24, 25].
Recognizing CAS is crucial. Continued use of aminophylline or β-blockers during active vasospasm can worsen myocardial ischemia, leading to infarction or arrhythmias [26].
Targeted testing is indicated when clinical suspicion is high, typically based on symptoms consistent with vasospastic angina, non-diagnostic Holter monitoring, inability to obtain an electrocardiogram (ECG) during pain episodes, negativity of functional test with exercise or pharmacological stress, and angiographic evidence of normal or near-normal coronary arteries.
In such carefully selected patients, the diagnostic yield of targeted testing is high, and the benefits outweigh the risks. Among the most effective and safe methods are:
When performed under continuous echocardiographic monitoring, these tests allow real-time detection of inducible RWMA, enabling early test termination before the onset of ECG changes or chest pain. This provides a critical safety margin compared to ECG-only monitoring. The presence of inducible RWMA is diagnostic of epicardial CAS.
In cases where coronary microvascular dysfunction is suspected, the test of choice is hyperventilation combined with Doppler imaging of the coronary flow velocity (CFV) in the mid-to-distal left anterior descending artery. This test reveals a paradoxical reduction in CFV, despite an increased rate-pressure product, indicating coronary microvascular disease despite the absence of inducible RWMA [30] (Table 2). The hyperventilation test is an efficient and highly specific diagnostic method for detecting epicardial CAS, with a sensitivity of 90% and specificity of 99% according to a recent meta-analysis of 6 studies using coronary angiographic verification [29]. However, its sensitivity remains relatively low for the detection of microvascular CAS when using RWMA as the only diagnostic criterion. Notably, sensitivity improves significantly when incorporating CFV reduction as an additional diagnostic endpoint [30]. Advantages and limitations of the noninvasive approach compared to invasive testing for CAS diagnosis are summarized in Table 2.
Diagnostic modalities for coronary vasospasm.
| Parameter | Cardiac catheterization | Stress echocardiography |
|---|---|---|
| Accessibility | Low | High |
| Economic cost | High | Low |
| Radiation exposure | High (10 mSv) | Absent |
| CO2 emissions | 100 kg | 1 kg |
| Stress | Acetylcholine | Hyperventilation, ergonovine |
| Marker | Coronary diameter change | RWMA, CFV |
| Sensitivity | Very high | High |
CFV: coronary flow velocity; RWMA: regional wall motion abnormality.
The practical implementation of hyperventilation and ergonovine testing in clinical practice has been limited to date for several reasons. Hyperventilation is considered safe but exhibits suboptimal sensitivity when relying solely on traditional ECG or echocardiographic RWMA, and its efficacy depends on patient cooperation. In contrast, ergonovine demonstrates higher sensitivity but faces widespread restrictions due to lack of commercial availability in many countries and legitimate safety concerns—primarily stemming from catastrophic events reported with high-dose administration outside the catheterization laboratory.
However, when low scalar doses are used in carefully selected patients outside the catheterization lab—excluding those with established CAD and limiting testing to individuals with normal resting left ventricular function, angiographically normal coronary arteries, and negative functional stress tests—ergonovine can be safely performed using RWMA as an endpoint [27].
When provocative tests are performed during coronary angiography, reported major complication rates are low, ranging from 0.2% to 4.7% with intravenous ergonovine, 0.4% to 0.8% with intracoronary ergonovine, and 0% to 4.9% with intracoronary acetylcholine, with a mean complication rate of 0.62%. To this risk of acute complications, one should add the risk of nephrotoxic iodinated contrast material and the cancer risk of radiation exposure around 10 mSv (corresponding to 500 chest X-rays).
According to the landmark study of Om et al. [27], among 14,012 completed tests, 15% (n = 2,144) showed positive results, with development of RWMAs. Significant arrhythmias were more frequent in patients with positive than in those with negative test results (1.8% vs. 0.4%, p < 0.05). The most frequent arrhythmias included transient ventricular tachycardia, sinus bradycardia, and atrioventricular block, which were promptly reversed with nitroglycerin. There was no mortality or development of myocardial infarction.
Limited data are available on hyperventilation, although a recent meta-analysis of studies encompassing over 1,000 patients studied with 12-lead ECG, echocardiography, or myocardial perfusion radionuclide imaging reported no major complications in properly selected patients [29].
The stepwise approach to the diagnosis of CAS is summarized in Figure 1.
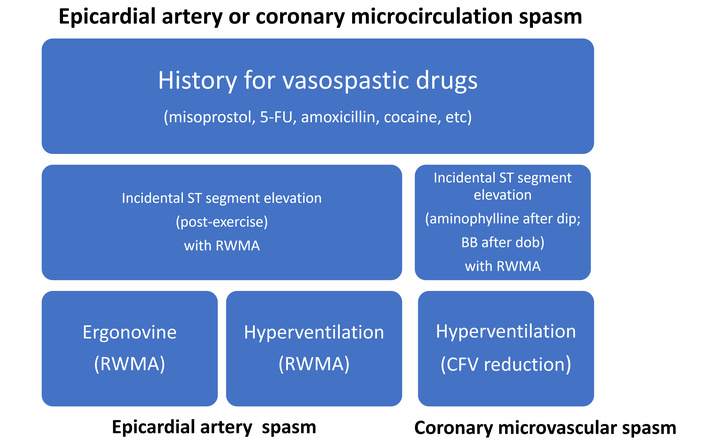
Three steps for CAS detection: clinical history, functional stress testing, and targeted testing. Epicardial CAS can be found incidentally during exercise or pharmacological testing for the diagnosis of chronic coronary syndromes with functional testing, and these findings are diagnostic of CAS and do not require further diagnostic testing: CAS has been proven. The progression to targeted noninvasive testing of coronary vasospasm with either ergonovine or hyperventilation is required only when there is a clinical suspicion, and the evidence of a CAS is still missing. BB: β-blocker; 5-FU: 5-fluorouracil; CAS: coronary artery spasm; CFV: coronary flow velocity; RWMA: regional wall motion abnormality.
Although lifestyle modifications and behavioural and pharmacological control of modifiable risk factors are crucial in patients with obstructive CAD, these have limited implications for the control of CAS and the prevention of CAS-related cardiac events. Clinical studies have shown that only smoking and the use of alcohol show a significant association with the occurrence of CAS. Indeed, smoking and alcohol use have also occasionally been associated with triggering CAS. Thus, avoiding smoking and alcohol abuse should be recommended to all patients with CAS [3, 9].
The diagnosis of coronary vasospasm identifies a specific endotype within the heterogeneous INOCA (ischemia with non-obstructive coronary arteries) population, representing a directly treatable target. Current European Society of Cardiology guidelines [2] recommend calcium channel blockers as first-line therapy, followed by nitrates, while advising against β-blockers. These may paradoxically exacerbate vasospasm by allowing unopposed α-adrenergic vasoconstriction without the counterbalancing β1-mediated vasodilation. Importantly, stenting of coronary stenoses is not recommended, as symptom recurrence frequently occurs even after technically successful percutaneous coronary intervention (PCI), suggesting that the underlying vasomotor dysfunction rather than fixed stenosis drives symptoms.
CAS should always be considered in the presence of a suggestive clinical history and a mismatch between coronary anatomy and functional findings. The diagnosis is particularly likely when myocardial ischemia is precipitated by drugs with known vasospastic potential, including recreational substances such as cocaine, or medications used in oncology (e.g., capecitabine), psychiatry (e.g., propranolol), neurology (e.g., triptans), pneumonology (e.g., methacholine), rheumatology (e.g., aspirin), or gynecology (e.g., misoprostol). Even when not specifically sought, CAS may be incidentally detected during evaluation for CAD, particularly in patients exhibiting ST-segment elevation during the recovery phase—after a negative peak stress—of exercise, dipyridamole, or dobutamine stress testing.
If CAS remains clinically suspected despite negative findings on ambulatory ECG monitoring and standard stress testing, a provocative test in ANOCA with ergonovine or hyperventilation may be performed in the echocardiography laboratory. In this context, the induction of an RWMA serves as both a diagnostic endpoint and a surrogate marker of epicardial CAS.
In patients with persistent symptoms and ANOCA, where the diagnosis remains elusive, a multi-stress, multi-marker approach can help differentiate microvascular etiologies. An abnormal CFV reserve < 2.0 during vasodilator stress indicates an underlying structural (organic) coronary microvascular disease, whereas a hyperventilation test with paradoxical reduction in CFV despite an increase in rate-pressure product is indicative of vasospastic coronary microvascular disease.
Overall, the proposed noninvasive approach holds significant potential to reduce costs, risks, and environmental impact compared to a catheterization lab-driven strategy, which carries inherent harms such as invasive procedures, coronary artery manipulation, intracoronary drug injection, nephrotoxic iodine contrast administration, and radiation exposure. Furthermore, the noninvasive method expands the diagnostic domain by overcoming the logistical and practical constraints of invasive techniques while enabling serial patient assessments to monitor therapeutic effects and disease progression. Large-scale multicenter validation is now required before clinical adoption, and this is currently underway as part of the SESPASM subproject within the Stress Echo 2030 study.
ANOCA: angina with non-obstructive coronary arteries
CAD: coronary artery disease
CAS: coronary artery spasm
CFV: coronary flow velocity
ECG: electrocardiogram
RWMA: regional wall motion abnormality
AV, GÁ: Conceptualization, Writing—original draft, Writing—review & editing. Both authors read and approved the submitted version.
Albert Varga, who is the Editorial Board Member of Exploration of Cardiology, had no involvement in the decision-making or review process of this manuscript. The other author declares that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 562
Download: 28
Times Cited: 0
