Affiliation:
1Children’s Orthopaedic Centre, Mumbai 400016, Maharashtra, India
ORCID: https://orcid.org/0000-0001-6227-5841
Affiliation:
2Department of Orthopaedics, All India Institute of Medical Sciences (AIIMS), Phulwari Sharif, Patna 801507, Bihar, India
Email: riteshpandey8262@yahoo.com
ORCID: https://orcid.org/0000-0001-7193-5514
Explor Musculoskeletal Dis. 2026;4:1007119 DOI: https://doi.org/10.37349/emd.2026.1007119
Received: October 31, 2025 Accepted: March 04, 2026 Published: March 29, 2026
Academic Editor: Fernando Pérez-Ruiz, Cruces University Hospital, Spain
The article belongs to the special issue Innovation in Orthopedics
Paediatric orthopaedics has evolved as a specialized branch of orthopaedics, reflecting the understanding of child development and paediatric musculoskeletal disorders. The push for creative ideas and innovations has expanded its scope and indications to a wide group of musculoskeletal disorders in children. This review discusses some of the most impactful innovations of the late 20th and 21st centuries in the field of paediatric orthopaedic surgery and highlights their relevance in the management of complex problems. It includes their evolution over time, current strengths and challenges, and the scope of further improvement in the future. In addition, it discusses the future perspective and possibilities for similar groundbreaking innovations in the field of paediatric orthopaedics.
Like all fields of surgical endeavour, paediatric orthopaedics has expanded in its scope and indications. This has been due to the thrust on innovations and the application of creative ideas to the field. Innovative surgical techniques like minimally invasive procedures and growth modulation have revolutionized the treatment of paediatric limb deformities. Biocompatible materials and modular designs in the preparation of prosthetics and implants have improved the outcomes in limb salvage and reconstructive surgeries. The focus of management has shifted to functionality, thereby leading to an increased emphasis on preserving joint function and growth. The use of three-dimensional (3D) imaging and 3D printing for preoperative planning and custom implants has proven its worth for better outcomes. While many innovations have been introduced over the last few decades, the current review aims to discuss some of the most significant innovations of the late 20th and 21st centuries in the field of paediatric orthopaedic surgery and to highlight their relevance in the management of complex problems (Table 1). This narrative review is based on what are widely considered impactful innovations in this field. Over a period of time, these innovations have undergone several modifications and have made a considerable impact on the management of musculoskeletal disorders in children. They still carry potential to improve further, and attempts in this direction are ongoing (Table 2).
List of innovations in paediatric orthopaedics based on their type.
| Technological | Biological | Both technological & biological |
|---|---|---|
|
|
|
3D: three-dimensional; EOS: early onset scoliosis.
Development of important innovations in paediatric orthopaedics.
| SR. NO. | Innovation | Year (inventor/country) | Uses | Modification (if any) over time | Status | Future perspective |
|---|---|---|---|---|---|---|
| 1 | Advanced paediatric musculoskeletal imaging [1–17] | 1980s (various contributors across the globe) | To make accurate and reliable diagnoses in all phases of patient care | Several modifications for making them safer, affordable, and accurate in making diagnosis | A pivotal role in impeccable clinical practice | Deep learning reconstructionUse of AI toolsImage-guided interventions in a 3D virtual reality environment |
| 2 | 3D printing [18–30] | 1983 (Charles Hull/USA) | Preoperative planning, getting patient-specific implants, instrumentation, and prosthesis | Modification focused on improving speed, material versatility, and precision | Though a promising tool in patient care, it has many challenges for use on a larger scale | Need for a modified technique that is cost-effective, readily available, and can deliver the product much faster |
| 3 | Ponseti technique [31–38] | 1952 (Ignacio Ponseti/USA) | Early treatment of clubfoot deformity | Accelerated Ponseti techniqueModified Ponseti technique for atypical clubfoot | Today, it is the standard method for treating early clubfoot deformity | Need to further reduce the duration of casting and improve the complianceOne-week accelerated Ponseti technique could be the next standard practice |
| 4 | Safe surgical dislocation of the hip [39–53] | 2001 (Reinhold Ganz/Switzerland) | Treatment of traumatic and non-traumatic paediatric hip conditions like DDH, SCFE, Perthes disease, and femoro-acetabular impingement | Smith’s modification for younger children. Instead of doing TFO, elevation of the cartilaginous sleeve from GT is preferred | Favored approach for hip preservation surgery | To see its continued evolution as a crucial joint preservation technique, augmented by advanced technologies like computer navigation and robotics |
| 5 | Use of botulinum toxin in cerebral palsy [54–67] | 1993 (A.L. Koman/USA) | Treatment of spasticity and early contractures in cerebral palsy | Increased precision with USG guided injections, attempts to lower the dosage, and combining injections with a comprehensive treatment plan | Standard treatment for dynamic contractures in cerebral palsy | Developing newer variantsExploring new indicationsTo increase the safetyTo reduce the long-term effects on the injected muscle |
| 6 | Three-dimensional instrumented gait analysis (3D-IGA) [68–81] | 1890 (Christian Braune & Otto Fischer/Germany) | Assessment of gait abnormalities | Integration of wearable sensors like inertial measurement units (IMUs) and electromyography (EMG) | It has evolved as an important tool in the assessment of ambulant children with cerebral palsy | Development of markerless systemsWearable sensorsUse of AI and smartphone apps for data analysis |
| 7 | Growth modulation with tension band plates [82–97] | 2007 (Peter Stevens/USA) | Correction of limb deformities before skeletal maturity | More minimally invasive techniques (plate is positioned over skin before inserting the guidewires, central wire is avoided), sleeper plate technique | Preferred choice for growth modulation | More minimally invasive techniques, better understanding of its use in complex deformities and younger children, and development of novel implants based on the “constant force” concept for better control of growth modulation |
| 8 | Ilizarov principles and distraction osteogenesis [98–103] | 1950 (Gavril Abramovich Ilizarov/USSR) | Correction of bone defect, complex limb deformity, and limb length discrepancy | Unilateral external fixators, hybrid techniques for bone transport, internal limb lengthening device, software-driven six-axis external fixation device, use of carbon fiber providing lighter weight and lower profile, improvement in pin and clamp design for better stability | Preferred choice for management of difficult nonunion, bone defect, limb deformity, and limb length discrepancy | Further improvements in the design of hardware and software, leading to more precision, fewer complications, and which will be more user-friendly. Its amalgamation with technology like robotics and augmented reality may help in expanding the indications to other areas beyond deformity correction. The construction of patient-specific frames through 3D printing may further improve the treatment outcome |
| 9 | Motorized internal limb lengthening [104–126] | 1992 (Rainer Baumgart & Alex Betz/Germany) | Limb lengthening for correction of limb length discrepancy | Initial nails required mechanical activation, whereas the currently available nails are motorized and remote-controlled | Motorized intramedullary lengthening nails have become the implant of choice for limb lengthening in many countries | Innovations in metallurgy and implant design for higher mechanical strength & biocompatibilitySmart nails with built-in sensorsPlate-based & extramedullary devices |
| 10 | Magnetically controlled growing rods for early onset scoliosis (EOS) [127–142] | 2004 (Arnaud Souberian/France) | Correction of EOS in children | The Phenix rod was the original invention. The current version available is the MAGEC rod, which is built upon the original concept, but is a different implant | MCGR is an important innovation in the management of EOS as it offers gradual, non-invasive, outpatient correction of spine deformity | Focus on enhanced longevity, improved biomechanics, and long-term safety |
SR. NO.: serial number; AI: artificial intelligence; DDH: developmental dysplasia of the hip; SCFE: slipped capital femoral epiphysis; TFO: trochanteric flip osteotomy; GT: greater trochanter; USG: ultrasonography; MAGEC: Magnetic Expansion Control; MCGR: magnetically controlled growth rods.
The field of paediatric musculoskeletal imaging has witnessed significant advancements. Recent innovations in technology combined with greater understanding of disease pathophysiology and the shifting focus on patient-centered care have revised the diagnostic panorama for many paediatric orthopaedic conditions like congenital anomalies, musculoskeletal injuries, infections, and tumors. The advent of the electron-beam X-ray imaging system (EOS) enables high-quality, low-dose biplanar radiographs and allows for 3D reconstructions. It has the advantage of giving rapid whole-body scans with highly replicable measurements [1]. This is particularly useful in paediatric musculoskeletal imaging, where frequent imaging is required for ongoing assessment and follow-up, thereby providing a safer and more effective alternative to conventional radiography.
The diagnostic potential of ultrasound has enlarged due to recent technological advancements like contrast-enhanced ultrasound (CEUS), sonoelastography, and ultra-high frequency ultrasound (UHFUS) [2–4]. This can be considered an important move in paediatric orthopaedics as ultrasound is a mainstay imaging modality for younger children due to its safety and ability for dynamic, real-time assessment. Similarly, efforts are ongoing to make computerized tomography (CT) scans safer and more useful by reducing the radiation exposure and enhancing the quality of scans. Some of the important advancements in this direction are the development of automated exposure control, lower tube voltage and current settings, high pitch acquisition, and iterative reconstruction algorithms [5, 6]. The incorporation of photon-counting detector CT (PCT) with deep learning-based reconstruction has potential for further reducing the radiation exposure and improving diagnostic accuracy in children [7]. The role of magnetic resonance imaging (MRI) in paediatric musculoskeletal imaging has also expanded due to recent advances like whole body MRI (WB-MRI) and CT-like MRI sequences [6, 8]. WB-MRI has emerged as a useful tool for assessment of multifocal and multi-system diseases like infections and tumors in paediatric orthopaedics, which need frequent follow-up [9, 10]. CT-like MRI sequences give images almost identical to those of CT without the risk of radiation and enable in-depth analysis of cortical bone and structural lesions [6]. Furthermore, the emerging MRI applications like diffusion tensor imaging, dynamic contrast-enhanced MRI, non-contrast perfusion MRI and MR spectroscopy highlight the continuing effort to expand its clinical applications in paediatric musculoskeletal imaging [11].
The support of artificial intelligence (AI) is further transforming paediatric musculoskeletal imaging by helping in the identification of pathology, quantification of disease severity, and characterizing the findings. The disease identification applications of AI are being used in the detection of challenging pathologies in children like subtle fractures, developmental dysplasia of the hip (DDH), and bone lesions [12–14]. Quantification applications are being used for the assessment of bone age and limb length discrepancy [15, 16]. A recent study has used the disease characterization potential of AI to differentiate Ewing sarcoma from osteomyelitis using a deep learning algorithm on paediatric radiographs [17]. Thus, the field of paediatric musculoskeletal imaging has undergone a significant revolution and is still witnessing metamorphosis through innovative technologies and approaches pointed towards better safety, accuracy, and efficiency.
3D printing, also known as additive layer manufacturing, is a novel technique to create a 3D object of a wide range of materials like plastic, rubber, metal, and ceramic using the reference of a digital model. It uses computer-aided technology for depositing successive layers of material to create an object of identical shape to that of the patient-specific part under consideration. Invented by Charles Hull in 1983, it has gained popularity over the last few decades and is being extensively used in different fields of medicine. It was reportedly first used in orthopaedics in 1999 by D’Urso et al. [18] to generate stereolithographic biomodels for patients undergoing surgery for complex spine deformities.
In reference to paediatric orthopaedics, 3D printing has established its usefulness in both traumatic and nontraumatic conditions. It is being used in preoperative planning and for getting 3D printed models and patient-specific instrumentation (Figure 1). Preoperative planning using a 3D printed model enables a better understanding of the pathoanatomy and helps in planning the surgical procedure more precisely. Different surgical steps like fracture reduction, osteotomies, and size and positioning of implants can be preplanned, thereby reducing the intraoperative struggle, operative time, fluoroscopic exposure, and blood loss [19]. Additionally, it ensures more accurate intraoperative corrections and hence better outcomes. It is also being used frequently in academic training and patient education [20]. Byrne et al. [21] used 3D printed patient-specific osteotomy guides and patient-specific implants for the correction of complex posttraumatic forearm deformities and reported significant improvement in alignment of bones, range of pronation and supination, grip strength, and pain. Similarly, the use of a 3D printed navigation template and osteotomy guide in the correction of cubitus varus deformity has been reported to achieve more accurate correction and better patient satisfaction [22, 23]. The use of 3D printing for complex paediatric hip and foot conditions is also well documented [24–28]. Its use in planning and executing osteotomies around the hip in cases of DDH, slipped capital femoral epiphysis (SCFE), and Legg-Calvé-Perthes (LCP) disease using patient-specific 3D navigation templates has reported better accuracy and precision and hence better outcome when compared to conventional approaches [24]. Similarly, the use of 3D printed patient-specific instrumentation has proved to be helpful in proper orientation of the blade to adequate depth, thereby ensuring complete resection of tarsal coalition and reducing the incidence of recurrence [28, 29]. Furthermore, the use of 3D printing is being explored for uses other than its surgical applications. Some of the reported applications include 3D printed limb prosthesis and the use of 3D printed fracture reduction devices instead of traditional plaster casts [30].
Thus, 3D printing is a promising tool and could improve patient care in the field of paediatric orthopaedics. However, at present, it has many limitations that restrict its use on a larger scale. The most important limitation is the need for modern equipment and resources, which may not be available at every center. Similarly, the cost involved in the application of 3D printing may not be comfortable for every healthcare center. Another important factor is the long production time of a 3D product, which limits the use of this method in an acute setting. Hence, for a wider application, it is the need of the moment to have a modified technique that is cost-effective, is readily available, and can give a 3D printed product much faster.
The treatment of CTEV was primarily invasive at the end of the last century. It consisted of extensive soft tissue releases in infancy with additional bony procedures in older children. However, the outcomes of these surgeries were not satisfactory as the feet were left weaker, stiffer, and painful. Dr. Ignacio Ponseti, after his extensive research, started treating these cases non-surgically with serial castings and tendo-Achilles tenotomy in 1952 and published this method for the first time in 1963. This new method suggested a noninvasive alternative that was effective in correcting clubfeet. Moreover, it denied the validity and effectiveness of Kite’s method, which was a popular technique in those days but did not give uniformly good results. Ponseti’s technique used the talar head as the fulcrum instead of the cuboid to correct the deformities around the talus. However, the technique was widely criticized and gained popularity only after 1996 when Ponseti published his book on congenital clubfoot and the non-surgical treatment method that he had developed. Today, the Ponseti technique has become the standard treatment for clubfoot deformity and has been consistently reported as a safe and effective method [31]. The method is simple, cheap, and doesn’t require hospitalization, which makes it useful for developing countries (Figure 2).
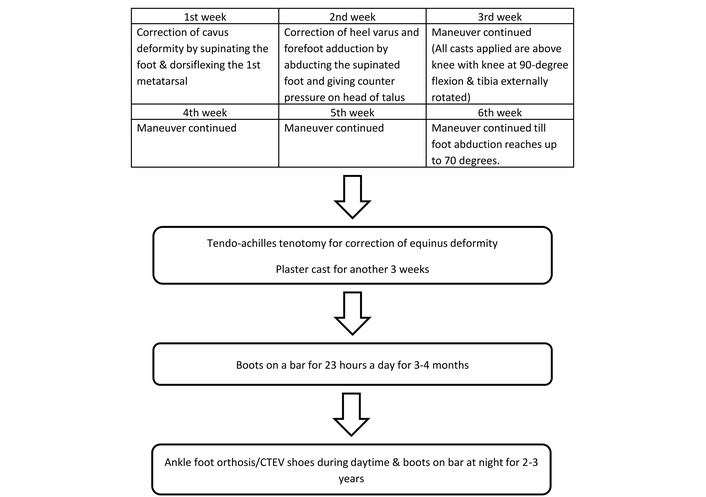
Outline of the Ponseti technique for management of clubfoot. CTEV: congenital talipes equinovarus.
However, the application of the Ponseti method involves several challenges related to technical execution, patient compliance, and logistical barriers. The learning curve is long, and it requires a certain level of skill and expertise for evaluating the deformity and applying the cast correctly. Some commonly observed complications of inappropriate casting are the “Kite” error, rocker-bottom foot, inadequate (below-knee) casting, skin issues, pressure sores, and development of atypical clubfoot. Incidents of inadequate Achilles tenotomy and neurovascular injuries while performing the tenotomy have also been reported. The clinicians should be able to differentiate syndromic and resistant clubfoot from the idiopathic ones to avoid disappointment and treatment failure. Parental non-compliance during casting and bracing protocol is a significant concern. Moreover, the management requires long-term monitoring, which may be challenging to follow. Concerns were also raised about the barriers to the uninterrupted implementation of the standard Ponseti technique [32, 33]. This includes the need for travelling every week and a relatively longer duration of casting, which may increase the dropout rates and increase the number of inadequately treated cases. With time, different accelerated regimens have been suggested to reduce the total duration of treatment, keeping the basic principles unchanged [34, 35]. Recently, a one-week accelerated Ponseti technique has been suggested to be equally effective and safe, and the correction of clubfoot is achieved in one week [36–38]. The lesser duration of casting avoids complications like disuse atrophy and osteopenia and offers faster and cost-effective correction of deformity. The children from remote areas and longer distances facing traveling issues can conveniently stay in or near the treating hospital and undergo initial correction and tenotomy in a single visit. This has been reported to improve their compliance with treatment and reduce the dropout rates [38]. Still, the expense of padding, casting, and tenotomy can be considerable for the families and a significant barrier to the implementation of this method in many countries.
The concept of SSD of the hip was initially suggested by Reinhold Ganz [39] in the early 1990s when he started working on a technique based on the hip’s vascularity, focusing on preserving the medial femoral circumflex artery (MFCA) [40]. The technique was finally introduced by Ganz et al. [39] in 2001. This method has proved its safety in preserving the vascularity of the femoral head and has become a revolution in paediatric and adolescent hip surgery [41–43]. An increasing trend in its application and indications over the last two decades has been reported [43]. The technique includes a trochanteric flip osteotomy (TFO) and ‘Z’ capsulotomy of the hip joint. The attachment of hip external rotators over the greater trochanter is preserved, thereby protecting the medial circumflex femoral artery and hence the vascularity of the femoral head. Smith et al. [44] proposed a modified technique for children less than eight years of age due to concerns of damaging the greater trochanter physis while doing the TFO. Instead of doing TFO, the modified technique consists of elevating a cartilaginous sleeve from the greater trochanter, along with the attachment of the vastus lateralis and hip abductors.
The approach of SSD of the hip offers several advantages, making it a favored approach for hip surgeries. It provides a 360° view of the femoral head and acetabulum, consequently making hip salvage possible in various traumatic as well as non-traumatic hip pathologies [45–47]. It allows evaluation and management of all types of labral, chondral, and bony abnormalities. Furthermore, when combined with the preparation of an extended retinacular flap over the femoral neck, it facilitates different femoral neck osteotomies without increasing the risk of avascular necrosis. The TFO allows relative femoral neck lengthening whenever indicated, thereby making the hip abductor mechanism better.
Even though it is a useful, safe, and favored technique, the SSD of the hip is known to have several significant complications. The most common is heterotopic ossification around the hip with an incidence as high as 37% [39, 48]. Incidence of nonunion of TFO and prominent lateral screws causing lateral thigh pain has also been reported [39, 48, 49]. These complications may be significant enough to require revision surgery [49]. To avoid these complications, some authors have suggested doing SSD through a modified Hardinge or Watson-Jones approach without TFO [50–52]. The use of SSD for addressing certain pathologies of the head and neck of the femur may lead to avascular necrosis of the femoral head in 10–20% of cases [53]. Additionally, the technique of SSD can be challenging due to its complexity and long learning curve, and hence a certain level of familiarity with the operative technique and hip anatomy, and adequate understanding of different hip pathologies is warranted for beginners.
Cerebral palsy is a nonspecific term and includes a group of permanent developmental disorders predominantly of movement and posture [54]. The brain insult is static and occurs in early childhood, but the musculoskeletal effects of this insult are dynamic and change with the growth of the child. The dynamic contractures due to spastic overactivity of muscles are known to progress into fixed contractures. Before the introduction of BoNT, different types of non-operative and operative interventions were in practice to address muscle overactivity. The non-operative measures included the use of oral relaxants (baclofen, tizanidine, and diazepam), muscle stretching exercises, use of plaster cast and splints, and the injection of neurolytic agents like phenol and alcohol into the involved muscles to produce chemo-denervation. The common neurosurgical interventions in practice were selective dorsal rhizotomy and insertion of intrathecal baclofen.
The first report of using BoNT in children with cerebral palsy was published by Koman et al. [55] in the United States in 1993. Very soon, a similar study was reported by Cosgrove et al. [56] in the United Kingdom in 1994. From then on, many reports in support of BoNT have been published, thereby establishing consensus for its role in the management of cerebral palsy [57–59]. Today, its use has become a standard treatment modality to treat dynamic contractures of both upper and lower limbs, thereby reducing the use of other treatment modalities substantially. Botulinum neurotoxins are naturally produced by bacteria of the Clostridium botulinum family and act on the neuromuscular junction to inhibit the release of acetylcholine from the presynaptic terminal (Figure 3). This produces a sort of chemical denervation of the injected muscle, thereby reducing spasticity. Its use has been reported to be safe and free from any central effect as it does not cross the blood-brain barrier. However, the incidence of systemic spread has been reported with the effects observed at other sites away from the actual site of injection. The effect is short-lived, and the muscle spasticity typically recurs after 3–6 months due to the nerve endings regaining their potential to produce acetylcholine by axonal sprouting. This leads to the need for repeated injections. Nevertheless, the muscle strength is reduced due to acute muscle atrophy, and the duration of decreased spasticity helps other rehabilitative measures to take over and produce considerable gains in the function and gait of the patient.
As of now, seven different types of botulinum neurotoxin have been described (BoNT-A to BoNT-G), but only BoNT-A and BoNT-B have been used in the treatment of cerebral palsy. The use of onabotulinum toxin A (Botox, Allergan) and abobotulinum toxin A (Dysport, Ipsen) has been extensively reported in the literature, whereas BoNT-B has been indicated in cases not responding to BoNT-A due to the presence of neutralizing antibodies. The optimal dose of BoNT depends on the type of preparation, the muscle volume in which it is being injected, the amount of spasticity, and the weight of the patient. Earlier studies have reported a total dose up to 25 units/kg body weight and a maximum total dose up to 400 units as safe for BoNT-A [60]. Similarly, appropriate doses for different muscle groups have been suggested, with the recommendation of not exceeding the dose of 50 units at a single site [61]. Furthermore, the once-a-year injection schedule has been reported to be equally effective and to have less incidence of adverse effects when compared to the more frequent injection schedules [62, 63]. The correct positioning of injecting needles is of paramount importance for achieving successful outcomes. The manual method of confirming needle placement is useful and commonly used. The use of objective methods like ultrasound supplemented with electromyography or electrical stimulation decreases the error, particularly in smaller muscle groups [64].
Even though BoNT has established its place in the management of spastic cerebral palsy, concerns have been raised from time to time regarding its overall safety and its long-term effects on the injected muscle [65–67]. Multani et al. [66] observed acute muscle atrophy after BoNT injection and reported that the reduced spasticity is not a primary effect but secondary to muscle atrophy. The injected muscle elements are partially replaced by fat and connective tissue, causing muscle fibrosis in the long term [67]. Due to these concerns, the enthusiasm for BoNT has waned in some parts of the world. The long-term studies to see these changes are missing, and hence, more work is needed to ensure the safety of this wonderful drug. As of now, the treatment should be optimized as per the available guidelines, and an attempt should be made to decrease the frequency of injections, along with an objective evaluation of muscle volume before every injection. Meanwhile, single-event multilevel surgery (SEMLS) has appeared as an important treatment modality for correcting malalignment in cerebral palsy. It involves a combination of soft tissue and bony procedures at multiple levels in the same sitting, thereby avoiding interval surgeries. SEMLS has been reported to improve gait and preserve the walking ability of children and has emerged as a standard of care for children with cerebral palsy.
The neurologic and orthopaedic abnormalities in cerebral palsy give rise to a variety of abnormal gait patterns in most of the children. These gait dysfunctions are complex to evaluate by clinical methods alone and are challenging to treat effectively in the absence of detailed relevant information. The treatment of these gait dysfunctions involves a thorough understanding of the child’s gait biomechanics and a multidisciplinary approach consisting of orthotics, physiotherapy, spasticity management, and surgery. Before the development of 3D-IGA, several methods were put into practice for the assessment of gait dysfunction, and are still being used where this facility is not available. These include observational gait analysis, standardized rating scales, and 2D digital videography. However, these methods lack sensitivity and are not reliable enough to analyze these complex gait patterns with accuracy [68, 69]. For example, 2D gait analysis cannot explain the movements that are misaligned from the observer’s point of view and misinterpret the combination of knee flexion and hip internal rotation as a valgus knee. Continuous efforts have been ongoing since the early 1980s to develop a system that can analyze and measure the gait dysfunction in a more objective way [70]. Braune and Fischer effected a breakthrough in 1890 by developing a method of 3D analysis for studying the gait kinematics (angular displacements) of lower limbs in humans [71]. The subsequent studies were able to do kinetic analysis of hip, knee, and ankle joints (forces acting across these joints) while walking [72, 73].
Over the last two decades, 3D-IGA has evolved as an important tool in the assessment of ambulant children with cerebral palsy [74]. It provides interdependent and objective data about typical and pathological gait in all three planes by defining anatomic joint and segment angles. 3D-IGA makes it possible to analyze gait kinematic and kinetic parameters simultaneously. The kinematic analysis is done with the help of reflective marker sets and infrared camera technology to track the 3D location of marker trajectories during movement. The positions of markers are then used by a biomechanical model to determine the orientation of 3D segments, which are then used to determine the joint angles. The kinematic data for each segment/joint can be shown as a series of graphs for every plane separately. Spatiotemporal gait parameters can also be calculated to measure gait speed, length, and width of step, stride length, and cadence. The force platform enables the calculation of ground reaction forces, thereby enabling the kinetic analysis of joint movement and power. Kinetic data can also be displayed in the form of graphs for each joint in different planes. Furthermore, simultaneous use of electromyography can calculate the activity of muscles by placing sensors over the surface of target muscles. The large and complex data of the overall gait can be further summarized into a single score, like the gait deviation index and gait profile score, thereby identifying specific gait deviation and its probable etiology [75, 76].
The obtained information can be used to understand the effect of abnormal gait patterns on function and can be useful to improve walking. 3D-IGA combined with clinical examination and other standardized evaluation measures leads to more appropriate treatment recommendations. It may point out those children who are not good candidates for surgery. For those requiring SEMLS, it further helps in determining the type and quantity of intervention. A good concurrence has been observed between the recommendation based on 3D-IGA and the actual surgery performed, and the use of 3D-IGA has been reported to improve the post-operative outcomes [77, 78]. Similarly, it improves the treatment outcome by helping in the formulation of a child-specific physiotherapy protocol [79]. Its role as a reliable tool for measuring the treatment outcome has also been reported [80, 81].
However, the use of 3D-IGA has certain challenges. It requires setting up a 3D gait lab, which may not be feasible at many centers due to cost issues. This makes it an out-of-reach and expensive tool for patients. The healthcare personnel may find it difficult to interpret the graphs and hence need prior training and education. A few technical limitations have also been reported. For example, measurement of kinetic data is difficult in children with small step length, and if they are unable to keep only one foot on the plate at a time. Similarly, it is difficult to perform on children with behavioral and cognitive issues as they won’t be able to follow the commands and may not be able to complete the gait analysis. It is also not possible to do kinetic analysis for children with assisted gait where the assisting device shares the load of the patient’s weight. Despite these limitations, 3D-IGA has evolved as an important armamentarium in the management of cerebral palsy. It has the potential to improve the functional outcome to a larger extent and may reduce the frequency and overall cost of surgical interventions. Performing 3D-IGA before an operative intervention may also boost the confidence of the family towards the benefits of that surgery and help them in shared decision-making for the child.
The technique of growth modulation for correcting limb deformities in skeletally immature children has undergone advancement for a century. It uses the child’s growth potential and has a definite advantage over a corrective osteotomy in being less invasive and allowing immediate weight bearing and faster restoration of activity. This technique and relevant implants have witnessed a lot of changes over the decades. Growth modulation using Blount staples served as an effective and cost-effective treatment option for many decades. However, its use declined due to higher rates of complications like implant failure, implant migration, and premature growth arrest. Since its first report by Stevens [82] in 2007, guided growth with a tension band plate (figure-of-8 plate) has become the preferred choice of most of the surgeons today and has given satisfactory results consistently [83–85]. The surgical procedure consists of applying a two-holed extra-periosteal plate and one non-locking screw on either side of the physis (Figure 4). The plate acts as a focal hinge at the periphery of physis and restricts growth on that side of the physis. Its indication has extended to correct the deformities secondary to a wide range of conditions like metabolic disorders, skeletal dysplasia, and those due to genetic and neurological disorders [86–88]. Similarly, the use has extended from correcting deformity in the coronal plane to those in the sagittal and transverse planes and has established itself as a promising option in correcting limb length discrepancy [89, 90]. Moreover, it has been tested to correct similar problems in the upper limb and has shown promising results [91, 92]. Tension band plate has been reported to be equally effective and safe when compared to staples, but offers the advantage of shorter operating time and no backout [93]. When compared to percutaneous epiphysiodesis using transphyseal screws (PETS), TBP offers the advantage of not violating the physis, showing better purchase, particularly in younger children, and having a comparable correction rate [94]. However, several studies have preferred PETS over TBP for correcting flexion contractures at the knee, as it causes less irritation when applied over the anterior aspect of the distal femur. Major applications of this technology have been the correction of coronal plane knee deformities like genu valgum and varum, similar ankle deformity of valgus, and sagittal plane deformities like flexion deformity of the knees. Recently, this technology has been applied to the hip to correct growth disorders of the femoral neck, like coxa valga and coxa vara, and for halting the growth of the greater trochanter by means of screw epiphysiodesis. The concept of rotational guided growth (RGG) has also emerged for correcting rotational deformities by applying the plates in an oblique orientation over the medial and lateral aspects of the physis [95]. Recently, an open novel device (6 plate) was described and used for RGG over the distal femur in lambs [96]. Thus, the idea of growth modulation is an important innovation in the field of paediatric orthopaedics, and this technique and relevant implants are undergoing consistent improvement.
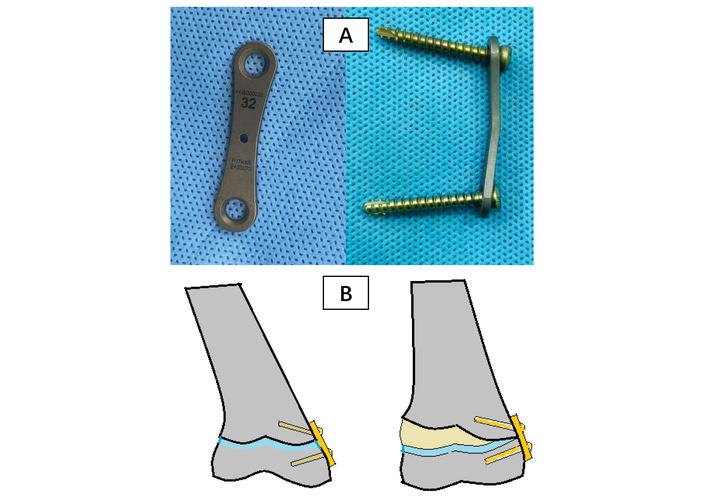
Growth modulation with tension band plate. (A) Figure-of-8 tension band plate and screws; (B) mechanism of action of tension band plate (growth modulation).
However, the procedure involves several technical, biological, and patient-related challenges that may lead to complications. The proper positioning of the implant needs an image intensifier, which may not be available in many centers. Complications related to the hardware include implant failure, screw back-out, and soft tissue irritation. The amount of correction may be unpredictable, particularly in pathological conditions. Incidence of overcorrection is frequently reported, and hence the need for regular follow-up and timely implant removal should be emphasized. Similarly, the possibility of recurrence of the original deformity (rebound phenomenon) after implant removal should be well communicated. For optimal results, the timing of growth modulation is important. The procedure requires an open growth plate and at least 1–2 years of remaining growth. While it has been used safely and effectively in children as young as 2 years old, the mean age at the time of procedure was reported as 9.5 years (2–16 years) in a systematic review by Kumar and Sonanis [97].
Distraction osteogenesis was known in the world literature since the 1900s through the work of Codivilla (1904) and later by Putti (1921). They suggested elongation of the bone through an osteotomy in a manner that allows callus formation. Between 1950 and 1960, Gavril Abramovich Ilizarov from the USSR performed several animal and clinical studies on biological principles and histologic mechanisms of distraction histogenesis in bone and soft tissues. The technique described by him used a ring fixator (registered in 1952 and known as Ilizarov fixator) to gradually separate the bone ends, thereby creating a controlled environment to stimulate new bone formation. However, the work of Ilizarov was not widely accepted initially and started getting recognition in the USSR after he successfully treated Valery Brumel (Soviet Olympic silver and gold medallist high jumper) in 1967 for nonunion of the leg bone. Similarly, it took about 30 years for his work to be introduced in Western countries after he treated the Italian journalist Carlo Mauri in 1980 [98].
The basic principles of distraction osteogenesis include a stable fixation, a low-energy osteotomy, followed by a latent period of 5–7 days and a distraction rate of 1 mm/day in 3–4 divided increments [99]. Regenerate bone develops between the cross-section of the distracted bone surfaces with a central fibrous interzone consisting of type 1 collagen. New bone trabeculae form directly from this fibrous interzone and are oriented parallel to the direction of distraction force. These trabeculae then consolidate and remodel into solid bone. Similarly, the mechanical principles of the Ilizarov frame are also well established [100]. Smaller rings are more stable than larger rings. Frame stiffness increases by 70% by 2 cm reduction in ring diameter. Other factors influencing the stability of the frame are the distance between two consecutive rings, the number of rings in each bone segment, the number of transfixing wires on each ring, the diameter and tension of transfixing wires, and the crossing angle between two wires. These principles are considered the bare minimum for achieving a good outcome and for avoiding the complications of the procedure.
Today, Ilizarov principles have become the mainstay for managing bone defects, limb length discrepancy, and for achieving gradual deformity correction [101]. The Ilizarov ring fixator provides all-around stable fixation of bone with the advantage of soft tissue preservation and adjustability. These features make it the preferred tool for managing many challenging orthopaedic problems in children like infected nonunion, bone tumors, and congenital limb disorders. Over the last two decades, the original Ilizarov method has been modified in various ways, but the principles remain unchanged. These modifications were brought out to circumvent the commonly seen drawbacks with the use of the frame. Different types of unilateral external fixators have been used to avoid heavy and cumbersome ring frames. Combined techniques of bone transport over an intramedullary nail or a plate were developed to shorten the total fixator duration. Other studies have performed bone transport with simultaneous use of antibiotic spacers to reduce the incidence of infection. Intramedullary limb lengthening devices were invented to replace the fixator, thereby avoiding the complications of the external frame. Furthermore, the original ring fixator itself has undergone several transformations in its design and application. Different designs using computer-based software have been developed. Today, six-axis external fixation devices are routinely used in children for correction of complex limb deformity and limb length discrepancy, and have added a new dimension to the mechanics of the traditional Ilizarov fixator. It provides better stability, thereby reducing instability-related issues like nonunion, delayed union, and secondary deformity [102]. The deformity correction can be planned and accomplished more accurately in a shorter time duration [103]. The use of computer-based software makes the procedure simple and allows correction of angulation, translation, rotation, and length discrepancy simultaneously without changing the hinge construct sequentially. The science and technology involved with the hexapod frame are evolving continuously. The possibilities in the future include further improvements in the design of hardware and software, leading to a more refined system with more precision, fewer complications, and which will be more user-friendly. Its amalgamation with other advancements in technology, like robotics and augmented reality, may help expand the indications to other areas beyond deformity correction. The construction of patient-specific frames through 3D printing may further improve the treatment outcome.
The science behind the six-axis external fixation device (hexapod device) started in the 17th century with the natural observation of multiple parallel limbs (quadruped, hexapod, octopod) providing better stability and maneuverability. A six-axis mechanism was designed by Stewart and Gough in the 1960s (Stewart Gough platform) and was extensively applied for flight simulators, amusement park rides, and robotics [98, 99].
The first patent of a hexapod circular frame for use in orthopaedics was submitted in 1985 by Philippe Moniot in France, followed by S.I. Pisler in 1989 in the Soviet Union. However, these devices didn’t use mathematical solutions to correct the deformity. The first hexapod device to use a computer-based program for deformity correction was developed by Harold Taylor and Charles Taylor in 1994 in Tennessee and became popular as the Taylor Spatial Frame Device. Since then, different designs using computer-based software have been developed and have revolutionized the treatment of limb deformity. In the SUV frame (Russia, 2010), only three struts are connected to each ring, while the other three struts are connected to the side of the earlier mounted strut. This offers a comparatively easier calculation independent of the size and shape of the mounted rings and is the most modular hexapod available as of now. The Adam frame (Turkey, 2009) is an octopod instead of a hexapod and has the advantage of allowing a greater range of movement between the two rings.
Today, six-axis external fixation devices are routinely used in children for correction of complex limb deformity and limb length discrepancy and have added a new dimension to the mechanics of the traditional Ilizarov fixator [100, 101]. It provides better stability, thereby reducing the instability-related issues like nonunion, delayed union, and secondary deformity [102]. The deformity correction can be planned and accomplished more accurately in a shorter time duration [103]. The use of computer-based software makes the procedure simple and allows correction of angulation, translation, rotation, and length discrepancy simultaneously without changing the hinge construct sequentially. Thus, the innovation of the six-axis fixation device has increased the versatility in paediatric deformity correction and limb length equalization. The science and technology involved with the hexapod frame are evolving continuously. The possibilities in the future include further improvements in the design of hardware and software, leading to a more refined system with more precision, fewer complications, and which will be more user-friendly. Its amalgamation with other advancements in technology, like robotics and augmented reality, may help expand the indications to other areas beyond deformity correction. The construction of patient-specific frames through 3D printing may further improve the treatment outcome.
Traditionally, limb lengthening was done with the help of external fixators like the Ilizarov frame and limb reconstruction system (LRS) [104, 105]. Though this method is highly effective and still very popular, there are many associated challenges and complications like pin tract infection, soft tissue tethering, joint stiffness, fracture of the regenerate, and a long fixator time [106, 107]. Moreover, external fixators are often cumbersome and unacceptable to the patients, subsequently affecting their routine activities and mental health adversely. Several attempts have been made to reduce the total fixator time during limb lengthening by innovative techniques like lengthening over a nail or plate and internal fixation after completion of distraction [108, 109]. However, issues related to the use of external frames persisted with these hybrid techniques, leading to the invention of intramedullary limb lengthening devices.
The initial internal limb lengthening nails were operated by a ratchet mechanism, where patients were required to rotate their limb manually at the osteotomy site to produce lengthening [110–112]. This maneuver was often painful, and the device also had issues of poor control over the rate of lengthening [113, 114]. Furthermore, the process was limited to the distraction of the osteotomy site, and compression, if needed, was not possible. The present-day technology of externally controlled motorized intramedullary lengthening nail was first introduced by Baumgart et al. [115] in 1997. The FITBONE (originally Wittenstein, Igersheim, Germany, and currently Orthofix, Inc., Lewisville, TX, USA) uses a subcutaneous transceiver connected to the intramedullary nail. An external transducer is placed directly over this receiver to pass radiofrequency waves, which in turn drives the nail motor to produce axial movements. The PRECICE NAIL (Nuvasive Inc, San Diego, California) was introduced in 2012 and there have been three generations of this device till now [116]. The telescopic rod is lengthened by a magnetic drive mechanism in the rod and is operated by an external remote controller containing two rotating earth magnets. Both these nails can be moved in either direction with accuracy to produce distraction and compression. Following the introduction of these nails, several studies have reported promising results and motorized intramedullary lengthening nails have become the implant of choice for limb lengthening in many countries [117–119].
Though the motorized intramedullary lengthening nail avoids most of the complications of external fixators, a few challenges and complications of this technique have been reported. The incidence of implant failure and stuck nail has been reported in several studies [120–122]. Moreover, the risk of soft tissue contractures and joint subluxation/dislocation persists. These complications can be avoided by spanning the fixation across the proximal and distal joints when using an external fixator, but this is not possible when using a nail. Use of a full-time dynamic brace, BoNT injection, and a slower rate of distraction has been suggested to avoid the incidence of joint complications [123]. Another challenge is regarding its use in skeletally immature children. The risk of developing avascular necrosis of femur head is a potential complication with antegrade nailing of the femur. Moreover, an open growth plate and a narrow canal diameter preclude its use in younger children. The smallest available nail diameter today is 8.5 mm, which is often difficult to use in children until they attain the age of 10 years. The concept of extra-medullary nailing for internal limb lengthening has been reported, where the nail is placed in a submuscular position near the lateral cortex of the femur and the posteromedial border of the tibia, respectively [124–126]. Though this concept seems attractive, it cannot be validated as of now due to limited evidence and needs further research on a larger sample with a longer follow-up. Furthermore, the issue of limited surgical training, higher implant cost, and implants not being easily available limits its use on a larger scale, particularly in developing countries.
The management of EOS remains challenging, and a variety of treatment modalities, both surgical and non-surgical, have been reported over the last century. Mehta’s cast has been accepted as a highly effective, non-operative treatment modality, particularly for children below four years of age [127]. The method utilizes elongation, derotation, and flexion, thereby applying 3D forces to correct the deformity. The notion of using growth rods for cases not responding to non-surgical methods has been put into practice since the first report of the Harrington rod for this purpose in 1962 [128]. Subsequently, several modifications were reported to improve the outcome of this technique [129–131]. The concept of traditional growing rods was introduced by Akbarnia et al. [132, 133], suggesting the use of two rods instead of a single rod, and was reported to be more effective in the management of EOS. However, traditional growth rods need serial distraction every six to nine months till the child attains maturity. This is a significant disadvantage as the distraction process is invasive and needs hospitalization and general anaesthesia. Moreover, a notable rate of procedure-related complications was reported with an additional 24% increased risk of complications with every successive distraction [134].
The magnetically controlled growing rods for EOS were introduced in 2004 with the objective of avoiding invasive serial distractions [135]. At present, Magnetic Expansion Control (MAGEC, NuVasive, San Diego, USA) is the only available magnetically controlled growth rod system approved by the Food and Drug Administration (FDA). The spinal distraction is executed as an outpatient procedure by a manually operated magnetic external remote controller. The controller is placed near the operating area over the internal magnet in the MAGEC rod to develop an external magnetic field. This produces rotation in the internal magnet, thereby causing lengthening of the rod. Since its invention, the effectiveness of MAGEC rods in producing satisfactory distraction and modeling of the curve and hence preserving the pulmonary function in EOS has been reported in many studies [136–138]. It has significant advantages as a noninvasive outpatient department (OPD) procedure and hence avoids the complications and cost associated with the serial distractions of traditional growth rods. This reduces the physical and psychological burden on the patients and has been observed to improve their health-related quality of life [139]. Despite the potential advantages, some complications have been observed with the technique of MCGR. The most reported complications are implant failure, prominence of the end of the rod, and loss of initial height. Failures of distraction, breakage of connection, and proximal junctional kyphosis have also been reported [140]. Another important concern is about the deposition of metal debris and the concentration of metal ions in the adjacent soft tissues [141]. However, the overall rate of complication was observed to be much less when compared to that of traditional growth rods [142]. Thus, MCGR is an important innovation in the management of EOS and provides better patient satisfaction by reducing the physical, socioeconomic, and psychological burden of repeated invasive distractions.
The field of paediatric orthopaedics has observed many other significant evolutions which cannot be overlooked and are mentioned here in brief, though equally important. Descriptions of minimally invasive surgical techniques like arthroscopy and endoscopy have brought a positive change by reducing the morbidity and allowing faster post-operative recovery. Today, the titanium elastic nail system (TENS) developed in Nancy (France) in 1988, is one of the widely accepted, minimally invasive treatment modalities for long bone fractures in children. It provides the required stability but allows micromotion at the fracture site, which helps in early union. Moreover, it offers several advantages like less soft tissue damage, less blood loss, early weight bearing and rehabilitation, easy implant removal, and fewer chances of physeal injury [143]. The availability of paediatric-specific specialized instruments allows surgeons to place smaller incisions and still have better visualization of the operative field. The evolution of computer-assisted surgical techniques has enabled surgeons to perform surgery with greater accuracy. Similarly, the availability of orthopaedic implants specifically designed for children has enabled better stability and hence improvement of the surgical outcomes. The use of biocompatible material in implant design encourages bone growth and reduces the risk of adverse reactions. These improvements in material science offer better durability and less wear, thereby reducing the incidence of implant failure and frequent replacements. The development of various intraoperative assistance technologies like navigation and robotics has made complex paediatric orthopaedic procedures like correction of spine deformities easier, more accurate, and safer. The advancements in intraoperative neuromonitoring (IONM) have enabled its use in real-time assessment of the nervous system during difficult spine and limb surgeries, and its use has become a standard practice during these surgeries [144].
Recent technological advances in the field of orthotics have benefited children in their non-operative management. The newer custom-made orthotic devices provide a perfect fit and are known to be more comfortable and lighter in weight. It therefore increases the confidence of children, improves their compliance, and enables them to engage in activities with minimal limitations. Physical therapy and rehabilitation sessions are now enriched using newer equipment for increasing muscle strength, joint flexibility, and overall mobility. Innovative treatment approaches like comprehensive care are now adjusted to the specific needs of patients and focus not only on the physical aspect but also on the psychological and emotional issues, thereby improving the overall quality of life. The availability of advanced technologies like telemedicine and remote monitoring has further improved the accessibility and continuous care of patients even in remote areas.
The arrival of AI and its subspecialties has added newer perspectives to the field of paediatric orthopaedics. It has significantly improved the accuracy of diagnosis, treatment outcome, and postoperative rehabilitation guidance, and can also help in the diagnosis and management of intraoperative complications. Besides that, it serves as an important tool helping the orthopaedic surgeons in other areas like research assistance, academic teaching, and improved communication.
The field of paediatric orthopaedics continues to evolve rapidly, and the ongoing research and growth have the potential to bring more revolutionary innovations in the future. Several domains, like regenerative medicine and genetics, offer exciting prospects for the future. Through ongoing research in stem cells and regenerative medicine, attempts are being made to regenerate tissues and improve healing. This could significantly alter the treatment of many paediatric orthopaedic conditions like bone defects, osteogenesis imperfecta, and cerebral palsy. Genetic therapies like gene editing and gene therapy can correct the underlying genetic abnormality through a targeted approach. Similarly, further advancements in biomedical engineering have the potential to improve the treatment outcomes further by availing innovative devices, implants, and orthotics. The sustained improvement in AI indicates the possibility of bringing more improvements in paediatric orthopaedic practices.
Despite having an encouraging time ahead, these innovative solutions face certain challenges in their application. Many of them are still not easily accessible. Similarly, these modern innovations may be expensive and unaffordable to a large population. Hence, efforts are needed to equilibrate between providing high-quality care and making it more cost-effective and readily available for all needy children. Ethical concerns in implementing these innovations should also be taken into consideration to ensure the safety of children. Parents should be informed and consented to before applying these modalities. Moreover, constant monitoring and surveillance are essential to warrant their long-term efficacy and safety. Taking these challenges into account, one can anticipate great potential for improvement in the field of paediatric orthopaedics with even more ground-breaking innovative solutions.
In summary, the recent innovations in the field of paediatric orthopaedics indicate a paradigm shift towards precision, preservation, and patient-focused individualized care. They have helped in reducing disability and improving function, thereby enhancing survival and quality of life for children worldwide. Looking forward, the future of paediatric orthopaedics lies in amalgamating genetic information, biophysical modelling, and modern health technologies to customize interventions to each child’s unique growth potential and distinct functional needs. Continued research, long-term outcome studies, and impartial access to innovation will be essential to sustain progress.
3D: three-dimensional
3D-IGA: three-dimensional instrumented gait analysis
AI: artificial intelligence
BoNT: botulinum toxin
CT: computerized tomography
CTEV: congenital talipes equinovarus
DDH: developmental dysplasia of the hip
EOS: early onset scoliosis
MAGEC: Magnetic Expansion Control
MCGR: magnetically controlled growth rods
MRI: magnetic resonance imaging
PETS: percutaneous epiphysiodesis using transphyseal screws
RGG: rotational guided growth
SEMLS: single-event multilevel surgery
SSD: safe surgical dislocation
TBP: tension band plates
TFO: trochanteric flip osteotomy
WB-MRI: whole body magnetic resonance imaging
ANJ: Conceptualization, Writing—original draft, Writing—review & editing. RAP: Data curation, Formal analysis, Writing—original draft, Writing—review & editing. Both authors read and approved the submitted version.
Ashok N. Johari, who is the Associate Editor and Guest Editor of Exploration of Musculoskeletal Diseases, had no involvement in the decision-making or the review process of this manuscript. The remaining author declares that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Ali Yüce ... Abdülhamit Misir
Tanja Neussl ... Johannes Dominikus Pallua
Ashok N. Johari
