Affiliation:
Department of Radiobiology and Molecular Genetics, VINČA Institute of Nuclear Sciences—National Institute of the Republic of Serbia, University of Belgrade, 11000 Belgrade, Serbia
ORCID: https://orcid.org/0000-0002-5486-0079
Affiliation:
Department of Radiobiology and Molecular Genetics, VINČA Institute of Nuclear Sciences—National Institute of the Republic of Serbia, University of Belgrade, 11000 Belgrade, Serbia
ORCID: https://orcid.org/0000-0002-4769-2652
Affiliation:
Department of Radiobiology and Molecular Genetics, VINČA Institute of Nuclear Sciences—National Institute of the Republic of Serbia, University of Belgrade, 11000 Belgrade, Serbia
Email: isenovic@yahoo.com
ORCID: https://orcid.org/0000-0002-0012-2636
Explor Med. 2026;7:1001384 DOI: https://doi.org/10.37349/emed.2026.1001384
Received: November 03, 2025 Accepted: January 27, 2026 Published: March 03, 2026
Academic Editor: Ferdinando Carlo Sasso, University of Campania “Luigi Vanvitelli”, Italy
Diabetes mellitus is one of the biggest public health issues of modern society, with a constant increase in prevalence. It is a complex metabolic disorder characterized by hyperglycemia, dyslipidemia, and impaired insulin signaling, leading to redox imbalance and, consequently, blood vessel dysfunction. One of the key factors in the regulation of vascular tone and contractility is the sodium/potassium adenosine triphosphatase (Na+/K+-ATPase), whose reduced expression and altered activity contribute to the development of vascular dysfunction in type 2 diabetes (T2D). Impaired redox balance and increased production of reactive oxygen species, which directly affect Na+/K+-ATPase activity, also affect the telomere-telomerase system, leading to telomere shortening, DNA damage, and cell apoptosis. Hyperbaric oxygen therapy is used to treat ischemic lesions and vascular complications of diabetes, but the molecular mechanisms underlying its effects on Na+/K+-ATPase and telomere length in T2D patients remain incompletely elucidated.
Diabetes mellitus (DM) is one of the most significant public health issues, affecting 529 million people worldwide in 2021, with the number predicted to reach 1.31 billion by 2050 [1]. DM is not a single cause and consequence disease, but a clinical disorder characterized by multiple metabolic disturbances, including hyperglycemia and dyslipidemia [2]. Vascular diseases caused by DM predominantly affect small blood vessels of the eyes, kidneys, peripheral nerves, and the cerebral vasculature [3, 4]. Hypo- and hyperglycemic states accompany inflammation and disturbances in redox balance, which directly influence microvascular complications. The reduction in sodium/potassium adenosine triphosphatase (Na+/K+-ATPase) activity is associated with hyperglycemia and DM states, which are intrinsically linked to vascular pathology, making Na+/K+-ATPase an important marker of vasculopathy [5, 6]. Besides, oxidative stress (OS) and chronic inflammation contribute to diabetic microvascular complications, which are accompanied by telomere shortening, loss of DNA protection, and cell apoptosis [7]. Abnormalities in the telomere-telomerase system usually occur in people with type 2 diabetes (T2D) and vascular complications [8].
The Na+/K+-ATPase is a complex of a highly conserved plasma membrane enzyme, consisting of three subunits: α1, β1, and γ, with multiple tissues- and cell-specific isoforms. The Na+/K+-ATPase plays a crucial role in regulating the Na+ and K+ ion gradient across the cell membrane, which is required for many cellular functions, including the control of contractility and cell volume, cell excitability, absorption processes, ion homeostasis, and systemic vascular hypertension. Regulation of the Na+/K+-ATPase is a complex process that occurs at multiple levels, including gene expression, phosphorylation, trafficking, and activity. Na+/K+-ATPase expression/activity is an essential mediator of vascular tone and contractility, and its abnormal regulation is implicated in several diseases. Decreased Na+/K+-ATPase abundance and altered isoform expression both contribute to vascular dysfunction [5, 6]. Reduced insulin sensitivity is a characteristic feature of vascular pathophysiological conditions such as T2D. Accumulating data indicate that insulin-impaired vascular sensitivity predisposes to increased tone and vascular constriction, the hallmark of hemodynamic abnormalities characteristic of DM and impaired glucose tolerance [2, 9]. A disturbed insulin signaling pathway in T2D is thought to decrease insulin receptor substrate/phosphatidylinositol-3-kinase (PI3K)/protein kinase B (Akt) signaling and increase mammalian target of rapamycin and the downstream signaling molecule ribosomal S6 kinase [10, 11]. Insulin regulates cation transport by altering Na+/K+-ATPase expression, a process known to be crucial for the normal function of the cardiovascular system [12]. Insulin influences Na+/K+-ATPase function via several signaling cascades, including PI3K/Akt, protein kinase A, protein kinase C, and mitogen-activated protein kinase. Obesity, accompanied by insulin resistance, decreased Na+/K+-ATPase expression/activity in rat hearts, associated with attenuation of PI3K/Akt signaling, increased angiotensin II, and cardiomyocyte hypertrophy [13, 14]. These findings emphasized the importance of the Na+/K+-ATPase as a marker of vascular complications in DM. Despite numerous studies investigating the role and regulation of Na+/K+-ATPase in vasculopathy, the exact molecular mechanism remains unclear. The reason for this may be related to the multiple functions of the Na+/K+-ATPase. Interestingly, Na+/K+-ATPase could function as a signal transducer and activator of the inositol 1,4,5-trisphosphate receptor via a direct interaction [15]. It was also found that the Na+/K+-ATPase plays a crucial role in the organisms’ adaptation to increased reactive oxygen species (ROS). Oxygen-sensitivity of the Na+/K+-ATPase is mediated by redox modifications of thiol groups, including S-nitrosylation, S-glutathionylation, and redox-sensitive phosphorylation, all of which affect enzyme activity and induce metabolic changes. Oxygen-derived free radicals and H2O2, nitric oxide (NO), and oxidized glutathione are the signaling messengers that make the Na+/K+-ATPase “oxygen-sensitive”. Besides stimulating Na+/K+-ATPase, the oxidant amplification loop increased expression of senescence markers, cell injury, and apoptosis, whereas inhibition of Na+/K+-ATPase downstream signaling (Src) attenuated these changes [16, 17]. Thus, the regulation of Na+/K+-ATPase expression/activity could be significant in both treatment and potential prevention of T2D-associated microvascular complications.
Another important marker of different pathophysiological conditions is telomere shortening to a critical length, mediated by the induction of a persistent DNA damage response at chromosome ends and loss of cellular viability [18]. Telomere shortening is associated with increased ROS because the high guanine content of telomeres makes them very sensitive to ROS-induced damage, leading to the formation of 8-oxo-7,8-dihydro-2′-deoxyguanosine [19]. Hyperglycemia and DM are associated with the activation of several molecular pathways that are all involved in increased OS, leading to damage across multiple cell lines of the vascular system and, consequently, microvascular complications. While population studies of DM patients indicate a correlation between shorter telomere length (TL) and worse OS, the mechanism underlying this association remains challenging to determine [19–21]. Thus, more convincing evidence is required to define how inflammation and ROS induce TL dynamics in vascular disease related to DM. Exploring the molecular mechanisms regulating telomere erosion in T2D and their link to microvascular complications requires further research.
Microvascular complications in T2D result in poor circulation, peripheral nerve damage, and dry skin prone to cracking, making infection control more difficult and patients more susceptible to ulcers [3]. The clinical management of T2D is complex, as T2D-associated microvascular complications are often not evident for prolonged periods. Current treatment for T2D patients is still focused on insulin, its analogs, and insulin action sensitizers with numerous side effects and insufficient efficacy [22, 23]. Also, a disadvantage of the current therapy for T2D includes limited knowledge of therapies, their efficacy, timing, and side effects [22, 23]. Hyperbaric oxygen therapy (HBOT) is widely used to treat ischemic lesions and vascular complications caused by DM [24]. HBOT is a promising medical treatment option for patients with T2D and associated microvascular complications. Despite treatment of diabetic foot with HBOT resulting in better results compared with the usual treatment for wounds, it is still unclear whether HBOT is better than conventional treatment to relieve pain or prevent the need for nursing care [25]. In addition, the absence of wound healing in certain DM patients underscores the need for detailed research into the molecular mechanisms underlying HBOT. There was no evidence that HBOT caused more side effects than conventional treatment for wounds, reduced the number of amputations, improved quality of life, shortened hospital stays, or increased life expectancy. Also, studies did not consider these aspects appropriately, in greater detail. Our previous findings in humans [26, 27] and those of others in rats [28] indicate that HBOT reduces inducible NO synthase activity/expression and NO generation in lymphocytes and improves the lipid profile. These findings are consistent with other studies reporting antioxidant and anti-inflammatory effects of HBOT [29–31]. Clinical outcomes of HBOT interventions are presented in Table 1. Although HBOT has the potential to be an effective adjunct to standard therapy for T2D patients, a more comprehensive understanding of the molecular mechanisms by which HBOT modulates vascular function in these patients is necessary. HBOT significantly affects the organism’s redox homeostasis by increasing the partial pressure of oxygen in tissues [37]. Increased oxygen availability leads to a temporary increase in ROS, which not only represents a potential for OS development but also serves as a signaling molecule that can activate the cell’s adaptive defense pathways [38]. HBOT activates antioxidant defense mechanisms and reduces oxidative damage in endothelial cells, which is crucial for preserving vascular function [39]. HBOT modulates redox-sensitive signaling pathways associated with Na+/K+-ATPase, potentially involving Src-dependent mechanisms, and influences transcription factors such as nuclear factor erythroid 2-related factor 2 (Nrf2) and hypoxia-inducible factor 1-alpha (HIF-1α), linking OS to adaptive cellular responses [37, 40, 41]. The redox-sensitive transcription factors, such as HIF-1α and Nrf2, can modulate Na+/K+-ATPase activity, whereby HIF-1α regulates cellular adaptation to hypoxia, while Nrf2 activates antioxidant defense mechanisms that can help in the protection of Na+/K+-ATPase from oxidative damage [42, 43]. At the same time, HBOT-induced changes in redox signaling may influence telomere stability and telomere-related pathways, potentially linking Na+/K+-ATPase activity to cellular aging processes [37, 40, 41].
Clinical outcomes of HBOT interventions.
| Condition | Number of patients | Duration of HBOT | Outcome | Ref. |
|---|---|---|---|---|
| T2D/Wagner grades I–III foot ulcers | 15 | Twice a day, 5 days a week for 2 weeks | The size of the ulcers decreased significantly | [32] |
| Diabetes mellitus/diabetic foot ulcers | 18 | Twice a day, 5 days a week for 2 weeks | Ulcer size reduction↑Malondialdehyde↑Superoxide dismutase↑Catalase | [33] |
| Stages III and IV diabetic foot ulcers | 20 | 10 weeks | 60% of ulcers showed complete healing | [34] |
| Chronic diabetic foot wounds (grade II or III on Wagner) | 15 | 20–40 sessions | Median ulcer surface area significantly reduced | [35] |
| T1D/T2D/Wagner grades III and IV foot ulcers | 30 | 10–30 sessions | 7 patients—finger amputations3 patients—foot amputations4 patients—below-knee amputation | [36] |
| T1D | 19 | 10 sessions | Reduction of inflammation (decreased inducible nitric oxide synthase) | [26] |
| T1D | 24 | 10 sessions | Improved lipid profile | [27] |
HBOT: hyperbaric oxygen therapy; T1D: type 1 diabetes; T2D: type 2 diabetes.
Investigation of the mechanisms by which HBOT exerts its effects on Na+/K+-ATPase expression/activity and TL in states of T2D and associated microvascular complications has the potential to improve therapy further. Optimizing HBOT timing could represent a step toward personalized medicine. Understanding an entirely new area of vascular biology and applying it to a pandemic disease is a significant step in basic research. There is a need for such an approach because glucose-lowering drugs have insufficient long-term benefits in T2D patients. Microvascular complications are common in patients with T2D. Indeed, it remains relevant to pursue fundamental research in this area and to broaden our understanding of the underlying mechanisms of T2D-related microvascular complications. Currently, most investigations focus on confirming the epidemiological links between T2D and vascular diseases. Only a limited number of studies have examined causality. The derangements of various metabolites do not explain their role in promoting microvascular disease in T2D. HBOT functions as a therapeutic stimulus that modulates redox-sensitive signaling pathways by increasing oxygen availability. In addition, by regulating redox homeostasis, HBOT may influence telomere dynamics. Repeated HBOT decreases OS and impairs ROS-induced guanine oxidation, a major driver of rapid telomere shortening, especially in telomeric regions rich in G-bases. By stabilizing telomerase activity and maintaining telomere structure, HBOT-induced modifications in redox-sensitive signaling pathways could also decrease the activation of the DNA-damage response and consequently postpone the onset of cell senescence [19, 44]. This perspective proposes HBOT not only as a therapeutic strategy but also as a regulator of potential biomarkers of therapeutic response, such as Na+/K+-ATPase and TL. Previously proposed approaches were predominantly focused on OS or metabolic effects of HBOT, whereas this perspective links redox-sensitive signaling to cellular aging processes. It provides a new hypothesis that HBOT can directly affect Na+/K+-ATPase and TL, which are proposed as potential biomarkers of HBOT response (Figure 1). HBOT’s effects on Na+/K+-ATPase regulation and TL in T2D patients and in patients with microvascular diseases could represent a novel treatment approach, leveraging newly identified biomarkers for T2D-related vascular diseases. Thus, unraveling the specific responses of Na+/K+-ATPase and TL to HBOT will be a significant step forward in understanding microvascular complications in T2D, toward a more integrated view of the treatment of this disease, and to foster improved evidence-based interventions. Future research priorities should focus on enhancing the therapeutic application of HBOT in T2D, including determining the optimal timing and duration of HBOT treatment, confirming reliable biomarkers for monitoring therapeutic efficacy, and investigating its incorporation into tailored treatment programs. Addressing these criteria is critical to transforming HBOT from a promising theoretical framework into a practical, tailored diabetes management solution. Other biochemical factors, including protein aggregation, pH-dependent proteome stability, and metformin-related molecular interactions, may also contribute to the complex pathophysiology of T2D, and all these factors will be explored in our future studies.
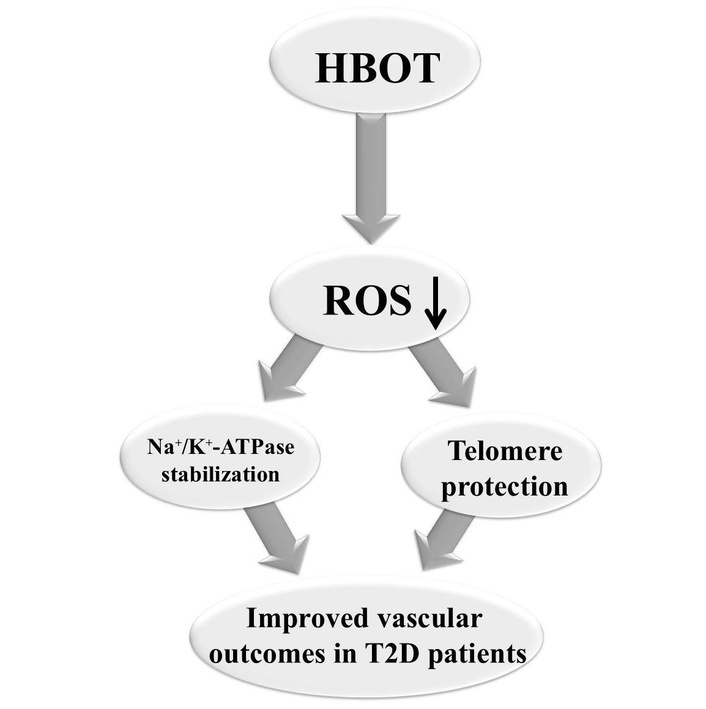
New hypothesis of HBOT effects. HBOT: hyperbaric oxygen therapy; Na+/K+-ATPase: sodium/potassium adenosine triphosphatase; ROS: reactive oxygen species; T2D: type 2 diabetes.
Akt: protein kinase B
DM: diabetes mellitus
HBOT: hyperbaric oxygen therapy
HIF-1α: hypoxia-inducible factor 1-alpha
Na+/K+-ATPase: sodium/potassium adenosine triphosphatase
NO: nitric oxide
Nrf2: nuclear factor erythroid 2-related factor 2
OS: oxidative stress
PI3K: phosphatidylinositol-3-kinase
ROS: reactive oxygen species
T2D: type 2 diabetes
TL: telomere length
SZ: Writing—original draft. MO: Writing—original draft. ERI: Conceptualization, Supervision, Writing—review & editing. All authors read and approved the submitted version.
Esma R. Isenovic, who is the Editorial Board Member of Exploration of Medicine, had no involvement in the decision-making or review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
This work was funded by the Ministry of Science, Technological Development, and Innovation of the Republic of Serbia [Contract No# 451-03-136/2025-03/200017]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1432
Download: 21
Times Cited: 0
