Affiliation:
1Department of Chemical Engineering, Z. H. Sikder University of Science and Technology (ZHSUST), Shariatpur 8024, Bangladesh
ORCID: https://orcid.org/0009-0002-0620-9297
Affiliation:
2Department of Chemical Engineering, College of Environmental Science and Forestry, State University of New York, Syracuse, NY 13210, USA
ORCID: https://orcid.org/0009-0007-5166-1764
Affiliation:
1Department of Chemical Engineering, Z. H. Sikder University of Science and Technology (ZHSUST), Shariatpur 8024, Bangladesh
2Department of Chemical Engineering, College of Environmental Science and Forestry, State University of New York, Syracuse, NY 13210, USA
Affiliation:
1Department of Chemical Engineering, Z. H. Sikder University of Science and Technology (ZHSUST), Shariatpur 8024, Bangladesh
3Center for Intelligent Manufacturing, Department of Civil and Mechanical Engineering, SUNY Polytechnic Institute, Utica, NY 13502, USA
Email: alamm7@sunypoly.edu
ORCID: https://orcid.org/0000-0002-2636-5038
Explor BioMat-X. 2026;3:101363 DOI: https://doi.org/10.37349/ebmx.2026.101363
Received: August 04, 2025 Accepted: March 04, 2026 Published: March 24, 2026
Academic Editor: Lalit Mohan Pandey, Indian Institute of Technology Guwahati, India
Electrochemical sensors have emerged as powerful tools for the detection and monitoring of neurotransmitters, offering high sensitivity, selectivity, and potential for real-time analysis. Neurotransmitters play a crucial role in regulating various physiological and neurological processes, and imbalances in their levels are linked to a wide range of neurological disorders, including Parkinson’s disease, depression, Alzheimer’s disease, and epilepsy. This review highlights recent advancements in electrochemical sensor technologies for neurotransmitter detection, focusing on innovations that enhance performance through the use of nanomaterials, wearable devices, and multiplexed sensing techniques. The integration of nanomaterials such as graphene, carbon nanotubes, and metal nanoparticles has significantly improved sensor sensitivity and selectivity, enabling more accurate detection even at low concentrations. Furthermore, the development of flexible, wearable, and implantable sensors is facilitating continuous, non-invasive monitoring of neurotransmitter levels in real time. Advances in multiplexed sensors are enabling the simultaneous detection of multiple neurotransmitters, providing a more comprehensive approach to disease diagnosis and management. Despite these promising developments, challenges remain, including issues of selectivity, stability, and long-term monitoring. Nevertheless, electrochemical sensors hold great potential for transforming the way neurological disorders are diagnosed and managed, offering opportunities for personalized, real-time monitoring and more effective treatment strategies.
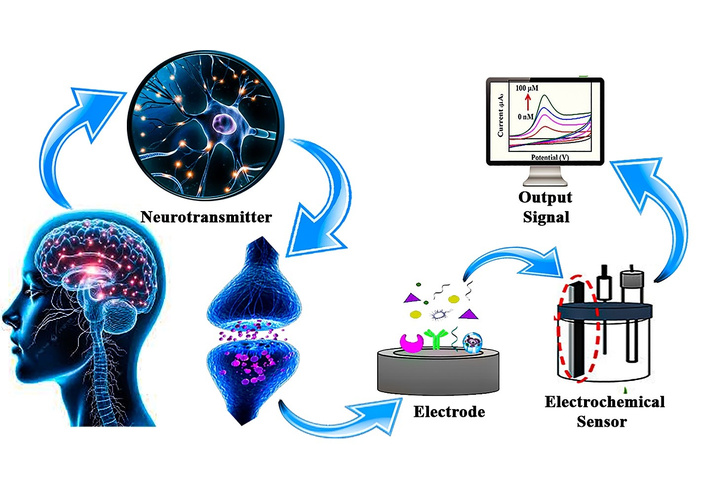
Neurotransmitters are chemical messengers that play a pivotal role in the transmission of signals within the brain and nervous system. They are involved in regulating a broad spectrum of physiological processes, including mood, cognition, behavior, and movement. Disruptions or imbalances in neurotransmitter levels are associated with a wide range of neurological and psychiatric disorders, such as Parkinson’s disease, Alzheimer’s disease, schizophrenia, depression, and epilepsy. These disorders not only affect the quality of life of millions worldwide but also pose significant challenges for clinicians in terms of accurate diagnosis, monitoring progression, and tailoring effective treatments [1, 2]. Thus, the measurement and monitoring of neurotransmitters in biological systems, especially in real time, are crucial for understanding the underlying pathophysiology of these conditions and for developing personalized treatment plans. Although techniques such as LC-MS offer high accuracy, they require complex instrumentation and are not suitable for real-time or portable sensing, where electrochemical nanosensors provide significant advantages [3] or enzyme-linked immunosorbent assays (ELISA) [4, 5], although highly accurate, are often expensive, time-consuming, require specialized equipment, and can only provide results in laboratory settings. These limitations underscore the need for more accessible, cost-effective, and rapid diagnostic tools for neurotransmitter analysis [6].
Electrochemical sensors have emerged as a promising solution to these challenges. These sensors offer numerous advantages, including high sensitivity, selectivity, real-time monitoring, and portability, making them ideal for point-of-care diagnostics and continuous monitoring of neurotransmitter levels. Electrochemical sensors operate based on the principle of measuring the electrical signal (current or voltage) generated during the oxidation or reduction of neurotransmitter molecules at the sensor’s electrode surface. This signal is directly related to the concentration of the neurotransmitter in the sample, allowing for rapid and quantitative analysis [7–9]. In recent years, significant advancements have been made in the development of electrochemical biosensors for neurotransmitter detection, driven by innovations in nanomaterials, wearable electronics, and multiplexed sensing platforms. Nanomaterials, such as graphene [10, 11], carbon nanotubes (CNTs) [12, 13], and metal nanoparticles [14], have been incorporated into sensor designs to enhance performance, improving both sensitivity and selectivity by increasing the surface area and facilitating better electron transfer [15]. Meanwhile, wearable electrochemical sensors offer the potential for continuous, non-invasive monitoring of neurotransmitters in sweat or interstitial fluid, a major step forward in enabling real-time, personalized treatment strategies for patients with neurological disorders [16, 17]. Additionally, multiplexed sensors that can simultaneously measure multiple neurotransmitters provide a more comprehensive approach to disease diagnosis and progression monitoring, further enhancing their utility in clinical settings. Figure 1 is a schematic illustration of neurotransmitter detection using a nanomaterials-based biosensor. Neurotransmitters released at the synapse bind to specific receptors, representing biochemical signaling in the brain. A biological sample (e.g., blood or related biofluid) containing neurotransmitters is collected and introduced to a sensor platform. The sensing element is functionalized with advanced nanomaterials such as CNTs, graphene, fullerenes, and other nanostructures which enhance sensitivity and selectivity. The interaction between neurotransmitters and the nanomaterial-based sensor is transduced into a measurable signal, enabling the detection and analysis of neurotransmitter levels relevant to neurological function and disorders.
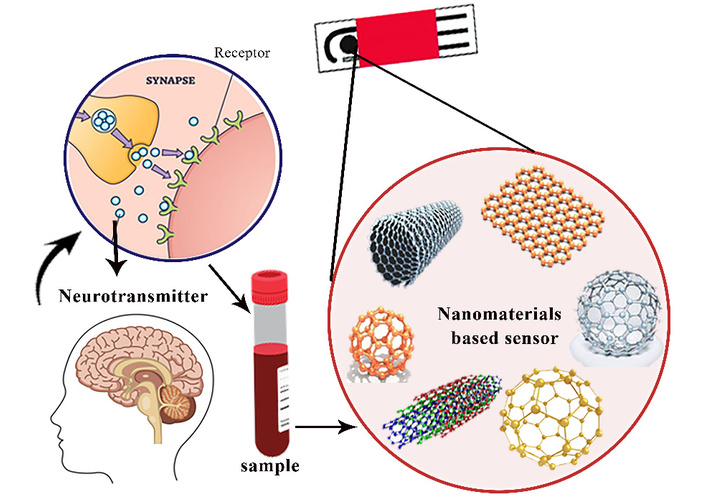
The electrochemical detection of neurotransmitter using nanomaterials-based sensor.
This review aims to explore the state-of-the-art in electrochemical sensors for neurotransmitter detection, focusing on the following areas: (i) the fundamental principles of electrochemical detection, (ii) recent advancements in sensor technologies, particularly the use of nanomaterials and wearable or implantable systems, (iii) the application of multiplexed sensors for simultaneous detection of multiple neurotransmitters, and (iv) the current and potential clinical applications of electrochemical neurotransmitter sensors in diagnosing and monitoring neurological diseases. Additionally, we will discuss the challenges faced in this field, such as selectivity, interference from other biomolecules, and long-term stability of sensors, as well as the future directions and potential impact of these technologies on neurological diagnostics and personalized medicine. By providing an overview of the latest advancements and ongoing challenges in electrochemical sensors for neurotransmitter diagnosis, this review will contribute to a deeper understanding of how these technologies can transform the landscape of neurological disease diagnosis, management, and treatment. With the potential for real-time, point-of-care monitoring, electrochemical sensors represent a promising tool for improving patient outcomes and providing personalized, precision healthcare for individuals living with neurological and psychiatric disorders.
Compared with conventional techniques such as HPLC (10–60 minutes per run) and microdialysis (5–30 minutes per sample), electrochemical sensor provide markedly advanced temporal resolution, enabling millisecond-scale monitoring of rapid neurotransmitter dynamics. They achieve comparable or lower detection limits in the pico-to nanomolar range while avoiding dilution effect essential to sample-based method. Additionally, their miniaturized probe dimensions allow minimally invasive, real-time in vivo measurement with superior spatial localization and reduced tissue disruption [13, 15]. Although electrochemical technique such as fast-scan cyclic voltammetry (FSCV) can achieve sub-second to millisecond temporal resolution, they primarily capture rapid changes in extracellular neurotransmitter concentration rather than individual synaptic release events within the synaptic cleft.
This approach was intended to provide a balanced and representative overview of current progress while highlighting key challenges and future research directions in the field. Articles were selected based on their relevance to sensor design, sensing mechanisms, performance metrics (e.g., sensitivity, selectivity, stability), and clinical or biomedical applicability. Studies not directly related to electrochemical detection or lacking sufficient technical detail were excluded. Priority was given to original research articles and authoritative review papers published in high-quality journals to ensure the reliability and relevance of the discussed findings. The search primarily covered studies published between 2017 and 2025, with particular emphasis on recent advances from the last decade to capture emerging trends and state-of-the-art developments in electrochemical neurotransmitter sensing.
Electrochemical sensors have emerged as powerful tools for real-time detection and monitoring of neurotransmitters, molecules that are central to brain function and communication within the nervous system. Imbalances in neurotransmitter levels are implicated in a wide range of neurological and psychiatric disorders, including Parkinson’s disease, Alzheimer’s disease, depression, schizophrenia, and epilepsy. Therefore, accurate, sensitive, and selective measurement of neurotransmitter levels can offer critical insights into the diagnosis, progression, and treatment of these conditions. Over the past few years, significant advancements have been made in electrochemical sensor technology, resulting in more effective, efficient, and versatile devices for neurotransmitter detection [18, 19]. This review explores the latest innovations in this field, including the integration of nanotechnology, advances in sensor materials, the development of wearable and implantable systems, and the use of artificial intelligence (AI) to enhance sensor performance.
Moreover, Y. Ou et al. (2019) [14] represent that, several successful exemplifications of multiplexed neurotransmitter detection in vivo have been demonstrated using electrochemical sensor platforms. FSCV with carbon-fiber microelectrodes has enabled the simultaneous detection of dopamine and serotonin in rodent brains by exploiting differences in their redox signatures. Electrode surface modification, such as CNT coatings, further enhance sensitivity and temporal resolution. In addition, implantable microelectrode arrays have been used to collect parallel electrochemical signals from multiple brain regions, supporting multiplexed measurements within a single device. Despite these advances, limitations remain. Electrochemical approaches are largely restricted to electroactive neurotransmitters and often struggle with overlapping oxidation potentials, which can compromise selectivity. Long-term in vivo measurements are also challenged by biofouling and tissue responses, reducing sensor stability and reliability over time [14].
The specificity of electrochemical neurotransmitter sensor is governed by the idiosyncratic electrochemical behavior of the neurotransmitter and the surface chemistry of the sensing material. For instance, dopamine and serotonin are redox-active molecules that can be directly oxidized on conductive nanomaterials such as graphene, CNTs, MXenes, and metal nanoparticle-decorated electrodes, as give fast electron-transfer pathways and strong adsorption sites [20]. Although, neurotransmitters such as glutamate and acetylcholine are electrochemically inactive and therefore enzyme-modified electrodes, where enzymes like glutamate oxidase or acetylcholinesterase convert the target molecules into electroactive products (e.g., H2O2) that can be measured amperometrically. Studies using nanostructured materials, including Ni nanowire arrays, MXene-based FETs, and polymer CNT composite, further demonstrate that increasing surface area, catalytic activity, and molecular interaction sites significantly enhances both sensitivity and selectivity [21]. Additionally, molecularly imprinted polymers and metal nanoparticles are frequently introduced to suppress interference from species such as ascorbic acid and uric acid, as abundant in biological fluids. Since selecting an appropriate sensing material requires careful consideration of the neurotransmitter’s redox activity, molecular structure, and interference environment, allowing the sensor design to be optimized for high specificity and reliable performance in complex biological samples [22].
Table 1 summarizes the major classes of electrochemical neurotransmitter sensors, highlighting their transduction mechanisms (amperometric, voltammetric, enzyme-based, and FET-based), fabrication techniques, and representative materials. For each sensor type, typical target neurotransmitters, and analytical performance [e.g., limit of detection (LOD) and temporal response]. Furthermore, the table is intended to give new and experienced researchers a concise overview of how different electrode architectures and fabrication approaches influence sensing performance, thereby facilitating systematic comparison and rational design of next-generation neurotransmitter biosensors.
Compression summary of representative electrochemical sensors for neurotransmitter detection and their fabrication strategies.
| Sensor type | Fabrication technique | Target neurotransmitter | Performance | Reference |
|---|---|---|---|---|
| Amperometric | rGO/Bi2S3 nanorods modified GCE | Dopamine | LOD ≈ 12.3 nM | [20] |
| Amperometric | AuNPs/polymer/rGO/GCE | Dopamine | LOD ≈ 11.5 nM, high selectivity | [21] |
| Voltammetric (DPV, MIP-based) | Electropolymerized molecularly imprinted polymer on GCE | Dopamine, adenine | LOD ≈ 0.12 µM | [22] |
| Enzyme-based amperometric | GlOx/MWCNT/PPy/Pt | Glutamate | LOD ≈ 0.3–140 µM | [23] |
| Voltammetric (DPV) | rGO/GCE | Dopamine | LOD ≈ 0.11 µM | [24] |
| Voltammetric (FSCV) | Carbon fiber microelectrode | Dopamine, serotonin | highly sensitive | [23] |
Electrochemical sensors are powerful analytical tools used to detect neurotransmitters by converting their chemical activity into measurable electrical signals. This process is based on redox (reduction-oxidation) reactions that occur when neurotransmitter molecules interact with the surface of a working electrode [24]. Depending on the nature of the neurotransmitter, it may either donate or accept electrons during this interaction, resulting in a flow of electric current. The magnitude of this current or, in some cases, a change in voltage is directly proportional to the concentration of the neurotransmitter present in the sample. Figure 2 is a schematic representation of a nanomaterial-modified electrochemical biosensor. The analyte from a biological sample interacts with the nanomaterial-modified electrode, generating a chemical signal that is converted by a transducer into an electrical signal, amplified, and recorded as an electrochemical response.
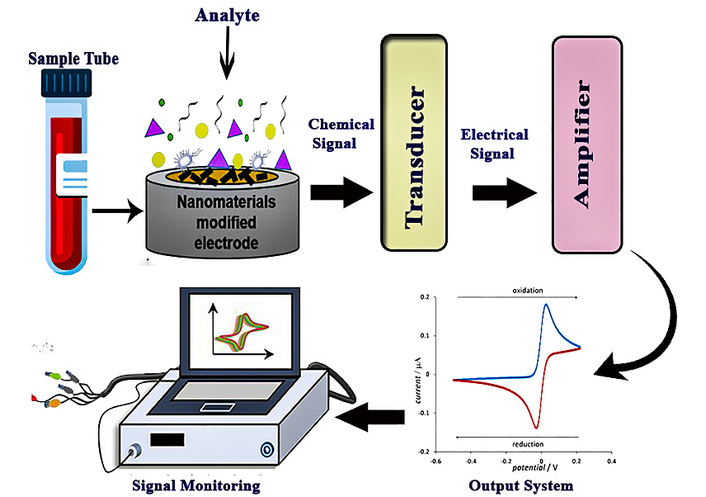
General working principle of an electrochemical sensor. Adapted from [24]. © The author(s) 2023. Licensed under a CC-BY 4.0.
A typical electrochemical sensor consists of three main components: the working electrode, the reference electrode, and the counter electrode. The working electrode is the primary site where the redox reactions take place. The reference electrode provides a stable and known potential against which the working electrode’s potential is measured, ensuring accurate readings. The counter electrode serves to complete the electrical circuit, allowing current to flow through the system [25]. The efficiency and reliability of electrochemical sensors are strongly influenced by several design and material factors. The choice of electrode material (such as carbon, gold, or platinum) can significantly affect sensitivity and electron transfer kinetics. Surface modifications, including the addition of nanomaterials or conductive polymers, can enhance the electrode’s surface area and facilitate better interaction with the target analytes [26]. Furthermore, signal transduction in electrochemical detectors refers to the process by which a chemical event, such as the interaction of an analyte with the sensing surface, is converted into an electrical response. When the target molecules come into contact with the electrode face, it undergoes a response frequently oxidation or reductions at initiates electron transfer. This electron exchange produces measurable changes in electrical parameters such as current, potential, conductivity, or impedance. For example, in an amperometric sensor, the current generated from the redox response is proportional to the analyte concentration, while a potentiometric sensor calculates the potential difference between electrodes in line with the Nernst equation. Conductometric sensors detect variations in the result conductivity caused by the product or consumption of charged species, and impedimetric detectors measure changes in interfacial impedance performing from analyte list. The effectiveness of this transduction process is determined by factors such as electrode material, surface modifications, electrolyte composition, and operating conditions. Eventually, the electrical signal is reused and identified with analyte concentration, enabling particular discovery and monitoring [27]. Moreover, incorporating biological recognition elements such as enzymes, antibodies, or aptamers greatly improves the sensor’s selectivity by allowing it to specifically bind and respond to particular neurotransmitters in complex biological environments [28]. These innovations make electrochemical sensors highly valuable for real-time monitoring of neurochemical activity in both research and clinical settings [29].
Recent developments in electrochemical sensors for neurotransmitter detection have focused on enhancing their sensitivity, selectivity, and stability, as well as enabling real-time monitoring. Key innovations include the use of advanced materials, miniaturized sensor designs, and integration with digital technologies [30–33].
Nanomaterials have revolutionized electrochemical sensors by improving key parameters such as surface area, electronic conductivity, and biocompatibility, which are critical for achieving high sensitivity and selectivity. Some of the most commonly used nanomaterials in neurotransmitter sensors include:
(i) Graphene: A single layer of carbon atoms arranged in a hexagonal lattice, graphene has remarkable electrical conductivity, high surface area, and flexibility, making it an ideal material for enhancing electrochemical sensor performance. Graphene-based electrodes can improve the sensitivity and selectivity of sensors for detecting neurotransmitters like dopamine, serotonin, and glutamate [34–39]. Recently, S. K. Arumugasamy and colleagues [38] developed an electrochemical sensor utilizing graphene quantum dots integrated with multi-walled CNTs for the detection of dopamine. The sensor demonstrated high sensitivity and was evaluated using the Differential Pulse Voltammetry (DPV) technique. Measurements were conducted over a broad linear concentration range of 0.25 to 250 μM, with the sensor achieving a low LOD of 95 nM [38]. Besides this, a novel electrochemical sensor combining chitosan and graphene oxide has been developed for the selective and highly sensitive detection of serotonin. This biomimetic sensor operates based on the direct electrochemical oxidation of the target analyte, measured using DPV [40]. It exhibited a wide dynamic detection range from 5 nM to 10 μM, with a low estimated LOD of 1.6 nM. The sensor demonstrated excellent performance characteristics, including high reproducibility, long-term stability, and reusability. It also showed strong selectivity against potential interfering substances and achieved good recovery rates in real sample analyses, highlighting its potential for use in biomedical applications [41]. Farther more, an ultra-sensitive electrochemical biosensor for glutamate detection has been developed using a reduced graphene oxide/nickel foam (rGO/NiF) nanocomposite film, fabricated via an electrochemical exfoliation technique employing graphite rods recovered from waste batteries. The enzyme-based rGO/NiF sensor demonstrated a linear response within the physiological glutamate concentration range of 5–300 µM (with normal levels typically between 30–80 µM). It exhibited high sensitivity and a low detection limit (~0.1 µM), along with excellent operational stability, indicating strong potential for long-term biomedical monitoring applications [32]. The GO-based detection of neurotransmitters including GABA, dopamine, glutamate, serotonin, and acetylcholine is demonstrated in Figure 3.
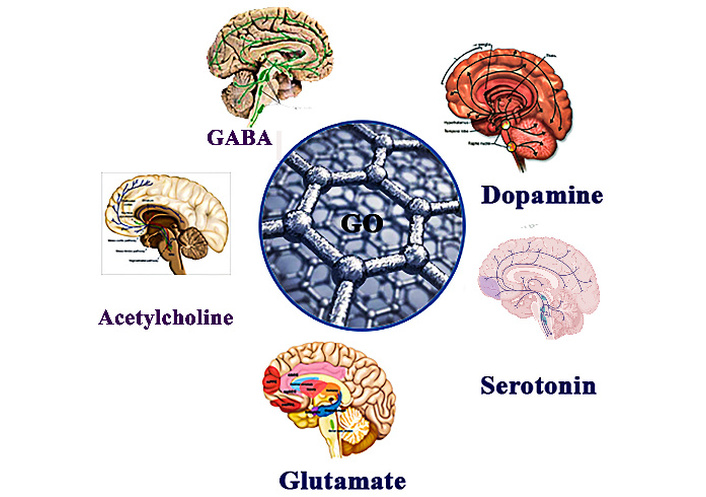
Schematic illustration of graphene oxide-based electrochemical detection of major neurotransmitters.
(ii) Carbon Nanotubes (CNTs): CNTs possess high electrical conductivity and a large electroactive surface area, which enhance electron-transfer kinetics and improve the electrocatalytic performance of electrodes for sensitive neurotransmitter detection when used as electrode modifiers K. Shekher et al. [41] developed a highly sensitive electrochemical sensor for serotonin detection, utilizing gold nanoparticles embedded within a polymer layer on CNT matrices. This sensor was specifically designed to detect serotonin in the presence of the common interferent dopamine under in-vitro conditions. Using Square Wave Voltammetry (SWV) under optimized parameters, a clear linear calibration curve was obtained between the peak current and serotonin concentration, achieving a low detection limit of 7.8 nM. The modified electrode, incorporating gold nanoparticles and poly (aminomethylthiophene) (nAu/pAMT) on functionalized CNTs, exhibited excellent selectivity for serotonin even in the presence of dopamine [42]. Figure 4 represents the various carbon-based nanomaterials which improve the electron transfer rate as well as stability during electrochemical sensing of neurotransmitters.
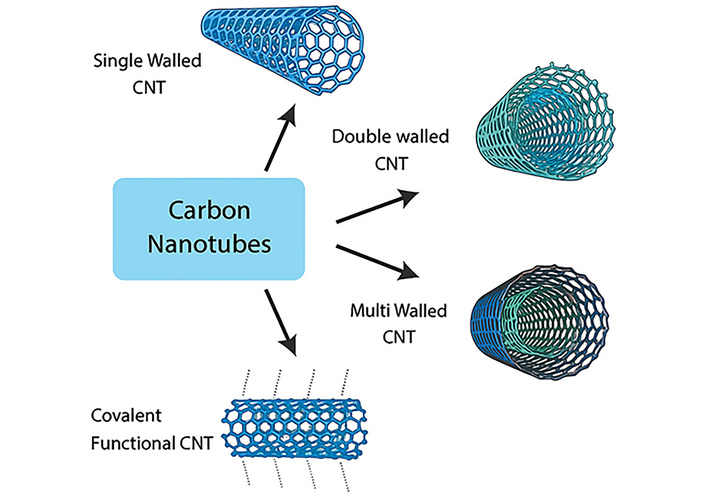
Various types of CNTs (carbon nanotubes) used to improve electron transfer rates and stability in a sensor.
To assess its practical applicability, the sensor was evaluated using human serum samples and demonstrated high accuracy in real-sample analysis [43]. In addition, a highly sensitive electrochemical sensor was developed using a composite of ferrocene-linked gold nanoparticles on multi-walled CNTs (FeC-AuNPs-MWCNT) for the detection of serotonin. The electrode was prepared by reducing gold nanoparticles onto MWCNTs, followed by covalent attachment of ferrocene. The sensor demonstrated a broad linear detection range, a low detection limit, excellent selectivity against common interferents, and good reproducibility. It was successfully applied to the detection of serotonin in urine or dopamine samples, indicating strong potential for clinical diagnostic applications [44]. Several types of CNTs used to improve electron transfer rates and stability in a sensor such as single walled CNTs, double walled CNTs, multiwall CNTs, and covalent functional CNTs.
(iii) Metal Nanoparticles: Gold, platinum, and silver nanoparticles have been widely used to enhance electro catalytic activity, leading to faster redox reactions and improved detection limits. Metal nanoparticles also help reduce sensor interference from other molecules in complex biological environments. A sensitive and versatile electrochemical sensor was developed using a carbon paste electrode modified with gold nanoparticles and Nafion for the detection of catecholamine compounds such as dopamine, epinephrine, norepinephrine, L-DOPA, and serotonin, even in the presence of common interfering substances like uric acid and ascorbic acid. The sensor’s electrochemical performance was thoroughly characterized using cyclic voltammetry, DPV, and electrochemical impedance spectroscopy (EIS) at physiological pH. Its practical utility was confirmed through successful detection of L-DOPA in human urine samples, highlighting its potential for clinical and biomedical applications [45].
Besides this, in 2017, N. Chauhan and colleagues [44] developed an electrochemical biosensor for the detection of the neurotransmitter acetylcholine, utilizing metal nanoparticles to enhance performance. The sensor exhibited a broad LDR from 4.0 nM to 800 μM, with a rapid response time of less than 4 seconds and a low detection limit of 4.0 nM (based on signal-to-noise ratio). It demonstrated excellent sensitivity, high selectivity, and long-term stability during storage. In addition to its wide detection range, the biosensor’s exceptionally low detection limit highlights its strong potential for sensitive neurochemical analysis [46]. The nanoparticles such as gold, platinum, coper and silver are used in advancing the detection of neurotransmitters are explored in Figure 5.
(iv) Emerging Nanomaterials: Beyond conventional nanomaterials, emerging hybrid systems based on metal-organic framework (MOFs) and two-dimensional MoS2 have demonstrated considerable potential for enhancing organic electrochemical transistor (OECT) performance. The integration of MOF–MoS2 hybrid nanosheets into PEDOT:PSS channels enables simultaneous enhancement of electronic and ionic transport, owing to the high charge mobility of MoS2 and the porous, high-surface area structure of MOFs. Correspondingly, synergistic effects lead to significantly enhanced transconductance, capacitance, and sensing sensitivity, offering a promising pathway toward ultra-sensitive, stable, and flexible OECT-based biosensors for non-invasive and wearable health monitoring applications. The performing OECT sensor exibits high selectivity and a wide detection range from 30 nM to 1 mM, demonstrating substantial responsiveness across physiologically relevant concentration. Such performance indicates that MOF–MoS2 integration not only enhances electronic and ionic transport but also enables ultra-sensitive detection with low limits of detection and broad linear range, emphasizing its promise for next-generation biosensor [46].
Electrochemical interference from endogenous species such as ascorbic acid and uric acid remains a crucial challenge in biological sensing. Recent strategies to address this include molecularly imprinted polymers that provide target-specific recognition, perms elective membranes as Nafion that suppress negatively charged interferents via electrostatic exclusion, and enzyme-assist layers that introduce biochemical selectivity. In addition, advanced voltammetric technique and hybrid multilayer architectures combining these approaches have proven effective for selective detection in complex biological fluids [28].
The miniaturization of electrochemical sensors has led to the development of compact, portable, and cost-effective devices that are ideal for point-of-care and at-home monitoring. Key trends in miniaturization include:
(i) Lab-on-a-chip (LOC) Platforms: LOC devices represent a powerful technological advancement by integrating microfluidic systems with electrochemical sensors on a single, compact platform. This integration enables the precise handling and manipulation of extremely small volumes of biological samples often in the microliter or nano liter range making it possible to detect multiple neurotransmitters simultaneously within a single test. The microfluidic component allows for controlled fluid flow and sample preparation, while the embedded electrochemical sensors offer high sensitivity and specificity in detecting various neurochemicals such as dopamine, serotonin, and glutamate. These platforms are designed to be lightweight and portable, which significantly enhances their utility in non-laboratory settings. Their ability to deliver real-time data means that clinicians and researchers can monitor changes in neurotransmitter levels as they happen, offering immediate insights into the patient’s neurological status. This is particularly beneficial in scenarios requiring continuous or on-site monitoring, such as during neurosurgical procedures, in emergency medical services, or for long-term outpatient care. Furthermore, the portability and user-friendly nature of LOC devices make them ideal candidates for point-of-care diagnostics. They can be deployed in remote or resource-limited environments where access to centralized laboratory infrastructure is minimal. In the context of neurological health, such devices hold promise for early detection and monitoring of conditions like Parkinson’s disease, epilepsy, and depression, ultimately supporting timely intervention and personalized treatment strategies.
Electrochemical neurotransmitter sensors are being monolithically integrated into microfluidic lab-on-chip platforms by co-fabricating nanostructured working electrodes (e.g., gold nanopillars or carbon nanofibers) directly within microchannels to enhance mass transport and sensitivity. These systems exploit laminar microfluidic flow to precisely control neurotransmitter delivery while minimizing diffusion noise, enabling sub-second temporal resolution. On-chip reference and counter electrodes, combined with antifouling surface chemistries, allow stable, multiplexed detection of dopamine and serotonin in physiologically relevant concentrations, supporting real-time neurochemical monitoring in organ-on-chip and ex vivo brain models.
The illustration of LOCs is given in Figure 6. In addition, this figure represents the clear idea of of LOCs including microfluidic devices, multianalyte detection, point of care ready, real-time data, portable device for detection sample of neurotransmitter and continuous monitoring.
(ii) Wearable and Flexible Sensors: Recent advances in flexible electronics have significantly contributed to the development of next-generation wearable electrochemical sensors, enabling continuous and non-invasive monitoring of biochemical markers, including neurotransmitters. These flexible sensors are typically fabricated on lightweight, stretchable substrates such as polymers or textiles, allowing them to conform seamlessly to the contours of the human body. This design ensures both comfort and long-term wearability, making them highly suitable for integration into everyday wearable devices like wristbands, skin patches, or smart clothing. These wearable electrochemical sensors are capable of detecting neurotransmitters such as dopamine, serotonin, and norepinephrine in various non-invasively accessible biofluids such as sweat, interstitial fluid, and saliva [16, 47]. These fluids contain trace levels of neurochemical markers that reflect central nervous system activity, enabling peripheral monitoring of brain function without the need for invasive sampling techniques like blood draws or spinal taps. Such technology holds particular promise in the management of neurological disorders, including Parkinson’s disease, where dynamic changes in dopamine levels serve as a critical biomarker for disease progression and therapeutic response. Traditional methods of monitoring these fluctuations are often episodic and require clinical visits, which can miss short-term variations and delay intervention. In contrast, wearable sensors provide real-time, continuous data that can be used to personalize treatment regimens, track symptom variability, and optimize drug delivery schedules. Moreover, the integration of these sensors with wireless communication technologies allows for remote data transmission to healthcare providers, enabling proactive disease management and telemedicine applications. As the field continues to evolve, wearable electrochemical sensors are poised to transform how neurological conditions are diagnosed, monitored, and managed bringing precision medicine closer to everyday life [48, 49].
Traditional electrochemical sensors often measure one neurotransmitter at a time, but in complex biological systems, multiple neurotransmitters can influence health outcomes. The development of multiplexed sensors allows for simultaneous detection of several neurotransmitters, providing a more complete view of brain chemistry. Multi-electrode arrays (MEAs) consist of several electrodes, each capable of detecting a different neurotransmitter. These arrays allow for the simultaneous detection of neurotransmitters such as dopamine, serotonin, and glutamate in real-time, improving the efficiency of diagnostics and monitoring in patients with neurological disorders [50, 51]. For example, MEAs can be used in studies of Parkinson’s disease, where dopamine levels fluctuate over time. In 2013, I. Suzuki and colleagues [51] developed planar CNT-based MEA chips capable of simultaneously measuring dopamine release and electrophysiological signals, including action potentials (APs) and field postsynaptic potentials (fPSPs). These chips were created by electroplating indium-tin oxide (ITO) electrodes with CNTs, significantly enhancing their electrochemical sensitivity. The CNT-MEA chips successfully detected dopamine at nanomolar concentrations and recorded synaptic release from mouse striatal slices with a high signal-to-noise ratio of 62. Additionally, APs and fPSPs were recorded from cultured hippocampal neurons with a 100-fold improvement in signal-to-noise ratio. This technology offers a robust platform for in vitro investigations of synaptic activity, drug screening, stem cell differentiation, and the study of neurological disorders [51].
In contrast, enzyme-based biosensors employing tyrosinase, choline oxidase, and monoamine oxidase provide high molecular selectivity for neurotransmitter detection through enzyme substrate specificity, enabling effective discrimination in complex biological environment. Although, their practical operation is frequently limited by enzyme denaturation, leaching, and sensitivity to pH and temperature, performing in reduced long-term stability [22]. However, nanomaterial based electrochemical sensors offer superior sensitivity, reproducibility, and functional durability due to enhanced electron transfer and structural robustness, but generally lack intrinsic selectivity among structurally similar neurotransmitters. Hybrid strategies combining enzymes with nanostructured supports have thus surfaced as a promising compromise, aiming to balance selectivity with long-term stability [38].
Recent advancements in electrochemical sensors have made it possible to monitor neurotransmitter levels continuously and non-invasively using wearable and implantable devices. These sensors provide valuable insights into disease progression and treatment efficacy, offering a more personalized approach to patient care. Implantable sensors, on the other hand, are designed for direct measurement of neurotransmitter levels within the brain or other tissues. These sensors are biocompatible, miniaturized, and capable of operating over long periods, making them suitable for long-term monitoring in clinical settings. For example, implantable sensors are being used in Parkinson’s disease to monitor dopamine levels in the brain, providing insights into disease progression and treatment effectiveness [52, 53]. A novel cortical surface implant has recently been developed to improve motor function in freely moving animal models of Parkinson’s disease. This minimally invasive device incorporates a graphene electrode array and stands out as the first fully integrated system offering biocompatibility, wearability, removability, target specificity, and wireless control.
Placed on the motor cortex, the implant delivers precise cortical stimulation to enhance therapeutic effects while reducing off-target impacts, all while continuously monitoring motor activity. In Parkinson’s disease models, stimulation through the implant successfully restored motor function and normalized brain wave patterns, accompanied by strengthened synaptic responses. These improvements were associated with the upregulation of genes involved in glutamatergic signaling, specifically metabotropic glutamate receptor 5 (mGluR5/Grm5) and dopamine D5 receptor (D5R/Drd5). This wireless neural interface presents a promising platform for real-time monitoring and targeted therapy, with potential applications in the treatment of Parkinson’s disease and other neurodegenerative diseases [54].
Although, during continuous monitoring with electrochemical neurotransmitter detectors, signal stability and quantitative reliability are significantly challenged by signal drift, baseline noise, and calibration variability. Signal drift generally arises from electrode surface fouling, biofouling, and gradual changes in electrode material properties, leading to time-dependent shifts in sensor response that are unrelated to actual neurotransmitter fluctuations [4]. Moreover, baseline noise is exacerbated by the inherently low concentration of neurotransmitters, physiological motion, and electrical interference, as together reduce the signal-to-noise rate and hinder the detection of subtle neurochemical events. These effects are farther compounded by calibration challenges, as dynamic biological surroundings alter sensor sensitivity over time while lacking stable in situ reference standards [17]. Accordingly, pre-calibrated sensor responses frequently degrade during prolonged operation, challenging frequent recalibration or advanced drift-compensation strategies. Addressing these interconnected challenges remains critical for achieving accurate, long-term electrochemical monitoring of neurotransmitters in real-world biological settings [25].
AI is increasingly being used to improve the performance of electrochemical sensors, particularly in the areas of signal processing, data analysis, and predictive analytics.
Electrochemical sensors generate large amounts of data, which can be difficult to interpret manually. AI algorithms, particularly machine learning (ML) techniques, can be applied to this data to enhance signal quality, filter out noise, and identify patterns in neurotransmitter levels. AI can also aid in distinguishing between neurotransmitter fluctuations and background signals from other biomolecules, improving sensitivity and accuracy [55]. AI algorithms can first be applied to ameliorate signal quality by filtering out noise and correcting birth drift, which is vital for directly interpreting the slight electrochemical changes associated with neurotransmitter exertion. Ways similar to adaptive filtering, denoising auto encoders, or neural networks can ameliorate the signal-to-noise rate, creating weak or lapping signals more distinguishable. Beyond noise reduction, AI excels in pattern recognition and point birth. By assaying temporal or spectral features of the detector data, ML models can identify patterns harmonious to oscillations in neurotransmitter situations. This capability allows the system to distinguish true biochemical changes from spurious signals as a result of other biomolecules, temperature shifts, and dopamine or serotonin can be separated from snooping motes, enhancing both perceptivity and particularity. Besides, AI enables predictive and quantitative analysis. When trained with known datasets, the algorithms can estimate neurotransmitter attention in real time, indeed, in complex or dynamic natural situations. This facilitates nonstop monitoring and provides dependable insight into physiological or pathological changes. By integrating these AI-grounded processing ways, electrochemical detectors become more robust, adaptive, and capable of delivering high-accuracy measures that would be problematic or insolvable to achieve with homemade analysis or traditional estimation styles alone [56].
By integrating AI-based models with real-time neurotransmitter data, healthcare providers can predict disease progression, response to therapy, and possible relapses in neurological diseases. For example, AI systems could use data from continuous monitoring of dopamine levels in Parkinson’s disease patients to predict on-off motor fluctuations, allowing for more personalized and effective treatment strategies [57, 58]. G. Costantini and colleagues [59] investigated the application of voice analysis paired with machine learning to differentiate between early-stage and advanced Parkinson’s disease patients. They compiled a bespoke dataset of high-quality vocal recordings from Italian subjects, encompassing healthy controls, early-stage Parkinson’s patients who had not begun medication, and mid-to-advanced patients undergoing L‑DOPA therapy [59]. Various ML pipelines were evaluated, employing different feature selection techniques and classification algorithms, along with a custom convolutional neural network (CNN) for deep learning. Workflow of neurotransmitter analysis using electrochemical sensing and AI integration. Neurotransmitter samples are electrochemically detected, generating high-density signals that are processed and interpreted using AI-enabled sensing platforms for advanced analysis and monitoring in Figure 7.
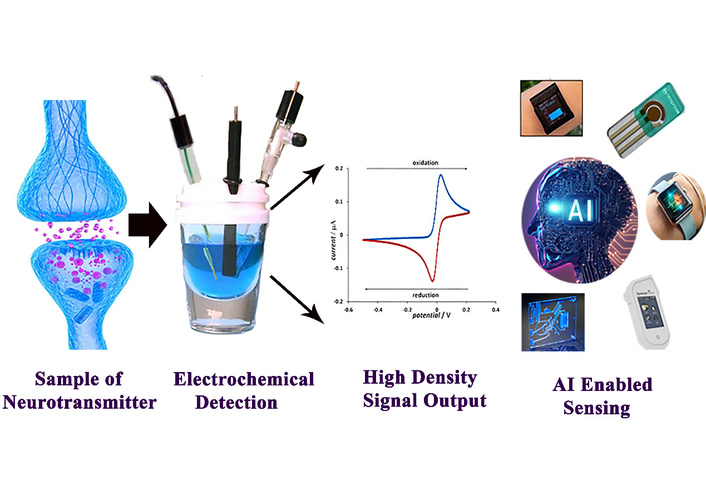
The working principle of AI integrated electrochemical sensor for the detection of neurotransmitters. Adapted from [24]. © The author(s) 2023. Licensed under a CC-BY 4.0.
The study found that both traditional ML models, such as KNN, SVM, and naïve Bayes and deep learning approaches delivered comparable performance, with KNN showing a slight edge. Among the feature selection methods, correlation-based feature selection (CFS) proved most effective. The selected vocal features served as reliable biomarkers, enabling clear differentiation between healthy individuals, early untreated Parkinson’s disease patients, and those in more advanced stages receiving treatment [59].
Electrochemical sensors for neurotransmitter detection hold significant promise for transforming clinical practice by offering real-time, precise insights into both the diagnosis and treatment of various neurological and psychiatric disorders. In Parkinson’s disease, continuous monitoring of dopamine levels can help assess disease progression and optimize dopaminergic therapy. For mood disorders such as depression and anxiety, tracking neurotransmitters like serotonin and norepinephrine can support the personalization of antidepressant treatments and improve therapeutic outcomes. In epilepsy, fluctuations in glutamate levels may serve as predictive markers for seizure activity, enabling early intervention. Similarly, in Alzheimer’s disease, monitoring imbalances in neurotransmitters such as acetylcholine could facilitate early diagnosis and more targeted therapeutic strategies. These applications highlight the potential of electrochemical sensors as powerful tools for enhancing the management of complex brain disorders.
Despite promising laboratory-scale performance, the clinical translation of electrochemical sensors remains limited by regulatory, standardization, and reproducibility barriers. Regulatory approval requires extensive evidence of long-term safety, stability, biocompatibility, and reliability under clinically applicable conditions, which is frequently lacking in proof-of-concept studies. In addition, the absence of standardized protocols for calibration, performance evaluation, and data reporting hampers cross-study comparison and regulatory assessment. Reproducibility is further challenged by batch-to-batch variability in nanomaterials and reliance on custom fabrication materials, complicating scalable manufacturing under Good Manufacturing Practice conditions. Inclusively, these factors decelerate the transition of electrochemical sensors from experimental platforms to clinically deployable technologies.
While electrochemical sensors for neurotransmitter detection have made significant strides, several challenges remain that must be addressed to further enhance their performance and clinical applicability. These challenges primarily concern selectivity, interference from other biomolecules, and the long-term stability of the sensors. Additionally, while these technologies hold great promise, their full potential in neurological diagnostics and personalized medicine will require overcoming a number of technical and practical obstacles.
One of the main challenges in neurotransmitter detection is ensuring selectivity for the target neurotransmitter. Neurotransmitters like dopamine, serotonin, and glutamate are chemically similar, and they often coexist in the same biological environments, such as the brain or blood. This makes it difficult to detect individual neurotransmitters without cross-reactivity or interference from other molecules. To overcome this, researchers have turned to selective recognition elements, such as antibodies, enzymes, or molecular imprinted polymers (MIPs), which can be used to functionalize electrodes. However, achieving high selectivity is still a significant hurdle, particularly in complex biological samples, where other biomolecules can interfere with the electrochemical signal [60, 61]. Moreover, electrode surface modification using nanomaterials like graphene, CNTs, or metal nanoparticles has shown promise in enhancing selectivity, but challenges remain in designing universal sensors that can accurately distinguish between neurotransmitters at low concentrations in the presence of interfering substances. Ensuring that these sensors can operate in real-world biological conditions, such as the brain’s complex chemical environment, is a critical ongoing challenge.
Despite significant progress in nanomaterial-based electrodes, selective discrimination of dopamine from structurally similar catecholamines such as norepinephrine and epinephrine remains challenging due to their closely overlapping redox potentials. Recent studies demonstrate that selectivity is increasingly achieved through recognition-driven and system-level strategies rather than material modification alone. For example, molecularly imprinted polymer-based sensors have shown preferential dopamine binding over norepinephrine and epinephrine [10]. Enzyme-assisted platforms employing tyrosine have further enhanced biochemical specificity. In addition, DPV combined with chemometric analysis has enabled reliable peak deconvolution of dopamine/norepinephrine mixtures [17, 20].
The human body contains a wide array of molecules, such as proteins, lipids, and other metabolites, that may interfere with the electrochemical detection of neurotransmitters. For example, ascorbic acid (vitamin C), uric acid, and other common biomolecules can produce similar electrochemical signals, leading to false positives or reduced sensor accuracy. This is particularly problematic when trying to measure neurotransmitter levels in complex samples like blood, saliva, or interstitial fluid, where these interfering molecules are often present at high concentrations. To mitigate this, researchers have developed more sophisticated selective sensing strategies, such as DPV, cyclic voltammetry, and amperometry [62–64], which can help distinguish neurotransmitter signals from background noise. Additionally, the incorporation of enzyme-based biosensors and MIPs that specifically target neurotransmitter molecules has been explored to reduce interference. Nonetheless, achieving high selectivity in the presence of potential interferences remains one of the major challenges in this field.
Another key challenge is ensuring the long-term stability of electrochemical sensors. For implantable sensors or wearable devices that continuously monitor neurotransmitter levels, long-term performance is crucial. Many electrochemical sensors, especially those that use organic or enzyme-based materials, suffer from sensor degradation over time due to factors like biofouling, material wear, and electrode surface contamination. Biofouling refers to the accumulation of proteins and other biological materials on the sensor surface, which can interfere with the electrochemical reactions and cause sensor drift or loss of signal. This is particularly problematic for implantable sensors placed in the body, where the biological environment can lead to rapid degradation of the sensor’s materials. To address this, researchers are exploring the use of biocompatible materials, protective coatings, and self-cleaning surfaces to extend the lifetime of these sensors [65, 66]. Furthermore, the stability of nanomaterials used in sensor electrodes can be affected by factors such as electrode material degradation and aggregation of nanoparticles. Developing more robust and durable materials for the electrodes, particularly those that are exposed to harsh biological environments, is crucial for long-term clinical use. The function of neurotransmitters in human is explored in Table 2.
The function of neurotransmitters in humans.
| Neurotransmitter | Main functions | References |
|---|---|---|
| Acetylcholine | Muscle contraction, memory, learning, attention | [67] |
| Dopamine | Reward, motivation, mood regulation, motor control | [68] |
| Serotonin | Mood, sleep, appetite, digestion, memory | [69] |
| Norepinephrine | Arousal, alertness, stress response, blood pressure regulation | [70] |
| Epinephrine | Fight or flight response, heart rate, energy, alertness | [71] |
| GABA | Inhibitory effect, reduces anxiety, relaxation, muscle tone | [72] |
| Glutamate | Excitatory effect, learning, memory, synaptic plasticity | [73] |
| Endorphins | Pain relief, pleasure, euphoria, stress reduction | [4] |
| Anandamide | Mood, memory, pain regulation, endocannabinoid system | [74] |
Table 3 summarizes the typical long-term operational stability and reproducibility of commonly employed electrode modification strategies, including enzyme-based, nanomaterial-based, polymer-modified, and hybrid systems. The comparison highlights general performance trends, dominant degradation mechanisms, and representative studies rather than direct numerical equivalence, as stability testing conditions vary significantly across studies. This qualitative assessment provides insight into trade-offs between selectivity, durability, and reproducibility in the design of electrochemical sensors for long-term and implantable applications.
Comparative long-term stability and reproducibility of different electrode modification strategies used in electrochemical neurotransmitter sensing.
| Electrode modification strategy | Typical operational stability | Reproducibility | Major degradation mechanism | References |
|---|---|---|---|---|
| Enzyme-based electrodes | Low–Moderate | Moderate | Enzyme denaturation, leaching, pH/temperature sensitivity | [22] |
| Carbon nanomaterial-based | High | High | Surface fouling, oxidation | [25] |
| Metal nanoparticle-based | Moderate | Moderate | Aggregation, surface oxidation | [26] |
| Polymer-modified electrodes | High | High | Mechanical degradation, swelling | [27] |
| Hybrid systems | Moderate–High | High | Enzyme instability mitigated by support matrix | [28] |
Beyond sensing performance, the practical deployment of wearable electrochemical sensors critically depends on wireless data transmission, miniaturized electronics, and sustainable power sources. In this context, self-powered and energy-harvesting strategies have emerged as key enablers of next-generation wearable systems [75]. Triboelectric nano-generators (TENGs) and piezoelectric nano-generators (PENGs) are particularly attractive, as they convert biomechanical energy from human motion, skin deformation, or physiological activities into electrical output. Recent studies have demonstrated flexible TENG or PENG powered electrochemical sensors capable of driving signal conditioning circuits and wireless transmission modules, enabling real-time monitoring without external batteries [76]. Moreover, thermoelectric generators (TEGs) offer a complementary approach by harvesting body heat through the temperature gradient between skin and ambient environments, providing continuous and passive energy generation. Although TEGs typically exhibit lower power density, advances in low-power electronics and efficient power management have made them viable for intermittent sensing and wireless data transfer. Collectively, the integration of mechanical and thermal energy harvesters with ultra-low-power electronics and wireless communication technologies such as Bluetooth Low Energy or NFC is paving the way toward fully autonomous, compact, and user-friendly wearable sensing platforms [77].
Despite the current challenges, the future of electrochemical sensors for neurotransmitter detection is highly promising, particularly with the integration of new technologies that can overcome these limitations. The key areas for future research and development include:
Future electrochemical sensors will likely see the continued development of novel nanomaterials and composite materials that provide enhanced selectivity, sensitivity, and stability. Materials such as graphene oxide, carbon nanofibers, quantum dots, and bio-inspired materials are being explored for their potential to improve sensor performance. By combining these advanced materials with bio recognition elements (e.g., enzymes, antibodies), researchers can create more specific sensors with reduced interference from other biomolecules. In addition, smart coatings that reduce biofouling and improve sensor longevity are also a promising avenue for ensuring long-term sensor stability. The development of self-healing materials that can recover from damage or degradation will further extend the lifetime of both implantable and wearable sensors.
As wearable and implantable electrochemical sensors continue to evolve, one of the next big steps is their integration with personalized healthcare systems. Non-invasive wearable devices capable of continuously monitoring neurotransmitter levels (e.g., in sweat or saliva) hold great potential for use in chronic disease management, particularly in conditions like Parkinson’s disease or epilepsy. Incorporating AI and ML into these systems will enhance their ability to predict disease states or therapy responses based on real-time neurotransmitter data. For example, AI-powered systems can identify patterns in neurotransmitter fluctuations, enabling early detection of seizures, disease progression, and response to medication. This would lead to more personalized and adaptive treatment strategies tailored to the individual’s unique neurochemical profile.
Although wearable and implantable neurotransmitter sensors have shown excellent performance in laboratory and preclinical studies, their clinical translation is expected to occur in a phased manner over the next 5 to 10 years. Short-term wearable systems for sweat and saliva analysis are likely to reach clinical use earlier, whereas implantable brain or subcutaneous sensors will require longer confirmation due to safety and regulatory conditions. Key challenges include long-term biocompatibility, biofouling, mechanical stability in dynamic tissues, and reliable calibration in vivo. In addition, regulatory approval by agencies such as the FDA or EMA will require extensive testing of toxicity, sterility, electrical safety, and long-term drift. Addressing these issues through advanced encapsulation, biocompatible nanomaterials, and standardized validation protocols will be essential for translating these technologies from laboratory prototypes to clinical neurodiagnostic tools.
Furthermore, wearable and implantable electrochemical sensors have shown considerable promise; long-term biocompatibility remains a critical challenge for clinical translation. Recent studies increasingly address immune response and tissue damage through the use of soft, mechanically compliant materials such as conducting polymers and hydrogel-based electrodes, which better match the modulus of biological tissues and reduce chronic inflammation. Anti-fouling surface coatings and drug-eluting interfaces have further been employed to mitigate protein adsorption and foreign body responses. Importantly, incorporate in vivo implantation studies and histological analyses, reflecting growing attention to ethical considerations, long-term safety, and regulatory requirements.
The development of compact, cost-effective electrochemical sensors for point-of-care diagnostics could revolutionize the way neurological and psychiatric conditions are diagnosed and monitored. Such systems could be deployed in clinics, at home, or in remote settings, providing real-time feedback to patients and healthcare providers. By enabling non-invasive, real-time monitoring, these sensors could replace traditional diagnostic methods, such as blood sampling, and allow for more frequent and less intrusive testing. For example, in the case of epilepsy, real-time monitoring of glutamate levels in interstitial fluid could enable predictive seizure detection, reduce the risk of seizures, and improve quality of life. Similarly, dopamine monitoring in Parkinson’s disease patients could help manage motor fluctuations and adjust medications on the fly.
As the demand for more comprehensive monitoring increases, the development of multiplexed electrochemical sensors capable of detecting multiple neurotransmitters in a single test will be crucial. These sensors could offer valuable insights into neurochemical imbalances, providing a more holistic view of a patient’s neurological health. Multiplexed sensors could help clinicians assess the interplay between neurotransmitters like dopamine, serotonin, and glutamate, which is critical in understanding complex conditions like Parkinson’s disease, depression, and schizophrenia. By landing these dynamics contemporaneously, multiplexed detectors offer a holistic view of brain chemistry, enabling clinicians to better estimate neurochemical relations and tailor treatments to individual cases. Beyond diagnostics, this technology holds promise for individualized drug, nonstop monitoring, and guiding medicine development, offering a clearer understanding of how curatives impact multiple neurotransmitter pathways. As exploration progresses, multiplexed discovery could transfigure how we approach brain health and psychiatric care.
Electrochemical sensors for neurotransmitter detection represent a transformative technology in the realm of neurological diagnostics and personalized medicine. Over recent years, significant advancements have been made in sensor materials, miniaturization, and integration with wearable and implantable systems, enhancing their sensitivity, selectivity, and potential for real-time monitoring. The integration of nanomaterials, AI-powered data analysis, and multiplexed sensing capabilities has significantly broadened the scope of these devices, enabling more comprehensive, non-invasive, and continuous monitoring of neurotransmitter levels. However, challenges remain, particularly in achieving high selectivity in complex biological environments, minimizing interference from other biomolecules, and ensuring the long-term stability of sensors, especially in implantable and wearable formats. Addressing these challenges will require continued innovation in biocompatible materials, smart coatings, and advanced signal processing techniques. Looking ahead, the potential impact of electrochemical neurotransmitter sensors is profound. They could revolutionize personalized treatment plans, enabling real-time feedback on disease progression and therapy responses. This would lead to more precise, adaptive, and individualized care, particularly for patients with neurological and psychiatric disorders such as Parkinson’s disease, epilepsy, depression, and schizophrenia. With ongoing research and development, electrochemical sensors are poised to become an integral tool in both clinical diagnostics and home-based healthcare, offering new possibilities for proactive and personalized management of brain health. Electrochemical dopamine sensors enable precision medicine by supporting real-time, patient-specific monitoring in Parkinson’s disease. Continuous neurochemical feedback could facilitate closed-loop drug dosing or adaptive neuromodulation, minimize motor fluctuations and side effects, while allow individualized therapeutic windows and dynamic treatment adjustment as disease progresses.
AI: artificial intelligence
APs: action potentials
CNT: carbon nanotubes
DPV: Differential Pulse Voltammetry
fPSPs: field postsynaptic potentials
FSCV: fast-scan cyclic voltammetry
LDR: linear detection range
LOC: lab-on-a-chip
LOD: limit of detection
MEAs: multi-electrode arrays
MIPs: molecular imprinted polymers
ML: machine learning
MOFs: metal-organic framework
OECT: organic electrochemical transistor
PENGs: piezoelectric nano-generators
rGO/NiF: reduced graphene oxide/nickel foam
TEGs: thermoelectric generators
TENGs: triboelectric nano-generators
MA: Methodology, Validation, Investigation, Resources, Writing—original draft. SA: Software, Validation, Formal analysis. MRU: Conceptualization, Validation, Data curation, Visualization. MMA: Conceptualization, Writing—review & editing, Supervision, Project administration. All authors have read and agreed to the published version of the manuscript.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2808
Download: 56
Times Cited: 0
