Affiliation:
1Biomedical Technology Wing, Sree Chitra Tirunal Institute for Medical Science and Technology, Thiruvananthapuram 695012, Kerala, India
Affiliation:
1Biomedical Technology Wing, Sree Chitra Tirunal Institute for Medical Science and Technology, Thiruvananthapuram 695012, Kerala, India
Affiliation:
1Biomedical Technology Wing, Sree Chitra Tirunal Institute for Medical Science and Technology, Thiruvananthapuram 695012, Kerala, India
Email: naresh.kasoju@sctimst.ac.in
ORCID: https://orcid.org/0000-0002-8700-0729
Explor BioMat-X. 2026;3:101364 DOI: https://doi.org/10.37349/ebmx.2026.101364
Received: December 16, 2025 Accepted: April 27, 2026 Published: April 29, 2026
Academic Editor: Diego Mantovani, Laval University, Canada
Aim: Cell sheet technology is a transformative approach in epithelial tissue engineering, offering scaffold-free constructs that preserve cell-cell and cell-matrix interactions, enabling better integration with host tissues. However, safe and efficient transfer of these fragile sheets remains a critical challenge, limiting their broader clinical adoption. This study aims to develop a facile method for the retrieval and transfer of epithelial cell sheets cultivated over thermo-responsive polymer surfaces (TRPS) using sacrificial films.
Methods: Three epithelial cell lines, HCE-S (cornea), HaCaT (skin), and A549 (lung), were cultured on poly(N-isopropylacrylamide-co-glycidyl methacrylate) (P(NIPA-GMA)) coated TRPS and conventional tissue culture surfaces. Upon reaching confluence, the dishes were incubated below the lower critical solution temperature to induce phase transition in TRPS. Subsequently, sacrificial films made of polyethylene oxide, gelatin and their blend were used to lift and transfer the cell sheets to new culture dishes containing a minimal amount of culture medium. Additional medium was then added to dissolve the film, allowing the cell sheet to settle gently onto the dish surface.
Results: In all three epithelial cell types, a continuous, confluent cell sheet was visible on the TRPS prior to transfer. Subsequent to temperature lowering and sacrificial film assisted transfer, the master TRPS dish exhibited a distinct void corresponding to the sheet removal, confirming successful detachment. The transferred sheets reattached successfully and maintained over a one-week observation period.
Conclusions: The sacrificial film-based transfer method provided a gentle, efficient and scalable alternative for handling cell sheets from TRPS. This approach enhances the translational potential of cell sheet engineering and supports its integration into clinical workflows for epithelial tissue regeneration.
Epithelial tissue engineering represents a crucial avenue in regenerative medicine, addressing the significant need for repairing or replacing damaged epithelial layers in various organs. Epithelial tissues are vital for maintaining homeostasis, serving as protective barriers and performing specialized functions, including absorption in the gastrointestinal tract, filtration in the kidneys, secretion in glands, and sensory perception in the cornea and skin [1]. However, epithelial injuries or diseases often lead to severe functional impairments and aesthetic concerns. The self-renewing capacity of epithelial tissues is often insufficient to repair large-scale damage, particularly in cases of chronic wounds, burns or corneal disorders [2, 3]. Conventional treatments, such as autologous or allogeneic grafting, face limitations like donor tissue scarcity, immune rejection and suboptimal integration. To overcome these challenges, epithelial tissue engineering focuses on developing bioengineered constructs that mimic native tissue characteristics, offering advanced solutions for functional and cosmetic restoration. The field has grown with innovations in biomaterials, cell culture techniques and scaffold designs, providing a foundation for more reliable and effective clinical therapies to come [4].
Cell sheet technology has emerged as a groundbreaking approach in epithelial tissue engineering, providing a scaffold-free method to fabricate tissue constructs with intact cellular and extracellular matrix (ECM) components. This technique involves cultivating cells to form cohesive monolayers that can be detached as intact sheets for transplantation [5–7]. Unlike scaffold-based methods, cell sheet technology preserves critical intercellular junctions and ECM proteins, which are essential for tissue integrity and functionality. These preserved structures facilitate better integration of the transplanted sheets with host tissues, promoting faster healing and improved outcomes [8]. The technology has shown remarkable potential in applications such as corneal reconstruction, esophageal repair, periodontal regeneration, and skin wound healing [9]. Moreover, it minimizes complications such as immune responses and inflammation that are often associated with scaffold degradation. Despite its promise, cell sheet technology faces challenges in scaling up for broader clinical applications, highlighting the need for innovation in the fabrication and transfer processes [10].
A major challenge in cell sheet technology lies in the transfer of delicate engineered cell sheets from culture surfaces to the intended target site. To this end, a few techniques have been developed to facilitate the transfer of intact cell sheets after detachment from thermo-responsive or other specialized substrates [11, 12]. Conventional methods employ membrane-assisted transfer, in which cell sheets are lifted using poly(vinylidene difluoride) or nitrocellulose membranes before being reattached to target surfaces [13]. Other strategies utilize plungers that allow gentle release of the cell sheet onto a new substrate [14]. Mechanical and pipette-based approaches have also been reported [15]. Although these approaches have advanced cell sheet manipulation, they present notable limitations [16]. Manual lifting often causes mechanical damage or folding, compromising sheet viability and function. Likewise, specialized transfer tools can be cumbersome and expensive, restricting their routine clinical use. These challenges highlight the need for alternative methods that simplify transfer while preserving the structural and functional integrity of the cell sheet [17, 18].
In this study, we propose the use of a sacrificial film as a novel method to assist in the transfer of in vitro engineered cell sheets (Figure 1). A sacrificial film, designed to dissolve in a quick manner, can serve as a transient substrate during the transfer of cell sheets. Once the cell sheet is transferred, the sacrificial film gets spontaneously dissolved, facilitating seamless detachment without compromising the integrity of the cell sheet [19]. We hypothesize that this method will simplify the transfer process, reduce mechanical damage, and preserve cell-cell and cell-ECM interactions, thereby enhancing the overall efficacy of cell sheet technology in epithelial tissue engineering. By addressing critical challenges in cell sheet transfer, this approach has the potential to expand the clinical applicability of cell sheet technology and improve patient outcomes in regenerative therapies.
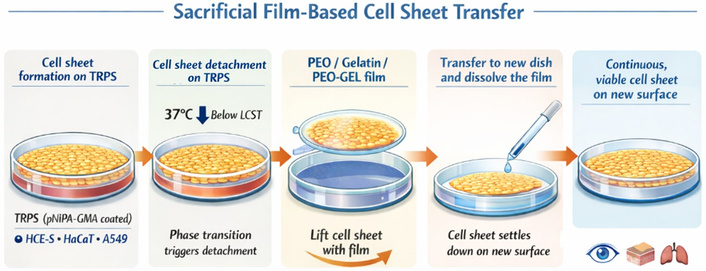
Schematic of sacrificial film-based transfer of intact epithelial cell sheets from TRPS surfaces via temperature-induced detachment, film-assisted lifting, and reattachment on a new substrate following film dissolution.
The chemicals polyethylene oxide (PEO, with an average molecular weight (Mv) of 900,000, cat. no. 189456-250G), gelatin (GEL, from porcine skin, Type A, gel strength 300, cat. no. G2500-500G), trypsin-ethylenediaminetetraacetic acid (EDTA) (0.25% w/v), and penicillin-streptomycin (100×) were procured from Sigma-Aldrich, Bangalore, India. Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12), fetal bovine serum (FBS), and Alamar Blue reagent were obtained from Thermo Fisher Scientific, Bangalore, India. Phosphate-buffered saline (PBS) and other cell culture reagents, unless otherwise specified, were purchased from HiMedia, Bangalore, India. Cell lines used in the study included human corneal epithelial cells–spontaneously immortalized (HCE-S), procured from Applied Biological Materials Inc., Canada (cat. T0737, lot no. C22C24UA), and adenocarcinomic human alveolar basal epithelial cells (A549) and human epidermal keratinocyte line (HaCaT), obtained from National Center for Cell Science (NCCS), Pune—a national repository in India. According to the supplier’s certification, cell lines were authenticated and tested for mycoplasma contamination prior to delivery. No further testing was performed in-house.
The sacrificial films were prepared using solutions of PEO, GEL, and a combination of PEO and GEL. A 5% (w/v) solution of PEO and a 2.5% (w/v) solution of GEL were prepared separately by dissolving the respective polymers in deionized water under constant stirring at room temperature until a homogeneous solution was obtained. Additionally, a blend solution of PEO and GEL was prepared by mixing equal volumes of the two solutions (1:1 v/v). For each type of film, 2.5 mL of the prepared solution was dispensed into clean 35 mm Petri dishes. The dishes were then placed in a mini-oven preheated to 60°C and maintained overnight to facilitate complete drying of the solution and ensure uniform film formation. Once the drying process was complete, the resulting films were carefully detached from the Petri dish surfaces using sterile forceps. The detachment process was performed gently to avoid any damage to the films. All procedures, including solution preparation, pouring, drying and film handling, were conducted under aseptic conditions to maintain sterility. The resultant films were stored under cool and dry conditions until further use.
The sacrificial nature of the films was assessed by measuring their dissolution rate under simulated physiological conditions. Colored films were prepared using food dye to enhance visibility and were cut into square pieces (1 cm × 1 cm dimensions). Each film was placed in a pre-weighed 35 mm Petri dish, and the weight of both the film and the empty dish was recorded prior to the experiment. To simulate physiological conditions, 2 mL of PBS was added to each dish, and the samples were incubated at 37°C. At predetermined time intervals (0, 1, 3, 6, 12, and 24 hours), the PBS solution was removed; care was taken to prevent the accidental loss of film fragments while pipetting. Following this, the Petri dishes containing any remaining portions of the film were placed in a hot air oven set at 60°C and dried until all residual moisture had evaporated. Once dried, the Petri dishes were re-weighed, and the weight of the remaining film was calculated by subtracting the weight of the empty Petri dish from the recorded weight. The percentage of weight loss was calculated as the ratio of the residual weight to the initial weight of the film at each time point.
Three epithelial cell lines, via HCE-S, HaCaT and A549, representing corneal, skin and lung epithelium, respectively, were cultured in DMEM/F12 medium supplemented with serum, bicarbonate and antibiotics and maintained at 37°C in a CO2 incubator (95% air, 5% CO2, and 90% relative humidity). Routine culturing was performed in standard tissue culture-treated polystyrene (TCPS) flasks and sub-confluent cultures were disaggregated using 0.25% trypsin-EDTA at 37°C for 2–3 minutes, and cells were counted using a hemocytometer before each experiment.
The copolymer P(NIPA-GMA) (poly(N-isopropylacrylamide-co-glycidyl methacrylate)), previously developed by our team [20], was used to prepare thermo-responsive polymer surfaces (TRPS). A 4% (w/v) stock solution of P(NIPA-GMA) was prepared in isopropanol. From this, 0.1 mL of the stock solution was diluted with 0.9 mL of isopropanol to obtain a 0.4% (w/v) working solution. Standard 35-mm culture plates were manually coated with the working solution using a gentle swirling motion and dried in a hot air oven at 60°C. The dried plates were rinsed with ice-cold water to remove any excess polymer and redried at 60°C. The plates were subsequently sterilized via ethylene oxide and stored in a cool and dry environment until use.
The three cell lines were seeded at a density of 50,000 cells per dish onto 35-mm plates coated with the TRPS as described earlier. The seeded cells were maintained in DMEM/F12 medium supplemented with serum and antibiotics and incubated at 37°C in a CO2 incubator until confluence was achieved. For comparison, cells were also cultured on standard TCPS. The growth of HCE-S, HaCaT and A549 cells on TRPS and TCPS was monitored daily using an inverted phase-contrast microscope. Besides, the Alamar Blue assay was employed to quantitatively assess cell proliferation over TRPS and TCPS over a period of 7 days.
P(NIPA-GMA), a thermo-responsive smart polymer, facilitates cell sheet retrieval through its phase transition from hydrophobic to hydrophilic at temperatures below its lower critical solution temperature [21]. HCE-S, HaCaT and A549 cells were cultured on TRPS, and upon reaching confluence, the culture plates were transferred to a refrigerator for 10 minutes. This temperature change induced the detachment of the monolayer cell sheets, which were observed and documented using an inverted phase-contrast microscope. To ensure comparability, cells cultured on TCPS substrates underwent similar treatment, and the observations were recorded.
To facilitate the transfer of detached cell sheets, a sacrificial film approach was employed. After incubating the TRPS plates in refrigerator for 10 minutes to induce the hydrophilic phase transition, the culture medium was removed. A sterile sacrificial film was gently placed over the detached cell sheet, allowing the hydrated, slightly sticky sheet to adhere to the film. The attachment process was completed by lightly pressing the film to ensure uniform contact, as required. The film carrying the cell sheet was lifted with sterile forceps and transferred onto a fresh TCPS dish containing a minimal volume of culture medium. Additional medium was then added to ensure complete dissolution of the sacrificial film, allowing the cell sheet to settle gently onto the dish surface. The dish was incubated at 37°C in a CO2 incubator to enable reattachment of the cell sheet to the new substrate. This method was successfully applied to HCE-S, HaCaT and A549 cell sheets, with observations made to evaluate its efficacy against conventional TCPS systems.
The impact of different transfer methods on cell viability and maintenance was studied using cell sheets prepared on both TRPS and TCPS substrates in 24-well plates. Detached cell sheets were transferred to new 24-well plates using sacrificial film, scraping, or trypsinization methods. Following transfer, the plates were incubated for seven days at 37°C in a CO2 incubator to allow reattachment and maintenance. Cell viability and metabolic activity were assessed using the Alamar Blue assay.
Statistical differences were analyzed by t-test and ANOVA as necessary, and differences were considered significant at p-value < 0.05 and denoted using a * mark. Qualitative images were representative of the respective group.
To support the transfer of in vitro engineered cell sheets, sacrificial films composed of PEO, GEL and a PEO/GEL blend were prepared and evaluated for their suitability as transient, water-soluble substrates. These films were prepared by solvent casting, wherein homogeneous polymer solutions were dispensed into 35 mm Petri dishes and dried at 60°C to yield uniform thin films, as shown in Figure 2a. The drying protocol ensured consistent film morphology and mechanical integrity, allowing for ease of handling during subsequent cell sheet transfer procedures. To validate the sacrificial functionality of the films, their dissolution profiles were examined under simulated physiological conditions. Film samples (1 cm square pieces) were incubated in PBS at 37°C, and weight loss over time was used as an indicator of dissolution. As shown in Figure 2b, the films demonstrated rapid dissolution, showing significant weight loss within the first 3 hours, indicating a fast-degrading profile. Such a rapid dissolution of films makes them ideal candidates for cell sheet transfer applications, where quick substrate removal is critical to maintaining cell sheet integrity and minimizing handling time.
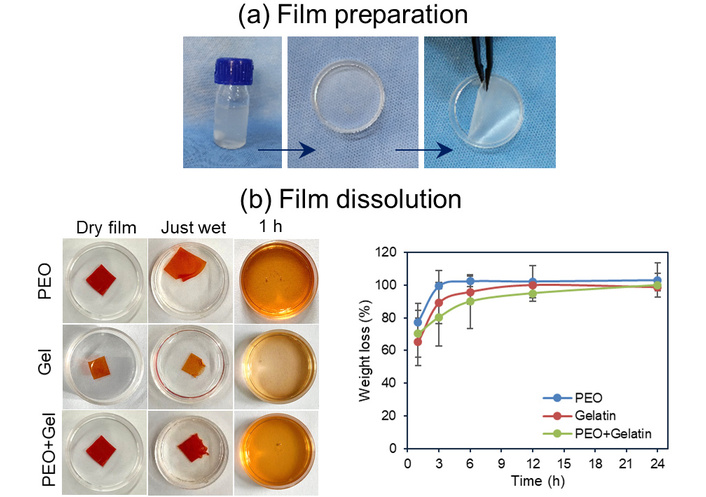
Preparation of films and confirmation of their sacrificial nature. (a) Representative images showing preparation of polymer films by solvent casting approach, and (b) data showing rapid dissolution of films including photographic images and weight loss% as a function of time.
To fabricate transferable epithelial cell sheets, three representative epithelial cell lines, HCE-S (corneal epithelium), HaCaT (skin epithelium) and A549 (lung epithelium), were cultured on TRPS and conventional TCPS as control. Cell proliferation and monolayer formation were monitored over a seven-day period using inverted phase-contrast microscopy. Figure 3a shows representative images of HCE-S cells that demonstrated good adhesion and progressive proliferation on TRPS. The morphological progression observed on TRPS closely mirrored that on standard TCPS controls, supporting the biofunctionality of the TRPS for epithelial cultures. Quantitative assessment of proliferation was performed using the Alamar Blue assay at days 1, 3, 5, and 7 (Figure 3b, representing that of HCE-S cells). The absorbance values showed a time-dependent rise, correlating well with the visual evidence of monolayer formation. The data validate the TRPS ability to support robust epithelial growth and confluence necessary for cell sheet engineering. Collectively, these results affirm that TRPS are conducive for generating intact epithelial monolayers. The preservation of morphology, along with sustained proliferation and viability, establishes TRPS as a suitable platform for the fabrication of detachable cell sheets.
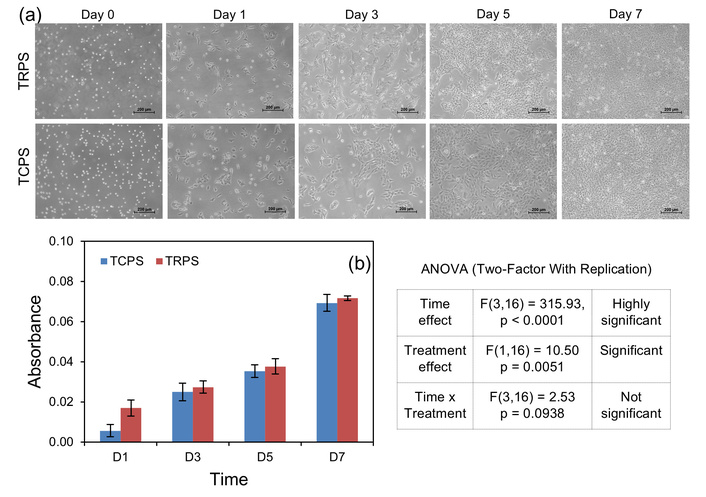
Preparation of cell sheets on thermoresponsive surfaces. (a) Phase contrast microscope images of HCE-S cells (scale bar: 200 µm) and (b) Alamar Blue assay data indicating proliferation of HCE-S cells on TRPS over a period of 7 days. A two-way ANOVA showed a significant effect of time (F(3,16) = 315.93, p < 0.0001), indicating increasing cell viability over time. Treatment also had a significant effect (F(1,16) = 10.50, p = 0.0051), with TRPS slightly higher than TCPS. However, time vs treatment interaction was not significant (F(3,16) = 2.53, p = 0.0938), indicating similar growth trends in both groups.
The retrieval and transfer of intact epithelial cell sheets were successfully achieved using a combination of TRPS and sacrificial film-assisted methodology. The underlying mechanism leveraged the temperature-sensitive phase transition behavior of the P(NIPA-GMA) copolymer. At physiological temperature (37°C), the TRPS remains hydrophobic and promotes cell adhesion and proliferation. Upon incubation in refrigerator for 10 minutes, the TRPS undergoes a hydrophobic-to-hydrophilic transition, resulting in spontaneous detachment of the confluent cell monolayer without the use of enzymatic agents. Note that incubation in the refrigerator ensured a gradual reduction in temperature, thereby avoiding sudden thermal shock that could affect junctional structures. To enable transfer, a sterile sacrificial film was gently placed on the detached cell sheet. Due to the moist and tacky surface of the cell sheet at low temperature, it readily adhered to the film. The film carrying the cell sheet was then lifted from the TRPS master dish and placed onto a fresh TCPS dish (Figure 4a). To visually confirm successful transfer, brilliant blue dye was used to stain the cell sheets prior to detachment and transfer (Figure 4b). The stained cell sheet clearly adhered to the sacrificial film, leaving behind a distinct, unstained area in the master dish, corresponding to the region where the sheet was lifted (note that the sacrificial film used for transfer was intentionally smaller than the 35-mm culture dish to allow clear visualization of the transferred and non-contacted regions). In the recipient dish, the presence of the blue-stained cell sheet confirmed successful transfer. In contrast, control experiments using TCPS showed no transfer: The stained monolayer remained fully attached to the master dish, and the new dish appeared devoid of cells or staining (Figure 4c). This clearly highlighted the critical role of the TRPS in enabling gentle, enzyme-free detachment.
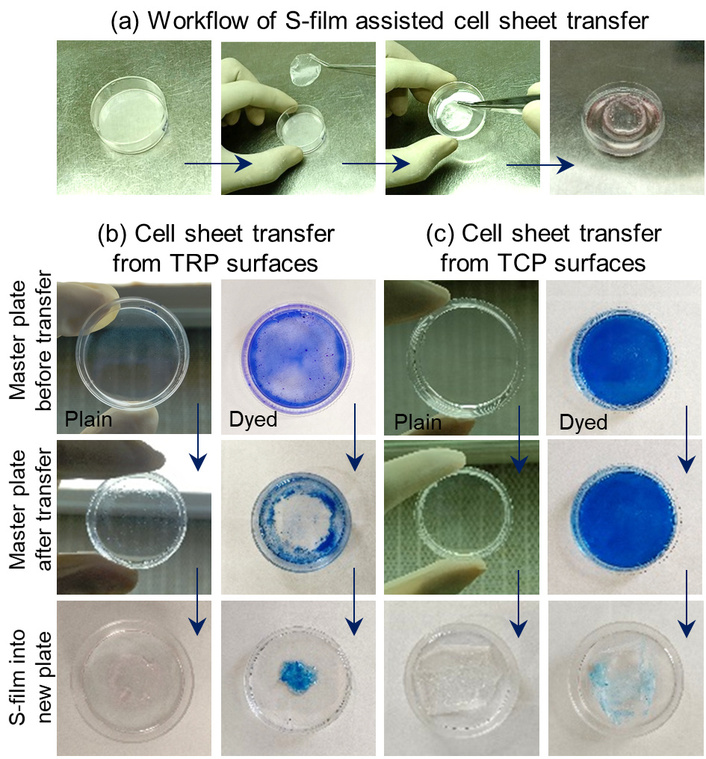
Representative images of sacrificial film (S-film) assisted cell sheet transfer. (a) Schematic of the overall workflow, (b) cell sheet transfer from TRPS, and (c) cell sheet transfer from TCPS.
Further validation was provided through inverted phase-contrast microscopy of HCE-S, HaCaT, and A549 cells before and after transfer (Figure 5a). In all three epithelial cell types, a continuous, confluent cell sheet was visible on the TRPS prior to transfer. Post-transfer, the master TRPS dish exhibited a clear void at the site of sheet removal, while the new TCPS dish displayed a successfully reattached monolayer. In contrast, cells cultured on control TCPS dishes showed no detachment, as expected. Both pre- and post-transfer images confirmed that transferred cell sheets remained viable and preserved overall morphology, indicating that the sacrificial film provided an effective and reproducible mechanism for sheet handling and transfer.
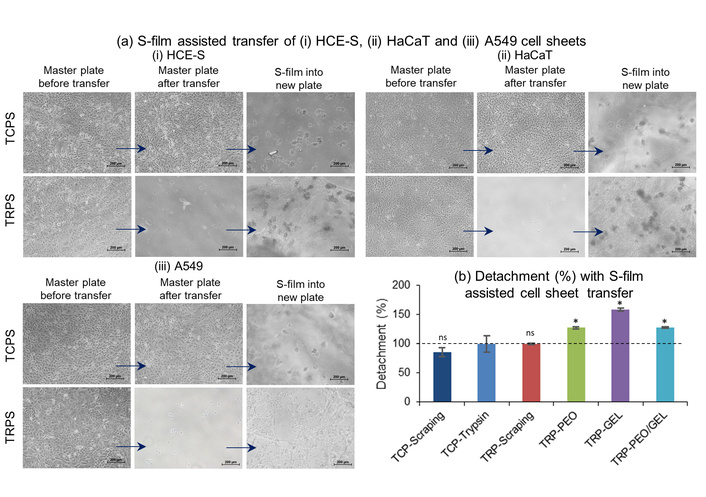
Assessment of cell sheet detachment using sacrificial film (S-film) assisted transfer approach. (a) Inverted phase contrast microscope images indicating successful transfer in TRPS in contrast to TCPS, of (i) HCE-S, (ii) HaCaT and (iii) A549 cells (scale bar: 200 µm), and (b) Alamar Blue assay derived detachment% indicating enhanced detachment of cell sheets using sacrificial films as compared to conventional methods via scraping and trypsinisation (representative of HCE-S cells). *: Denotes statistically significant differences between control TCP-trypsin and the rest of the others; ns: non-significant.
To further quantify the efficiency of cell sheet retrieval across different methods, detachment percentages were assessed using the Alamar Blue assay (Figure 5b). This assay measured the metabolic activity of cells remaining in the master plate (post-transfer), thereby indirectly indicating the extent of detachment. Cell detachment achieved via trypsinization on a TCPS substrate was used as the reference standard (100%). Manual scraping in TCPS yielded ~86% detachment efficiency, slightly lower—probably due to stronger attachment of the cell monolayer over the TCPS surface. Manual scraping on TRPS yielded complete detachment efficiency (~100%), likely due to the thermoresponsive effect of TRPS, which aided cell sheet detachment more effectively than TCPS. Among the sacrificial film-assisted methods, all three film types, PEO, PEO/GEL blend and GEL, showed higher detachment percentages compared to trypsinization. The PEO and PEO/GEL blend films both resulted in ~128% detachment efficiency, while the GEL film achieved the highest efficiency (~159%). These results suggest that sacrificial films not only enable gentle, non-enzymatic transfer but also outperform conventional methods in terms of completeness of cell sheet retrieval.
To assess the recovery and viability of the transferred cell sheets, inverted phase-contrast microscopic imaging was performed at regular intervals (days 1, 3, 5, and 7) following transfer to fresh culture dishes (Figure 6, shows representative images of HCE-S cells). This evaluation was conducted across all experimental groups, including those transferred via enzymatic (trypsin), mechanical (scraping) and sacrificial film-assisted methods, using both TCPS and TRPS as master plates. In control groups, where cell sheets were detached using trypsin or scraped off from either TCPS or TRPS, transferred cells adhered well to the new substrate and were well-maintained over the 7-day culture period. As expected, sacrificial film-assisted transfer from TCPS master plates did not result in successful cell transfer, thus, no cellular adherence or growth was observed in the new dish. In contrast, when sacrificial films were applied to TRPS cultured cell sheets, successful transfer was clearly evident. The cells were observed to reattach in the new dish within 24 h post-transfer and were well-maintained through days 3, 5, and 7. The ability of cells to reattach and maintain effectively following sacrificial film-assisted transfer suggests that this method preserves essential biological interactions such as cell-cell and cell-matrix adhesion, critical for post-transfer functionality.
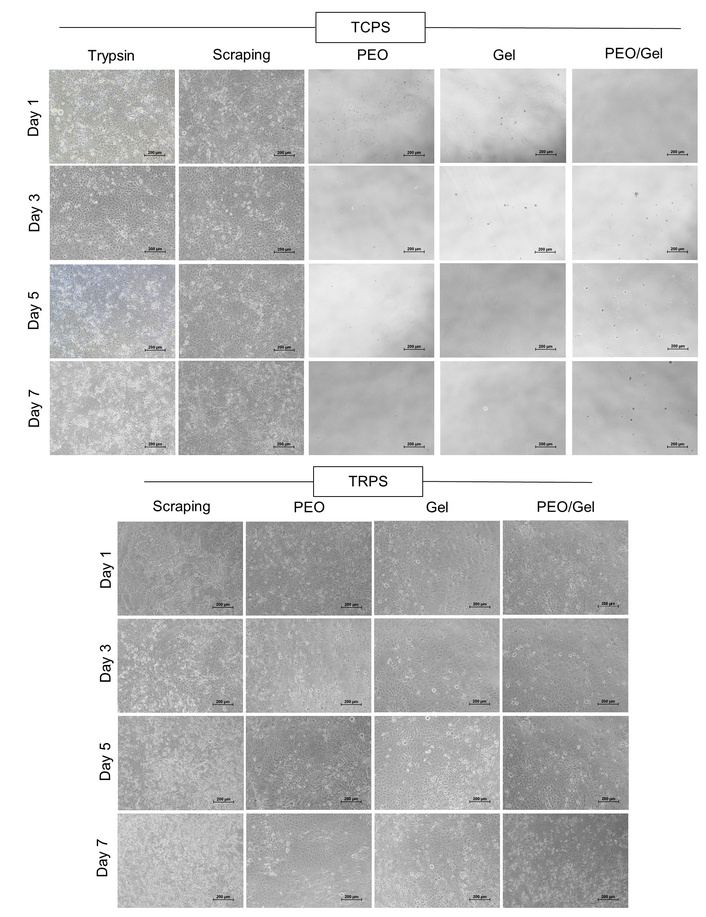
Inverted phase contrast microscope images showing reattachment and subsequent maintenance of HCE-S cells or cell sheets that were transferred from TRPS and TCPS dishes to the new dishes (scale bar: 200 µm).
The present study introduces a sacrificial film-assisted strategy to facilitate the retrieval and transfer of epithelial cell sheets cultured on TRPS. This dual system leverages the temperature-sensitive behavior of P(NIPA-GMA) for gentle detachment, combined with rapidly dissolvable sacrificial films for transfer [21]. The resulting method demonstrated high detachment efficiency while preserving cell viability and overall morphology. These findings mark a significant advance over conventional approaches such as enzymatic detachment or manual scraping, which are often associated with partial cell loss, ECM disruption or altered cell phenotype. The work aligns with earlier studies employing smart polymer coatings for cell sheet detachment [17, 22], yet enhances the transfer step, a known bottleneck in clinical translation, through a simple, low-cost and scalable sacrificial layer approach [23].
Importantly, the sacrificial films used in this study via PEO, GEL and their blends demonstrated rapid dissolution under physiological conditions, confirming their transient nature and suitability for biomedical handling. Among them, GEL-based films showed the highest detachment efficiency, potentially due to their greater hydrophilicity and better surface adherence to moist cell sheets. This observation is consistent with reports emphasizing GEL’s favorable surface energy and protein-interactive properties for biomedical applications [19, 24]. Notably, the films supported the seamless reattachment of cell sheets on fresh substrates, with no evidence of sheet fragmentation. This is critical in epithelial regeneration, where junctional continuity underpins barrier function and paracrine signalling [25]. Our results were comparable to previous studies, where thrombin-functionalized porous polycaprolactone mats were developed as cyto-compatible carriers enabling efficient transfer of cell sheets via clot-mediated adhesion [26]. Overall, sacrificial film-assisted transfer may offer a viable route for improving clinical workflow, particularly for tissues such as corneal epithelium, skin, airway lining and beyond.
In comparison to state-of-the-art transfer techniques, such as electro-responsive carriers [27], hydrogel plungers [28] and mechanical stamp-based systems [29], the proposed method offers notable advantages in simplicity, cost-efficiency and compatibility with existing cell culture infrastructure. Additionally, unlike hydrogel or tool-based systems [30], which may require post-transfer removal or may interfere with cell sheet integration, the sacrificial films used here fully dissolve, eliminating the need for secondary intervention. This could be particularly advantageous for automation in cell sheet biomanufacturing, where handling delicacy, scalability and minimal user intervention are critical. Moreover, the applicability of this method across three epithelial cell types representing distinct anatomical tissues highlights its versatility and potential generalizability to other monolayer-forming cell systems.
Looking ahead, further refinement of film composition could optimize parameters such as adhesion strength, dissolution kinetics and mechanical stability during transfer. Future work should also focus on functional assays post-transfer, such as transepithelial resistance, tight junction integrity or in vivo engraftment, in order to validate the physiological performance of the transferred sheets. The incorporation of bio-markers into the sacrificial layer could also allow for real-time monitoring of sheet orientation or quality control during transfer. Additionally, integration of this technique into closed-loop or robotic culture systems could expedite its transition to clinical-grade manufacturing. Overall, the sacrificial film-assisted method developed in this study represents a promising step toward standardizing and scaling cell sheet engineering, thereby bridging laboratory success and real-world clinical application.
This study presents a novel sacrificial film-assisted transfer method for retrieving and transferring engineered epithelial cell sheets from TRPS. Three epithelial cell monolayers (HCE-S, HaCaT and A549) were successfully cultivated on TRPS, and three types of sacrificial films (PEO, GEL and PEO/GEL blend) were prepared and tested for their detachment efficiency. The films enabled efficient, non-invasive transfer of cell sheets, outperforming traditional methods. Post-transfer, cells reattached and maintained successfully on new substrates. This approach addresses critical challenges in cell sheet engineering, enhancing its clinical applicability for regenerative therapies and automated biomanufacturing.
A549: adenocarcinomic human alveolar basal epithelial cells
DMEM/F12: Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12
ECM: extracellular matrix
EDTA: trypsin-ethylenediaminetetraacetic acid
GEL: gelatin
HaCaT: human epidermal keratinocyte line
HCE-S: human corneal epithelial cells–spontaneously immortalized
P(NIPA-GMA): poly(N-isopropylacrylamide-co-glycidyl methacrylate)
PBS: phosphate-buffered saline
PEO: polyethylene oxide
TCPS: tissue culture-treated polystyrene surfaces
TRPS: thermo-responsive polymer surfaces
The authors thank SCTIMST Thiruvananthapuram and Department of Science and Technology (Ministry of Science and Technology, Govt. of India) for the financial support.
AI disclosure statement: During the preparation of this manuscript, author(s) used Grammarly and ChatGPT for grammar editing, overall language improvement and preparation of Figure 1. After using the tool/service, author(s) thoroughly reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
SVS: Formal analysis, Investigation, Methodology, Writing—original draft. AKPR and CR: Formal analysis, Methodology, Writing—review & editing. NK: Conceptualization, Formal analysis, Funding acquisition, Methodology, Project administration, Supervision, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interests.
Not applicable.
Not applicable.
Not applicable.
The datasets supporting the findings of this study are available from the corresponding author upon reasonable request.
The study was funded by the Department of Science and Technology, Ministry of Science and Technology, Govt. of India, through TRC scheme at SCTIMST Thiruvananthapuram (TRC-P8223). The funder(s) had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 440
Download: 19
Times Cited: 0
