Affiliation:
Division of Respirology, Department of Medicine, Cumming School of Medicine, University of Calgary, Calgary, AB T2N 2T8, Canada
Email: Amanda.Stanton1@ucalgary.ca
ORCID: https://orcid.org/0009-0004-8892-2938
Affiliation:
Division of Respirology, Department of Medicine, Cumming School of Medicine, University of Calgary, Calgary, AB T2N 2T8, Canada
ORCID: https://orcid.org/0000-0002-6682-4155
Explor Asthma Allergy. 2026;4:1009124 DOI: https://doi.org/10.37349/eaa.2026.1009124
Received: December 22, 2025 Accepted: March 24, 2026 Published: April 21, 2026
Academic Editor: Erminia Ridolo, University of Parma, Italy
The article belongs to the special issue The Complex Interactions Between Lifestyles and Asthma
Obesity is a determinant of the risk of developing various diseases, including asthma. It can also contribute to asthma severity. Obvious determinants of the risk of developing obesity and conditions associated with obesity are the diet an individual consumes and their energy expenditure, as determined by activity. Therefore, diet and exercise are important non-pharmacological components in the management and prevention of many diseases. Several individual elements in diet, including certain fatty acids and vitamins, as well as types of diets, notably the Mediterranean diet, have been studied in asthmatic patients, but the literature is not consistent. This review explores the relationship between asthma and obesity, exercise, and multiple dietary components and regimens, including the Mediterranean diet; polyunsaturated fats; vitamins A, C, D, and E; flavonoids; probiotics; and sodium intake in the published randomized clinical trials. Overall, the data have many shortcomings, but there is no single component of diet that is consistently associated with improved asthma outcomes, nor any component found to be clearly harmful. However, a diet that helps an individual lose weight may indirectly improve their lung function and asthma control, even if the diet itself does not impact asthma outcomes. Exercise, now known to be safe and widely recommended in asthma, has various forms. This review looked at meta-analyses, as well as recently published data addressing this question, categorizing exercise as aerobic activity, pulmonary rehabilitation, and yoga. The most evidence for benefit is for aerobic exercise, but yoga also has potential for modest improvement in asthma symptoms. There is conflicting data as to whether supervised exercise programs are superior to unsupervised physical activity. Overall, exercise is helpful in asthma, but it is still unclear how much exercise should be done, and this should be tailored to each individual.
Obesity, diet, and exercise are important determinants of health and disease. ‘You are what you eat’ is an old adage. Obesity increases the risk of numerous diseases including diabetes, cardiovascular disease, some malignancies, and recently, it has been recognized that obesity increases the risk of asthma and asthma severity. Diet affects the risks of developing various diseases, including diabetes, obesity, inflammatory bowel disease, rheumatoid arthritis, cardiovascular disease, and cancer [1–3]. The association with lung diseases is not well established [4]. The overwhelming risks of other factors, including exposure to cigarette smoke, allergens, respiratory infections, and air quality may obscure the impact of diet.
Exercise was once felt to be possibly harmful for asthmatics due to the risk of precipitating asthmatic symptoms, but it is now widely accepted to be beneficial, with significant supporting data. In July 2025, Liu et al. [5] reviewed systematic reviews from 2000 to 2024 and found that physical activity significantly improved Asthma Quality of Life Questionnaire (AQLQ), Asthma Control Questionnaire (ACQ), forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC), peak expiratory flow (PEF), six-minute walk distance (6MWD), and maximal oxygen uptake (VO2max). Regular exercise is recommended for the general population, including patients with a myriad of medical conditions, such as cardiovascular disease, metabolic syndrome, and respiratory conditions, including asthma. Unfortunately, despite this, asthmatic patients are less active on average than their non-asthmatic counterparts [6].
This review serves to present and summarize the data connecting obesity, diet, and exercise with outcomes in patients with asthma to highlight how these non-pharmacological aspects of lifestyle could contribute to overall asthma management principles.
The aim was to identify randomized controlled trials (RCTs) of various dietary nutrients in asthma and the risk of developing asthma. We did a preliminary PubMed search for meta-analysis to serve as background. A PubMed search using the MeSH terms ‘diet’ and ‘asthma’ was done to determine which nutrient supplements were the ones that had been most commonly studied in asthma. Polyunsaturated fatty acids (PUFAs), vitamin D, Mediterranean diet, the antioxidant vitamins A, C, and E, flavonoids, probiotics, and minerals selenium, zinc, and dietary sodium were the nutrients identified. The PubMed database was queried with the following terms: polyunsaturated fatty acids and asthma, polyunsaturated fatty acid supplementation and asthma, vitamin D and asthma, and vitamin D supplementation and asthma (see Figure 1). Moreover, the databases were also queried with “Mediterranean diet” and asthma, flavonoids and asthma. The PubMed database was queried for omega-3 (n-3) fatty acids and asthma, omega-6 (n-6) fatty acids and asthma, vitamin A and asthma, vitamin C and asthma, vitamin E and asthma, dietary sodium and asthma, probiotics and asthma, selenium and asthma, zinc and asthma, and obesity and asthma (see Figure 2). We did not place limitations on the size of the subject cohort, dose of supplement, or duration of therapy because of the paucity of studies of the various interventions.
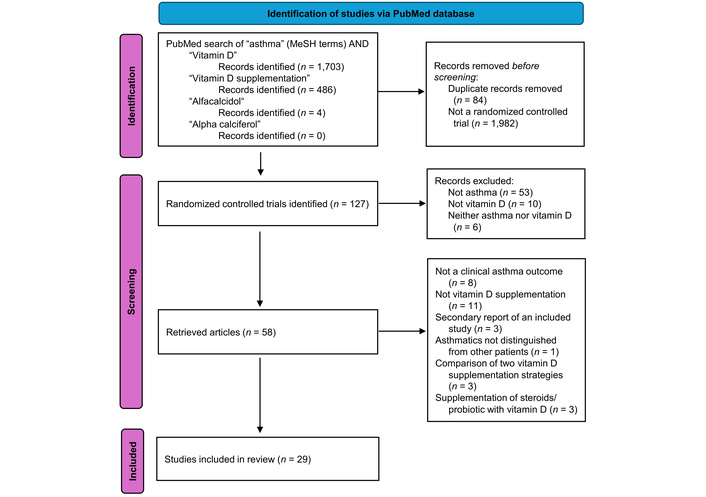
Flowchart of PubMed randomized controlled trials of vitamin D supplementation in asthma. Adapted from [7]. © 2021 The Author(s). Licensed under CC BY 4.0.
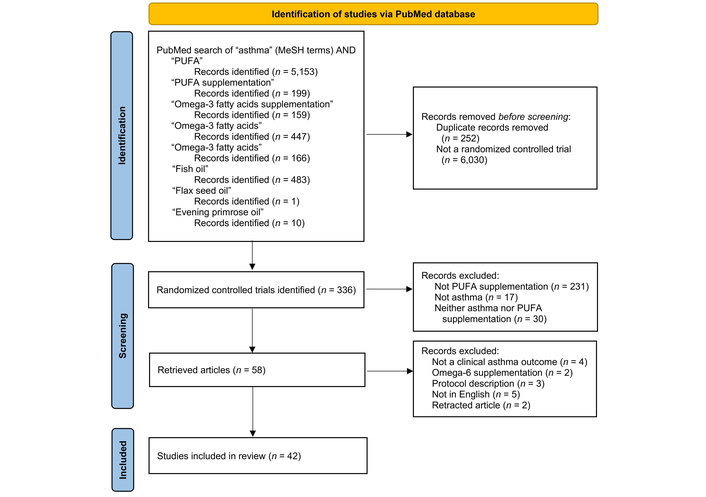
Flowchart of PubMed randomized controlled trials of PUFA supplementation in asthma. PUFA: polyunsaturated fatty acid. Adapted from [7]. © 2021 The Author(s). Licensed under CC BY 4.0.
A search of PubMed and the Cochrane databases was conducted using the MeSH terms “exercise” and “asthma.” For the former, there were 2,827 articles retrieved. For this reason, only meta-analyses were included, which resulted in the elimination of 2,706 articles; there were 36 Cochrane reviews that included these search criteria, resulting in 157 articles. Articles that did not include adult patients or did not include both asthma and exercise were also excluded, resulting in 18 meta-analyses and reviews in total. On review of these studies, three additional meta-analyses that did not explicitly include “asthma” and “exercise” were added. Figure 3 demonstrates this approach in more detail.
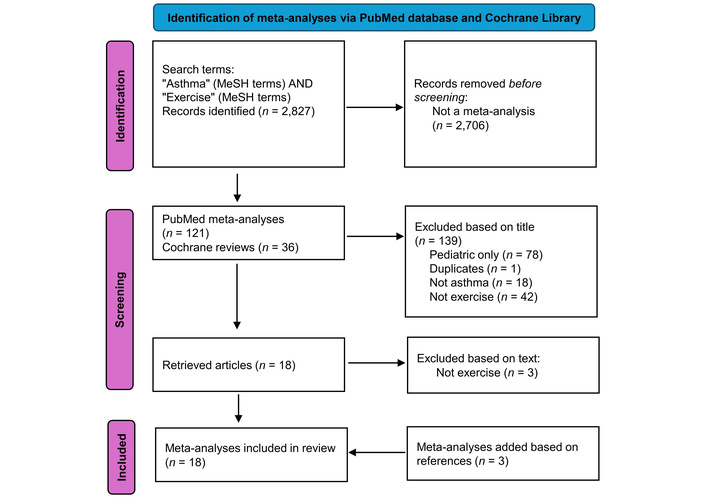
Flowchart of meta-analyses in the PubMed database and Cochrane Library review articles addressing asthma and exercise included in the review. Adapted from [7]. © 2021 The Author(s). Licensed under CC BY 4.0.
In order to ensure this review included the most recent data, and considering that a similar review was done in 2020, as mentioned earlier, the initial PubMed search of “asthma” and “exercise” was repeated. This time, any articles published prior to 2020 were excluded. All studies, rather than only meta-analyses, were included, and the same exclusion criteria were used, removing articles that did not include adults, as well as those that did not address asthma and exercise. This is contained within Figure 4. All searches were performed during October 2025.
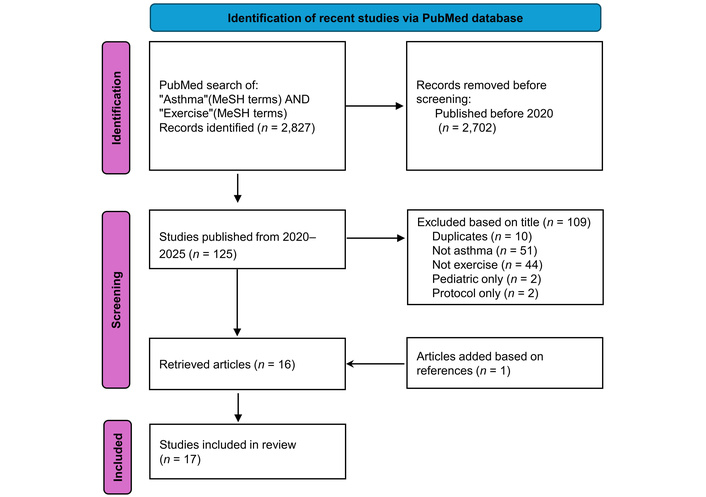
Flowchart of new publications in the PubMed database from 2020 to October 2025 addressing asthma and exercise included in the review. Adapted from [7]. © 2021 The Author(s). Licensed under CC BY 4.0.
Obese individuals are more likely to be diagnosed with asthma [8]. A specific phenotype called obesity-associated asthma has also been described, which is a late-onset, rather severe, non-type 2 driven disease, present mostly in women [9]. The obvious question is whether they are more likely to be asthmatic or more likely to be misdiagnosed as asthmatic, being more likely to experience shortness of breath [10]. In a large Canadian RCT, nonobese physician-diagnosed asthmatics, with a body mass index (BMI) of 20–25, were compared to obese asthmatics with BMI ≥ 30 [11]. All underwent spirometry before and after a bronchodilator. Those who did not demonstrate reversibility underwent a methacholine challenge. If the challenge was negative, they were weaned off their asthma medications, and the methacholine challenge was repeated. If the methacholine challenge off medications was negative, subjects remained off medication and were followed for a further 6 months. An asthma diagnosis was excluded in 31.8% of the obese group and 28.7% of the non-obese group, p = n.s. [11]. Overdiagnosis of asthma was no more likely in obese asthmatics, indicating that asthma was indeed more common among obese individuals [11]. In a Norwegian study, non-asthmatic subjects were followed for 11 years to see whether obesity, defined as BMI ≥ 30, increased the risk of developing asthma [12]. The odds ratio of obese subjects developing asthma during the 11-year follow-up was nearly 1.5 times greater than that of nonobese subjects [12]. Childhood obesity also increases the risk of asthma [13].
Adipocytes are the predominant cell type in adipose tissue [14]. They are not just passive lipid storage sites but have hormonal activity and produce leptin and adiponectin. Leptin plays an important role in appetite control and metabolism. Adiponectin has anti-inflammatory and antiapoptotic activities, inhibits hepatic gluconeogenesis, increases glucose uptake, and promotes lipid oxidation, stimulating insulin sensitivity [14, 15]. Levels of adiponectin decrease with increasing obesity, resulting in increased inflammation. Adiponectin “suppresses the expression of pro-inflammatory mediators while inducing the expression of anti-inflammatory mediators” [16]. It also “suppresses the expansion of antigen-specific T cells and their expression of cytokines, while enhancing T cell apoptosis” [16]. Adipocytes also contain aromatase, which has a feminizing effect, converting androgens to estrogens [17]. Resistin is another peptide secreted from adipose tissue and is associated with inflammatory diseases [18].
Obesity and an obesogenic diet can alter immune function and are proinflammatory [19]. An obesogenic diet alters the gut microbiome and facilitates microbial breaching of the gut lining and entering the circulation, thereby promoting systemic inflammation [20]. Adipose tissue has proinflammatory actions, including the production of cytokines interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), and monocyte chemoattractant protein-1 [21, 22]. Elevated IL-6 levels are seen in patients with more severe asthma and worse lung function [22].
Obesity can reduce chest wall compliance, in turn reducing lung volumes [23, 24]. There is also evidence that obesity can affect asthma severity [25]. Proposed causes include facilitating eosinophilic and neutrophilic inflammation and alterations in the gut microbiome [26]. In one study, obese subjects with a mean BMI of 45.7 were randomized to a 3-month weight reduction diet or maintained their regular diet [27]. Subjects in the intervention arm lost an average of 16.5 ± 9.9 kg, and controls had a mean weight gain of 0.6 ± 2.6 kg. There were significant improvements in PC20, FEV1, and FVC in the intervention group, and these parameters did not change in the control group [27]. In an earlier study, the same group demonstrated that weight loss can improve spirometry in obese female asthmatics [28]. Another group reported an improvement in asthma symptoms with weight loss [29]. In summary, asthma is more common, more severe, and asthma symptoms are worse in obese individuals.
Asthma is an inflammatory condition, and some foods reduce the formation of inflammatory mediators [30, 31]. Various dietary components have been reported to reduce the risk of developing asthma or impact its severity, including PUFAs, both n-3 and n-6 PUFAs, plant-based diets, vitamin D, antioxidant vitamins A, C, and E, flavonoids, probiotics, the Mediterranean diet, and various dietary supplements, but the literature is contradictory [8, 32].
Some processed foods are proinflammatory and may contribute to disease [33]. If nutrition plays a role in asthma, it may be due to its effects on various body tissues, indirectly by the effects of obesity, the gut and/or lung microbiome, or by epigenetics affecting the expression of various genes [26, 34–36]. Poor nutrition may cause dietary deficiencies, and toxins or pollutants in some diets may be deleterious [37]. The benefit of a ‘healthy diet’ may be that it substitutes for unhealthy foods, i.e., processed foods with too much salt, refined sugars, trans or saturated fats, or cured meats. Another consideration is that only individuals with certain genotypes might benefit from dietary modification or supplementation [38].
There are major challenges in trying to study the impact of diet on asthma. Maintaining subjects on a particular diet for an extended period is difficult. Epidemiologic studies identified possible associations between diet and the risk of developing asthma and asthma outcomes, but do not confirm causality [39]. Problems with epidemiologic data are that they are often based on retrospective surveys, and there are so many potential confounders. Moreover, most dietary histories are based on memory and may not be accurate [20]. Individuals may report recent consumption patterns that may not be reflective of long-term dietary trends, or there may be recall bias, and diets may evolve over time because of preference or food availability [40]. Income and socioeconomic status will also affect asthma outcomes, so even within individual investigations, diets may vary between individuals [41]. Most reviews of the effects of nutrition on asthma emphasized the high degree of clinical diversity in the interventions, heterogeneity in the methodology, and pointed out the risks of bias [42]. Despite these limitations, there is an enormous literature on the effects of diet on asthma.
The intervention trials, even RCTs, may be challenging to decipher. Studies of particular diets, e.g., the Mediterranean diet, assume that diets are similar between studies. Although diets may fall within a certain category, the types of food, proportions of different categories of food, total caloric intake, duration of the intervention, and whether patients’ weights remain stable, increase, or decrease during the study period are not standardized and vary between studies [43].
Another challenge is that strictly following any diet may lead to weight loss, which can improve asthma symptoms and spirometry, but whether this just represents the benefits of losing weight improving chest wall mechanics, represents an actual improvement in asthma control, or both is unclear [44]. Obese asthmatics who lost an average of 14.5% of their body weight experienced an improvement in FEV1 of 7.2% and 8.6% in FVC [45].
The role of various diets in asthma severity and control has been studied. The following is a review of some of the more commonly studied interventions.
Compared to the ‘Western diet’, the Mediterranean diet has been shown to be effective in preventing obesity both in the Mediterranean area and in other parts of the world where it has been adopted [45, 46]. This diet is rich in antioxidants and PUFAs that are anti-inflammatory. Generally, it includes ‘fish, monounsaturated fats from olive oil, fruits, vegetables, whole grains, legumes, nuts, and moderate alcohol consumption’ and is lower in saturated fats, meat, and dairy products [47].
Whether a particular diet can reduce the risk of developing asthma or affect its severity is controversial, with numerous conflicting reports in the peer-reviewed literature. A large cross-sectional Korean study did not find a relationship between asthma prevalence and consuming a Mediterranean-style diet [48]. In a cross-sectional Greek study of 105 adults, asthma control did not improve significantly with a Mediterranean diet [49]. A Swedish study looked at diet at 8 years of age and asthma outcomes at 24 years. There was no consistent association between diet and asthma, allergic/nonallergic phenotypes, or lung function [50].
Others have reported that the Mediterranean diet can improve asthma. An Iranian report with data on 7,667 children reported that the closer their diet was to the Mediterranean diet ideal, the less likely they were to experience wheezing [51].
Studies of the Mediterranean diet are hard to interpret. Although a diet may be called Mediterranean, it may vary considerably between reports. The types of food, proportions of different categories of food, total caloric intake, duration of the intervention, and whether patients’ weight remains stable are not standardized [48]. Only a few studies provided subjects with a prepared diet that ensured they ate a particular diet consistently through the study period [32, 52, 53]. Even some of the studies providing food to the intervention arm subjects were flawed in that food was not supplied to subjects in the control arm. Some studies provided a dietary item or suggested adding a dietary item to the intervention group, but did not control the rest of their diet or the control group’s diet [32, 46]. Another consideration is that, regardless of its effect on asthma, it may have unrelated benefits such as reducing the risks of obesity, developing diabetes, and cardiovascular disease [54].
Vertebrates do not produce PUFAs but must derive them from their diet. PUFAs are found in some plants, including flax and evening primroses, or originate in algae and plankton and move up the food chain in marine environments [55–58]. Fish oil contains high levels of n-3 fatty acids, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA).
PUFAs provided in the diet replace saturated fatty acids in some biochemical processes, including the production of leukotrienes and other oxylipins, leading to a less inflammatory profile [59]. EPA competes with arachidonic acid (AA) for metabolism by the lipoxygenase pathways, producing cysteinyl leukotrienes that have only 1–10% of the activity produced when AA is the substrate. Leukotrienes are proinflammatory, producing bronchospasm, bronchial inflammation with migration of eosinophils into the bronchial mucosa, increased mucus production, increased vascular permeability causing bronchial swelling, and impairing mucociliary clearance [60]. Patients fed n-3 fatty acids preferentially produce leukotriene C5, D5, and E5 rather than the more proinflammatory leukotriene C4, D4, and E4 [61]. Given the effects of PUFAs on inflammation, a number of investigators studied the effects of fatty fish consumption or PUFA supplementation in asthma (Table S1) [53, 56, 62–99].
As early as the 1980s, data emerged to suggest that a diet high in n-3 fatty acids might reduce the risk of asthma. The risk of asthma was reported to be less in Inuit living in Greenland, whose diet was largely based on fish and marine mammals, than in Inuit living in Denmark, who consumed less fish [100]. The pattern of serum PUFAs was different between Inuit living in Greenland and those living in Denmark, whose serum fatty acid profile was similar to that of other Danes [101, 102]. However, a Norwegian study did not find that a diet with higher fish content was protective [103]. A serious limitation of the observational studies was that they varied in design and dietary intakes and often had contradictory findings.
Supplementation trials, primarily with n-3 fatty acids, were undertaken in adults and adolescents, young children, infants, and pregnant women to study the effects on their offspring (Table S1). Most studies involved relatively small numbers of subjects and various amounts of PUFA supplements taken for variable durations, making comparisons between trials difficult (Table S1). Moreover, reported asthma outcomes varied between studies and could range from parents commenting on their child’s nocturnal cough, asthma symptoms, ACQ results, quality of life (QoL) questionnaires, rescue inhaler use, exhaled nitric oxide (NO) concentrations, various spirometric parameters, responses to various inhalational challenges, changes in maintenance or rescue inhaler use, exacerbations, systemic steroid use, emergency department (ED) visits or hospitalizations.
The reported effects of diets containing fish oil or n-3 PUFAs on asthma have been inconsistent since a possible relationship was first proposed. Some studies and meta-analyses reported a reduction in wheeze and/or asthma in children and improvements in symptoms or lung function. Others, however, have not.
Papamichael published a review of the role of fish consumption on asthma in children and identified 23 reasonable studies concluding that early introduction of fish into the diet and regular fish consumption reduce the risk of asthma and wheeze [32]. Among those 23 studies, 15 studies of the early introduction of fish, between the ages of 6 and 9 months, found that it improved symptoms and reduced the risk of asthma, 6 showed no benefit, and 2 suggested an adverse effect. Another study suggested that dietary intake of n-3 PUFAs may influence bronchodilator response in patients with poorly controlled asthma and obesity [104]. However, there did not appear to be any relationship between fish consumption and asthma in other populations. A study in Norway found no association between fish consumption and respiratory symptoms in young adults [103].
A two-period crossover trial where diets were completely controlled in adult asthmatics reduced serum cholesterol levels but had no consistent effect on asthma [105]. In a recent study, physician-diagnosed asthmatics with a blood eosinophil count ≥ 0.15 × 109/L requiring treatment with inhaled corticosteroids (ICS) were randomized to salmon oil with 270 mg of PUFAs or placebo capsules containing medium-chain triglycerides for 6 months [63]. There was no statistical difference in asthma exacerbations between the two groups [63]. Another recent study found that PUFA intake did not correlate with asthma severity in obese asthmatics [62]. A Cochrane database review in 2002 that included 9 RCTs satisfying their inclusion criteria found no evidence that dietary fish oil supplementation was either beneficial or detrimental in asthma [106]. Specifically, there were no consistent effects on FEV1, peak expiratory flow rate (PEFR), asthma symptoms, asthma medication use, or bronchial hyperreactivity [106].
A Mendelian randomization study of the causal association of PUFAs with asthma was unable to find a relationship between asthma and DHA, EPA, linolenic acid, or the ratio of n-3 to n-6 PUFAs in plasma [107]. Others have reported that n-3 PUFAs may be protective against the effects of indoor air pollution and second-hand smoke exposure [108, 109].
Among studies that suggested a potential benefit to PUFA supplementation, some have reported improved bronchial hyperreactivity [53, 65, 66, 68]. A study found that exercise-induced bronchospasm improved in the absence of other asthma parameters [67]. It is hypothesized that since cysteinyl leukotrienes C4, D4, and E4 and prostaglandin D2 (PGD2) are continuously released in the airways after an exercise challenge, by reducing their production, n-3 PUFAs are beneficial. Fish oil supplementation compared to olive oil supplementation reduced the severity of exercise-induced bronchoconstriction in elite athletes in a double-blind 3-week crossover study [68]. The same investigators showed that fish oil reduced serum concentrations of leukotrienes, PGD2, IL-1β, and TNF-α [68]. However, not all investigators reported a reduction in exercise-induced bronchospasm after exercise with PUFA supplementation [56, 64, 69]. Perhaps the inclusion of n-6 PUFAs in some investigations obviated the benefit of n-3 supplements [109–111]. Although supplementation with fish oil attenuated the late response to allergen challenge, it did not lead to symptom improvement or any objective changes in peak flows or bronchodilator usage [112]. In a crossover study, results with fish oil were similar to those of olive oil supplementation in asthma [112].
The inconsistency in the reports of fish consumption or PUFA supplementation may be due to variability in the sensitivity to leukotrienes among asthmatics. Some patients respond favourably to leukotriene receptor antagonists, but others do not [113]. Some of the variability in response to fish consumption/PUFAs may reflect the different sensitivity to cysteinyl leukotrienes. The meta-analyses of PUFAs in asthma conclude that the data did not support the existence of an effect on asthma outcomes or were felt to be inconclusive. Although it is not clear how beneficial fish oil supplementation is, there are no obvious harmful effects [114].
Investigations of maternal dietary supplementation with n-3 PUFAs on asthma outcomes in their offspring have looked at various outcomes, including impact on wheeze and other asthma symptoms, spirometry, and exacerbations. Some studies demonstrated benefits to supplementation, whereas others have not. One meta-analysis, including 8 RCTs, with 3,037 mother-infant pairs, did not find PUFA supplementation reduced the risk of asthma or allergic asthma [115]. Despite the inconsistencies, there is some evidence that maternal supplementation with larger amounts of fish oil than one would expect from a normal fish-containing diet may be protective in offspring. In a more recent Danish trial, women were randomized to receive 2.4 g of fish oil or olive oil daily through the 3rd trimester and were followed for 3 years afterwards [93]. This amount of fish oil is 15 to 20 times the average intake of American women [93]. Children of mothers who received fish oil were less likely to experience asthma, wheezing, or lower respiratory tract infections [93]. However, there was no effect on asthma exacerbations, the probability of developing eczema, or allergic sensitization. Other studies have also reported a reduction in asthma or recurrent wheeze in children of mothers supplemented with n-3 PUFAs [94]. Not surprisingly, the potential benefit of n-3 PUFA supplementation was greatest in the children of women who had the lowest serum levels of DHA and EPA [92].
However, other studies have reported conflicting results [97, 98, 116]. A study in the Seychelles, in a population that consumes a lot of fish, maternal fish consumption and fish consumption in their 7-year-old offspring did not correlate with asthma risk [116]. A Cochrane review found no evidence that PUFA supplementation in infancy had an effect on asthma [106].
In summary, despite numerous studies going back to the 1980s, whether there is a role for PUFA supplementation in preventing or improving asthma is unclear. The asthma manifestation most commonly benefiting from n-3 PUFA supplementation is a reduction in bronchial hyperresponsiveness to a variety of triggers.
Contrary to supplementation with n-3 PUFAs, supplementation with n-6 PUFAs, which includes AA, would be expected to be pro-inflammatory and possibly contribute to the risk of developing asthma and asthma severity. As with n-3 PUFAs, the literature on the effect of n-6 PUFAs on asthma is contradictory. One study reported that n-6 PUFAs led to a worsening of asthma outcomes in response to indoor air pollution in children [108]. Self-reported dietary intake of n-6 PUFA at 8 years was associated with an increased risk, and n-3 PUFA intake was associated with a reduced risk of asthma at age 24 in a Swedish study [117]. Others have not reported a benefit or detriment to dietary n-6 on asthma [77, 118]. Another study found an inverse relationship between maternal prenatal n-6 PUFA plasma levels and the risk of asthma in their offspring [119].
Vitamin D is immunomodulatory, playing a role in both innate and adaptive immunity, so it could theoretically be beneficial in asthma [120]. It may also have a mild antioxidant effect [120]. Some reports have suggested that vitamin D deficiency is a risk for asthma. Children with low vitamin D levels benefited less from ICS therapy and had poorer lung function [121]. Those with lower serum vitamin D levels were more likely to be African-American and have higher BMIs, risks for poorer lung function [121]. Asthmatic mothers with adequate serum vitamin D levels prenatally were less likely to have children with asthma or recurrent wheeze at age three than those with low serum vitamin D levels [122]. Children with mild-to-moderate persistent asthma who were vitamin D insufficient were more likely to experience a severe exacerbation [123]. Other studies were unable to demonstrate a relationship between asthma and vitamin D levels [124].
Although the role of vitamin D deficiency in asthma is controversial, others have studied the effects of vitamin D supplementation in asthma (Table S2) [125–154]. Most vitamin D supplementation studies have looked at pediatric populations or the effects of maternal prenatal supplementation on their offspring to determine if it affected the risk of wheeze or asthma in early childhood or if it reduced the risk of exacerbations [155].
A few studies reported that the risk of wheeze but not asthma in infants and small children was reduced with vitamin D supplementation [156], whereas others found no relationship to wheeze or asthma [124, 136, 157]. A large analysis that paired vitamin D data from over 4 million individuals found that vitamin D levels did not correlate with the risk of allergic or nonallergic asthma [124].
Children were randomized to placebo, low dose, or high dose vitamin D, and the prevalence of physician-diagnosed asthma was 11%, 0%, and 4%, respectively [141]. A systematic review and meta-analysis that identified 7 studies, 3 in children and 4 in adults, found that vitamin D serum levels increased with supplementation, and there was a trend to fewer exacerbations, but the difference was not statistically significant [158]. There were no differences in FEV1%, fractional exhaled NO (FeNO), or Asthma Control Test (ACT) [159]. Two other meta-analyses concluded that vitamin D reduced the risk of exacerbations [155, 160]. A Cochrane review meta-analysis concluded that in mild-to-moderate asthma, vitamin D reduced the risk of severe asthma exacerbations and healthcare use [161]. Another potential dimension to the vitamin D and asthma saga is that there is at least one report that found that the benefits of vitamin D in asthma may be related to individuals with certain genotypes [162].
Two recent Cochrane database systematic reviews of vitamin D supplementation in pregnant or breastfeeding women or children were unable to show that vitamin D prevented asthma, improved asthma control, or reduced the risk of asthma exacerbations [163, 164]. Some studies found that prenatal supplementation improved outcomes in those with vitamin D deficiency but did not in those with adequate vitamin D levels.
Vitamin D plays an important role in immunity. Acute respiratory tract infections are a common cause of asthma exacerbations in children, and there are reports that vitamin D in daily doses of 100–1,000 international units (IU) reduces the risk of acute respiratory infections [42, 165]. One review of 25 eligible randomised controlled trials with 10,933 participants (aged 0 to 95 years) found that vitamin D supplementation reduced the risk of acute respiratory tract infection [42]. The benefit was greatest in very vitamin D-deficient patients [42, 166].
There are several reports that vitamin A levels are lower in asthma, but supplementation has not been shown to be beneficial [167, 168].
There is very little data available about the effects of vitamin C on asthma outcomes, but the little available data did not demonstrate a clinically significant benefit or harm from supplementation [169–175]. One study found that vitamin supplementation was associated with a reduced risk of wheeze in offspring [176]. Another study reported that vitamin C supplementation may reduce the risk of childhood wheeze if the mother smoked during pregnancy [172]. A meta-analysis comparing high to low dietary vitamin C intake found no difference in the risk of asthma [171]. Another meta-analysis of maternal vitamin C intake found that the offspring of mothers with the highest consumption during pregnancy were no less likely to experience wheeze than offspring of mothers with the lowest vitamin C consumption [167].
Vitamin E levels have been reported to be lower in patients with severe asthma [176]. A few observational and small studies reported that vitamin E supplementation during pregnancy reduces the risk of wheeze and asthma in offspring, which led to a number of trials of prenatal vitamin supplementation [177]. A meta-analysis found that higher maternal prenatal consumption of vitamin E compared to the lowest vitamin E consumption reduced wheeze and asthma in offspring [167]. Several studies were unable to show that prenatal vitamin E supplementation had any effect on the incidence of asthma [176–180]. One study showed no benefit during the first year after birth, but in the absence of ‘colds’, the risk of wheeze was less in the second year [180].
However, other studies did not demonstrate a benefit from prenatal supplementation [176]. A Danish study with 44,594 pregnant women was unable to show any benefit of prenatal vitamin E supplementation on asthma in their offspring [181]. Prenatal vitamin E supplementation correlated with increased crown-rump length (CRL), and CRL correlated with lung function and inversely with wheeze and asthma in offspring, but when corrected for CRL, there were no associations between vitamin E and lung function or reduced asthma symptoms [179]. In a cross-sectional study of National Health and Nutrition Examination Survey (NHANES) data on 18,118 children between the ages of 3 and 18, 11.3% had asthma, but did not find a significant association between a composite dietary antioxidant index and asthma [170]. The antioxidant index was based on the consumption of 6 dietary antioxidants over the previous 24 hours: vitamin A, vitamin C, vitamin E, zinc, selenium, and carotenoids [170].
A report using Mendelian randomization applied to a large European database found no relationship between vitamin E or the other antioxidant vitamins, vitamin A and C, and asthma risk [171].
Flavonoids are polyphenolic molecules found in fruit, vegetables, wine, and tea [182]. They have antioxidant and immune-modulating activities that are beneficial in animal asthma models [183]. Several studies have suggested that flavonoid consumption is protective against asthma in adults and children [182, 184]. Cross-sectional survey data from the NHANES database suggested that greater flavonoid intake was associated with a reduced risk of asthma [185]. This study also found that consumption of some flavonoid subclasses, particularly flavanone, anthocyanidin, flavonol, and flavone, was negatively associated with asthma prevalence, whereas isoflavone and flavan-3-ol subclasses were positively associated with asthma prevalence. The obvious limitations of this report are that it is a cross-sectional study and not necessarily reflective of diet over time, dietary information was based on recall, and there was no confirmation of the asthma diagnosis. Moreover, there may be other reasons why some asthmatics consume a diet richer in flavonoids including socio-economic status, living location, and race.
A Cochrane review of a flavonoid, Pycnogenol, an extract of maritime pine bark, in chronic disorders included 2 small trials in asthma; one with 44 subjects reported an improvement in lung function and asthma symptoms, and another with 60 subjects reported that more were able to stop using their albuterol inhalers [185]. Their conclusion was that “small sample sizes, limited numbers of RCTs per condition, variation in outcome measures, and poor reporting of the included RCTs mean no definitive conclusions regarding the efficacy or safety of pine bark extract supplements are possible.”
Although the α-diversity of the gut microbiome varies from normal in chronic obstructive pulmonary disease (COPD) patients, the gut microbiota of asthmatics is similar to that of healthy individuals [186]. Despite the findings that the gut microbiome of asthmatics is comparable to that of healthy individuals, some reviews have concluded that probiotics improve asthma, including a reduction in exacerbations [187–189]. Commonly used interventions are with Lactobacillus and Bifidobacterium [187]. Not all reviews concluded that probiotics were beneficial. One review of 17 RCTs composed of 5,264 children found that pooled data for the risk of developing asthma after probiotic supplementation showed no significant reduction compared with controls [190]. The Cochrane review also reported shortcomings with the trials and felt that it was premature to recommend their routine use. They also point out that there were no concerns about adverse effects [191].
Epidemiologic evidence from the United Kingdom suggested that increased salt intake increased the risk of asthma [192]. In a separate report, the same author found a correlation between bronchial reactivity to histamine and urinary sodium excretion, reflective of sodium consumption [193].
Increased dietary salt might increase bronchial reactivity by inhibiting bronchial smooth muscle sodium-potassium ATPase or by reducing circulating catecholamines [194]. In a crossover study comparing a high salt diet to a low salt diet in subjects with exercise-induced asthma, the high salt diet increased airway inflammation [195]. In another study comparing a low to high salt diet, pre-exercise lung function was not affected, but post-exercise FEV1 declined more on a high salt diet [196]. Two Cochrane database reviews concluded that dietary sodium reduction did not improve asthma control, but the more recent review suggested that it might improve lung function in exercise-induced asthma [197, 198].
One of the challenges with delineating the relationship between exercise and asthma is that the term “exercise” describes a wide spectrum of activities and intensities. Furthermore, exercise can be supervised, such as in pulmonary rehabilitation programs, or unsupervised. Several meta-analyses have been published in the last 5 years, assessing the evidence for exercise’s effects on various asthma outcomes. Often, these refer to certain types or categories of exercise, specifically aerobic exercise, yoga, or resistance training, or various combinations of these. There are also articles that specifically examine pulmonary rehabilitation, and those that also include unsupervised exercise.
Resistance training is a known component of pulmonary rehabilitation and is usually included in exercise prescriptions. However, unfortunately, there is almost no data that looks at resistance training in isolation in asthmatics, and so no conclusions regarding this could be made in this review. When the term “resistance” was included in the search above, this resulted in various articles looking at “inspiratory muscle training,” rather than what is classically considered “resistance training,” which involves muscle strengthening or weight training. A Cochrane review investigating inspiratory muscle training was published in 2013, and an update with a meta-analysis was published in 2023 [199]. The challenge with this is that this training involved an external resistive device, which can be viewed as a medical therapy rather than exercise, and is not easily adopted into a patient’s lifestyle. In fact, one meta-analysis excluded articles with any form of “exercise therapy” [200]. As such, we will not include these interventions when referring to “exercise” in this review.
Similarly, there are articles that touch on “breathing techniques,” or “breathing training,” which, in our opinion, also encompass part of a grey area. Though these would meet the criteria for the definition of “physical activity,” they are not universally accepted as “exercise.” The goal of breathing training or breathing techniques is to modify a behaviour, rather than to enhance fitness through strengthening muscles or improving speed or endurance when performing an activity [201].
Aerobic exercise is the most common form of exercise studied. Multiple articles mention exercise only, not specifically aerobic exercise; however, it is a reasonable assumption that they refer to aerobic exercise. The 2013 Cochrane review by Carson et al. [202] reviewed 23 studies that looked at “physical training” in asthma, which included “running, gymnastics, cycling, swimming, weights, and walking.” Training was defined as “whole body exercise lasting more than 20 minutes, twice a week, for at least 4 weeks” [202]. This definitively debunked the notion that patients with stable asthma should be wary of exercise out of concern for worsening of their symptoms. The review specifically recommended encouraging these patients to pursue regular physical activity, with none of the studies reviewed documenting an exacerbation of asthma symptoms with physical training. Universally, exercise is well tolerated without worsening of asthma control in the literature since this review.
The 2013 Cochrane review demonstrated that exercise results in a significant improvement in VO2max of nearly 5 mL/kg/min and a 3.7 bpm increase in maximal heart rate compared to control [202]. However, there were no significant differences in FEV1, FVC, PEFR, or 6MWD. Multiple studies included showed a significant improvement in health-related QoL (HRQoL), but there were insufficient data to formally assess this in the meta-analysis. An important notion to acknowledge is that exercise will have variable effects on different individuals, and not all individuals will show a significant benefit. A 2020 study examining the effects of HIIT showed clinically meaningful improvements in asthma control in 35% of patients in the intervention group [203].
Hansen et al. [204] published a systematic review in 2020, with the aim of further investigating the role of aerobic exercise as a form of therapy in asthma. It provided an update of the evidence since the 2013 Cochrane review, including 11 studies with at least 8 weeks of aerobic exercise training. Although heterogeneity was noted in all outcomes assessed, they found that aerobic exercise was associated with an improvement in ACQ scores and HRQoL. There was also a non-significant trend towards improved FEV1. There was no improvement in local airway inflammation markers, namely, FeNO or sputum eosinophils; no other studies have found a significant association between exercise alone and local airway inflammation.
Interestingly, only one 2023 RCT looked at the effect of exercise on ICS requirement and showed a non-significant trend towards a larger reduction in ICS dose in the intervention group, equating to almost a 25% reduction in dose from baseline, which was maintained 1 year later [205]. Finally, Liu et al.’s comprehensive 2025 meta-analysis [5] also demonstrates that aerobic exercise improves AQLQ scores, FEV1, FVC, and PEFR. In summary, the improvements in HRQoL, lung function, and asthma control seen with aerobic exercise support the unanimous recommendation for all asthmatics to remain physically active.
Yoga has been increasingly popular as a form of physical activity and is now considered an adjunct or complementary therapy for asthmatics. Yoga consists of multiple components, including breathing exercises, meditation, and postures [206]. The first meta-analysis that looked at the use of yoga as an intervention in asthma was not able to conclude that yoga could be considered “routine for asthmatic patients” [207]. The main limitation is the lack of high-quality evidence due to small “sample sizes, allocation bias, selective reporting of outcomes, and inconsistent results” [206]. This was also echoed in the 2016 Cochrane review, entitled “Yoga and Asthma,” which demonstrated “moderate evidence for [a] small improvement in quality of life and [asthma] symptoms” [208].
A more recent meta-analysis in 2023, using data from 15 experimental studies in adult patients in the past decade, found a significant improvement in PEFR and HRQoL in asthmatic patients who practiced yoga, compared to controls; this was corroborated by another study in 2024 [209]. The meta-analysis also favoured an improvement in FEV1, FVC, and FEV1/FVC with yoga, but was inconsistent in its conclusions as to whether these met the criteria for statistical significance. There was no statistically significant improvement in overall asthma control.
Analyzing the individual components of yoga is even more limited due to the paucity of data. The 2016 Cochrane review compared articles that studied yoga breathing alone to the combination of yoga techniques and did not find significant differences between them. However, the authors warned that this finding should be interpreted cautiously. Subsequent studies that reviewed meditation and breathing exercises individually have not shown a significant improvement in any asthma parameters; therefore, newer evidence indicates the benefit of the combination of yoga [206]. A very recently published pilot study of 30 patients used a sham program as their control to compare with an online yoga program. This showed a statistically significant difference in asthma control and QoL scores in the subjects in the experimental group compared to the control group, further supporting that yoga has a positive effect on asthma outcomes [209].
Pulmonary rehabilitation programs are widely heterogeneous, with one meta-analysis reporting that most of the programs included aerobic exercise with or without resistance or breathing/stretching exercises. Most commonly, these supervised programs consisted of moderate-to-vigorous intensity aerobic exercise in thrice weekly 30-to-45-minute sessions. Even then, there is marked variability as to how intensity was defined in the programs.
A 2022 Cochrane review assessed the impact of formal pulmonary rehabilitation programs in asthmatic patients, using data from 10 trials. A number of outcomes were reported, including VO2max, 6MWD, and scores on various questionnaires, including the ACQ, AQLQ, St. George’s Respiratory Questionnaire (SGRQ), and the Chronic Respiratory Disease Questionnaire (CRQ). Secondary outcomes of note included severe asthma exacerbations, distance on field walk tests, levels of physical activity, and adverse events.
Overall, the review found that there was a possible short-term increase in VO2max after pulmonary rehabilitation in individuals with asthma, but evidence for this is uncertain at best. However, there was evidence for an improved 6MWD in the short term, surpassing the minimally clinically important difference (MCID). Long-term data for this is lacking. Interestingly, the one study that looked at physical activity levels showed that those subjects who underwent pulmonary rehabilitation on average had significantly higher daily steps than those in the usual care group [210].
With respect to asthma control, there was evidence for a modest improvement in ACQ scores; however, this did not exceed the questionnaire’s MCID and was not sustained. An RCT in 2022 showed that the mean change in ACQ-6 was below the MCID; nearly 55% of respondents reached this MCID in the pulmonary rehabilitation group vs. 23% in usual care, which met statistical significance, again suggesting that patients respond variably to the intervention [211]. Only one study reported an exacerbation rate, for which there was no significant difference between the two groups. Similar to aerobic exercise, pulmonary rehabilitation was “generally… well-tolerated.”
There was a “large improvement” in QoL; the overall mean difference in the SGRQ was 18.5 points, exceeding the MCID of 4 points, but, interestingly, this was not corroborated in the AQLQ scores. The review also performed subgroup analyses for some measures, based on whether the program was at least 8 weeks in duration, as well as whether the patients had severe or non-severe asthma. For the most part, there were no significant differences noted.
Generally, evidence is in favour of pulmonary rehabilitation programs, though a recent 2025 meta-analysis reports that pulmonary rehabilitation did not have any impact on HRQoL compared to breathing exercises and education in asthmatic subjects [212]. As such, it is plausible that those patients who did not complete the pulmonary rehabilitation program were active but not supervised. Furthermore, as a structured program, pulmonary rehabilitation times and meetings may be inconvenient for individuals with erratic schedules. Exercise in individuals not in the pulmonary rehabilitation programs could explain a smaller difference with respect to HRQoL.
It is probable that “self-guided pulmonary rehabilitation” in asthma patients that involves the same activity provided by a pulmonary rehabilitation program would have similar outcomes with respect to cardiorespiratory fitness. Pulmonary rehabilitation, however, may be more helpful for those patients who may not have much experience with exercise, prefer a structured regimen, have difficulties maintaining motivation, or have limitations to exercise (such as injury or significant comorbidities). Ultimately, the main goal is for the patient to exercise, and it most likely does not matter whether or not exercise is supervised, as long as it happens.
The effects of many different diets on the risk of developing asthma or improving asthma control have been studied. Maintaining subjects on a particular diet during a trial for a prolonged period is challenging. To ensure that individuals maintain a particular diet throughout a study period obliges the investigators to provide all meals and possibly snacks and monitor all the food consumed for many subjects for an adequate period, ideally months. The cost and challenges of supervising this type of study would be prohibitive. There have been a few small, relatively short studies where meals were provided, but the studies weren’t large or long enough [90]. Therefore, most studies of the effects of diet on asthma have relied on survey data despite its limitations; most were cross-sectional, therefore did not necessarily capture subjects’ diets over time. Although the current data have numerous shortcomings, it does not suggest that dietary supplements have a significant effect on the risk of developing asthma or control in individuals with established disease.
Most studies relied on the patient providing the asthma diagnosis. Even patients reporting their doctor’s diagnosis of asthma may not be reliable. In a cross-Canada trial, approximately a third of patients with physician-diagnosed asthma were able to stop and remain off asthma medications for a year [213]. Dietary histories are also questionable, relying on the subject’s recall. Another confounding factor is whether the reported diet reflects the dietary effect or socioeconomic influences. Wealthier individuals are more likely to maintain a healthier diet, have better access to health care, and live in an area with better air quality, which affects the risk of developing asthma, asthma control, and outcomes. Other concerns with the studies included in this review are that many were small, did not provide power estimates for their primary outcomes, therefore susceptible to type 2 errors, and very few included corrections for multiple comparisons, like Bonferroni, when calculating statistical significance [214].
Reports of the effects of the Mediterranean diet on asthma are inconsistent. Some studies demonstrated a benefit, whereas others have not. The lack of consistency in diet content, both of the Mediterranean diet and the control diet, in studies makes interpretation difficult, if not impossible. Although it is not clear whether it affects the risk of having asthma or asthma control there is no evidence that the Mediterranean diet is harmful and the benefit on the risk of developing asthma or worsening existing asthma of a Mediterranean diet and other diets is that they lead to weight loss or prevent the development of obesity.
If there is a benefit to n-3 PUFA supplementation, it is relatively modest. However, there are no serious concerns about dietary PUFA supplementation, and there appear to be other benefits, particularly a reduction in cardiovascular risks. Moreover, a diet rich in n-3 PUFAs is likely to be low in saturated or trans fats and ultra-processed foods. The conflicting data on the effect of n-6 PUFAs on asthma suggest that if there is either a beneficial or an adverse effect, it is likely relatively small.
The studies of vitamin D supplementation are challenging to interpret. Some limited supplementation to subjects who were vitamin D deficient, while others did not. Differences in the population studied, sample sizes, outcomes studied, differences in vitamin D dosing, timing selected for supplementation in the pediatric studies, and duration of therapy make comparisons between studies challenging. Many, if not all, of the studies were likely underpowered. Studies of the offspring of prenatally vitamin D-supplemented women have similar deficiencies. The reliability of vitamin D systematic reviews is limited, and interpretation is challenging because of the risk of bias and heterogeneity.
In summary, the data do not support routine vitamin D supplementation in asthma patients or prenatally to pregnant women to prevent or improve asthma control. However, there may be a benefit related to supplementation of pregnant women and children who are vitamin D-deficient, and there are other reasons to correct vitamin deficiency at any age, such as ensuring proper bone development.
Interpretation of the studies of the antioxidant vitamins A, C, and E is also disappointing. The conflicting data suggest that if there are benefits to supplementation, they are relatively modest, and it makes the most sense to focus supplementation on individuals who are deficient. Another way to look at the data is that it emphasizes the importance of a healthy, balanced diet being provided during pregnancy and to all individuals with asthma.
The data surrounding flavonoids and probiotics are also limited. Despite some advocating for their use for asthma or to prevent asthma, there is insufficient evidence to support those claims. There is limited data on the role of dietary sodium. However, there is a suggestion that it might reduce bronchial hyperreactivity in asthmatics.
The effects of diet on asthma may not reflect what individuals are eating but what they are not. Obesity is a risk for having asthma and for making existing asthma worse. The Mediterranean diet, diets containing PUFAs or flavonoids, may result in weight loss, which can improve lung function, therefore asthma control, and likely reflects lower consumption of processed foods containing more calories, more sugar, trans fats, saturated fats, sodium, and less vitamins. Whether the recommended diets have a direct effect on the risk of developing asthma or asthma control, diets similar to the ideal Mediterranean diet, including fish, fruit and vegetables, vitamin D content, and are relatively low in salt, sugar, saturated and trans fats, and cured meats, are safe and better for a patient’s overall health.
There is stronger evidence that supports the benefits of exercise in asthmatics than that of diet. New evidence continues to strengthen this argument, with particular emphasis on aerobic exercise, as well as yoga. Of note, there is no data to suggest any difference between supervised exercise, such as in pulmonary rehabilitation, rather than unsupervised exercise. So, what type of exercise should we choose? And how much is enough?
Unlike with dietary interventions, where the amount of the intervention can be quantified in standard units, such as with vitamins and sodium, and where there are recommended amounts of nutrients that humans should take in, this is harder to define with exercise. So, how much exercise is enough, and how would this even be calculated with the heterogeneity of exercise and how it impacts the individual? Surely, there must be a dose-response relationship. The benefit of asthma probably plateaus after a certain level of exercise is reached, and this threshold is likely unique to each patient.
A 2024 study addressed how much exercise should be recommended in patients with moderate-to-severe asthma. Walking at least 7,500 steps per day was associated with having better control, as measured by ACQ scores. Interestingly, the study also categorized subjects as “sedentary,” defined as being sedentary for at least 8 hours per day, or “non-sedentary” [215]. Akin to the theory that diet’s impact on asthma may actually be driven by what individuals are not eating, perhaps it is the sedentary time that impacts an individual’s asthma outcomes, as this has also been related to obesity, which has clear negative implications on asthma. They found that the amount of sedentary time was not associated with asthma control; that is, the association between walking more than 7,500 steps daily and better asthma control was sustained regardless of whether patients were “sedentary” or not [215]. The authors concluded essentially that the priority should be increasing physical activity, as opposed to reducing sedentary time.
Regarding the “ideal” exercise, one meta-analysis from 2022 found that aerobic exercise resulted in superior improvements in cardiorespiratory fitness, compared to stretching and/or breathing exercises. A meta-analysis from Xing et al. [216] divided exercise into five groups: breathing training, aerobic training, relaxation training, yoga training, and “Breathing training and Aerobic training.” All five groups of exercise training resulted in an increase in FEV1 and PEFR, with no significant difference between groups, but the authors concluded that combined breathing and aerobic training, as well as yoga training, may be more effective with respect to improvement in lung function.
Ultimately, Liu et al. [5] conclude that “specific interventions provide targeted advantages in various domains.” Specifically, aerobic exercise improved AQLQ scores, FEV1, FVC, and PEFR; and yoga also increases AQLQ, FEV1, and FVC. This is consistent with the findings in the 2023 network analysis. Overall, they deemed the evidence to be “inadequate” despite these “favourable findings.” They rated the strength of the data from the individual studies and found that the overwhelming majority were either classified as “low” or “critically low,” indicating that there is still a need for larger studies and higher-quality data addressing the effects of exercise in asthma as well. Their recommendation was that a tailored approach to exercise prescriptions in asthmatics is ideal, taking into account the patient’s goals and preferences.
Similar to exercise, considering diet as a lifestyle choice, it seems clear that whichever diet is chosen, it should be effective in preventing or combating obesity, which is established as having a negative impact on asthma outcomes. However, the ideal diet should also be compatible for the patient, such that they view it as a long-term investment in their asthma health and health in general.
As obesity rates increase across the globe, it is inevitable that obesity will be an ongoing problem. Obesity is associated with various metabolic signals that have the potential to affect the risk of developing asthma and contribute to asthma severity. Unless the currently available and future obesity drugs can control the obesity epidemic, we need to find alternative ways to treat obesity-associated asthma. Future studies of the various adipocyte products are required to determine which of these play a role in asthma, and appropriate treatments need to be developed to counter their effects.
The available studies of the various nutrients and diets have several shortcomings. We did not find power estimates in most of the reports, and many of the studies were likely underpowered. The available studies of various nutrients in asthma have several important deficiencies. There are no criteria for target serum levels, when an intervention should be initiated, nor how long an intervention needs to be maintained to impact the risk and/or severity of asthma. Moreover, criteria need to be defined for control groups. Diets apart from the tested supplements need to be controlled to ensure that the intervention and control groups are otherwise similar. Another consideration is that the studies do not ensure that total calorie intake is similar in both the intervention and control groups. The studies also do not report whether both the intervention and control groups both gain or lose a similar amount of weight during the studies. Future studies of dietary supplementation need to address these shortcomings.
Now that exercise is an established recommendation for asthmatic patients, with more and more data to support this, other questions arise. The type, intensity, and amount of exercise required to reap the benefits are still unknown and likely depend on the individual. This makes it challenging to derive a blanket recommendation for the asthmatic population as a whole. The data for exercise is convincing, but there remains a paucity of high-quality studies to answer some of the aforementioned questions, which should be a priority for further research in this area. As of now, an appropriate exercise regimen that is enjoyable for the patient and is compatible with their lifestyle long-term seems to be the best regimen.
6MWD: six-minute walk distance
AA: arachidonic acid
ACQ: Asthma Control Questionnaire
AQLQ: Asthma Quality of Life Questionnaire
BMI: body mass index
CRL: crown-rump length
DHA: docosahexaenoic acid
EPA: eicosapentaenoic acid
FeNO: fractional exhaled nitric oxide
FEV1: forced expiratory volume in 1 second
FVC: forced vital capacity
HRQoL: health-related quality of life
ICS: inhaled corticosteroids
IL-6: interleukin-6
MCID: minimally clinically important difference
n-3: omega-3
NHANES: National Health and Nutrition Examination Survey
PEFR: peak expiratory flow rate
PGD2: prostaglandin D2
PUFAs: polyunsaturated fatty acids
QoL: quality of life
RCT: randomized controlled trial
SGRQ: St. George’s Respiratory Questionnaire
TNF-α: tumor necrosis factor-α
VO2max: maximal oxygen uptake
The supplementary tables for this article are available at: https://www.explorationpub.com/uploads/Article/file/1009124_sup_1.pdf.
AS: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. SKF: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The datasets that support the findings of this study are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 561
Download: 24
Times Cited: 0
