Affiliation:
1Department of Biomedicine, Neurosciences and Advanced Diagnostics, University of Palermo, 90127 Palermo, Italy
ORCID: https://orcid.org/0009-0008-5011-0335
Affiliation:
2Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Riad El-Solh/Beirut 1107 2020, Lebanon
ORCID: https://orcid.org/0009-0003-6039-3408
Affiliation:
2Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Riad El-Solh/Beirut 1107 2020, Lebanon
Affiliation:
2Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Riad El-Solh/Beirut 1107 2020, Lebanon
ORCID: https://orcid.org/0000-0002-9570-307X
Affiliation:
2Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Riad El-Solh/Beirut 1107 2020, Lebanon
Affiliation:
1Department of Biomedicine, Neurosciences and Advanced Diagnostics, University of Palermo, 90127 Palermo, Italy
Affiliation:
2Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Riad El-Solh/Beirut 1107 2020, Lebanon
ORCID: https://orcid.org/0000-0002-9568-8092
Affiliation:
3Department of Chemistry and Biochemistry, Faculty of Arts and Sciences, Holy Spirit University of Kaslik, Jounieh 446, Lebanon
ORCID: https://orcid.org/0000-0003-3033-5330
Affiliation:
1Department of Biomedicine, Neurosciences and Advanced Diagnostics, University of Palermo, 90127 Palermo, Italy
ORCID: https://orcid.org/0000-0001-9288-1148
Affiliation:
1Department of Biomedicine, Neurosciences and Advanced Diagnostics, University of Palermo, 90127 Palermo, Italy
ORCID: https://orcid.org/0000-0001-6904-5141
Affiliation:
2Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Riad El-Solh/Beirut 1107 2020, Lebanon
Email: aj00@aub.edu.lb
ORCID: https://orcid.org/0000-0002-5955-656X
Explor Dig Dis. 2026;5:1005117 DOI: https://doi.org/10.37349/edd.2026.1005117
Received: January 07, 2026 Accepted: March 04, 2026 Published: April 02, 2026
Academic Editor: Jose C. Fernandez-Checa, Institute of Biomedical Research of Barcelona (IIBB), CSIC, Spain
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic immune-mediated condition typically requiring invasive endoscopy for monitoring. The bidirectional oral-gut axis suggests that saliva may serve as a non-invasive diagnostic fluid for studying gut inflammations. Studies of salivary biomarkers have shown varying results. Inflammatory biomarkers such as IL-6 are the most robust salivary biomarker, consistently correlating with endoscopic activity. In contrast, salivary calprotectin lacks the reliability of its fecal counterpart, showing diagnostic value primarily in pediatric cases with oral manifestations. Microbial analysis indicates reduced salivary diversity, specifically an enrichment of Prevotella and Veillonella alongside a depletion of core commensals like Streptococcus. While oxidative stress markers such as advanced oxidation protein products (AOPPs) can distinguish disease severity, they lack long-term prognostic utility. Conversely, recent shifts toward exosome-based transcriptomic analysis have improved the stability of salivary microRNAs, offering high precision in differentiating IBD phenotypes. Despite these advancements, clinical integration is currently hindered by small cohort sizes, the confounding effects of local oral health, and a lack of standardized collection protocols. To establish saliva as a reliable tool in the IBD clinical toolkit, future research must prioritize multi-panel biomarker approaches and longitudinal studies to validate diagnostic accuracy across diverse patient populations.
Inflammatory bowel disease (IBD) consists of a chronic inflammation of the gastrointestinal tract that comprises two subtypes: Crohn’s disease (CD) and ulcerative colitis (UC) [1, 2]. Although they have some clinical similarities, their tissue involvement is distinct. CD can affect any region of the gut and is characterized by a non-contiguous, transmural inflammation that can result in complications such as fibrotic strictures, fistulas, and abscesses [1, 3]. In contrast, UC is limited to the colon and the rectum, characterized by superficial mucosal inflammation that extends proximally in a contiguous manner, and can lead to ulcerations and severe bleeding [1, 3].
While the global burden of IBD was once focused only in North America and Europe, the incidence rate is rising rapidly across Asia and South America, in newly industrialized countries, due to environmental shifts like urbanization, change in diets, and social stress [1, 2, 4, 5]. This rising incidence rate has led to a significant rise in pediatric-onset IBD, a form often characterized by repetitive extensive inflammation and a change of clinical course to a more aggressive course that demands appropriate diagnostic strategies [4].
It is well documented that the pathophysiology of IBD is complex and immune-mediated [1]. Current evidence points to a combination of genetic susceptibility, environmental triggers, alterations in gut microbiome, epithelial barrier dysfunction, and dysregulated innate and adaptive immune responses [5]. This multifactorial aspect underlies the relapsing-remitting course requiring continual therapeutic adjustments to achieve and maintain remission [1, 6].
Despite its gastrointestinal origin, the inflammatory pathways of IBD extend beyond the intestinal mucosa, manifesting extraintestinal manifestations, making it a multisystem disease [7, 8]. The oral cavity serves as a vital diagnostic extension of IBD due to the bidirectional oral-gut axis [9, 10], where oral lesions often precede gastrointestinal symptoms and reflect intestinal pathology [11, 12]. This relationship positions saliva as a high-potential, non-invasive alternative to invasive standards like endoscopy and biopsy or costly imaging such as Magnetic Resonance Enterography [13, 14]. While clinicians currently rely on serum C-reactive protein and fecal calprotectin to monitor activity, these markers are frequently limited by poor specificity and can be influenced by unrelated inflammation [13, 14]. Consequently, the high frequency of late diagnosis and subsequent surgical intervention underscores the necessity for more reliable, accessible biomarkers in saliva to advance clinical monitoring [13, 14].
Among these extraintestinal sites affected, the oral cavity has gained increased attention due to its diagnostic relevance. Emerging research shows that the oral and gut microbiomes form an interconnected system, where dysbiosis in one site can influence inflammatory manifestations in the other, reinforcing the concept of bidirectional oral-gut axis [9, 10]. This axis suggests that oral changes may mirror or amplify intestinal disease activity. In fact, IBD patients exhibit poorer oral health, and in some cases, multiple oral lesions develop before gastrointestinal symptoms [11, 12]. These findings, supported by emerging models of the oral-gut axis, position the mouth as a clinically informative extension of intestinal pathology and highlight its potential, in particular saliva, as a non-invasive diagnostic tool.
In the context of IBD, frequent late diagnoses lead to adverse outcomes and increased surgical rates [13], primarily because current practice relies on invasive endoscopy with biopsy or cross-sectional imaging like Magnetic Resonance Enterography [14]. While serum C-reactive protein and fecal calprotectin are commonly used as accessible monitoring tools, their lack of specificity for IBD can lead to potential misdiagnosis or confusion with unrelated inflammatory processes [13, 14].
In response to these diagnostic challenges, contemporary medicine is witnessing a paradigm shift toward non-invasive, patient-centred tools. Thus, an optimal biomarker must be non-invasive while also offering high diagnostic precision and feasibility for repeated monitoring [13]. This framework has positioned saliva as a promising alternative, given its ability to capture immunological, microbial, and molecular signatures that mirror systemic and intestinal inflammation [10, 15, 16].
Saliva is a complex biological fluid containing a rich matrix of proteins, enzymes, and immunoglobulins that make it uniquely informative for diagnostic purposes [17, 18]. Its diagnostic potential for oral and systemic diseases is further enhanced by components from gingival crevicular fluid, which enrich the fluid with immune mediators that reflect both local and broader inflammatory states [18–21]. A schematic representation of the importance of saliva as a diagnostic biological fluid is presented in Figure 1.
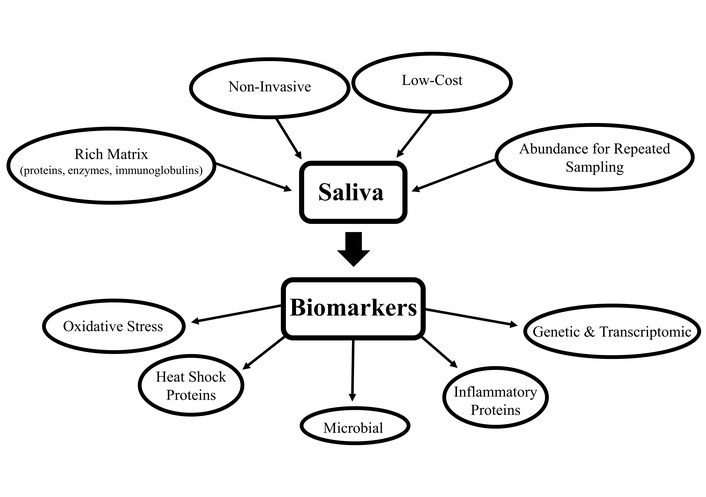
Schematic representation of the importance of saliva as a diagnostic biological fluid.
In this perspective, proteomic markers such as inflammatory cytokines have been studied in different conditions ranging from periodontal inflammation to oncologic diseases [21]. Genetic and transcriptomic biomarkers such as DNA variants, mRNA, and microRNAs provide additional insights about systemic conditions and have shown clinical relevance among hereditary diseases and in oncology [21]. In addition, metabolic biomarkers such as glucose, neurotransmitter metabolites, and oxidative stress molecules have also demonstrated utility in monitoring endocrine, neuropsychiatric, and inflammatory disorders [20, 21].
Together, these biomarkers highlight saliva’s capacity to reflect systemic athophysiology, a potential further strengthened by its non-invasive collection, low cost, and suitability for the repeated sampling required for early detection [20, 21]. Within this emerging paradigm, researchers are extensively exploring salivary diagnostics for IBD as a direct response to the limitations and patient burden associated with current invasive tools [20, 22].
Inflammatory proteins constitute a particularly informative category of salivary biomarkers due to their ability to mirror immune activity at both local and systemic levels [20]. Their presence in saliva arises through multiple physiological pathways.
Circulating cytokines and chemokines can reach saliva through passive diffusion or active transport across the highly vascularized salivary glands, and in some cases, their salivary levels correspond to serum concentrations [23]. During active IBD, systemic inflammatory signaling increases epithelial and vascular permeability, thereby facilitating the transfer of these immune mediators into saliva [20, 23, 24].
In parallel, immune cells within the oral mucosa, particularly neutrophils, release inflammatory proteins through exocytosis as part of the innate immune activation [20, 23, 24].
Moreover, a major source of these mediators also derives from the gingival crevicular fluid, which carries cytokines, antimicrobial peptides, and neutrophil granule components that reflect both periodontal host responses and broader systemic inflammation [25].
While research on salivary biomarkers has extended over the past few years, a 2020 systematic review emphasized that these inflammatory proteins remained among the least studied categories, with only a small number of heterogeneous investigations available at that time [26]. Among the inflammatory markers examined so far, IL-6 continues to be the most extensively studied salivary cytokine, with multiple investigations converging on its relevance in IBD [23, 26, 27]. In stimulated saliva, IL-6 was found to be higher in patients with IBD compared to controls, and showed a strong correlation with endoscopic activity [27]. This finding aligns with a broader review that describes IL-6 as the most stable salivary cytokine associated with systemic inflammation in IBD, making it the most robust inflammatory protein currently identified [23, 27]. This consistency among studies reflects the fact that IL-6 is released early during gut inflammation and sustains a broader systemic signal, which increases the possibility of its detection in saliva compared with cytokines that have shorter or more localized activity [27–29].
From a broader perspective, IL-6 was found elevated again alongside several Th17-derived cytokines, although none of the Th17 markers retained significance after adjusting for confounders such as oral inflammation and demographic factors [30].
Beyond IL-6, the salivary profile of other cytokines remains inconsistent and is frequently shaped by oral rather than intestinal inflammations [23]. This pattern was reflected in comparison to the healthy population. Clinical findings indicate that salivary TNF-α increases in UC and CD primarily when periodontal inflammation is present, that IL-10 is reduced in CD, IL-1β is higher in UC than in CD, and that IL-17A shows no group-wise differences, underscoring the strong influence of oral disease on salivary cytokines [31]. Proteomic analyses further reinforce this pattern, identifying IL-6 alongside MMP-10 as potential salivary biomarkers rising alongside serum levels, suggesting a clearer link between systemic inflammation and their appearance in stimulated saliva [27, 31].
Beyond cytokine alterations, neutrophil-derived proteins have also been explored as salivary biomarkers of intestinal inflammation. Among these, calprotectin has received the most attention due to its established role in fecal diagnosis [32]. In fact, calprotectin, predominantly released from activated neutrophils, is widely utilized as an adjunctive specific biomarker and demonstrates accuracy in distinguishing IBD from functional disorders as well as predicting relapse [32].
However, in saliva, calprotectin does not uphold its diagnostic properties observed in stool. While early reviews had identified it as a potentially promising salivary biomarker [26], recent studies demonstrate that it lacks diagnostic value in IBD. In 2022, a study by Bos et al. [33] showed that no differences were observed in comparison with healthy controls, nor were any associations detected with fecal calprotectin or disease activity [33]. Furthermore, while a 2025 investigation showed a slight elevation in CD, this increase was not statistically significant and did not correlate with the Crohn’s Disease Activity Index [34]. Furthermore, a larger study including 100 patients with IBD (60 with CD and 40 with UC) demonstrated no differences in salivary calprotectin concentrations between IBD patients and healthy controls, and no variation between active and inactive disease states [24].
However, a more nuanced pattern emerges in a 2025 pilot study in early-onset IBD, where salivary calprotectin was significantly elevated only in children with oral manifestations and showed a positive correlation with fecal calprotectin and relapse risk within this subgroup, while remaining inconsistent in patients without oral involvement [35]. These findings suggest that salivary calprotectin may hold prognostic value in pediatric cases characterized by associated oral disease, despite its overall lack of reliability in broader IBD populations. Saliva shows an abundance of inflammatory protein biomarkers, with varying specificity to IBD (Table 1), with only IL-6 having a high specificity to IBD, necessitating the identification of novel biomarkers in saliva specific to IBD, such as heat shock proteins (HSPs).
Salivary biomarkers in IBD.
| Salivary biomarker | Findings | Specificity | References |
|---|---|---|---|
| IL-6 | Consistently elevated, correlates with endoscopic activity | High specificity, least affected by oral inflammation among cytokines | [13, 23, 26–29] |
| MMP-10 | Identified as elevated alongside IL-6 in proteomic analysis | Potential systemic specificity | [31] |
| Th17-derived cytokines | Elevated alongside IL-16 | Low specificity, influenced by oral inflammation and demographic factors | [30] |
| TNF-α | Elevated mainly in the presence of periodontal inflammation | Low specificity largely reflects oral inflammatory status | [23, 31] |
| IL-10 | Reduced levels of CD compared with controls | [23, 31] | |
| IL-1β | More elevated in UC than in CD | [23, 31] | |
| IL-17A | No differences were observed in comparison with healthy controls | - | [31] |
| Calprotectin | No differences between IBD patients and controls | Limited specificity in saliva, except in pediatric IBD with oral manifestations | [26, 32–35] |
IBD: inflammatory bowel disease; CD: Crohn’s disease; UC: ulcerative colitis.
Beyond classical inflammatory mediators, proteins involved in cellular stress responses associated with immune activation have also been investigated, notably HSPs.
HSPs constitute a family of molecules that are expressed in all cells [36]. They primarily act as molecular chaperones to support protein folding and stability, and they preserve cellular homeostasis under physiological conditions and pathological stress [36]. When cellular homeostasis is disrupted by inflammation or oxidative stress, HSP expression is upregulated as part of an adaptive response to limit damage, but in chronic states, this develops into sustained alterations in HSP regulation rather than a transient reaction. In inflammatory and autoimmune diseases, induced families such as HSP60, HSP70, and HSP90 modulate cell cycle regulation, apoptosis, and immune responses, acting either as cytoprotective molecules or as drivers of persistent inflammation [36, 37].
In IBD, altered expression of several HSP families, including HSP70 and HSP90, has been reported primarily in intestinal tissue and in the systemic circulation of patients with CD and UC [37]. These alterations have been linked to intestinal epithelial stress responses, immune activation, and the maintenance of the inflammatory state [37]. Although HSPs are involved in IBD at tissue and systemic levels, their salivary presence remains largely unexplored, as existing evidence is restricted to local conditions like chronic periodontitis [38]. Furthermore, while anti-HSP70 autoantibodies have been detected in the saliva of healthy individuals [39], these observations lack a systemic disease context, providing a clear rationale for investigating salivary HSPs as potential non-invasive biomarkers in IBD.
With the emergence of studies based on the oral-gut axis, salivary microbial biomarkers have been extensively studied in the last few years [26]. Dysbiosis in the oral cavity and gastrointestinal tract is increasingly viewed as interconnected, with microbial shifts in one site influencing mucosal immunity in the other [10]. This framework supports the study of salivary microbial communities alongside inflammatory proteins, since fluctuations in bacterial profiles may reflect ecological instability that coincides with, or precedes, intestinal inflammation.
In the last five years, a reduction in salivary microbial diversity has been repeatedly investigated in the literature. A large prospective cohort reported a significant reduction in richness in both UC and CD compared with healthy controls [40]. Whereas in a systematic review and meta-analysis including 976 IBD patients, the decrease in Shannon diversity of microbial community was confined particularly to patients with high disease activity, while those with low activity showed diversity levels comparable to healthy controls [41].
In contrast, Park et al. [42] in 2022 reported contradictory findings. Using whole saliva sequencing and extracellular vesicle analysis, which captures only vesicle-associated microbial material, the study did not identify any reduction in salivary microbial diversity [42]. These contradictory findings can be explained by the difference in sample size and the use of broader community-level 16S rRNA sequencing in the larger studies [41], which may have been more sensitive to detecting diversity shifts.
Beyond overall diversity change, recent evidence increasingly converges on a pattern of salivary dysbiosis with Prevotella and Veillonella consistently emerging as enriched genera, while core oral commensals such as Streptococcus, Neisseria, and Haemophilus appear depleted [41, 43, 44]. This emerging evidence contrasts with findings illustrated in a 2020 systematic review, which had suggested a possible increase in Streptococcus [26], likely reflecting the smaller cohorts and methodological limitations of earlier studies.
Advances in analytic methodologies have further broadened the dysbiotic profile, with more sensitive detection of additional oral species reported alongside elevated local inflammatory cytokines [13], thereby reinforcing their potential relevance as biomarkers in IBD.
Furthermore, comparisons between disease phenotypes highlight how these microbial alterations manifest within clinical subgroups. UC has been described as showing relatively higher Streptococcus and Enterobacteria, whereas CD presents more pronounced enrichment in Veillonella [43]. Similarly, a more recent large-scale salivary profiling study demonstrated that the microbial signatures of CD and UC are sufficiently distinct for machine learning models to differentiate between them [45]. These phenotype-dependent variations further support the emerging value of salivary microbial patterns as potentially disease-specific diagnostic markers in IBD.
In contrast, when examining broader taxonomic groups, elevations in Bacteroidetes and Firmicutes have been documented in salivary profiles of patients with IBD, yet no distinctions have been reported at the level of clinical subgroups [46]. This seems to reflect a general state of dysbiosis rather than phenotype-specific microbial signatures.
Disruptions in the gut microbiota have been shown to stimulate reactive oxygen species (ROS) production, thus leading to oxidative injury to the mucosal barrier, increases in permeability, reinforces dysbiosis, and sustains inflammation in CD [47]. This bidirectional relationship underscores the need to examine oxidative stress alongside microbial alterations in the context of salivary biomarkers in IBD. In fact, oxidative stress has emerged as a central pathway in IBD pathogenesis, arising when ROS accumulate beyond the capacity of antioxidant defences, leading to structural and functional cellular damage and further sustaining the cycle of chronic inflammation [47]. Substantial clinical and experimental evidence demonstrates that IBD involves prominent oxidative modifications to colonic lipids, proteins, and DNA, signifying a chronic imbalance in redox homeostasis [48]. Within this framework, several oxidative stress biomarkers—including malondialdehyde, 8-hydroxy-2’-deoxyguanosine, and albumin-free thiols—are consistently elevated in blood and stool, showing a strong correlation with disease severity that supports their use for diagnosis and monitoring [49].
Salivary oxidative-stress markers have been investigated since the early 2000s, with studies assessing nitric oxide, total antioxidant status, albumin, and uric acid in IBD [50]. These early findings suggested that redox alterations could be detected in saliva, yet most results were considered preliminary and required further validation [50, 51]. Amid the earlier heterogeneous investigations of salivary oxidative imbalance, Janšáková et al. (2021) [52] reported higher salivary advanced oxidation protein products (AOPPs) and advanced glycated end-products (AGEs) together with reduced antioxidant capacity in CD, demonstrating that oxidative protein modification can be captured in oral fluid [52]. Consistent patterns were also reported in active disease state in CD, where disturbances in lipid peroxidation and antioxidant status were found in both saliva and serum, further reinforcing the potential of oral fluid in mirroring systemic redox alterations [49].
Although redox disruption appears consistently across earlier salivary studies, more recent data point to a narrower diagnostic contribution. In a 2025 cohort using standardized saliva collection and predefined clinical endpoints, AOPPs and AGEs were elevated at baseline, but only AOPPs distinguished mild from moderate to severe endoscopic activity, and none predicted one-year outcomes [51]. This contrasts with earlier heterogeneous findings and clarifies the limited prognostic value of salivary oxidative-stress markers in IBD.
MicroRNAs represent a class of small, noncoding RNA molecules that adjust gene expression in response to environmental and inflammatory stresses, and their inherent stability has positioned them as promising biomarkers across multiple diseases [29]. In IBD, numerous miRNAs have been reported as altered in pathways related to epithelial barrier integrity, autophagy, and inflammatory signaling, supporting the idea that these molecules reflect key biological disturbances associated with disease activity and therapeutic response [29]. Their detectability in extracellular fluids, including saliva, positions them as a potential non-invasive tool for capturing transcriptomic alterations relevant to IBD [53].
Despite the diagnostic potential of microRNAs, the available evidence in IBD is still very limited, with only a small number of studies having examined them in saliva. The earliest dataset, originating from a 2015 study cited through a 2024 review, described differences in salivary miRNA expression between CD and UC, noting an alteration of miR-101 in CD and variations of miR-21, miR-31, miR-142-3p, and miR-142-5p in UC [53].
More recently, nearly a decade later, a study published in 2025 introduced a different approach by concentrating on salivary exosomes rather than whole saliva, aiming to protect RNA from degradation and improve analytical consistency [22]. In this cross-sectional study, exosomes were isolated through size exclusion chromatography, verified by nanoparticle tracking analysis and transmission electron microscopy, and then subjected to miRNAs through sequencing followed by RT-qPCR validation [22]. This methodology revealed clear differences between individuals with IBD and healthy controls [22]. The same exosomal miRNA also varied according to clinical activity and, in some cases, correlated more closely with clinical activity indices than standard blood biomarkers, underscoring the value of targeting exosomal fractions when investigating salivary biomarkers [22].
Taken together, these findings show that salivary transcriptomic research in IBD remains at an early stage. However, the recent incorporation of exosome-based methodology suggests a developing field with the potential to enhance the clinical relevance of salivary RNA biomarkers.
Saliva is a complex and dynamic fluid with the potential of detecting ranges of biomarkers (Table 2). Saliva composition is prone to vary with the stimulation method, oral hygiene, dietary habits, sleep patterns, and local oral health, making it sensitive to pre-analytical variation [54]. Studies involving saliva have shown that differences in collection approach, the presence of blood contamination, variations in flow rate, and recent behaviors such as eating, drinking, or smoking can alter measurable molecular profiles, thereby reducing their diagnostic reliability [54]. Furthermore, additional technical variability arises at the processing stage, including the use of preservatives, which can further influence analytical outputs [55]. These issues represent the most recurrent methodological barriers across salivary biomarker research, highlighting the need for standardized collection and handling protocols.
Summary.
| Type | Salivary biomarkers | IBD diagnostics | References |
|---|---|---|---|
| Inflammatory proteins | IL-6, MMP-10, Th17, TNF-α, IL-10, IL-1β, IL-17A, calprotectin | IL-6 showed the highest specificity among inflammatory biomarkers, with calprotectin showing high specificity in pediatric IBD | [13, 23, 26–35] |
| Heat shock proteins (HSP) | HSP60, HSP70, HSP90, anti-HSP70 | Although HSP concentrations are altered in IBD intestinal tissues, more studies should be done on their concentration in the saliva of IBD patients | [37–39] |
| Microbial | Prevotella, Veillonella, Streptococcus, Enterobacteria | IBD patients showed reduced salivary diversity, specifically an enrichment of Prevotella and Veillonella alongside a depletion of core commensals like Streptococcus | [26, 41–45] |
| Oxidative stress | AOPPs, AGEs | Oxidative stress salivary markers are used to determine the severity of the gut inflammation, but they lack long term prognostic abilities | [49–52] |
| Genetic and transcriptomic | miR-21, miR-31, miR-101, miR-142-3p, miR-142-5p,exosomal miRNA | Salivary miRNAs showed a difference between UC and CD. Exosome-based transcriptomic analysis has improved the stability of salivary microRNAs, offering high precision in differentiating IBD phenotypes | [22, 53] |
AGEs: advanced glycated end-products; AOPPs: advanced oxidation protein products; CD: Crohn’s disease; IBD: inflammatory bowel disease; UC: ulcerative colitis.
Beyond methodological sources of variation, current salivary studies in IBD remain constrained by limited sample sizes, the absence of longitudinal follow-up, and inconsistent reporting or control of treatment status. Additionally, the scarcity of empirical studies and the lack of multi-panel salivary investigations further restrict the ability to assess reproducibility and to establish consistent biomarker patterns.
Taken together, these factors indicate that while saliva represents a promising non-invasive biomarker, its application in IBD still requires further investigation before reliable integration into clinical assessment can be achieved.
Salivary biomarker research in IBD is further advanced with the emerging potential of technology-driven approaches.
Advances in artificial intelligence and machine-learning models have demonstrated improved diagnostic performance when applied to genomic and transcriptomic datasets, successfully distinguishing CD from UC and differentiating IBD from non-IBD conditions [56].
While not yet applied directly to salivary biomarkers, these findings suggest that integrating salivary biomarkers with advanced computational technologies may support the creation of more precise, multimodal tools for disease diagnosis and monitoring.
Emerging as a non-invasive tool, salivary diagnostics reflect systemic IBD processes through miscellaneous domains, with consistent findings including IL-6 increase and microbial change, specifically the enrichment of Prevotella and Veillonella alongside depletion of core commensals. While oxidative stress markers like AOPPs show promise despite limited prognostic utility, calprotectin lacks reliability in saliva compared to stool samples, except in a few cases, such as pediatric cases with oral manifestations or with stable transcriptomic biomarkers such as exosomal microRNAs. Despite encouraging progress, the clinical application of salivary biomarkers in IBD remains limited by methodological heterogeneity, small cohort sizes, insufficient longitudinal designs, and the substantial impact of oral health on molecular profiles. Considering these limitations, establishing better standardized collection and processing protocols, together with expanding investigations using multi-panel biomarker approaches, will be crucial to advance the translational potential of saliva.
AGEs: advanced glycated end-products
AOPPs: advanced oxidation protein products
CD: Crohn’s disease
HSPs: heat shock proteins
IBD: inflammatory bowel disease
ROS: reactive oxygen species
UC: ulcerative colitis
AC: Conceptualization, Writing—original draft, Writing—review & editing. CAK: Conceptualization, Writing—original draft, Writing—review & editing. DB: Conceptualization, Writing—original draft, Writing—review & editing. AGG: Validation, Supervision. LD: Validation, Supervision. LGL: Validation, Supervision. SAK: Validation, Supervision. NO: Validation, Supervision. FC: Validation, Supervision. AL: Validation, Supervision. AJ: Validation, Supervision. All authors read and approved the submitted version.
Abdo Jurjus, who is the Editorial Board Member of Exploration of Digestive Diseases, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1083
Download: 19
Times Cited: 0
