Affiliation:
1Department of Internal Medicine, ESIC Post Graduate Institute of Medical Sciences and Research, New Delhi 110015, India
Affiliation:
1Department of Internal Medicine, ESIC Post Graduate Institute of Medical Sciences and Research, New Delhi 110015, India
Email: dramit_67@yahoo.com
ORCID: https://orcid.org/0000-0001-9348-7601
Affiliation:
1Department of Internal Medicine, ESIC Post Graduate Institute of Medical Sciences and Research, New Delhi 110015, India
Affiliation:
2Indian Council of Medical Research, New Delhi 110029, India
ORCID: https://orcid.org/0000-0002-9129-4686
Explor Musculoskeletal Dis. 2026;4:1007118 DOI: https://doi.org/10.37349/emd.2026.1007118
Received: January 05, 2026 Accepted: February 11, 2026 Published: March 26, 2026
Academic Editor: Joan M. Nolla, University of Barcelona, Spain
Aim: Peripheral neuropathy is a frequent but often under-recognized extra-articular manifestation of rheumatoid arthritis (RA), frequently linked to chronic systemic inflammation. The neutrophil-to-lymphocyte ratio (NLR) has emerged as a simple marker of systemic inflammatory burden. This study aimed to investigate the association between NLR and peripheral neuropathy in RA.
Methods: This cross-sectional study included 230 RA patients. Peripheral neuropathy was identified through clinical evaluation and nerve conduction studies. Demographic and clinical data, serological status, disease activity [28-joint Disease Activity Score (DAS28)], inflammatory markers, and complete blood counts were obtained. NLR was calculated from absolute neutrophil and lymphocyte counts. Multivariable logistic regression was used to identify factors associated with peripheral neuropathy. Discriminatory performance of NLR was evaluated using receiver operating characteristic (ROC) curve analysis.
Results: Peripheral neuropathy was present in 93 of 230 patients (40.4%). Patients with neuropathy exhibited significantly higher NLR compared with those without neuropathy (median 3.8 vs. 2.3; P < 0.001). In multivariable logistic regression adjusting for age, disease duration, disease activity (DAS28), C-reactive protein (CRP), serological status, and glucocorticoid use, elevated NLR remained independently associated with the presence of peripheral neuropathy [adjusted odds ratio (OR) = 1.92, 95% confidence interval (CI): 1.48–2.49; P < 0.001]. Other factors significantly associated with neuropathy included older age, longer disease duration, higher DAS28, and seropositive status. CRP and glucocorticoid use were not significantly associated with neuropathy in the adjusted model. In the overall cohort, the model including NLR demonstrated significantly improved discrimination for peripheral neuropathy compared with the base model without NLR, with the area under the ROC curve (AUC) increasing from 0.75 (95% CI: 0.69–0.81) to 0.83 (95% CI: 0.77–0.89) after including NLR (P < 0.001).
Conclusions: Elevated NLR is independently associated with the presence of peripheral neuropathy in RA after adjustment for major confounders, and it demonstrates incremental discriminatory value for distinguishing neuropathy status in RA.
Rheumatoid arthritis (RA) is a systemic autoimmune disease characterized by chronic synovial inflammation and progressive joint destruction. Peripheral neuropathy is a frequent extra-articular manifestation of the disease. Electrophysiological studies report prevalence rates of approximately 30–40% in RA; however, substantial variability exists across cohorts, with some studies reporting lower prevalence (around 8%) and others demonstrating higher rates up to 75%, with many cases remaining subclinical and detected only on nerve conduction testing [1–5]. Clinically, neuropathy in RA most often presents with predominantly sensory symptoms such as numbness, tingling, or pain in the hands and feet and may also include sensorimotor involvement, mononeuritis multiplex, or focal entrapment neuropathies such as carpal tunnel or tarsal tunnel syndromes [4–6]. These manifestations can overlap with arthritis symptoms, significantly affecting the function and quality of life.
Chronic inflammation is central to the pathogenesis of RA. Conventional markers such as erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and composite indices like 28-joint Disease Activity Score (DAS28) are widely used to assess inflammatory burden and disease activity. However, these measures often fail to capture persistent, subclinical inflammation that contributes to long-term complications. Emerging evidence highlights the neutrophil-to-lymphocyte ratio (NLR) as a simple, cost-effective, and readily available biomarker that reflects systemic immune dysregulation by integrating both innate and adaptive immune responses. Elevated NLR typically indicates heightened inflammation, characterized by neutrophilia alongside relative lymphopenia. In RA, NLR is consistently higher than in healthy controls and shows strong correlation with disease activity [7–9]. Previous research suggests that NLR demonstrates a graded association with increasing disease activity and severity [10].
Despite growing interest in NLR as a marker of disease activity in RA, its role in identifying extra-articular complications such as peripheral neuropathy has not been systematically investigated. In other chronic diseases, including diabetic peripheral neuropathy, elevated NLR has been linked to the presence and severity of nerve dysfunction, supporting the concept that subclinical inflammation contributes to neural injury [11].
Serological markers such as rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP) are established indicators of autoimmune status in RA and have been associated with more severe disease and extra-articular manifestations. These autoantibodies may identify patients with higher inflammatory burden and therefore greater risk of complications [12, 13].
In this context, we proposed that elevated NLR would be associated with the presence of peripheral neuropathy in RA. The aim of this study was to examine the association between NLR and the presence of peripheral neuropathy in RA and to evaluate its potential as a simple biomarker for characterizing neural involvement. We also explored the relationship between NLR and serological status to determine whether autoimmune profiles influence the inflammatory environment linked to neuropathy.
This cross-sectional observational study was conducted at the Rheumatology Division of Department of Internal Medicine, ESIC Post Graduate Institute of Medical Sciences and Research, Basaidarapur, New Delhi, between April 2024 and July 2025. Consecutive adult patients (≥ 18 years) fulfilling the 2010 American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) classification criteria for RA were recruited [14]. Exclusion criteria included diabetes mellitus, chronic kidney disease, hypothyroidism, vitamin B12 deficiency, alcohol use disorder, known neurotoxic drug exposure, active infection, malignancy, or any other condition known to cause peripheral neuropathy. The institutional ethics committee approved the protocol, and all participants provided written informed consent prior to enrolment. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Baseline demographic data (age, sex), disease characteristics (duration of RA, current medications), and clinical features were documented. Disease activity was assessed using the DAS28 incorporating ESR. Serological status was determined by testing for RF and anti-CCP antibodies. Patients were classified as seropositive if either RF or anti-CCP was positive and seronegative if both were negative. Information on ongoing medications was documented, including glucocorticoids (with doses expressed as daily prednisolone equivalents), conventional disease-modifying antirheumatic drugs (DMARDs), biologic agents [including tumor necrosis factor-α (TNF-α) inhibitors and anti-interleukin-6 (IL-6) agents], targeted synthetic DMARDs [e.g., Janus kinase (JAK) inhibitors], and non-steroidal anti-inflammatory drugs (NSAIDs).
All participants underwent standardized nerve conduction studies (NCS) performed by experienced neurophysiologists blinded to clinical and laboratory data. Sensory and motor nerve studies were conducted in the upper and lower limbs, including bilateral median, ulnar, peroneal, and sural nerves, according to established protocols. Peripheral neuropathy was defined based on abnormal conduction velocities, prolonged distal latencies, or reduced amplitudes, in accordance with normative values adjusted for age and limb temperature. Patients with abnormalities in NCS but no neurological symptoms were categorized as having subclinical neuropathy, while those with sensory symptoms, weakness, or reflex changes attributable to neuropathy were classified as clinical neuropathy.
Peripheral venous blood samples were collected at baseline before electrophysiological testing. Complete blood counts were obtained using an automated hematology analyzer (Mythic 70, Cormay Diagnostics, Poland). NLR was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count. ESR was measured by the Westergren method, and CRP was quantified using a high-sensitivity immunoturbidimetric assay. Serological testing for RF and anti-CCP was performed using standard immunoassays.
All analyses were conducted using SPSS version 25.0 (IBM Corp., USA). Continuous variables were expressed as mean ± standard deviation (SD) or median with interquartile range (IQR), depending on distribution. Categorical variables were reported as frequencies and percentages. Comparisons were performed using Student’s t-test or Mann–Whitney U test for continuous variables and chi-square test for categorical variables. Comparisons of NLR across neuropathy subgroups (no neuropathy, subclinical neuropathy, clinical neuropathy) were conducted using Kruskal-Wallis tests with post hoc pairwise analysis. Multivariable logistic regression was performed to assess factors independently associated with peripheral neuropathy. Glucocorticoid use was included in the model due to its potential influence on NLR. Adjusted odds ratios (ORs) with 95% confidence intervals (CIs) were reported. To evaluate the ability of clinical models to distinguish between patients with and without peripheral neuropathy, we conducted discrimination and reclassification analyses. Receiver operating characteristic (ROC) curves were constructed to assess the discriminative performance of a base model comprising CRP, DAS28-ESR, disease duration, and serological status, and a combined model that additionally incorporated NLR. The area under the ROC curve (AUC) and corresponding 95% CIs were calculated for the overall cohort as well as within serological subgroups. Differences in AUC between models were compared using DeLong’s test. To quantify the incremental value of adding NLR to the base model, net reclassification improvement (NRI) and integrated discrimination improvement (IDI) statistics were computed for the overall cohort and within seropositive and seronegative subgroups. These metrics estimate the extent to which the combined model more accurately reclassifies individuals relative to the base model. All discrimination and reclassification analyses were performed using R (version 4.3.2; R Foundation for Statistical Computing, Vienna, Austria) with appropriate packages. All statistical tests were two-tailed, and a P value < 0.05 was considered statistically significant.
Of the 230 patients with RA included in the study, 93 (40.4%) had peripheral neuropathy confirmed by clinical and electrophysiological assessments, while 137 (59.6%) had no neuropathy. Table 1 demonstrates the demographic, clinical, serological, and treatment characteristics of the study cohort. Patients with neuropathy were significantly older than those without neuropathy (mean age 56.2 ± 10.9 years vs. 47.4 ± 12.8 years, P < 0.001) and had a longer median disease duration (10.9 vs. 5.1 years, P < 0.001). No significant differences were observed in sex distribution between the two groups (P = 0.60). Disease activity, as measured by DAS28, was significantly greater among patients with peripheral neuropathy (5.7 ± 0.9 vs. 4.5 ± 1.0, P < 0.001). When stratified by serological status, 155 patients (67.4%) were seropositive, and 75 (32.6%) were seronegative. Seropositivity was more frequent in the neuropathy group than in patients without neuropathy (76.3% vs. 61.3%, P = 0.02). Traditional inflammatory markers were also elevated in patients with neuropathy. Mean ESR was significantly higher in the neuropathy group compared with those without neuropathy (59.8 ± 18.1 mm/h vs. 44.2 ± 17.3 mm/h, P < 0.001), and median CRP levels were likewise greater among patients with neuropathy (13.4 vs. 6.8 mg/L, P < 0.001). Patients with peripheral neuropathy exhibited significantly higher neutrophil counts and lower lymphocyte counts compared with those without neuropathy, which translated into a markedly elevated NLR (median 3.8 vs. 2.3, P < 0.001). With respect to treatment characteristics, a significantly greater proportion of patients with peripheral neuropathy were receiving glucocorticoids compared with those without neuropathy (62.4% vs. 44.5%, P = 0.006), and the median daily prednisolone equivalent dose was significantly higher in the neuropathy group (P < 0.001). There were no significant differences between the groups in the use of methotrexate, other non-biologic DMARDs (including sulfasalazine, leflunomide, hydroxychloroquine, etc.), biologic DMARDs, or JAK inhibitors. The majority of the patients in both groups were prescribed NSAIDs.
Baseline characteristics of the study cohort (n = 230).
| Characteristics | Neuropathy(n = 93) | No neuropathy(n = 137) | P value |
|---|---|---|---|
| Age, years (mean ± SD) | 56.2 ± 10.9 | 47.4 ± 12.8 | < 0.001 |
| Female sex, n (%) | 74 (79.6) | 105 (76.6) | 0.60 |
| Male sex, n (%) | 19 (20.4) | 32 (23.4) | 0.60 |
| Disease duration, years (median, IQR) | 10.9 (6.0–16.0) | 5.1 (2.3–8.4) | < 0.001 |
| DAS28 (mean ± SD) | 5.7 ± 0.9 | 4.5 ± 1.0 | < 0.001 |
| Seropositive (RF and/or anti-CCP), n (%) | 71 (76.3) | 84 (61.3) | 0.02 |
| ESR, mm/h (mean ± SD) | 59.8 ± 18.1 | 44.2 ± 17.3 | < 0.001 |
| CRP, mg/L (median, IQR) | 13.4 (7.0–24.1) | 6.8 (3.2–12.5) | < 0.001 |
| NLR (median, IQR) | 3.8 (2.7–5.0) | 2.3 (1.6–3.1) | < 0.001 |
| Treatment | |||
| Glucocorticoid use, n (%) | 58 (62.4) | 61 (44.5) | 0.006 |
| Daily prednisolone equivalent dose, mg (median, IQR) | 7.5 (5–10) | 5.0 (2.5–7.5) | < 0.001 |
| Methotrexate use, n (%) | 63 (67.7) | 96 (70.1) | 0.70 |
| Other non-biologic DMARDs, n (%) | 42 (45.2) | 54 (39.4) | 0.34 |
| Biologic DMARDs, n (%) | 22 (23.7) | 31 (22.6) | 0.85 |
| JAK inhibitors, n (%) | 12 (12.9) | 15 (10.9) | 0.64 |
| NSAIDs, n (%) | 81 (87.1) | 115 (83.9) | 0.50 |
Continuous variables were compared using Student’s t-test (normally distributed) or Mann–Whitney U test (non-normal). Categorical variables were compared using the chi-square test. SD: standard deviation; IQR: interquartile range; DAS28: 28-joint Disease Activity Score; RF: rheumatoid factor; anti-CCP: anti-cyclic citrullinated peptide; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; NLR: neutrophil-to-lymphocyte ratio; DMARDs: disease-modifying antirheumatic drugs; JAK: Janus kinase; NSAIDs: non-steroidal anti-inflammatory drugs.
When stratified by serological status, NLR differed significantly according to the presence of neuropathy (Table 2). Among seropositive subjects without neuropathy, the median NLR was 2.4 (IQR: 1.7–3.2). In contrast, seropositive patients with neuropathy had a significantly higher median NLR of 4.1 (IQR: 3.0–5.4) (P < 0.001 vs. seropositive without neuropathy). Similarly, within the seronegative subgroup, individuals with neuropathy exhibited a higher median NLR of 3.3 (IQR: 2.5–4.5) compared with seronegative patients without neuropathy, who had a median NLR of 2.2 (IQR: 1.5–3.0) (P < 0.001).
Neutrophil-to-lymphocyte ratio (NLR) according to neuropathy status stratified by serological subgroup.
| Subgroup | n | NLR (median, IQR) | P value (vs. no neuropathy) |
|---|---|---|---|
| Seropositive without neuropathy | 84 | 2.4 (1.7–3.2) | - |
| Seropositive with neuropathy | 71 | 4.1 (3.0–5.4) | < 0.001 |
| Seronegative without neuropathy | 53 | 2.2 (1.5–3.0) | - |
| Seronegative with neuropathy | 22 | 3.3 (2.5–4.5) | < 0.001 |
NLR comparisons between groups were performed using the Mann–Whitney U test. IQR: interquartile range.
Further analysis by neuropathy subtype showed a stepwise increase in NLR (Table 3). Patients with subclinical neuropathy (n = 39) had a significantly higher median NLR of 3.2 (IQR: 2.4–4.1) than those without neuropathy (P < 0.001). Median NLR was even greater in patients with clinical neuropathy (4.6, IQR: 3.3–5.8), and this difference was significant compared with the subclinical neuropathy group (P < 0.001).
Neutrophil-to-lymphocyte ratio (NLR) according to neuropathy severity.
| Neuropathy status | n | NLR (median, IQR) | P value |
|---|---|---|---|
| No neuropathy | 137 | 2.3 (1.6–3.1) | - |
| Subclinical neuropathy | 39 | 3.2 (2.4–4.1) | < 0.001 vs. no neuropathy |
| Clinical neuropathy | 54 | 4.6 (3.3–5.8) | < 0.001 vs. subclinical |
Differences across the three groups were assessed using the Kruskal-Wallis test. Pairwise comparisons were performed with the Mann–Whitney U test. IQR: interquartile range.
A multivariable logistic regression analysis was performed to identify factors independently associated with peripheral neuropathy (Table 4). In the model adjusted for age, disease duration, DAS28, CRP, serological status, and glucocorticoid use, higher NLR was significantly associated with increased odds of peripheral neuropathy (adjusted OR = 1.92, 95% CI: 1.48–2.49; P < 0.001). Older age (adjusted OR = 1.02, 95% CI: 1.00–1.04; P = 0.02), longer disease duration (adjusted OR = 1.07, 95% CI: 1.02–1.12; P = 0.005), higher DAS28 (adjusted OR = 1.45, 95% CI: 1.11–1.89; P = 0.005), and seropositive status (adjusted OR = 1.51, 95% CI: 1.01–2.32; P = 0.04) were also significantly associated with neuropathy. CRP and glucocorticoid use were not significantly associated with peripheral neuropathy in the adjusted model.
Multivariable logistic regression analysis of factors associated with neuropathy.
| Variable | Adjusted OR | 95% CI | P value |
|---|---|---|---|
| NLR | 1.92 | 1.48–2.49 | < 0.001 |
| Age | 1.02 | 1.00–1.04 | 0.02 |
| Disease duration | 1.07 | 1.02–1.12 | 0.005 |
| DAS28 | 1.45 | 1.11–1.89 | 0.005 |
| CRP | 1.02 | 0.98–1.06 | 0.28 |
| Seropositive status | 1.51 | 1.01–2.32 | 0.04 |
| Glucocorticoid use | 1.28 | 0.80–2.06 | 0.30 |
Adjusted odds ratios (ORs) were estimated using multivariable logistic regression analysis. NLR was included as a continuous variable. Statistical significance of each variable was assessed using the Wald chi-square test. CI: confidence interval; NLR: neutrophil-to-lymphocyte ratio; DAS28: 28-joint Disease Activity Score; CRP: C-reactive protein.
To evaluate the incremental value of NLR for discriminating between patients with and without peripheral neuropathy, we compared models with and without NLR (Table 5). In the overall cohort (Figure 1A), the base model including DAS28-ESR, CRP, serological status, and disease duration yielded an AUC of 0.75 (95% CI: 0.69–0.81). The addition of NLR significantly improved the model performance, as reflected by a higher AUC (0.83, 95% CI: 0.77–0.89; P < 0.001) and better reclassification and discrimination metrics. When the cohort was analyzed by serological status, the pattern remained consistent. In both seropositive (Figure 1B) and seronegative (Figure 1C) subgroups, adding NLR to the base model led to significant increases in the AUC and better classification metrics, indicating that NLR enhanced the model’s ability to discriminate patients with peripheral neuropathy regardless of serological status.
Incremental diagnostic value of NLR in discriminating peripheral neuropathy in rheumatoid arthritis (RA).
| Model/Subgroup | AUC(95% CI) | P value vs. base | NRI(95% CI) | IDI(95% CI) |
|---|---|---|---|---|
| Overall cohort | ||||
| Base model | 0.75 (0.69–0.81) | - | - | - |
| Base model + NLR | 0.83 (0.77–0.89) | < 0.001 | 0.18 (0.07–0.29) | 0.06 (0.02–0.10) |
| Seropositive RA | ||||
| Base model | 0.74 (0.67–0.81) | - | - | - |
| Base model + NLR | 0.82 (0.75–0.88) | < 0.001 | 0.16 (0.05–0.27) | 0.05 (0.02–0.09) |
| Seronegative RA | ||||
| Base model | 0.71 (0.62–0.80) | - | - | - |
| Base model + NLR | 0.79 (0.71–0.86) | 0.002 | 0.15 (0.04–0.26) | 0.04 (0.01–0.08) |
Base model included CRP, DAS28-ESR score, disease duration, and serological status. Model discrimination was assessed using ROC curve analysis. NRI and IDI were calculated to evaluate the improvement in predictive performance after the inclusion of NLR. NLR: neutrophil-to-lymphocyte ratio; AUC: area under the receiver operating characteristic curve; CI: confidence interval; NRI: net reclassification improvement; IDI: integrated discrimination improvement; CRP: C-reactive protein; DAS28: 28-joint Disease Activity Score; ESR: erythrocyte sedimentation rate; ROC: receiver operating characteristic.
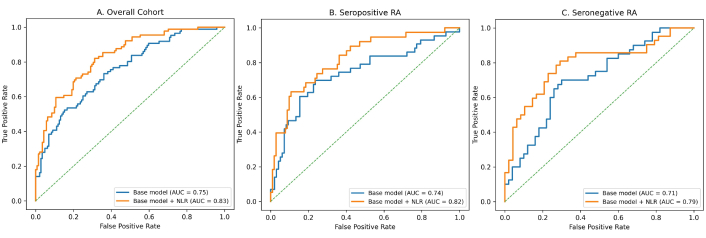
ROC curves of the base model and the base model augmented with NLR across the overall cohort and RA subgroups. ROC curves comparing the discriminatory performance of the base model and the model augmented with NLR for: (A) overall cohort, (B) seropositive RA, and (C) seronegative RA. For each cohort, the true positive rate (sensitivity) is plotted against the false positive rate (1 – specificity). The AUC for the base model and the base model plus NLR is indicated in the legend of each panel. The diagonal dashed line represents the reference line for random classifier performance. AUC: area under the receiver operating characteristic curve; NLR: neutrophil-to-lymphocyte ratio; RA: rheumatoid arthritis; ROC: receiver operating characteristic.
RA is a chronic autoimmune disease characterized by persistent inflammation, joint damage, and increased morbidity and mortality. Inflammatory markers such as CRP and ESR are commonly used to assess disease activity, but they have significant limitations and may not reflect true inflammatory burden. Imaging studies frequently demonstrate active synovitis despite normal or low levels of these markers, indicating the need for additional, accessible indicators of systemic inflammation [15, 16].
In this cohort of patients with RA, we found that an elevated NLR was independently associated with the presence of peripheral neuropathy, including subclinical involvement, even after adjustment for key confounders such as age, disease duration, disease activity, conventional inflammatory markers, serological status, and glucocorticoid use. These findings support a role for systemic inflammatory burden in neural involvement and underscore the potential utility of NLR as a potential surrogate marker of systemic inflammation and immune dysregulation. This association remained consistent across serological subgroups, with both seropositive and seronegative patients with neuropathy demonstrating significantly higher NLR values compared with their respective counterparts without neuropathy.
The overall prevalence of peripheral neuropathy in the present study was approximately 40%, with subclinical involvement identified in 41.9% of patients. Previous studies have reported a wide range of prevalence estimates, largely attributable to variations in diagnostic criteria, study populations, and assessment methodologies. Despite this heterogeneity, electrophysiological evaluations consistently indicate a substantial burden of peripheral neuropathy in RA, encompassing both overt and subclinical forms [1–5]. Our findings are concordant with this literature and underscore the frequency of neural involvement even in the absence of overt neurological symptoms.
Seropositivity was observed more frequently among patients with neuropathy, consistent with prior literature indicating that extra-articular manifestations are more common in seropositive RA than in seronegative disease. In multiple cohorts, autoantibody positivity has been associated with a higher burden of systemic manifestations beyond the joints, including neurologic, pulmonary, and vasculitic complications, compared with patients lacking these antibodies [17–20]. The higher prevalence of neuropathy and elevated NLR observed among seropositive patients in our cohort suggests that autoimmune mechanisms may be linked to heightened systemic inflammatory activity, thereby increasing susceptibility to neural involvement.
We also observed that NLR was significantly elevated not only in patients with clinically overt neuropathy but also in those with subclinical involvement. The progressive increase in NLR across patients without neuropathy, those with subclinical involvement, and those with clinical neuropathy suggests a graded relationship between systemic inflammatory imbalance and the severity of neural involvement. This stepwise pattern strengthens the biological plausibility of the observed association and supports a potential causal pathway linking sustained inflammation to neural injury. Further, multivariable logistic regression analysis revealed that NLR was independently associated with the presence of peripheral neuropathy in RA. In the incremental analysis, inclusion of NLR substantially improved the discriminative performance of the model for distinguishing patients with peripheral neuropathy. Previous research has consistently demonstrated that NLR correlates with established disease activity indices such as DAS28, ESR, and CRP, and is elevated in patients with active RA compared with those in remission or healthy controls [16, 21–23]. However, the present findings extend this literature by demonstrating that NLR contributes independent diagnostic value for peripheral neuropathy in RA, even after adjustment for established correlates such as disease activity, serological status, and disease duration. This suggests that NLR not only reflects general systemic inflammation but may also be sensitive to inflammatory pathways linked with microvascular and neural injury in RA, offering incremental information beyond conventional markers.
The pathophysiological association between NLR and peripheral neuropathy in RA is supported by established immunological mechanisms underlying the disease. Neutrophils play a central role in RA pathogenesis through the release of pro-inflammatory cytokines (such as IL-1, TNF-α, and IL-6), reactive oxygen species, and proteolytic enzymes, which can contribute to endothelial dysfunction and microvascular injury [24, 25]. In parallel, relative lymphopenia reflects impaired immune regulation, particularly an imbalance between regulatory T cells (Tregs) and pro-inflammatory Th17 cells that sustains systemic inflammation [26–30]. Peripheral nerves are especially vulnerable to chronic ischemic and inflammatory damage, with microvascular injury promoting axonal degeneration and demyelination even in the absence of overt vasculitis. By capturing both innate immune activation and adaptive immune dysregulation, NLR may reflect this pathogenic milieu more effectively than ESR or CRP alone.
This study has certain limitations that merit consideration. The cross-sectional design precludes causal inference, and longitudinal studies are required to determine whether elevated NLR predicts the development or progression of neuropathy over time. While we adjusted for major confounders, including age, disease duration, disease activity (DAS28), conventional inflammatory markers (ESR, CRP), serological status, and glucocorticoid use in our multivariable analyses, residual confounding by unmeasured factors or treatment-related effects cannot be entirely excluded. ROC analysis was performed to evaluate the incremental discriminatory value of NLR; however, an optimal NLR cutoff was not derived. A specific threshold for NLR has not been established for peripheral neuropathy in RA, and cutoffs can vary across populations. Additionally, categorizing a continuous variable can reduce statistical power and obscure the underlying association. Future longitudinal and validation studies are warranted to establish clinically meaningful NLR thresholds for neuropathy assessment. Nevertheless, the relatively large sample size and systematic electrophysiological assessment strengthen the validity of the observed associations.
In conclusion, this study provides robust evidence that an elevated NLR is independently associated with the presence of peripheral neuropathy in RA, including subclinical involvement, after adjustment for key confounders such as age, disease duration, disease activity, conventional inflammatory markers, serological status, and glucocorticoid use. The observed association in both seropositive and seronegative subgroups highlights the relevance of systemic inflammatory balance in neural involvement in RA. The improvement in model discrimination observed with the inclusion of NLR beyond conventional clinical and laboratory parameters supports its potential utility as a simple and accessible biomarker for characterizing neural involvement in clinical assessments of RA patients. Integration of NLR into routine evaluation frameworks may enhance recognition of patients with concurrent neuropathy and contribute to more comprehensive clinical management.
anti-CCP: anti-cyclic citrullinated peptide
AUC: area under the receiver operating characteristic curve
CIs: confidence intervals
CRP: C-reactive protein
DAS28: 28-joint Disease Activity Score
DMARDs: disease-modifying antirheumatic drugs
ESR: erythrocyte sedimentation rate
IL-6: interleukin-6
IQR: interquartile range
JAK: Janus kinase
NCS: nerve conduction studies
NLR: neutrophil-to-lymphocyte ratio
NSAIDs: non-steroidal anti-inflammatory drugs
ORs: odds ratios
RA: rheumatoid arthritis
RF: rheumatoid factor
ROC: receiver operating characteristic
TNF-α: tumor necrosis factor-α
BKS: Conceptualization, Investigation, Writing—review & editing, Supervision. AK: Formal analysis, Validation, Visualization, Writing—original draft, Writing—review & editing. SC: Investigation, Writing—original draft. ST: Validation, Visualization, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
The study was approved by the Ethics Committee of ESIC Post Graduate Institute of Medical Sciences and Research, Basaidarapur, New Delhi, India (Approval No. ESIPGIMSR-IEC/2024011).
Informed consent to participate in the study was obtained from all participants.
Informed consent to publication was obtained from relevant participants.
The data are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 563
Download: 13
Times Cited: 0
