Affiliation:
Faculty of Medicine, Tbilisi State Medical University, 0186 Tbilisi, Georgia
Email: jui.gaikwad@gmail.com
ORCID: https://orcid.org/0009-0000-1027-1507
Affiliation:
Faculty of Medicine, Tbilisi State Medical University, 0186 Tbilisi, Georgia
ORCID: https://orcid.org/0009-0003-1719-5879
Affiliation:
Faculty of Medicine, Tbilisi State Medical University, 0186 Tbilisi, Georgia
ORCID: https://orcid.org/0000-0003-4534-2111
Affiliation:
Faculty of Medicine, Tbilisi State Medical University, 0186 Tbilisi, Georgia
ORCID: https://orcid.org/0009-0007-6659-519X
Explor Cardiol. 2026;4:1012100 DOI: https://doi.org/10.37349/ec.2026.1012100
Received: November 06, 2025 Accepted: February 10, 2026 Published: March 08, 2026
Academic Editor: Réda Ibrahim, Institut de Cardiologie de Montreal, Canada
The aim is to evaluate the transannular patch (TAP) repair and valve-sparing repair (VSR) techniques following tetralogy of Fallot (TOF) correction, focusing on post-operative complications and cardiac function. A comprehensive search was performed in PubMed, EMBASE, and Scopus using relevant terms like “Tetralogy of Fallot, right ventricular outflow tract (RVOT), VSR, pulmonary valve replacement, transannular-patch repair”. Results indicated that VSR is favored due to its shorter cardiopulmonary bypass duration, preservation of the pulmonary valve, less demanding surgical requirements, shorter post-operative hospital stays, lower mortality rates, survival of at least 30 years, reduced pulmonary regurgitation, decreased right ventricular dysfunction, and improved physical activity tolerance and neurodevelopment. While TAP alleviates RVOT obstruction (RVOTO), it is associated with long-term pulmonary regurgitation. Both TAP and VSR are effective in managing TOF, but VSR provides better valve function preservation and long-term outcomes.
Tetralogy of Fallot (TOF) is a congenital heart defect characterized by a ventricular septal defect (VSD), pulmonary stenosis, right ventricular (RV) hypertrophy (RVH), and an overriding aorta [1]. Currently, surgical correction of TOF is critically important, with timely and effective surgery being essential for achieving favorable long-term outcomes [2].
TOF is present in 3 out of every 10,000 live births and is the most prevalent cause of cyanotic heart disease in individuals beyond the newborn stage, representing up to 10% of all congenital heart anomalies [3].
Globally, TOF constitutes 3–5% of congenital heart defects, with its occurrence varying from 0.4–2.1% in Europe, 3–8% in China, and approximately 6.7% in the USA. In contrast, Sub-Saharan Africa exhibits higher rates, ranging from 5–20% across various regions, underscoring the necessity for improved regional data to inform healthcare planning [4].
Due to the complexity and prevalence of TOF, comparing various surgical repair methods is crucial to enhance patient care. Specifically, evaluating the traditional use of transannular patches (TAPs) against valve-sparing (VS) repair (VSR) techniques is a key research focus [5]. TAPs are commonly employed to alleviate RV outflow tract (RVOT) obstruction (RVOTO) during surgery, but are often linked to postoperative issues like pulmonary regurgitation and RV dysfunction [6]. Conversely, VS procedures have emerged as a potential solution to mitigate these adverse effects, necessitating comparison to identify the optimal surgical approach [7].
This review aims to summarize current findings regarding the advantages and disadvantages of TAP versus VSRs, thereby establishing the role of each method in contemporary practice [8]. Identifying the most effective surgical technique for TOF treatment in clinical settings is crucial. The choice of surgical method directly affects patient outcomes, including mortality, quality of life, and the occurrence of postoperative complications such as RV dysfunction and pulmonary valve insufficiency [9]. Therefore, a thorough analysis of existing literature not only informs surgical practices but also guides healthcare providers in making decisions for treating congenital heart defects [10]. Ultimately, clarifying the outcomes of VSRs compared to TAP will enhance understanding of the best management strategies for TOF patients [11].
TOF is a cyanotic congenital heart defect characterized by four anatomical abnormalities. These include a large, non-restrictive VSD, an RVOTO that can be subvalvular, subinfundibular, valvular, or supravalvular, the aortic root overriding the ventricular septum, and RVH [3]. It consists of four distinct heart defects that primarily affect infants and children. Babies with TOF struggle to get sufficient blood to their lungs, resulting in the body receiving blood with low oxygen levels. The four defects in TOF include: 1. pulmonary stenosis, where a narrowed or stiffened pulmonary valve hinders the pulmonary artery from delivering adequate blood from the heart to the lungs; 2. VSD, which is a hole between the ventricles allowing the mixing of oxygen-rich and oxygen-poor blood; 3. overriding aorta, where the aorta is positioned above the ventricles, leading to insufficient oxygen-rich blood reaching the body; and 4. RVH, where the right ventricle is abnormally thick, making it difficult for blood to pass through the pulmonary valve (Figure 1). These defects result in poor blood oxygenation, causing cyanosis, a bluish discoloration of the lips, skin, and nails [11]. Notably, TOF has favorable survival rates following surgical intervention. Additionally, the age of the patient is crucial in surgical planning, with complete repair associated with better outcomes than staged repair [12]. The causes of TOF are multifactorial, with genetics playing a significant role. It is linked to the deletion of the 22q11.2 gene [13], along with other mutations such as trisomy 21, Alagille syndrome, Cat Eye Syndrome, or CHARGE and VATER/VACTERL syndromes. Variants in the key cardiac transcription factor NKX2.5 were the first to be identified as a genetic cause of TOF [14].
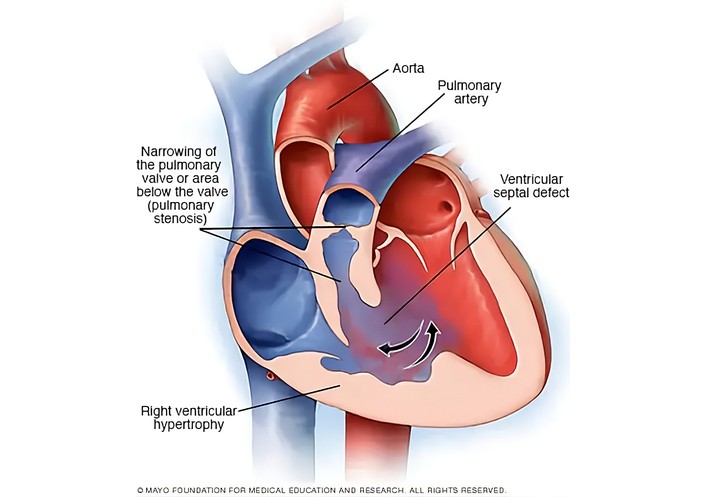
Tetralogy of Fallot illustration. Reprinted with permission from [14]. © 2019 British Journal of Anaesthesia.
In addition to genetic abnormalities, a rubella infection in the mother is a significant factor contributing to TOF. If the mother contracts the infection during the first trimester, it can result in congenital rubella syndrome, which includes serious birth defects like congenital heart defects, such as TOF [15]. The precise role of prenatal rubella infection or genetic factors in causing TOF remains unclear [15]. The severity of the right-to-left shunt is influenced by the pressure difference between the right and left ventricles and the RVOTO. Dynamic RVOTO, often caused by muscle spasms, can worsen under stress, leading to cyanotic episodes. A reduction in systemic vascular resistance (SVR) or an increase in pulmonary vascular resistance (PVR) can further exacerbate the shunt. Despite the desaturation, cardiac output is usually maintained initially. RVH results in diastolic dysfunction, making the heart dependent on venous return and sensitive to tachycardia, which decreases filling and raises oxygen demand [14].
TOF typically manifests with mild to moderate cyanosis, which becomes apparent as the obstruction in the right ventricle worsens. Patients experience cyanosis without signs of respiratory distress or symptoms of congestive heart failure, such as poor feeding, failure to thrive, or sweating, because the obstruction affects only pulmonary blood flow. Due to the RVOTO, the second heart sound is heard as a single, loud sound accompanied by a harsh systolic ejection murmur [3].
The primary clinical feature of TOF is the occurrence of hypercyanotic episodes, also known as “tet spells”. These episodes are characterized by a sudden and severe drop in oxygen saturation, along with symptoms like intense cyanosis, rapid and shallow breathing (hypercapnia), fainting, lethargy, and limpness. These symptoms typically appear between 2 and 6 months of age, especially during periods of agitation, crying, or dehydration, which exacerbate the RVOTO [3]. Infants often adopt a knee-to-chest position to increase SVR and improve blood flow to the lungs.
In older children, exertional dyspnea and gradually worsening cyanosis are observed, and they may squat during physical activity to alleviate symptoms [16]. In adults, if TOF remains uncorrected, complications such as heart failure, atrial and ventricular arrhythmias, and infective endocarditis can develop [16, 17].
The TAP repair method has existed since the 1990s for addressing RVOTO. It is particularly favored for patients with significant pulmonary annulus hypoplasia and severe RVOTO [11, 18]. The TAP procedure typically involves making an incision from the main pulmonary artery to the infundibulum, allowing surgeons to access the constricted outflow tracts. To gain deeper access into the infundibulum, the RV muscle is excised, and the pulmonary valve leaflet tissue is either partially or entirely removed. Patch material is then used to reconstruct the RVOT, extending to the bifurcation of the pulmonary arteries. While this procedure effectively removes the obstruction, it results in notable insufficiency in pulmonary circulation. Postoperative ICU stay was on average 1–2 days longer for TAP patients compared with those undergoing VSR in the cohort of 11,723 patients, due to underlying RVH, diastolic dysfunction, and the low cardiac output associated with diastolic dysfunction [5, 18–20]. A significant benefit of TAP is its capacity to immediately address RV hypertension [21, 22]. Although TAP can alleviate RV tract obstruction, pulmonary regurgitation remains the most common complication [23].
In response to the limitations of TAP and its long-term effects, VSR techniques have been developed as a superior alternative, aiming to preserve the native valve function and minimize post-operative dysfunctions. Surgeons perform VSR surgery to address slightly narrowed pulmonary valves, keeping the valves intact [24]. To preserve the valves, several techniques are employed, such as commissurotomy, delamination plasty, and T-shaped infundibulotomy [11, 25]. Compared to the TAP procedure, VSR offers significant advantages, including a reduced risk of pulmonary regurgitation, a lower likelihood of future reoperations, and a decreased risk of developing arrhythmias [11, 24]. Patients who underwent VSR did not require additional surgical or interventional procedures [11].
Valve-sparing procedure is intended to preserve the annulus integrity of the pulmonary valve and can be used for infants without severe annular hypoplasia [26]. Patients who received VSR were associated with shorter cardiopulmonary bypass time, shorter duration of ventilation, shorter ICU stay, and shorter hospital stay [5, 26–28].
Infants who required more intensive methods of surgery to enlarge the valvular area did not undergo VSR [27–29]. In a meta-analysis of 11,723 patients (TAP: 6,171, VS: 5,045), the VS group demonstrated a 60% lower mortality risk (RR = 0.40). This implies that the mortality risk in the TAP group was comparatively higher [5].
Moreover, the pulmonary regurgitation did occur in patients after the treatment using VSR, but the regurgitation was mostly mild [25], as compared to patients treated with TAP, the ratio for severe pulmonary regurgitation was 6.9 [30].
Planning for management and surgery of TOF is done on the basis of two major goals: complete relief of the RVOTO and long-term function of the right ventricle. The option during surgery to use either a TAP or a VSR is found to be influenced by several determinants, like pulmonary annulus dimension, pulmonary valve thickness, patient age, long-term surgical outcome, and ventricular function preservation as observed in Table 1 [31]. TAP is the most commonly utilized technique for the delivery of free RVOT flow, as seen in Figure 2. Performing an incision through the PV with a patch reserved for maintaining low post-operative ventricular pressure is utilized. However, this can result in pulmonary regurgitation, thus resulting in volume pressure in the right ventricle [32, 33]. Despite this, the long-term effects are pulmonary regurgitation and RVH. Chronic pulmonary regurgitation usually leads to RV dilation, arrhythmias, and eventually, later-in-life pulmonary valve replacement [19, 20, 34]. These have underscored the need for VS procedures.
Comparison of post-op effects of TAP repair vs. VSR repair.
| Parameter | Transannular patch (TAP) repair | Valve-sparing repair (VSR) |
|---|---|---|
| Cardiopulmonary bypass duration | Longer | Shorter |
| Pulmonary valve | Destroyed | Preserved |
| Operative demands | Difficult | Less intensive |
| Ventilation duration | Higher | Lower |
| Postoperative ICU stay | Longer | Shorter |
| Mortality rate | Higher | Lower |
| Pulmonary regurgitation | High incidence | Significantly lower |
| Right ventricular dysfunction | Higher risk | Lower risk |
| Right ventricular outflow tract gradients | Stable | Comparatively unstable |
| Postoperative arrhythmias | Higher | Lower |
| Reoperations | Higher | Lower |
| Physical activity tolerance | Reduced | Increased |
| Cognitive and motor development | Inferior | Superior |
| Pulmonary artery aneurysms | Present | Absent |
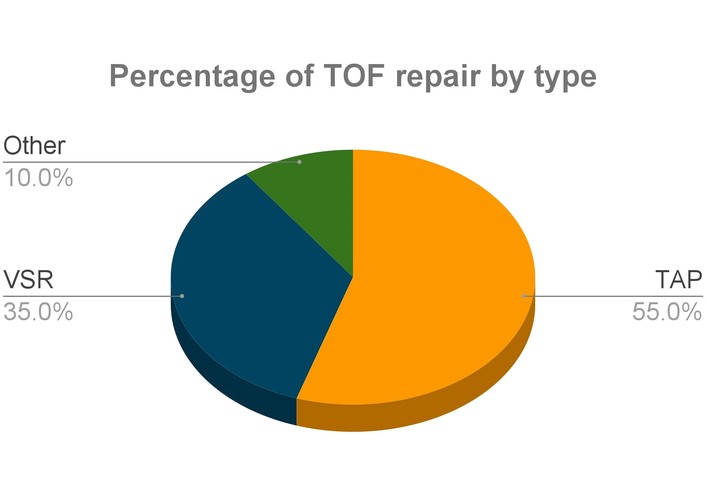
Percentage of TOF repair by type. 55.0% of TOF repairs were operated by TAP, 35.0% by VSR, and the other repair techniques utilized were 10.0% [35]. TOF: tetralogy of Fallot; TAP: transannular patch; VSR: valve-sparing repair.
VSRs are superior to TAP repairs in preserving RV function. VSR patients had constant RVOT gradients and minimal regurgitation [15]. VSR is directed at the relief of the RVOTO and preservation of the native valve leaflets. This is a surgical approach that is reserved for patients with mobile and functioning valve leaflets [3]. VSR significantly reduces chronic pulmonary regurgitation and, therefore, maintains RV dilation and minimizes the need for future PV repairs and interventions. Conversely, patients who received TAP repair developed increased postoperative arrhythmias due to chronic overload by pulmonary regurgitation. The duration of cardiopulmonary bypass was also much lower for patients undergoing VSR. In addition, patients undergoing VSR required shorter ventilation times with ~15.3 h shorter ventilation on average as compared to TAP [5]. There was no significant difference in the rate of residual RVOTO between the TAP and VS groups [36]. Reoperations due to pulmonary regurgitation were more common in TAP repair patients. TAP repair commonly results in pulmonary valve incompetence, causing pulmonary regurgitation and volume overloading of the right ventricle [27]. Since VSR maintains pulmonary valve competence, patients showed enhanced exercise tolerance and increased physical activity compared with TAP repair patients [36]. Infants who underwent VSR had superior cognitive and motor development compared to infants who underwent TAP repair [26]. Patients with VSR experienced fewer rehospitalizations due to cardiac-related reasons and had better late outcomes [19]. However, VSR is technically more challenging and needs advanced surgical techniques. Different studies have reported better outcomes in VSR patients, as it has a lower reintervention rate [26].
VSR presents a higher level of technical difficulty and complexity, yet this challenge can be mitigated by enhancing the surgeon’s experience and support, which in turn boosts the performance score [37]. For patients with pulmonary annular hypoplasia or anatomical constraints, VSR remains a viable option due to its ability to preserve the native valve. In a study involving 19 patients, valve preservation was achieved in 11 cases (58%), while the others experienced only mild stenosis during follow-up [38]. Although VSR can be financially burdensome, particularly in low-income countries, it can still be prioritized for patients who are suitable and eligible, given its safe valve preservation. If the surgery does not demand extensive surgical expertise, TAP could serve as an alternative.
TAP repair usually involves pulmonary valve destruction or removal to reduce obstruction in RV volume, resulting in free pulmonary regurgitation and chronic overload of the right ventricle. The overload, in the long run, can result in RV dilation and dysfunction, potentially progressing to heart failure and arrhythmias. Research indicates that patients who have had TAP repair have an increased risk of significant pulmonary regurgitation with serious long-term consequences. Due to distortion of physiological pulmonary valve hemodynamics, chronic pulmonary regurgitation-induced pulmonary artery aneurysms and increased outflow volumes have also been noted in patients after TAP repair [27]. Table 1 provides an overview of the differences between the two methods.
While the preoperative pulmonary valve annulus Z-score is a valuable tool, it is not sufficient by itself to determine whether to opt for VSR or a TAP. Recent studies and reviews indicate that many centers have successfully performed VSR on patients with preoperative Z-scores that were once deemed too low. Factors such as intraoperative functional evaluation (including residual gradient, RV pressure, and TEE), leaflet morphology (extent of dysplasia), the mechanism of RVOTO (whether infundibular or valvar/annular), coronary anatomy, and the experience of the surgeon and center are equally or more critical in making the final decision. When VSR is chosen correctly, it tends to result in less late pulmonary regurgitation and better preservation of RV function compared to TAP, although selection bias and the experience of the center are significant confounding factors.
Although the pulmonary valve annulus Z-score is a widely used preoperative measure for deciding between VSR and TAP in TOF, recent findings suggest that relying solely on the Z-score is inadequate for determining VSR suitability. In a cohort study by Siddiqi et al. [39], numerous patients with Z-scores below −2 successfully underwent VSR, and the Z-score was not significantly linked to the risk of reintervention, highlighting the importance of intraoperative valve function assessment and residual gradients. Similarly, Guariento et al. [40] discovered that very low Z-scores (≤ −2.85) combined with moderate to severe postoperative pulmonary regurgitation predicted poorer midterm valve competence, indicating that postoperative functional evaluation should complement preoperative assessments.
Choosing VSR involves considering several factors: the preoperative Z-score of the pulmonary valve annulus (typically greater than −2, although preservation is feasible with Z-scores as low as −3), the morphology of the valve leaflets (tricuspid, bicuspid, or dysplastic), the site and cause of RVOTO (whether infundibular or valvar), and the anatomy of the coronary arteries. An intraoperative evaluation using TEE, along with checking residual gradients and RV pressures, is crucial to verify if valve preservation is appropriate [39, 40].
Surgical treatment of TOF has increasingly focused on preserving or reconstructing the pulmonary valve to minimize long-term pulmonary regurgitation, RV enlargement, and the need for further interventions. VSR methods are used when the native pulmonary valve can be preserved, based on the morphology of the leaflets, the size of the annulus, and intraoperative evaluation, rather than solely on Z-scores. A frequently employed technique is the T-shaped infundibulotomy with resection of RVOT muscle, involving a longitudinal cut through the infundibulum and a transverse cut across the anterior annulus to alleviate obstruction. The infundibular muscle bundles are removed, the VSD is closed, and a commissurotomy is performed to enhance leaflet opening. An anteriorly positioned shield-shaped patch enlarges the RVOT without cutting through the annulus, encouraging native valve growth and maintaining function in patients with moderate stenosis or mild hypoplasia [25].
When valve leaflets are fused or slightly dysplastic, commissurotomy combined with leaflet thinning or delamination is used to restore mobility and increase the effective orifice. Serial Hegar dilators may be used intraoperatively to assess valve opening, guiding the decision on whether patch augmentation is necessary. This technique is appropriate for patients with flexible leaflets and mild to moderate stenosis and can often avoid large ventriculotomy or a TAP [4, 37].
In situations where the pulmonary annulus is severely underdeveloped or the valve leaflets are dysplastic and immobile, modified monocusp repair or patch-based reconstruction is performed. This involves cutting through the pulmonary annulus and reconstructing the RVOT with a pericardial or synthetic (ePTFE) patch shaped into a single cusp (monocusp) valve. Standard RVOT muscle resection and VSD closure are also carried out. This approach provides a functional valve substitute when a simple VSR is not possible, reducing the pulmonary regurgitation associated with a non-valved TAP [38, 41].
The selection of these techniques is guided by anatomical considerations: surgeons evaluate the shape of the pulmonary leaflets, the functional valve opening, the annulus size, and the site of RVOTO (whether infundibular or valvular). A step-by-step approach is often employed, starting with less invasive preservation methods such as commissurotomy, delamination, and dilation, and resorting to patch augmentation or reconstruction only if necessary [41, 42].
Studies on mid-term and long-term outcomes indicate that when VSR or reconstruction procedures are carefully chosen, they lead to lasting valve function, decreased RV dilation, and low rates of reintervention, even in patients with borderline annular sizes [40].
Overall, the literature highlights a move away from solely relying on Z-scores towards a detailed intraoperative evaluation of valve morphology, combined with personalized, anatomy-based strategies that range from simple VS techniques to complex monocusp reconstruction when needed. This customized approach enhances long-term pulmonary valve function and patient outcomes [4, 41].
TOF is a complex congenital heart condition with various symptoms that tend to worsen over time. Accurate surgical procedures with minimal complications are essential. One such symptom, RVOTO, can be promptly addressed with TAP repair. However, this method may lead to complications such as pulmonary regurgitation and RV dysfunction. Although valve sparing is currently popular and frequently utilized, the TAP repair remains the most commonly employed method for correcting TOF. This dominance is largely attributed to its historical significance as one of the earliest surgical techniques for TOF correction, making it a well-established and familiar approach, particularly in developing and underdeveloped countries. This is due to factors such as limited access to modern and advanced surgical equipment, cost-effectiveness, and the ease with which it can be implemented. VSRs offer significant advantages, including improved long-term survival, maintained valve function, and a lower risk of arrhythmias, ultimately enhancing the patient’s quality of life. Consequently, VSR is considered a superior surgical option compared to TAP.
RV: right ventricular
RVH: right ventricular hypertrophy
RVOT: right ventricular outflow tract
RVOTO: right ventricular outflow tract obstruction
SVR: systemic vascular resistance
TAP: transannular patch
TOF: tetralogy of Fallot
VS: valve-sparing
VSD: ventricular septal defect
VSR: valve-sparing repair
JRG: Conceptualization, Writing—original draft, Writing—review & editing, Supervision, Validation. JA: Writing—original draft, Writing—review & editing. KS: Writing—original draft, Writing—review & editing. LTDC: Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 846
Download: 65
Times Cited: 0
