Affiliation:
1Department of Internal Medicine, ESIC Postgraduate Institute of Medical Sciences and Research, New Delhi 110015, India
ORCID: https://orcid.org/0000-0001-6570-8577
Affiliation:
1Department of Internal Medicine, ESIC Postgraduate Institute of Medical Sciences and Research, New Delhi 110015, India
Email: dramit_67@yahoo.com
ORCID: https://orcid.org/0000-0001-9348-7601
Affiliation:
1Department of Internal Medicine, ESIC Postgraduate Institute of Medical Sciences and Research, New Delhi 110015, India
Affiliation:
1Department of Internal Medicine, ESIC Postgraduate Institute of Medical Sciences and Research, New Delhi 110015, India
ORCID: https://orcid.org/0009-0006-2566-4999
Affiliation:
2Indian Council of Medical Research, New Delhi 110029, India
ORCID: https://orcid.org/0000-0002-9129-4686
Explor Dig Dis. 2026;5:1005121 DOI: https://doi.org/10.37349/edd.2026.1005121
Received: February 03, 2026 Accepted: March 31, 2026 Published: April 27, 2026
Academic Editor: Han Moshage, University of Groningen, Groningen, The Netherlands
The article belongs to the special issue Gastrointestinal Diseases, Cholesterol, Oxysterols, and Bile Acids
Aim: Chronic liver disease (CLD) is characterized by progressive impairment of hepatic function and frequent lipid metabolism abnormalities, with reductions in high-density lipoprotein cholesterol (HDL-C) and other lipoprotein fractions shown to parallel worsening liver dysfunction and predict adverse clinical outcomes such as decompensation and mortality. Established prognostic scores like Model for End-Stage Liver Disease (MELD) and Child-Turcotte-Pugh (CTP) capture aspects of disease severity, but composite lipid indices such as the non-HDL/HDL-C ratio (NHHR), which balance atherogenic and protective lipoproteins, have emerged as potentially informative biomarkers in metabolic and liver disorders. This study evaluated the association of NHHR with clinical decompensation in CLD.
Methods: This cross-sectional study included 220 adults with CLD of mixed etiologies. Baseline demographics, liver disease severity scores, and fasting lipid profiles were obtained. NHHR was calculated, and patients were categorized into tertiles. Spearman correlation coefficients were calculated to examine relationships between NHHR and clinical severity markers. Multivariable logistic regression was used to evaluate the association between NHHR and clinical decompensation. Model performance was compared using receiver operating characteristic curves, net reclassification improvement (NRI), and integrated discrimination improvement (IDI).
Results: Among 220 patients with CLD (mean age 54.5 ± 11.9 years, 63% male), 96 (43.6%) had decompensated disease. Higher NHHR tertiles were associated with increasing MELD-3.0 scores (P = 0.028) and lower serum albumin (P < 0.001). NHHR correlated positively with MELD-3.0, bilirubin, and international normalized ratio (INR) and inversely with albumin and platelet count. Decompensation prevalence rose across NHHR tertiles (31.1% to 53.4%, P < 0.001). NHHR was independently associated with decompensation (adjusted OR 1.55, 95% CI 1.21–1.98, P < 0.001) and improved model discrimination (AUC 0.79 vs. 0.73).
Conclusions: NHHR is independently associated with clinical decompensation in CLD and provides incremental prognostic value beyond traditional predictors, suggesting its potential utility in clinical risk assessment and stratification.
Chronic liver disease (CLD) imposes a substantial and increasing global health burden, affecting millions of individuals and accounting for significant morbidity and mortality worldwide. Estimates from the Global Burden of Disease Study indicate that in 2021, there were more than 58 million incident cases of cirrhosis and other CLDs, with approximately 1.43 million deaths attributable to these conditions globally, highlighting the rising absolute burden of liver pathology despite some improvements in age-standardized rates [1]. Data from multiple epidemiologic analyses further show that liver diseases collectively account for nearly two million deaths annually, representing about 4% of all global mortality and making them a leading cause of death across regions [2].
The progression of CLD from compensated stages to clinical decompensation represents a pivotal inflection point in the natural history of the disease and is accompanied by marked increases in morbidity, mortality, and healthcare utilization. Accurate stratification of disease severity and early identification of patients at higher risk of advanced or decompensated liver disease are central to effective management and prognostication. Alterations in lipid metabolism are well recognized in CLD due to impaired hepatocellular synthesis and disrupted lipoprotein processing, leading to characteristic reductions in circulating total cholesterol, low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C), with declining lipid levels consistently observed as liver disease severity increases across Child-Turcotte-Pugh (CTP) classes and Model for End-Stage Liver Disease (MELD) strata [3–9]. Among these alterations, reductions in HDL-C and its associated apolipoproteins appear particularly pronounced and have been documented across diverse etiologies of CLD, including viral, alcohol-related, and metabolic liver diseases [10–14]. In contrast, changes in triglyceride levels have been more heterogeneous, underscoring the complex and multifactorial relationship between hepatic injury, systemic inflammation, and lipid homeostasis [7, 15].
Beyond descriptive associations, emerging evidence suggests that lipid fractions and lipid-derived indices may carry prognostic relevance in CLD. Several studies have demonstrated that lower HDL-C levels are independently associated with adverse clinical outcomes, including mortality and hepatic decompensation, even after adjustment for established prognostic scores such as MELD and CTP [16, 17]. These observations imply that lipid parameters may capture dimensions of hepatic dysfunction and systemic derangement not fully reflected by conventional scoring systems.
More recently, attention has shifted toward composite lipid indices that integrate information from both atherogenic and protective lipoprotein fractions. The non-HDL/HDL-C ratio (NHHR) represents one such metric, incorporating all apoB-containing, potentially atherogenic lipoproteins relative to HDL-C, which exerts anti-inflammatory and immunomodulatory effects. In cardiovascular and metabolic research, NHHR has demonstrated superior discriminatory ability compared with isolated lipid measures [18–20]. Emerging data also link elevated NHHR to metabolic liver disease and hepatic fibrosis; however, its relationship with clinical severity and decompensation in established CLD remains insufficiently characterized [21–24]. In this context, the present study examined the association between NHHR and clinical decompensation in patients with CLD and evaluated whether the inclusion of NHHR enhances discrimination of clinical status beyond conventional prognostic indicators.
This cross-sectional observational study was conducted at the Department of Internal Medicine, ESIC Postgraduate Institute of Medical Sciences and Research, Basaidarapur, New Delhi, from March 2024 to August 2025. The study protocol was approved by the institutional ethics committee, and all participants provided written informed consent prior to enrolment. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Consecutive adult patients (≥ 18 years) with a confirmed diagnosis of CLD, irrespective of etiology, were screened for eligibility. Diagnosis of CLD was based on clinical, imaging, serological tests, and/or histopathology criteria consistent with chronic liver pathology. Patients were excluded if they had cholestatic liver disease, hepatocellular carcinoma or other malignancies, acute cardiovascular events within the preceding 3 months, active systemic infections, or current use of lipid-lowering medications.
At enrolment, demographic data (age, sex), anthropometric measurements, and clinical history were recorded. Liver disease severity was assessed using the CTP classification and the MELD score [25]. Standard laboratory investigations were obtained, including liver biochemistry, coagulation profile, complete blood count, and a fasting lipid profile.
Decompensation was defined by the presence of at least one of the following: new or worsening ascites requiring intervention, variceal hemorrhage requiring endoscopic variceal ligation/sclerotherapy, hepatic encephalopathy, spontaneous bacterial peritonitis, hepatorenal syndrome, or clinically significant jaundice with a total bilirubin level exceeding 3 mg/dL [26]. Non-hepatic causes of hyperbilirubinemia were excluded through clinical evaluation and laboratory assessment, including review of clinical history and hemolysis parameters.
Fasting serum samples were analyzed for total cholesterol, HDL-C, and triglycerides using standardized enzymatic colorimetric assays based on cholesterol esterase, cholesterol oxidase, and peroxidase reactions. LDL-C was calculated using the Friedewald formula [LDL-C = total cholesterol – HDL-C – (triglycerides/5)] for samples with triglyceride levels < 400 mg/dL, while direct LDL-C measurement was used when triglycerides were ≥ 400 mg/dL, as the Friedewald equation is unreliable at higher triglyceride concentrations. Non-HDL-C was calculated by subtracting HDL-C from total cholesterol. The main exposure measure, the NHHR, was then computed as the ratio of non-HDL-C to HDL-C.
To examine associations across levels of NHHR, patients were stratified into tertiles based on the distribution of NHHR values in the cohort. Each tertile contained approximately one-third of the study population, ensuring near-equal group sizes. These tertile groups were used for comparative analyses of clinical characteristics and decompensation status.
All statistical analyses were conducted using SPSS v28.0 (IBM Corp., Armonk, NY, USA). Continuous variables are presented as mean ± standard deviation (SD) or median [interquartile range (IQR)] depending on distribution; categorical variables are presented as counts and percentages. Differences across NHHR tertiles were assessed using one-way analysis of variance (ANOVA) or Kruskal-Wallis tests for continuous variables and chi-square tests for categorical variables. Between-group comparisons (e.g., decompensated vs. compensated) were performed using independent t-tests or Mann-Whitney U tests, as appropriate. Associations between various clinical and biochemical variables and decompensation status were assessed using binary logistic regression analysis. Univariate analyses were followed by multivariable logistic regression models adjusted for age, sex, MELD score, and platelet count. Adjusted odds ratios (ORs) with 95% confidence intervals (CIs) were reported. Spearman correlation coefficients (ρ) were calculated to examine relationships between NHHR and clinical severity markers.
To evaluate the ability of clinical models to distinguish between decompensated and compensated patients, we conducted discrimination and reclassification analyses. Receiver operating characteristic (ROC) curves were constructed to evaluate the discriminative performance of a base model comprising age, sex, and MELD-3.0 score, and a combined model that additionally included NHHR. The area under the ROC curve (AUC) and corresponding 95% CIs were calculated for the overall cohort as well as within each NHHR tertile. Differences in AUC between models were compared using DeLong’s test. To quantify the incremental value of adding NHHR to the base model, net reclassification improvement (NRI) and integrated discrimination improvement (IDI) were calculated for the overall cohort and across tertile groups. Continuous NRI and IDI were used to evaluate the improvement in model performance after the inclusion of NHHR. These measures estimate the extent to which the combined model improves the correct classification of individuals with and without decompensation compared with the base model. All discrimination and reclassification analyses were performed using R (R Foundation for Statistical Computing, Vienna, Austria). All statistical tests were two-tailed, and a P value < 0.05 was considered statistically significant.
A total of 220 patients with CLD were included in the study, with a mean age of 54.5 ± 11.9 years, of whom 138 (63%) were male. Figure 1 demonstrates the age distribution of the study population. The majority of patients were in the 50–59 year age group (33.6%), followed by 40–49 years (25.5%) and 60–69 years (21.8%). Younger patients aged 18–39 years accounted for 10%, while 9.1% were aged ≥ 70 years. Overall, 96 patients (43.6%) had decompensated liver disease. Participants were categorized into tertiles according to baseline NHHR. Baseline characteristics according to NHHR tertiles are presented in Table 1. Mean NHHR increased significantly across tertiles (P < 0.001), while age and sex distribution were comparable among groups. HDL-C levels decreased progressively, and non-HDL-C levels increased across increasing NHHR tertiles (both P < 0.001). Higher NHHR was also associated with greater liver disease severity, reflected by a stepwise increase in MELD-3.0 scores (P = 0.028) and a corresponding decrease in serum albumin levels (P < 0.001). In contrast, international normalized ratio (INR), platelet counts, and serum bilirubin did not differ significantly across tertiles. The distribution of CLD etiology was similar among NHHR categories. Although a trend toward a more advanced Child-Pugh class was observed with increasing NHHR, this did not reach statistical significance. Importantly, the proportion of patients with decompensated cirrhosis increased significantly across NHHR tertiles, rising from 31.1% in tertile 1 to 53.4% in tertile 3 (P < 0.001).
Baseline characteristics of study participants stratified by NHHR tertiles (n = 220).
| Characteristic | Tertile 1 (NHHR ≤ 2.0) (n = 74) | Tertile 2 (NHHR 2.1–3.0) (n = 73) | Tertile 3 (NHHR > 3.0) (n = 73) | P value |
|---|---|---|---|---|
| NHHR, mean ± SD | 1.79 ± 0.13 | 2.57 ± 0.27 | 3.80 ± 0.35 | < 0.001 |
| Age, years, mean ± SD | 52.0 ± 10.5 | 55.1 ± 12.3 | 56.4 ± 11.7 | 0.178 |
| Male sex, n (%) | 48 (65) | 42 (58) | 48 (66) | 0.423 |
| HDL-C, mg/dL, mean ± SD | 47.2 ± 10.1 | 38.7 ± 8.8 | 31.5 ± 7.1 | < 0.001 |
| Non-HDL-C, mg/dL, mean ± SD | 111.0 ± 16.0 | 123.6 ± 18.7 | 138.2 ± 19.8 | < 0.001 |
| MELD-3.0 score, mean ± SD | 12.3 ± 4.2 | 13.1 ± 4.5 | 15.0 ± 5.1 | 0.028 |
| Serum albumin, g/dL, mean ± SD | 3.4 ± 0.7 | 3.1 ± 0.8 | 2.7 ± 0.8 | < 0.001 |
| Total bilirubin, mg/dL, mean ± SD | 2.3 ± 1.7 | 2.8 ± 2.0 | 3.1 ± 2.1 | 0.092 |
| INR, mean ± SD | 1.42 ± 0.31 | 1.45 ± 0.34 | 1.50 ± 0.38 | 0.166 |
| Platelet count, × 109/L, mean ± SD | 155 ± 60 | 148 ± 58 | 142 ± 55 | 0.212 |
| Etiology of CLD, n (%) | 0.357 | |||
| Alcohol-related | 33 (45) | 35 (48) | 38 (52) | |
| Metabolic (MASLD) | 21 (28) | 19 (26) | 16 (22) | |
| Viral (HBV/HCV) | 16 (22) | 15 (21) | 15 (21) | |
| Other* | 4 (5) | 4 (5) | 4 (5) | |
| Child-Pugh class (A/B/C), n | 40/26/8 | 34/28/11 | 30/32/11 | 0.052 |
| Decompensated disease, n (%) | 23 (31.1) | 34 (46.6) | 39 (53.4) | < 0.001 |
*: Includes autoimmune, Wilson’s disease, cryptogenic, and other less common etiologies. CLD: chronic liver disease; HDL-C: high-density lipoprotein cholesterol; INR: international normalized ratio; MASLD: metabolic dysfunction-associated steatotic liver disease; MELD: Model for End-Stage Liver Disease; NHHR: non-high-density lipoprotein/HDL-C ratio; SD: standard deviation.
Table 2 summarizes the distribution of specific decompensation events across NHHR tertiles. The proportion of patients experiencing any decompensation increased significantly with higher NHHR (31.1%, 46.6%, and 53.4% for tertiles 1, 2, and 3, respectively; P < 0.001). Among individual complications, ascites, hepatic encephalopathy, and jaundice were significantly more common in patients in the higher NHHR tertiles (P = 0.008, 0.026, and 0.047, respectively). Although variceal bleeding and spontaneous bacterial peritonitis were also more frequent in the higher tertiles, these differences did not reach statistical significance.
Decompensation events by NHHR tertiles (n = 220).
| Decompensation event | Tertile 1 (NHHR ≤ 2.0) (n = 74) | Tertile 2 (NHHR 2.1–3.0) (n = 73) | Tertile 3 (NHHR > 3.0) (n = 73) | P value |
|---|---|---|---|---|
| Any decompensation, n (%) | 23 (31.1) | 34 (46.6) | 39 (53.4) | < 0.001 |
| Ascites, n (%) | 15 (20.3) | 24 (32.9) | 30 (41.1) | 0.008 |
| Variceal bleeding, n (%) | 6 (8.1) | 8 (11.0) | 10 (13.7) | 0.432 |
| Hepatic encephalopathy, n (%) | 5 (6.8) | 9 (12.3) | 14 (19.2) | 0.026 |
| Spontaneous bacterial peritonitis, n (%) | 3 (4.1) | 5 (6.8) | 7 (9.6) | 0.305 |
| Jaundice (bilirubin > 3 mg/dL), n (%) | 8 (10.8) | 13 (17.8) | 18 (24.7) | 0.047 |
NHHR: non-high-density lipoprotein/high-density lipoprotein cholesterol ratio.
Spearman correlation analysis was conducted to examine the relationship between NHHR and clinical severity markers (Table 3). NHHR showed a moderate positive correlation with MELD-3.0 score (ρ = 0.32; P < 0.001) and total bilirubin (ρ = 0.30; P < 0.001). NHHR was also positively correlated with the INR (ρ = 0.26; P = 0.003). Inverse correlations were observed with serum albumin (ρ = –0.29; P < 0.001) and platelet count (ρ = –0.18; P = 0.014). A weaker inverse correlation was seen with serum sodium (ρ = –0.15; P = 0.028).
Correlation of NHHR with key clinical variables.
| Variable | Spearman’s ρ | P value |
|---|---|---|
| MELD-3.0 score | 0.32 | < 0.001 |
| Albumin (g/dL) | –0.29 | < 0.001 |
| INR | 0.26 | 0.003 |
| Platelet count (× 109/L) | –0.18 | 0.014 |
| Total bilirubin (mg/dL) | 0.30 | < 0.001 |
| Serum sodium (mmol/L) | –0.15 | 0.028 |
INR: international normalized ratio; MELD: Model for End-Stage Liver Disease; NHHR: non-high-density lipoprotein/high-density lipoprotein cholesterol ratio; ρ: Spearman correlation coefficient.
Table 4 presents the results of the univariate and multivariable logistic regression analyses evaluating factors associated with decompensation. In the univariate logistic regression analysis, higher NHHR was significantly associated with the presence of decompensation (OR 1.62, 95% CI 1.30–2.02; P < 0.001). A higher MELD-3.0 score was also significantly associated with decompensation. Platelet count showed a trend toward an inverse association but was not statistically significant (OR 0.97, 95% CI 0.94–1.01; P = 0.146). Age and male sex were not significantly associated with decompensation.
Univariate and multivariable logistic regression analysis of factors associated with decompensation.
| Predictor | Univariate OR (95% CI) | P value | Adjusted OR (95% CI) | P value |
|---|---|---|---|---|
| NHHR | 1.62 (1.30–2.02) | < 0.001 | 1.55 (1.21–1.98) | < 0.001 |
| Age | 1.01 (0.99–1.03) | 0.252 | 1.01 (0.99–1.04) | 0.214 |
| Male sex | 1.18 (0.74–1.88) | 0.495 | 1.12 (0.69–1.83) | 0.641 |
| MELD-3.0 score | 1.15 (1.09–1.21) | < 0.001 | 1.10 (1.04–1.16) | < 0.001 |
| Platelet count | 0.97 (0.94–1.01) | 0.146 | 0.98 (0.94–1.02) | 0.338 |
CI: confidence interval; MELD: Model for End-Stage Liver Disease; NHHR: non-high-density lipoprotein/high-density lipoprotein cholesterol ratio; OR: odds ratio.
In the multivariable logistic regression model adjusting for age, sex, MELD-3.0 score, and platelet count, NHHR remained independently associated with decompensation (adjusted OR 1.55, 95% CI 1.21–1.98; P < 0.001). MELD-3.0 score also demonstrated an independent association with decompensation (adjusted OR 1.10, 95% CI 1.04–1.16; P < 0.001).
Discrimination and reclassification performance of the predictive models for decompensation are summarized in Table 5. In the overall cohort, the base model incorporating age, sex, and MELD-3.0 score demonstrated moderate discrimination for the presence of decompensation, with an AUC of 0.73 (95% CI 0.67–0.79). The addition of NHHR to the base model resulted in a significant improvement in discrimination, increasing the AUC to 0.79 (95% CI 0.73–0.85; P < 0.001), along with significant gains in reclassification as reflected by positive NRI = 0.20 and IDI = 0.041. In analyses stratified by NHHR tertiles, the combined model consistently outperformed the base model across all groups. The improvement in discrimination was modest and not statistically significant in tertile 1 (P = 0.138), but was significant in tertile 2 (P = 0.045) and greatest in tertile 3, where the AUC increased from 0.75 to 0.81 (P < 0.001), accompanied by substantial improvements in reclassification (NRI = 0.27; IDI = 0.053). Overall, these findings indicate that incorporation of NHHR enhances the ability of clinical models to distinguish between decompensated and compensated CLD patients, particularly among those with higher NHHR levels. These results are also demonstrated in Figure 2 through the ROC curves, which show the improved discriminatory performance of the model after the addition of NHHR.
Classification performance of base and combined models for identifying decompensated vs. compensated patients by NHHR tertiles.
| Group | Model | AUC (95% CI) | ΔAUC vs. Base | P (DeLong) | NRI | IDI |
|---|---|---|---|---|---|---|
| Overall cohort (n = 220) | Base Model | 0.73 (0.67–0.79) | - | - | - | - |
| Base + NHHR | 0.79 (0.73–0.85) | + 0.06 | < 0.001 | 0.20 | 0.041 | |
| NHHR tertile 1 (≤ 2.0) (n = 74) | Base Model | 0.70 (0.58–0.82) | - | - | - | - |
| Base + NHHR | 0.74 (0.62–0.85) | + 0.04 | 0.138 | 0.08 | 0.010 | |
| NHHR tertile 2 (2.1–3.0) (n = 73) | Base Model | 0.74 (0.63–0.85) | - | - | - | - |
| Base + NHHR | 0.78 (0.68–0.88) | + 0.04 | 0.045 | 0.17 | 0.026 | |
| NHHR tertile 3 (> 3.0) (n = 73) | Base Model | 0.75 (0.65–0.85) | - | - | - | - |
| Base + NHHR | 0.81 (0.72–0.89) | + 0.06 | < 0.001 | 0.27 | 0.053 |
Base Model comprised age, sex, and MELD-3.0 score. AUC: area under the receiver operating characteristic curve; CI: confidence interval; NRI: net reclassification improvement; IDI: integrated discrimination improvement; MELD: Model for End-Stage Liver Disease; NHHR: non-high-density lipoprotein/high-density lipoprotein cholesterol ratio.
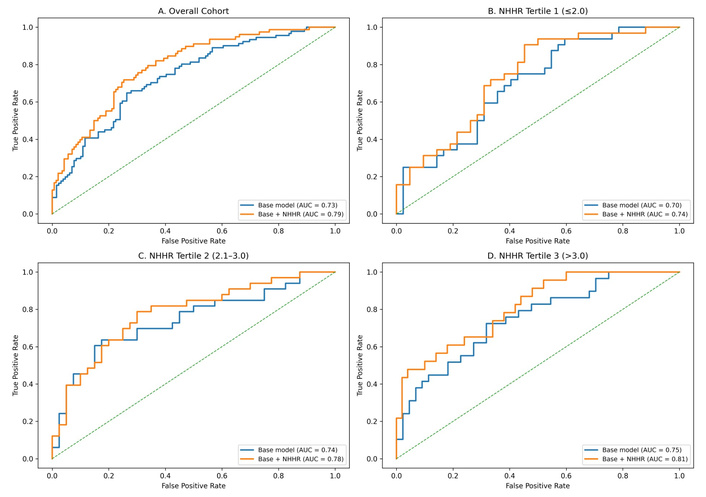
ROC curves of the base model and base model plus NHHR across NHHR tertiles. ROC curves comparing the discriminatory performance of the base predictive model (incorporating age, sex, and MELD-3.0 score) and the model augmented with NHHR across chronic liver disease cohorts: (A) overall cohort, (B) NHHR tertile 1 (≤ 2.0), (C) NHHR tertile 2 (2.1–3.0), and (D) NHHR tertile 3 (> 3.0). For each panel, the true positive rate (sensitivity) is plotted against the false positive rate (1 – specificity). The AUC for each model is indicated in the legend. The diagonal dashed line represents the reference line for random classifier performance. AUC: area under the receiver operating characteristic curve; MELD: Model for End-Stage Liver Disease; NHHR: non-high-density lipoprotein/high-density lipoprotein cholesterol ratio; ROC: receiver operating characteristic.
In this cross-sectional study of patients with CLD, we found that an elevated NHHR was associated with markers of advanced liver disease and with the presence of clinical decompensation. Patients with higher NHHR tertiles had progressively greater incidence of ascites, hepatic encephalopathy, and overall decompensation. A higher NHHR correlated moderately with MELD-3.0 and total bilirubin and inversely with serum albumin and platelet count. NHHR remained independently associated with decompensation after adjustment for key confounders. Furthermore, NHHR significantly enhanced discrimination and reclassification for decompensation when added to established clinical predictors such as the MELD-3.0 score. These findings highlight the potential utility of NHHR as a simple, accessible biomarker to complement conventional risk stratification in CLD.
A substantial body of literature demonstrates profound disturbances in lipid metabolism in CLD. Hepatic synthesis and secretion of lipoproteins are impaired in cirrhotic patients due to reduced apolipoprotein production and defective lipoprotein assembly, leading to progressive decreases in total cholesterol, HDL-C, and LDL-C with advancing liver dysfunction. Several observational studies have confirmed that lipid parameters, particularly HDL-C, decline with worsening Child-Pugh and MELD scores, reflecting impaired hepatic synthetic capacity and more severe disease states. Reduced HDL-C has been linked to poorer outcomes in cirrhosis. Previous research demonstrated that HDL-related biomarkers correlate inversely with disease severity and are independent predictors of survival in CLD, suggesting that HDL-C may serve as a prognostic indicator. Trieb et al. [17] demonstrated that HDL-related biomarkers reflect deterioration in liver function and reliably predict disease progression, development of complications, and survival in patients with cirrhosis. In particular, HDL-C and apoA-I decrease with increasing severity of the disease regardless of the etiology of cirrhosis. The authors further emphasized that HDL-C and apoA-I show excellent diagnostic accuracies for 90-day and 12-month mortality, which are very similar to those of composite scores such as Child-Pugh and MELD [17]. Similarly, Habib et al. [27] established HDL-C as a liver function test and prognostic factor in non-cholestatic cirrhosis. In patients hospitalized with spontaneous bacterial peritonitis, Llovet et al. [28] found that HDL-C was one of the parameters independently associated with short-term prognosis. In line with these observations, we found that patients in the highest NHHR tertile, where HDL-C levels were lowest, had significantly higher decompensation rates.
While HDL-C has been widely studied in CLD, composite lipid indices such as NHHR have been less extensively evaluated in this context. HDL-C has recognized anti-inflammatory and endothelial protective properties, whereas non-HDL fractions reflect atherogenic and proinflammatory lipoproteins; the NHHR therefore captures a balance between potentially protective and harmful lipid fractions. Studies in metabolic and cardiovascular populations show that ratios incorporating non-HDL and HDL cholesterol capture this balance more effectively than individual lipid measures [18–20]. In our cohort, NHHR demonstrated consistent associations with markers of hepatic dysfunction, correlating positively with MELD-3.0 score, total bilirubin, and INR, and inversely with serum albumin. These relationships indicate that higher NHHR aligns with more advanced liver dysfunction, reflecting impaired synthetic capacity and cholestatic injury. Importantly, our cohort comprised patients with CLD of diverse etiologies, including alcoholic liver disease, metabolic dysfunction-associated steatotic liver disease (MASLD), viral hepatitis-related liver disease, and other causes, thereby enhancing the generalizability of these findings across the etiologic spectrum of CLD. Notably, NHHR was independently associated with clinical decompensation in multivariable logistic regression models, underscoring its potential utility as an integrated indicator of disease severity. Our findings resonate with emerging evidence linking lipid and lipoprotein disturbances to hepatic outcomes. An et al. [29] reported that the NHHR was associated with worse liver function and independently predicted adverse outcomes in liver disease cohorts, suggesting that composite lipid ratios capture metabolic and lipoprotein derangements beyond conventional measures. Similarly, in non-alcoholic fatty liver disease populations, studies have demonstrated positive associations between NHHR and the severity of steatosis and fibrosis, reinforcing the concept that dysregulated lipoprotein balance parallels progressive hepatic injury [24, 30]. Kim et al. [20] further showed that NHHR outperformed the apolipoprotein B/apolipoprotein A1 ratio in identifying metabolic syndrome and insulin resistance. Population-based analyses from NHANES have also demonstrated significant associations between higher NHHR and the presence of MASLD and hepatic steatosis [21, 22]. Extending these observations to prognostic outcomes, Zhang et al. [23] identified elevated NHHR as a predictor of mortality in patients with MASLD, particularly cardiovascular mortality in those without advanced fibrosis, indicating prognostic implications beyond liver biochemistry alone. Shi et al. [13] demonstrated that low HDL-C independently predicted 90-day transplant-free mortality in patients with hepatitis B virus (HBV)-related acute-on-chronic liver failure, underscoring the critical role of HDL-related pathways in immune dysfunction and hepatic failure. Likewise, Mo et al. [14] developed a prognostic model based on serum apolipoprotein A-I that accurately stratified mortality risk in patients with HBV-related acute-on-chronic liver failure and acutely decompensated cirrhosis, highlighting the loss of protective lipoproteins as a marker of hepatic reserve and systemic inflammation. Although these studies primarily focused on metabolic and virus-related liver disease, their findings align closely with our observations in a broader CLD population. This convergence suggests that composite lipid measures such as NHHR may reflect a shared pathophysiologic axis linking impaired hepatic synthetic function, systemic inflammation, and metabolic stress across diverse etiologies and stages of liver disease. The positive associations between higher NHHR and adverse liver outcomes likely reflect multiple interrelated mechanisms. Specifically, an imbalance between non-HDL and HDL cholesterol may signify an excess of remnant and atherogenic lipoproteins relative to protective HDL particles, fostering systemic inflammation and endothelial dysfunction that can contribute to progressive hepatic injury and the development of portal hypertension. At the same time, a relative deficiency of functional HDL may compromise reverse cholesterol transport. Experimental and clinical studies have shown that CLD can alter HDL composition and reduce cholesterol efflux capacity, indicating impaired HDL functionality rather than merely decreased HDL concentrations [10, 17]. This functional impairment may diminish HDL-mediated reverse cholesterol transport and weaken its antioxidant and anti-inflammatory effects, thereby contributing to hepatocellular injury and systemic inflammation [31]. Although the precise mechanisms linking dysregulated lipoprotein balance to clinical decompensation remain incompletely understood, these alterations likely represent the combined effects of impaired hepatic synthesis and systemic metabolic stress inherent to advanced liver disease. Collectively, these data support the concept that NHHR integrates pro-inflammatory and protective lipoprotein signals in a manner that reflects hepatic reserve and the overall systemic metabolic burden across the spectrum of CLD.
We also demonstrated that incorporation of NHHR into established prognostic models resulted in a significant improvement in risk discrimination and patient reclassification, as reflected by higher AUC values and favorable NRI and IDI indices compared with a base clinical model comprising age, sex, and MELD-3.0 score. Notably, the gain in predictive performance was most pronounced within the highest NHHR tertile, suggesting that its added value is greatest among patients with substantial lipoprotein imbalance and more advanced metabolic derangement. From a clinical perspective, these results underscore the potential utility of NHHR as a pragmatic risk stratification tool beyond established clinical severity scores. Because NHHR can be readily derived from routinely available lipid panels, it represents a low-cost and accessible biomarker that may help identify patients who warrant closer surveillance and timely clinical intervention. This simplicity and broad availability may be particularly advantageous in resource-limited settings, where access to advanced imaging modalities or repeated comprehensive scoring assessments is often constrained.
This study has certain limitations that merit consideration. First, the cross-sectional design precludes causal inference. Consequently, although higher NHHR was associated with the presence of decompensation, it remains unclear whether elevated NHHR contributes to the development of decompensation or whether advanced liver dysfunction and metabolic alterations accompanying decompensation lead to changes in lipid profiles and consequently a higher NHHR. This possibility of reverse causality should be considered when interpreting the findings, and prospective longitudinal studies are required to clarify the temporal relationship between NHHR and hepatic decompensation. Second, this was a single-center study conducted at a tertiary-care hospital, which may limit the generalizability of the findings to other populations or healthcare settings. Although the cohort included patients with diverse etiologies of CLD, including alcohol-related liver disease, MASLD, viral hepatitis, and other causes, the study population may be more representative of patients with moderate to advanced disease typically seen in tertiary centers. Therefore, multicenter studies involving more diverse and community-based populations are needed to confirm the broader applicability of these findings. Third, detailed lipid characterization was not available. Specifically, data on apolipoproteins, LDL and HDL subclasses, and direct measures of HDL functionality were not assessed. These parameters may provide additional insights into lipid metabolism and inflammatory processes in CLD and could further clarify the biological mechanisms underlying the observed association between NHHR and decompensation. In addition, although key clinical confounders were adjusted for in multivariable analyses, residual confounding from unmeasured factors cannot be entirely excluded. NHHR should therefore be interpreted in the appropriate clinical context, as lipid levels may be influenced by factors such as acute infection, systemic inflammation, nutritional status, and metabolic comorbidities. Nevertheless, the relatively large sample size, systematic assessment of liver disease severity, and integration of discrimination and reclassification analyses strengthen the validity of the findings.
In conclusion, this study provides robust evidence that an elevated NHHR is independently associated with the presence of clinical decompensation in patients with CLD. The consistent correlations observed between NHHR and established liver function markers, including albumin, INR, total bilirubin, and MELD-3.0, reinforce the relevance of systemic metabolic imbalance in advanced hepatic dysfunction. The incremental discriminatory value conferred by NHHR beyond traditional clinical and biochemical parameters supports its consideration as a simple and accessible biomarker for risk stratification and targeted clinical evaluation in CLD. Integration of NHHR into routine clinical assessment may help identify patients with decompensation in CLD and support more comprehensive clinical evaluation and management.
AUC: area under the receiver operating characteristic curve
CIs: confidence intervals
CLD: chronic liver disease
CTP: Child-Turcotte-Pugh
HBV: hepatitis B virus
HDL-C: high-density lipoprotein cholesterol
IDI: integrated discrimination improvement
INR: international normalized ratio
LDL-C: low-density lipoprotein cholesterol
MASLD: metabolic dysfunction-associated steatotic liver disease
MELD: Model for End-Stage Liver Disease
NHHR: non-high-density lipoprotein/high-density lipoprotein cholesterol ratio
NRI: net reclassification improvement
ORs: odds ratios
ROC: receiver operating characteristic
RC: Conceptualization, Investigation, Writing—review & editing, Supervision. AK: Validation, Writing—original draft, Writing—review & editing. BKS: Writing—original draft, Writing—review & editing. AD: Conceptualization, Investigation, Writing—original draft. ST: Validation, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
The study was approved by the Ethics Committee of ESIC Postgraduate Institute of Medical Sciences and Research, Basaidarapur, New Delhi, India (Approval No. ESIPGIMSR-IEC/2024013). It was conducted in accordance with the principles of the Declaration of Helsinki.
Informed consent to participate in the study was obtained from all participants.
Informed consent to publication was obtained from relevant participants.
The data are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 587
Download: 12
Times Cited: 0
Diana Abu-Halaka ... Oren Tirosh
