Affiliation:
1Diffuse Laboratory for Interdisciplinary Research Applied to Medicine (DREAM), ASL-LE/University of Salento, 73100 Lecce, Italy
2Cardionair-Research in Telecardiology Center, 73048 Nardò, Italy
3Department of Cardiovascular Diseases, University of L’Aquila, 67100 L’Aquila, Italy
Email: marcellocostantini9@gmail.com
ORCID: https://orcid.org/0000-0002-7330-7869
Affiliation:
4Department of Cardiovascular Diseases, University of Insubria, 21100 Varese, Italy
ORCID: https://orcid.org/0000-0002-2321-2319
Affiliation:
3Department of Cardiovascular Diseases, University of L’Aquila, 67100 L’Aquila, Italy
ORCID: https://orcid.org/0000-0002-9669-1330
Affiliation:
3Department of Cardiovascular Diseases, University of L’Aquila, 67100 L’Aquila, Italy
5Cardiovascular Diseases Unit, “San Carlo di Nancy” Hospital—GVM, 00118 Roma, Italy
ORCID: https://orcid.org/0000-0003-3325-6910
Affiliation:
8Academia Nacional de Medicina, Buenos Aires 7311, Argentina
ORCID: https://orcid.org/0000-0003-4800-0678
Explor Cardiol. 2026;4:1012102 DOI: https://doi.org/10.37349/ec.2026.1012102
Received: January 20, 2026 Accepted: February 24, 2026 Published: March 23, 2026
Academic Editor: David S.H Bell, University of Alabama, USA
Aim: Cardiac memory is classically expressed as persistent T wave changes following restoration of normal ventricular activation. Its manifestations in idiopathic intermittent left bundle branch block (ILBBB) remain incompletely characterized. The aim of this report is to describe a distinct electrocardiographic phenotype potentially related to cardiac memory.
Methods: We retrospectively analyzed a homogeneous series of 14 patients with idiopathic, rate-dependent ILBBB, absence of demonstrable structural heart disease, homophasic T waves in leads V5–V6 during left bundle branch block (LBBB), and pseudo-ischemic T wave inversion in the right precordial leads during phases of normal conduction.
Results: In all patients, memory-related negative T waves were observed during normal conduction, while concordant (homophasic) T waves were consistently present during LBBB. A close relationship emerged between the width of the intraventricular conduction zone and the severity of repolarization abnormalities: narrower conduction zones were associated with deeper T wave inversion during normal conduction. During follow-up, most patients progressed to permanent LBBB, without documented major arrhythmic events.
Conclusions: These findings describe a distinctive and reproducible electrocardiographic phenotype within idiopathic ILBBB, likely related to cardiac memory. This pattern, provisionally designated homophasic idiopathic intermittent left bundle branch block and cardiac memory (OIL-CAME), warrants further confirmation in larger prospective studies.
Nowadays, worldwide, Xylella fastidiosa is widely recognized as responsible for the devastation of large territories through the destruction of millions of centuries-old olive trees, as occurred in Salento (Southeastern Italy). Over several decades, the memory of extraordinary landscapes and of an olive oil of unparalleled quality has almost been lost. With a metaphorical leap, from environmental memory to cardiac physiology, the concept of cardiac memory (CM) has become increasingly familiar to cardiologists. CM generally refers to persistent T wave changes—most commonly T wave inversion—that appear after a period of abnormal ventricular activation, once normal ventricular depolarisation is restored [1]. This phenomenon was first described in 1969 by Chatterjee et al. [2], who observed T wave inversion following transient periods of artificial ventricular pacing. Nearly a decade later, Denes et al. [3] reported deep T waves inversion in the right precordial leads during normal conduction in patients with intermittent left bundle branch block (ILBBB). However, the mechanisms and clinical implications of these and other electrocardiographic findings associated with ILBBB remained poorly understood until Rosenbaum and colleagues [4] introduced the concept of CM in their seminal work. In that landmark study, two fundamental principles were established: 1) during normal conduction, the direction of the T wave reflects (remembers) the direction of the QRS complex during a preceding period of abnormal ventricular activation; and 2) both the amplitude and persistence of memory-related T wave abnormalities are proportional to the duration of the preceding abnormal activation [4]. This conceptual framework clarified several previously unexplained electrocardiographic phenomena and subsequently became the subject of extensive investigation [5]. Despite these significant advances, important aspects of ILBBB—particularly when idiopathic—remain unresolved. These include its complex electrocardiographic manifestations, its relationship with CM, its clinical significance, and its long-term evolution. In particular, the interpretation of repolarization abnormalities associated with ILBBB continues to pose diagnostic challenges, as memory-related changes may closely mimic a primary ischemic or cardiomyopathic pattern. In this retrospective study, drawing on more than four decades of clinical and electrophysiological experience, we aim to contribute further insight into CM associated with idiopathic ILBBB. Specifically, we describe a distinctive electrocardiographic pattern characterized by memory-related T wave inversion during normal conduction and concordant (homophasic) T waves during left bundle branch block (LBBB), which we propose represents a recognizable electrocardiographic phenotype.
From a total cohort of 104 patients with intermittent intraventricular conduction disturbances, retrospectively collected by the first author at the Cardiology Departments of Pavia (1973–1986), Tortona (1987–1996), and Galatina (1997–2020), a subgroup of 14 patients was identified based on homogeneous clinical and electrocardiographic characteristics. The inclusion criteria were as follows:
presence of rate-dependent ILBBB [6];
positive T waves in leads V5–V6 during LBBB, corresponding to concordance between QRS polarity and T wave polarity in the lateral precordial leads [6], so-called homophasic LBBB [7];
negative, symmetrical, pseudo-ischemic T waves in the right precordial leads during phases of normal conduction, with a minimum amplitude of 0.1 mV [8];
referral for routine evaluation or atypical chest pain;
absence of demonstrable significant structural heart disease.
All 14 patients were hospitalized and, after informed consent, underwent a comprehensive diagnostic evaluation including baseline electrocardiographic recordings, transthoracic echocardiography, exercise stress testing, electrophysiological study, hemodynamic assessment, and coronary angiography.
During hospitalization, all patients underwent repeated multichannel electrocardiographic recordings over consecutive days during spontaneous and induced variations in sinus rate. Sinus tachycardia was induced by hyperventilation and inhalation of amyl nitrite, whereas bradycardia was obtained through vagal maneuvers, including carotid sinus massage. These maneuvers allowed day-to-day definition of the temporal zones of conduction and block along the left bundle branch. Particular attention was paid to identifying any symptoms temporally related to transitions between normal conduction and LBBB, and vice versa. Homophasic (concordant) T waves during LBBB were defined as T waves with polarity concordant to that of the QRS complex in leads V5–V6, with a minimum amplitude of ≥ 0.1 mV.
Pseudo-ischemic T wave inversion during normal conduction was defined as symmetrical negative T waves in the right precordial leads (V1–V3/V4), temporally related to preceding phases of abnormal ventricular activation and reversible with changes in intraventricular conduction, in the absence of clinical, biochemical, or imaging evidence of myocardial ischemia.
Endocavitary electrophysiological studies were performed after informed consent, in wash out from any cardioactive medications. Two electrode catheters were introduced:
a bipolar 6F catheter inserted via the right femoral vein and positioned across the tricuspid valve to record the His bundle’s potential;
a tetrapolar 6F catheter introduced through the right basilic vein and positioned at the junction between the superior vena cava and lateral wall of the right atrium, used for atrial stimulation and recording of atrial electrical activity.
Atrial pacing at progressively increasing and decreasing rates was performed, thus defining the temporal zones of block and conduction through the left bundle branch. When necessary, bradycardia was induced by vagal stimulation maneuvers. The tachycardia-dependent block zone was further explored using premature atrial stimulation during sinus rhythm or following eight-beat pacing trains at cycle lengths corresponding to a rate of 70–100 beats per minute.
Hemodynamic evaluation and coronary angiography study were performed using standard catheterization techniques. All patients underwent transthoracic echocardiography, including M-mode, two-dimensional imaging, Doppler, and color Doppler evaluation, in accordance with established guidelines. Exercise stress testing was performed in all patients using a cycle ergometer and a standard incremental workload protocol.
Clinical and electrocardiographic follow-up was available in 11 of the 14 patients, with a duration ranging from 12 to 65 months. At each follow-up visit, heart rate modulation was again induced using the same non-invasive methods described above, with multichannel electrocardiographic recording to reassess intraventricular conduction behaviour.
All electrocardiographic and electrophysiological data were jointly analyzed by the authors.
Table 1 summarizes the main clinical and electrocardiographic characteristics of the 14 patients included in the study. Ages ranged from 27 to 58 years (mean 46 ± 8.4 years); eight were male and six female. Ten patients were referred for atypical chest pain, usually described as precordial and stabbing in nature, whereas four were asymptomatic and underwent evaluation for a routine check-up. At first observation, the initial electrocardiographic diagnosis was ILBBB associated with suspected myocardial ischemia in six patients, isolated myocardial ischemia in five, and ILBBB associated with anteroseptal myocardial infarction in three. ILBBB was exclusively tachycardia-dependent in eight patients, whereas in the remaining six, it was both tachycardia- and bradycardia-dependent. In all 14 patients, homophasic T waves were observed during LBBB, defined as concordance between the polarity of the QRS complex and that of the T wave. In addition, all patients exhibited negative, pseudo-ischemic T waves in the right precordial leads during phases of normal conduction. In six patients (five with exclusively tachycardia-dependent ILBBB and one with both tachycardia and bradycardia-dependent ILBBB), electrophysiological study demonstrated the presence of a narrow conduction zone interrupting the tachycardia-dependent block zone. This conduction zone lasted no more than 150 ms and was consistent with supernormal conduction (Figure 1). In all these six cases, atrial pacing readily produced 2:1 conduction through the left bundle branch.
The main clinical and electrocardiographic characteristics of the 14 patients included in the study.
| Category | Characteristics |
|---|---|
| Case number | 14 |
| Sex | 8 males, 6 females |
| Age | 27–58 years (mean 46 ± 8.4 years) |
| Symptoms at presentation | -Atypical angina: 10-None (check up): 4 |
| Initial ECG diagnosis | -ILBBB and myocardial ischemia: 6-Myocardial ischemia: 5-ILBBB and myocardial infarction: 3 |
| Electrocardiographic characteristics of ILBBB | -Homophasic and tachycardia dependent: 3-Homophasic and tachycardia dependent with supernormal conduction during EPS: 5-Homophasic and tachycardia and bradycardia-dependent: 5-Homophasic and tachycardia and bradycardia-dependent with supernormal conduction during EPS: 1 |
ILBBB: intermittent left bundle branch block; EPS: electrophysiological study.
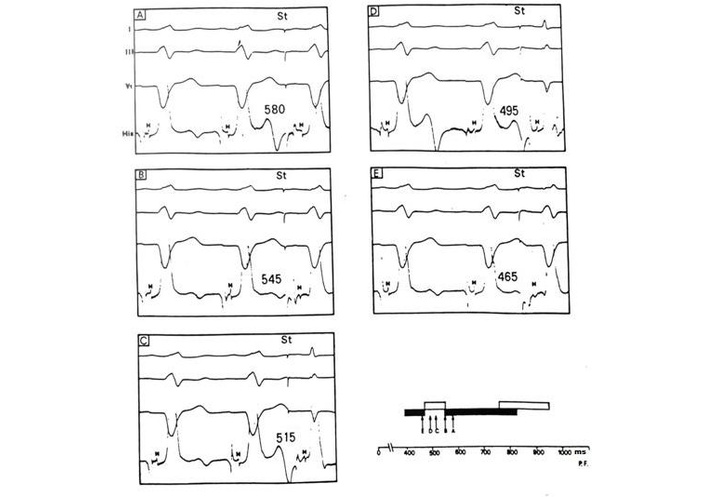
Premature atrial stimulation during sinus rhythm in a case with tachycardia-dependent left bundle branch block. The bundle branch block is evident during sinus rhythm and disappears when the cardiac cycle exceeds 840 ms during vagal stimulation, as shown in the diagram in the lower right, where black bars indicate cycles conducted with left bundle branch block and white bars indicate normally conducted cycles. Premature atrial stimulation (panels A–E) shows that the tachycardia-dependent block zone is interrupted by a narrow window of supernormal conduction (panels C and D). H: potential of His bundle; St: premature atrial stimulation during sinus rhythm.
Figure 2 illustrates a representative case from the study population. At the first evaluation, the patient was diagnosed with ILBBB and suspected myocardial ischemia. The electrocardiogram shows: 1) ischemic-like negative T waves in the right precordial leads up to V4, concordant with the negative QRS complexes in the same leads during LBBB; 2) concordance between QRS and T waves in the left precordial leads during LBBB, consistent with homophasic LBBB.
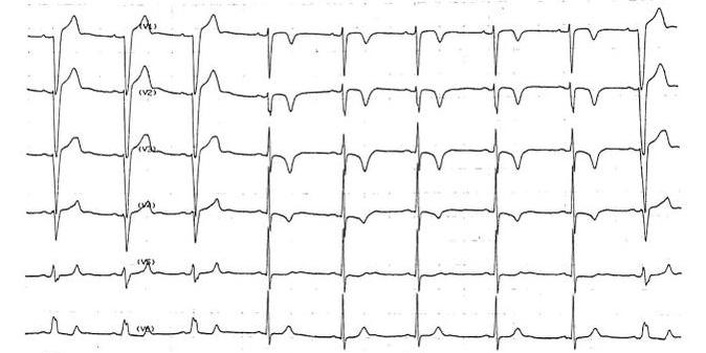
The figure illustrates a representative case from the study population, showing intermittent tachycardia-dependent left bundle branch block. In the left portion of the figure, intraventricular conduction is characterized by left bundle branch block; note the concordant T wave morphology (lead V6) in the presence of left bundle block. When the cardiac cycle lengthens from 560 to 640 ms, left bundle branch conduction resumes. In the presence of normal conduction, symmetric negative T waves are evident in the precordial leads until V4. In the right portion of the figure, shortening of the cardiac cycle from 600 to 560 ms is associated with reappearance of left bundle branch block. For further details, see the text.
Figure 3 depicts the relationship between intraventricular conduction patterns and cardiac cycle length in the 14 patients, based on a representative recording period for each case. Black bars indicate cycles conducted with complete LBBB, white bars indicate cycles with normal conduction, and dashed bars (cases 10, 12, and 13) represent cycles with incomplete LBBB [6]. A partial overlap between conduction and block zones within the left bundle branch was observed. As noted above, eight patients exhibited exclusively tachycardia-dependent LBBB, whereas six showed both tachycardia- and bradycardia-dependent conduction disturbances. In only two of the eight patients with apparently isolated tachycardia-dependent LBBB were the diastolic pauses sufficiently long to reliably exclude the presence of a bradycardia-dependent block. In the remaining cases, the pauses obtained were too short to definitively rule out bradycardia-dependent LBBB.
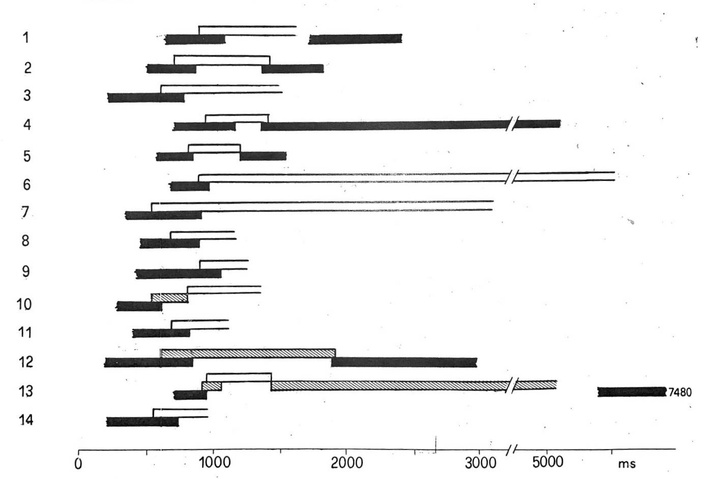
The diagram depicts the relationship between intraventricular conduction and cardiac cycle length in 14 patients. Black bars indicate cycles conducted with left bundle branch block, white bars indicate normally conducted cycles, and dashed bars represent cycles with incomplete left bundle branch block. For further details, see the text.
A strong relationship emerged between the width of the conduction zone and the degree of repolarization abnormalities in the right precordial leads. Specifically, the narrower the conduction zone, the more pronounced was T wave inversion during normal conduction, and vice versa. This singular variability is also observable in the same subject, in relation to the variation over time of the areas of blockage and conductivity across the left bundle branch.
Coronary angiography revealed no significant coronary artery disease in any patient. Right and left ventriculography demonstrated normal chamber size and morphology, with preserved and homogeneous segmental contraction in all cases.
Transthoracic echocardiography showed mild left ventricular hypertrophy in three patients; no other relevant abnormalities were detected. Exercise stress testing did not reveal clinical or electrocardiographic evidence of inducible myocardial ischemia in any patient. Among the ten patients presenting with chest pain, no temporal relationship was observed between the occurrence of symptoms and the presence or absence of LBBB.
Clinical and electrocardiographic follow-up was available only in 11 of the 14 patients, with a duration ranging from 12 to 65 months. Throughout follow-up, variability in temporal zones of conduction and block within the left bundle branch was documented. Figure 4 illustrates a representative example of this variability (case 2 in Figure 3) showing a relatively wide conduction zone between tachycardia- and bradycardia-dependent block zones at the initial evaluation, which narrows in a non-progressive way and eventually disappeared during subsequent follow-up examinations.
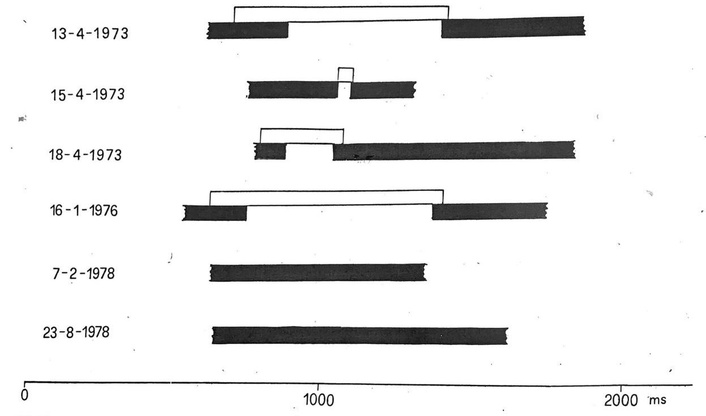
This figure depicts the variability in the temporal zones of conduction and block within the left bundle branch during follow-up. Black bars indicate cycles conducted with left bundle branch block and white bars indicate normally conducted cycles. For further details, see the text.
For analytical purposes, the 11 patients with follow-up were divided into two groups (Table 2): those followed for 12 to 30 months (n = 6) and those followed for 31 to 65 months (n = 5). Among patients with longer follow-up, all but one progressed to permanent LBBB. In the shorter follow-up group, LBBB became permanent in one patient, remained intermittent in three, and was no longer detectable in two. No definite arrhythmic events or progressive cardiac functional deterioration were observed in ten patients, whereas one patient developed echocardiographic features suggestive of dilated cardiomyopathy.
Follow-up in 11/14 patients.
| Category | Characteristics | |
|---|---|---|
| Duration of follow-up | ≤ 30 months (12–30) | ≥ 31 months (31–65) |
| Patient number | 6 | 5 |
| Clinical picture | Unchanged in all | -Unchanged in 4-Dilated cardiomyopathy in 1 |
| Characteristics of left bundle branch block | -Stable in 1-Intermittent in 3-No more evident in 2 | -Stable in 4-Intermittent in 1 |
In this study, we report a case series of 14 patients with idiopathic ILBBB characterized by memory-related repolarization abnormalities, with the aim of better defining a distinctive electrocardiographic behavior. Accordingly, the present discussion focuses specifically on these aspects and does not address other electrophysiological features of intermittent tachycardia-dependent (phase 3) and/or bradycardia-dependent (phase 4) bundle branch block, such as mechanisms of intermittence, supernormal conduction, alternating conduction, or overlap between conduction and block zones which have been extensively examined in previous reports, including in similar patient populations [9–11]. It should be briefly noted here that although, based on our experience, the presence or absence of supernormal conduction does not appear to influence the electrocardiographic phenotype described, this possibility cannot be completely ruled out and warrants further investigation.
Transient alterations in the sequence of ventricular activation are well known to induce apparent worsening or, in some circumstances, apparent improvement of ventricular repolarization. Such pseudo-worsening phenomena have been described in the setting of rate-dependent bundle branch block, rate-dependent fascicular block, wide-QRS tachycardia, ventricular ectopy, intermittent ventricular pre-excitation, ablation of accessory atrioventricular (AV) pathway, and temporary or intermittent ventricular pacing [1]. In all these conditions, a change in the direction of ventricular activation may give rise to CM-related repolarization changes, manifesting not only as the classic inverted T waves, but also as positive T waves or pseudo-normalization of previously abnormal T waves [1]. Among the various manifestations of CM, the occurrence of deep negative T waves during normal conduction in patients with ILBBB is of particular clinical relevance. In this setting, memory-induced T wave inversion in the right precordial leads may closely mimic “primary repolarization abnormalities” [8], such as those observed in Wellens’ syndrome, which reflects transient proximal left anterior descending coronary artery occlusion and requires urgent intervention [12]. Similarly, right precordial T waves inversion may also be an expression of underlying myocardial disease, including arrhythmogenic cardiomyopathy [13]. Therefore, distinguishing memory-related repolarization changes from primary pathological T wave abnormalities is of crucial diagnostic importance. Beyond the clinical context, an important electrocardiographic clue supporting a memory-related mechanism is the concordance between the polarity of the T waves during normal conduction and the polarity of the QRS complex during LBBB. When such concordance is present, a memory phenomenon is strongly suggested, whereas a lack of coherence between these polarities should raise suspicion of a primary repolarization abnormality [14]. In this regard, our series consistently demonstrated memory-related negative T waves during normal conduction in all patients.
In addition to this well-recognized manifestation of CM, our study highlights another distinctive and previously underappreciated electrocardiographic feature: the presence of homophasic T waves during LBBB. This finding clearly differentiates our patients from typical cases of LBBB, in which heterophasism—namely, discordance between QRS and T wave polarity—is usually observed [6, 15]. Although for many years CM was thought to become evident only after restoration of normal QRS duration, accumulating evidence suggests that memory-related changes in repolarization may, under certain conditions, become apparent even in the presence of abnormal depolarization. From a pathophysiological perspective, the surface ECG represents the summation of the cardiac myocyte action potentials, which display regional heterogeneity in duration. An inverse relationship between activation time and action potential duration (APD) is well established: myocardial regions activated early exhibit longer APD, whereas later-activated regions show progressively shorter durations [16, 17]. This relationship, attributed to electrotonic interactions through gap junctions [17–19], intrinsic heterogeneity of APD [17], and ephaptic coupling [20], allows ventricular repolarization to proceed in a direction opposite to that of depolarization [1, 4], resulting in an upright T wave on the normal ECG.
Any alteration in the activation sequence produces immediate secondary changes in repolarization, typically oriented opposite to the direction of abnormal depolarization, thus explaining the usual discordance between QRS and T waves during LBBB [1, 4]. However, during sustained abnormal activation, additional non-secondary repolarization changes gradually develop. These changes remain masked by secondary repolarization abnormalities and become evident only when normal ventricular activation is restored, constituting the classical CM phenomenon [1, 4]. Importantly, experimental and clinical data indicate that, in some circumstances, memory-related repolarization changes may overcome secondary changes and manifest even during ongoing abnormal depolarization [21]. As proposed by Shvilkin and colleagues [21, 22], memory-related recovery changes may supersede secondary repolarization changes in the presence of wide QRS complexes. This mechanism offers a plausible explanation for the homophasic T wave observed during LBBB in our patients. Why this unusual phenomenon was consistently present in our series remains uncertain; however, it is noteworthy that none of our patients had demonstrable structural heart disease at the time of first observation. Although LBBB is not traditionally regarded as a heritable disorder, several studies have implicated genetic abnormalities involving ion channel genes (including SCN5A, PRKAG2, and HCN4), gap junction proteins, desmosomal components, and cardiac transcription factors in the development of bundle branch block [23–28]. In particular, SCN5A mutations have been shown to produce progressive impairment of intraventricular conduction [27], consistent with idiopathic progressive cardiac conduction disease originally described by Lenegre and Moreau [29, 30]. It cannot be excluded that, in idiopathic LBBB of presumed genetic origin, non-secondary repolarization changes may overlap with secondary ones, thereby accounting for both T wave homophasism during LBBB and the T wave inversion during normal conduction. However, it is worth highlighting here some subtle differences between Lenegre disease and the phenotype we described. Scientific evidence suggests that the pathophysiology of the hereditary Lenegre disease is related to SCN5A and involves haploinsufficiency of the cardiac Na+ channel gene, together with an additional, yet unidentified, mechanism that alters cardiac conduction in an age-dependent manner [30]. Classically, in Lenegre disease, a chronic conduction defect progressively develops over decades, leading to complete AV block [29, 30]. Surprisingly, in our case series, we never observed AV block, nor any involvement of the right bundle branch in the disease, despite the fact that, theoretically, the right bundle branch should be more vulnerable than the left [23].
Regarding clinical evolution, the development of echocardiographic features suggestive of dilated cardiomyopathy in one patient during long-term follow-up deserves consideration. Experimental and clinical studies [23] have demonstrated that LBBB is associated with electromechanical dyssynchrony, which may promote adverse ventricular remodeling over time [23, 31, 32]. Accordingly, patients with LBBB and initially preserved ventricular function may subsequently develop systolic dysfunction [23, 31], although it cannot be excluded that, in our specific case, the electrocardiographic phenotype described was initially an expression of a subclinical cardiomyopathy.
Finally, our observations do not support a consistent relationship between the occurrence of chest pain and the presence of LBBB, in contrast to reports describing the so-called “painful LBBB syndrome” [33, 34]. In our series, no temporal association between symptoms and conduction changes was observed.
At a broader level, the mechanisms underlying CM likely extend beyond purely electrotonic interactions. Rosen and colleagues [5, 35, 36] have suggested that CM reflects a form of electrical remodeling induced by altered activation patterns, involving ion channels, proteins, second messengers, and gene expression, and displaying features reminiscent of short- and long-term memory in the neural system. CM is therefore increasingly regarded as a universal adaptive property of the heart, reflecting dynamic adjustment of repolarization to changes in activation sequence [22]. Whether CM or the associated pseudo-primary repolarization abnormalities have clinical implications beyond their diagnostic relevance remains to be fully clarified. Some studies have suggested a potential proarrhythmic role of CM under specific conditions [37, 38], underscoring the need for further investigation. Additional studies are warranted to confirm our findings and to better define the characteristics and clinical significance of the electrocardiographic phenotype described here, which we have designated with the acronym homophasic idiopathic intermittent left bundle branch block and cardiac memory (OIL-CAME). According to this spirit, we hope that both this electrocardiographic pattern and the centuries-old olive trees of Salento will not remain merely a faint memory.
Future studies integrating high-density body surface electrocardiographic imaging, intracardiac electrophysiological mapping, cardiac magnetic resonance with tissue characterization, and comprehensive genetic testing will be essential to confirm the mechanisms underlying this electrocardiographic phenotype. Such approaches may help distinguish pure electrical remodeling related to CM from subtle structural or genetic substrates potentially involved in idiopathic ILBBB.
This study describes a homogeneous series of patients with idiopathic ILBBB characterized by two consistent and interrelated electrocardiographic features: memory-induced negative T waves during normal conduction and homophasic T waves during LBBB. Therefore, in addition to the well-recognized manifestation of CM represented by negative T waves during normal conduction, our study highlights a second, distinctive electrocardiographic feature: the consistent presence of homophasic T waves during LBBB. This finding clearly differentiates our patients from typical LBBB, in which secondary repolarization abnormalities usually result in discordant QRS-T polarity. Although CM was originally thought to become evident only after restoration of normal ventricular activation, accumulating experimental and clinical evidence suggests that, under specific conditions, memory-related repolarization changes may partially or completely overcome secondary repolarization abnormalities and become apparent even during ongoing abnormal depolarization. Together, these findings characterize a novel electrocardiographic pattern, provisionally designated OIL-CAME, which may represent a distinct electrophysiological entity warranting further prospective study. Therefore, further studies are needed to confirm these observations, clarify the underlying mechanisms, and determine the clinical and prognostic implications of this electrocardiographic pattern.
One important limitation of the present study concerns the proposed mechanistic interpretation of T wave homophasism in idiopathic ILBBB. Our hypothesis assumes that CM-related repolarization changes may outweigh the secondary repolarization abnormalities classically associated with LBBB. According to the concept of CM as an accumulation-dependent phenomenon [4], the earliest beats exhibiting LBBB would theoretically be expected to show discordant repolarization, with progressive development of concordant T waves as memory develops. However, in this case series, it was not possible to record the very first beats during the onset of LBBB. Consequently, we could not directly assess whether initial repolarization in lateral precordial leads (particularly V6) was discordant or concordant, nor could we observe the temporal evolution of T wave morphology during the development of CM. This limitation prevents direct confirmation of the proposed sequence of repolarization changes and underscores the need for future studies.
APD: action potential duration
AV: atrioventricular
CM: cardiac memory
EPS: electrophysiological study
ILBBB: intermittent left bundle branch block
LBBB: left bundle branch block
OIL-CAME: homophasic idiopathic intermittent left bundle branch block and cardiac memory
We thank Mauricio Rosenbaum and Pablo Chiale, whose work truly inspired this study; the article is dedicated to their memory. We also thank Luigi Tavazzi and Marcello Chimienti for their support and guidance.
MC: Writing—original draft, Writing—review & editing, Conceptualization, Data curation. JASU: Data curation, Supervision, Validation. LC: Conceptualization, Formal analysis, Supervision. LS: Conceptualization, Methodology. AS: Software, Validation. MP: Formal analysis, Data curation. ATR: Writing—review & editing, Supervision. MVE: Supervision, Validation, Visualization. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
This is a retrospective study spanning more than 40 years, based on data collected and archived at multiple centers; according to local regulation and to the best of our knowledge, specific ethical committee approval was not required.
This study is retrospective and based on anonymized archived data collection over more than 40 years. At the time of data collection, patients had provided consent for clinical evaluation. Given the historical nature of the dataset, obtaining additional informed consent for the present analysis was not feasible and was therefore waived.
Not applicable.
The datasets generated and/or analyzed during the current study are not publicly available due to privacy and ethical restrictions, but are available from the corresponding author upon reasonable request and with permission of the participating institutions.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 669
Download: 51
Times Cited: 0
