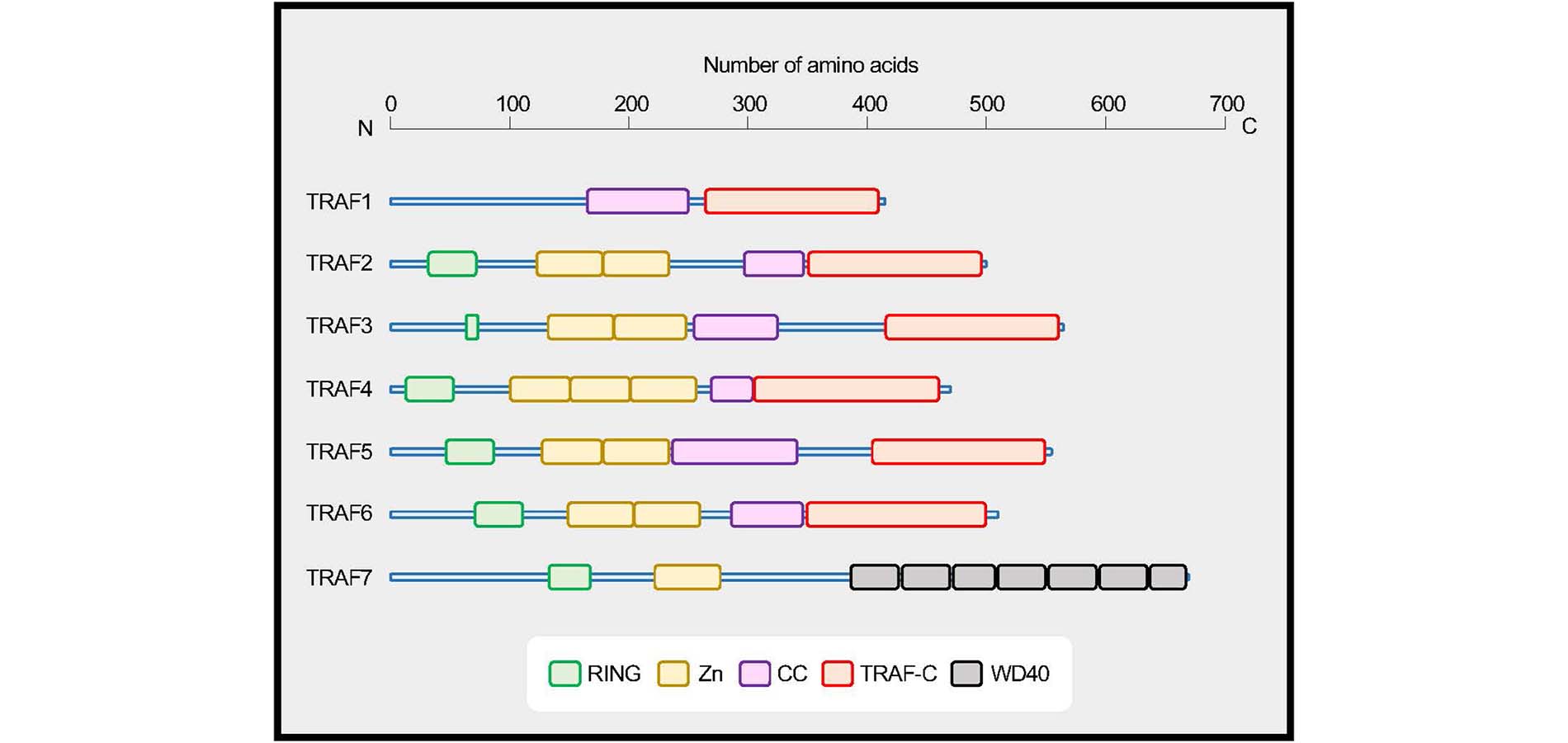
TRAF family proteins. TRAF1 to TRAF6 share a conserved coiled-coil (CC)/TRAF-C domain, and TRAF7 has a series of 40 amino acid peptide repeats terminating in tryptophan (W) and aspartic acid (D) residues (WD40) domains. TRAFs, with the exception of TRAF1, have an N-terminal really interesting new gene (RING) domain and zinc finger motifs (Zn). Data are based on UniProt (https://www.uniprot.org). TRAF1, Q13077; TRAF2, Q12933; TRAF3, Q13114; TRAF4, Q9BUZ4; TRAF5, O00463; TRAF6, Q9Y4K3; TRAF7, Q6Q0C0