Affiliation:
1Department of Internal Medicine, Medeor Hospital, Abu Dhabi 40330, United Arab Emirates
Email: M.AhmedT600@Gmail.com
ORCID: https://orcid.org/0009-0003-1098-607X
Affiliation:
2Department of Medicine, Caucasus International University, Tbilisi 0141, Georgia
ORCID: https://orcid.org/0009-0004-7465-6296
Affiliation:
3Department of Medicine, Ivane Javakhishvili Tbilisi State University, Tbilisi 0179, Georgia
ORCID: https://orcid.org/0009-0008-6593-5364
Affiliation:
3Department of Medicine, Ivane Javakhishvili Tbilisi State University, Tbilisi 0179, Georgia
ORCID: https://orcid.org/0009-0005-4215-1178
Affiliation:
3Department of Medicine, Ivane Javakhishvili Tbilisi State University, Tbilisi 0179, Georgia
Affiliation:
3Department of Medicine, Ivane Javakhishvili Tbilisi State University, Tbilisi 0179, Georgia
ORCID: https://orcid.org/0009-0003-8433-9676
Explor Dig Dis. 2026;5:1005112 DOI: https://doi.org/10.37349/edd.2026.1005112
Received: December 11, 2025 Accepted: February 03, 2026 Published: February 26, 2026
Academic Editor: Jose C. Fernandez-Checa, Institute of Biomedical Research of Barcelona (IIBB), CSIC, Spain
The article belongs to the special issue Inflammatory Diseases of the Gastrointestinal Tract
This review goes over the impact of caffeine consumption in inflammatory bowel disease (IBD), examining epidemiology, clinical outcomes, mechanistic studies, and translational research. Caffeine, a widely consumed methylxanthine, exerts diverse physiological effects on the gastrointestinal tract. Mechanistic and preclinical data offer plausible biological pathways by which caffeine could influence the IBD course. Caffeine’s antagonism of adenosine receptors may modulate immune cell activation and cytokine release; its effects on gut motility and secretion can alter symptom perception, and caffeine-mediated changes in intestinal epithelial barrier function, oxidative stress, and the gut microbiome have been demonstrated. These effects make it a lucrative investigational option, and various studies have demonstrated that caffeine intake may reduce the incidence of IBD and may even have disease-modifying effects in regular consumers. However, differences in caffeine source (coffee, tea, soda), dose, concurrent dietary patterns, and disease subtype (Crohn’s disease versus ulcerative colitis) limit definitive causal inference. Clinical implications remain cautious: while moderate caffeine intake may be tolerable and even helpful for many patients, individualized assessment is advisable, particularly for those with symptom-triggering sensitivity or overlapping functional bowel disorders. Future research should target mechanistic links and clinically meaningful outcomes to inform evidence-based dietary guidance for people with IBD.
Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory condition of the gastrointestinal tract characterized by immune dysregulation, intestinal inflammation, and impaired quality of life. IBD includes the categories of ulcerative colitis (UC) and Crohn’s disease (CD). The incidence of IBD has been increasing over the last two decades. This rise was initially observed in industrialized countries, but the trend is starting to develop in developing countries as well, as they become increasingly more industrialized [1, 2]. As of 2019, approximately 4.9 million cases have been recorded worldwide, with the USA and China having the highest number of cases [3].
Historically, UC has been more common than CD in various regions around the world, but now the gap is narrowing, especially in Asia. For instance, in Malaysia, the UC:CD ratio dropped from 5:1 (1980–1990) to 1.7:1 (2010–2018) [4]. A similar trend can be observed in more industrialized Asian countries such as Japan, Korea, and China, where the incidence of CD is rapidly increasing; the same pattern is reflected in Africa and South America [2, 5, 6].
As the incidence of IBD increases, the burden of increased healthcare costs and morbidity also increases. In the US, the annual healthcare costs for IBD increased from $6.4 billion in 1996 to $25.4 billion in 2016, averaging nearly $14,000 per patient, while the United Kingdom alone spends approximately £720 million annually according to NHS estimates, costing £3,000 to £6,000 per patient [7, 8]. This clearly emphasizes the need for interventions and also overall improved understanding of the disease itself [9].
Diet plays a key role in the development and symptomatology of IBD, and it is important to monitor it as it is a modifiable exposure; many patients frequently complain of certain associations between certain dietary habits and the symptoms they experience [10].
Caffeine is one such dietary factor that has attracted significant interest from both patients and healthcare providers. Caffeine exerts its pleiotropic effects primarily by blocking the adenosine receptor, leading to enhanced neuronal activity, alertness, and metabolic stimulation [11]. Aside from its systemic effects, caffeine also has gastrointestinal effects that might potentially be useful in ameliorating the symptoms of IBD. These effects include stimulation of gastric acid secretion, increased intestinal motility, and modulation of bile acid metabolism [11–13]. By stimulating gastric acid secretion and increasing intestinal motility, it can potentially promote more efficient digestion and relieve constipation, which IBD patients usually experience during treatment or remission. Modulation of bile acid metabolism can support gut homeostasis and exert anti-inflammatory effects [11, 12].
The relationship between caffeine consumption and IBD is complex. Some studies show that caffeine can be a potential trigger for gastrointestinal symptoms, with reports of diarrhea, abdominal discomfort, and exacerbation of disease flares [14]. Observational studies have presented mixed results: certain cohorts found no clear relationship between coffee or caffeine intake and disease risk or activity, while others show possible protective effects of moderate coffee consumption on disease development or progression [15, 16]. A proposed explanation for these varying results could be due to differences between UC and CD, the proposed metrics of disease activity that were reported (patient symptoms vs. mucosal healing or stool biomarkers), as well as inherent differences between populations with varying dietary and cultural backgrounds [17]. An additional explanation could be due to adenosine’s anti-inflammatory effects on the gut, and by inhibiting adenosine receptors, caffeine may tip the balance towards a more pro-inflammatory milieu. Interventional data are inadequate, with only a handful of randomized controlled trials investigating caffeine specifically in IBD.
From a clinical standpoint, the unclear effects of caffeine’s role in IBD pose challenges, as patients often seek dietary guidance; contradictory results may lead to unwanted dietary restriction or overconsumption without awareness of potential complications. This review intends to delve into the available evidence on caffeine and its relevance to IBD, clarify consistent findings, explain the inconsistent evidence, and outline priority areas for future research.
IBD is characterized by persistent inflammation of the gastrointestinal tract, leading to mucosal injury and damage, causing intestinal and extra-intestinal symptoms that can severely affect a patient’s quality of life. Although UC and CD can share several clinical features, they are different in terms of anatomic distribution, histopathology, complications, as well as treatment responses; however, disease monitoring and treatment protocols are often more similar than not [18–20]. The global burden of IBD has steadily risen in recent years, rapidly affecting both the Western and Eastern hemispheres, which reflects the impact of environment, diet, and lifestyle on disease pathogenesis [2, 9, 21].
Intestinal permeability and interactions between the immune system and bacterial products have been proposed to play a crucial role in the pathogenesis of IBD. The intestinal barrier consists of epithelial cells, tight junctions, mucus layers, and immune cells that prevent luminal contents from translocating into the bloodstream [22–25]. In IBD, this barrier is compromised, leading to an increase in intestinal permeability, bacterial translocation, and an exaggerated immune response. These mechanisms are backed by human biopsy studies, animal models, and in vitro epithelial systems, which demonstrate barrier dysfunction during active disease [26, 27]. As mentioned previously, the intestinal microbiota plays a critical role in maintaining gut immune tolerance and homeostasis. Dysbiosis, which is characterized by a decrease in bacterial diversity and an overrepresentation of pro-inflammatory bacterial species, has been consistently observed in IBD patients [26, 27]. Lloyd-Price et al. [28] reported that among IBD patients, beneficial commensals such as Faecalibacterium prausnitzii and Bifidobacterium species are depleted, as well as an expansion of Enterobacteriaceae, which promote inflammation and oxidative stress being reduced [29].
The association between the gut microbiome and IBD can be supported by studies that aimed to alter the microbiome to restore intestinal equilibrium. A randomized controlled trial demonstrated that kefir (a fermented milk product rich in probiotics) caused modest improvements in microbial composition, inflammatory markers, and symptoms in patients with IBD [29]. Other studies have tested fecal microbiota transplantation with variable success in both UC and CD [30, 31]. While microbiome-targeted therapies remain a more experimental treatment option, these findings highlight the intimate and important relationship between gut flora and mucosal health.
In UC, inflammation is generally limited to the colonic mucosa and submucosa, beginning in the rectum and extending proximally and continuously [18]. In CD, inflammation is transmural and may affect any part of the gastrointestinal tract, frequently involving the terminal ileum, with lesions separated by normal and healthy bowel [19]. This pattern of transmural involvement explains why CD often leads to strictures, fistulas, and penetrating complications, whereas UC more commonly results in superficial ulceration, bloody diarrhea, rectal urgency, and tenesmus [18, 24]. Patients often report weight loss and fatigue, especially during active flares. In CD, symptoms are more heterogeneous due to the variable anatomical involvement. Common symptoms in CD include chronic abdominal pain, non-bloody diarrhea, weight loss, fever, strictures, and malabsorption due to small bowel involvement and perianal disease such as fissures, fistula-in-ano, and abscesses [19]. When compared to CD, UC carries a significant risk of colorectal cancer (CRC), especially in patients with long-standing or extensive disease. Other complications include severe bleeding, toxic megacolon, and bowel perforation [25, 30]. Extraintestinal manifestations are also common in both forms, including arthritis, uveitis, erythema nodosum, and primary sclerosing cholangitis, though the pattern and prevalence may differ [31].
The management of IBD is tailored to disease phenotype and severity and is typically aimed at inducing and maintaining remission as well as preventing complications. In UC, treatment often begins with aminosalicylates (5-ASA) for mild disease, escalating to corticosteroids for flares, and early utilization of immunomodulators or biologics targeting tumor necrosis factor α (TNF-α), or interleukin (IL) pathways such as combined IL-12/23 inhibitors (e.g., Ustekinumab, a p40 subunit inhibitor) or IL-23 inhibitors (e.g., p19 inhibitors such as risankizumab, guselkizumab) for more severe cases [32]. In CD, 5-ASA agents are less effective, and as a result, treatment relies heavily on corticosteroids for induction, followed by immunomodulators such as azathioprine or methotrexate and biologics [33]. Recent advances include the use of Janus kinase (JAK) inhibitors and gut-selective therapies (such as integrin inhibitors, S1P modulators), which offer new opportunities for targeted intervention [34].
IBD management increasingly reiterates the importance of diet and lifestyle factors in disease expression and patient well-being. Dietary patterns may influence intestinal inflammation directly through effects on the microbiome, barrier function, as well as gut immune responses [10, 15]. High-fat, high-sugar diets are associated with worsening symptoms, while diets rich in fiber and plant-based nutrients may promote a favorable microbial environment and reduce disease activity [35, 36]. Given the global rise of IBD, the role of diet as both a risk factor and therapeutic adjunct is an important frontier for research, naturally leading into a discussion of nutrition-focused strategies for disease prevention and management.
Caffeine and its associated compounds have various effects within the gastrointestinal tract, affecting the gut microbiota, stimulating intestinal barrier function, modulating motility, and attenuating inflammation (Figure 1). In addition, other bioactive compounds found in coffee, such as diterpenes, chlorogenic acids, and melanoidins, synergistically act with caffeine to produce systemic effects on gut and metabolic health [37]. Coffee consumption is prevalent across various populations and age groups, with studies showing that a significant percentage of adults consume caffeine daily, primarily through coffee [38]. This widespread consumption has marked considerable interest in its potential impact on the gastrointestinal tract.
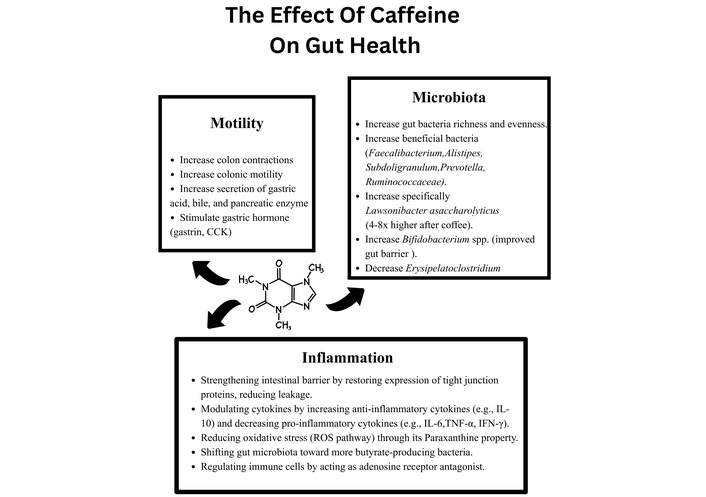
This illustration is a simplified mechanism model of the multifaceted effects of Caffeine on gut health, highlighting the influence on motility, microbiota, and inflammation. Caffeine enhances colonic contractions, motility, and digestive secretions. It promotes a healthier gut microbiota profile by increasing the number of “good” bacteria, supports intestinal barrier function, reduces oxidative stress, and modulates the immune response by upregulating anti-inflammatory cytokines and downregulating pro-inflammatory ones. Caffeine PubChem CID: 2519. https://pubchem.ncbi.nlm.nih.gov/compound/2519.
Caffeine exerts many of its effects on the body and the gastrointestinal tract through non-selective antagonism of adenosine receptors (A1, A2, and A3), which are widely expressed in enterocytes, immune cells, enteric neurons, and smooth muscle cells, leading to modulating absorption, secretion, inflammation, and motility [37, 39]. Inhibiting adenosine receptors can be either pro- or anti-inflammatory, since upregulation of the CD73/adenosine pathway is seen in inflammatory states [27, 37]. It is believed that adenosine, through its various effects on the gastrointestinal tract, may act as an “alarm signal”, signifying inflammation and driving forward mechanisms to modulate and resolve inflammation by increasing the concentration of cAMP [40]. It is interesting to note that despite its properties as an adenosine antagonist, caffeine increases cAMP levels by inhibiting phosphodiesterase enzymes, which could paradoxically potentiate adenosine’s anti-inflammatory effects in the gut [39, 41, 42].
Irrespective of their effects on adenosine receptors, caffeine and its metabolites, paraxanthine, theobromine, and theophylline can directly regulate intestinal inflammation and preserve epithelial barrier function. In vitro studies demonstrated that caffeine and paraxanthine modulate the expression of tight junction proteins (OCLN, ZO-1, CLDN1) and inflammatory cytokines (TNF-α, CXCL9), serving to restore intestinal barrier integrity and reducing reactive oxygen species [43, 44]. This is mostly mediated by their direct inhibition of CHI3L. In a pre-clinical study by Lee et al. [42] caffeine administration in dextran sodium sulfate (DSS)-induced colitis models improved body weight recovery, reduced histological damage, suppressed chitinase-3-like protein 1 (CHI3L1) expression (which degrades the intestinal barrier and promotes bacterial translocation), decreased pro-inflammatory cytokines (TNF-α, IL-17F, FN-Y, IL-4), and elevated anti-inflammatory IL-10, demonstrating both anti-inflammatory effects and barrier protection [45, 46]. CHI3L is such a potent driver of chronic intestinal inflammation that synthetic methylxanthines that are more selective to the gut and directly inhibit CHI3L1 are under investigation for the treatment of IBD [42]. This effect on the chitinase pathway could be a key mechanism as to why caffeine demonstrates anti-inflammatory effects on the gut.
Multiple human studies have demonstrated that habitual caffeine intake significantly influences the gut microbial environment. Higher intake of caffeine has been linked to greater microbial diversity and enrichment of beneficial short-chain fatty acid (SCFA)-producing genera such as Faecalibacterium, Roseburia, and Alistipes, which contribute to epithelial integrity and anti-inflammatory signaling. Conversely, potentially pathogenic or pro-inflammatory taxa, including Thomasclavelia ramosa (formerly known as Erysipelatoclostridium ramosum), tend to decrease among higher caffeine consumers, indicating a shift toward a more favorable microbial ecosystem [47]. Population-based observations further support these associations, showing correlations between caffeine intake, bowel habit patterns, and markers of intestinal inflammation such as fecal calprotectin, highlighting the potential of microbiota-mediated pathways linking caffeine to IBD risk [48, 49].
In a study analyzing colonic tissue from 34 individuals with normal colonoscopy results, higher caffeine consumers exhibited greater microbial diversity and increased abundance of beneficial genera such as Faecalibacterium, Roseburia, and Alistipes, while potentially harmful genera like Thomasclavelia ramosa (formerly known as Erysipelatoclostridium ramosum) were reduced [47, 50]. Similar findings were reported in adults consuming three or more cups of coffee daily, where increases in Bifidobacterium and Lactobacillus species and other SCFA-producing bacteria were observed. SCFAs such as butyrate and propionate exert potent anti-inflammatory effects and help restore intestinal permeability [13, 51, 52]. In addition, experimental animal models reinforced these observations, demonstrating reductions in Escherichia coli, Enterococcus, and Clostridium populations alongside enhanced Lactobacillus and Bifidobacterium, suggesting a shift toward a more favorable colonic environment [53, 54].
A key factor to recognize is that these effects may not be attributed to caffeine alone, since polyphenols found in coffee such as caffeic acid and chlorogenic acid also exert antioxidant and neuroprotective effects on the myenteric plexus, they also serve to enhance colonic motility via SCFAs generated by microbiota fermentation, emphasizing the prebiotic potential of coffee residues apart from caffeine’s effects alone [11, 37]. Coffee increases colonic motor activity within minutes of ingestion, independent of the gastrocolic reflex. This is observed empirically as coffee consumption can induce the urge to defecate in some individuals, supporting its role as a bowel stimulant [55, 56]. Habitual caffeine intake may improve bowel regularity by stimulating colonic contractions, particularly in populations prone to constipation. This effect has also been observed in decaffeinated coffee, suggesting that caffeine may not be the only stimulator of colonic motility in caffeinated beverages [55, 57].
Another important limiting factor is the sheer heterogeneity of source of caffeine, variability in preparation, fiber content, and study designs, which can complicate the mechanistic understanding of these effects [37]. Evidence regarding caffeine intake and CRC risk remains mixed, with some cohorts demonstrating decreased CRC risk with moderate coffee intake, while others report no association or possible site-specific cancer promotion, once again reflecting heterogeneity in population, study design, and coffee composition [37, 39, 58, 59].
Despite the reported benefits of caffeine on gut health, it is important to note that caffeine’s effects are not universally positive. Excessive intake has been associated with gastrointestinal discomfort, including symptoms such as acid reflux, heartburn, and abdominal pain [11, 60]. Caffeine stimulates gastric acid secretion, which may exacerbate conditions such as gastroesophageal reflux disease (GERD) and peptic ulcer disease in susceptible individuals. Moreover, its stimulatory effect on colonic motility, while beneficial for constipation, can lead to diarrhea or loose stools in sensitive populations, which is highly relevant in the IBD population [49, 57, 61].
Having discussed the various effects of caffeine on the gastrointestinal tract, there has been considerable interest in the effects of caffeine in patients with IBD.
Recent studies suggest that caffeine or coffee consumption does not consistently show a harmful link in relation to UC incidence and may, in fact, reduce the incidence of UC [48, 62, 63]. This trend was reported in the literature in both Eastern and Western countries. A 2017 systematic review and meta-analysis by Nie and Zhao [64] found that across several studies, the intake of tea and coffee was associated with a decreased incidence of UC [48, 65, 66]. In addition, a Mendelian randomization study by Dong et al. [67] found that patients with genetically predicted elevated plasma caffeine concentrations were consistently associated with a decreased risk of IBD; the decreased risk was similar in both UC and CD, suggesting a possible causal relationship.
Several studies demonstrated that in UC, caffeine intake did not significantly alter their bowel habits or gastrointestinal symptoms [49, 60, 61]. Barthel et al. [61] found that amongst IBD patients, the majority of UC patients did not report a change in their symptoms or bowel habits; almost 1 out of 5 UC patients (in contrast to 1 out of 2 CD patients) reported that coffee worsened their symptoms; however, the majority of them reported no attempts to refrain from coffee. A cross-sectional study published by Yang et al. [60] demonstrated that among patients with IBD, caffeine intake (< 204 mg/day) was negatively associated with chronic diarrhea and constipation; however, higher caffeine consumption (≥ 204 mg/day) was actually associated with an increased risk of constipation despite caffeine’s well-recognized pro-motility effects.
Despite the aforementioned neutral effects of caffeine on reported UC symptoms, it is well recognized that patients’ reported symptoms alone do not directly correspond to intestinal inflammation and disease activity; measuring the levels of inflammatory markers such as erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), fecal calprotectin, or endoscopic evaluation is utilized to monitor disease activity and relapses in IBD [61, 68, 69]. In this context, Neamți et al. [49] reported that while most patients in their cohort, with UC who regularly consumed coffee, did not report worse symptoms, and were found to have significantly lower fecal calprotectin levels, reflecting lower disease activity amongst coffee drinkers. It is important to note that this effect cannot be attributed solely to caffeine, and it is plausible that the other compounds that are present in coffee, such as the various polyphenols, could play a role; as such, further investigational studies are required to confirm this association.
To date, there have been no studies evaluating the effect of caffeine on endoscopic improvement, frequency of flares, or severity of flares. It is important to note that most of the evidence available is from observational studies and that there is a considerable scarcity of direct investigational studies, limiting direct causality. What can be gleaned so far is that caffeine and beverages containing caffeine have not been reported to directly worsen symptoms in UC, have been associated with improved fecal biomarkers; however, randomized controlled trials are required to clearly establish a direct link between these findings.
It’s well-established that diet plays a crucial role in the development and pathogenesis of IBD; this is a little more relevant in CD as it’s often associated with nutritional deficiencies and malabsorption. The evidence is somewhat mixed when it comes to the effect of caffeine on CD. This could be due to the heterogeneity of CD’s manifestations on the gastrointestinal tract, CD’s predilection to affect the small intestines, where most of the caffeine content in caffeinated beverages is absorbed [70].
Most of the available evidence so far is from observational studies. A case control study by Ng et al. [71] found that daily tea consumption was significantly associated with a decreased risk of developing CD, however coffee consumption had no significant effect on the risk of developing CD, it’s interesting to note that in the same study both daily tea and daily coffee consumption were significantly associated with a decreases risk for the development of UC. Overall, the study reported that the conventional “western diet”, which is high in refined sugars and processed foods, contributed negatively to the development of IBD, and that the recent dietary trends of developing nations towards a more westernized diet could be a potential explanation as to why IBD rates are increasing in both Eastern and Western hemispheres [1, 2, 71]. Since data is derived from cross-sectional and observational studies, reverse causation must be considered while interpreting it. Patients’ ability to tolerate coffee may simply be linked to milder disease or lower inflammatory burden, while those with active disease tend to avoid it. This behavioral pattern can therefore occur without implying a true disease-modifying effect.
Additional insight can be drawn from outcomes reported by patients according to Neamți et al. [49]. The rate of regular coffee consumption in both CD and UC was high and did not differ significantly between the two groups, and most patients did not report worsening of symptoms with regular coffee consumption. As mentioned previously, it is important to consider that in IBD, the absence or presence of symptoms does not directly correlate with intestinal inflammation and disease progression [24, 25].
In contrast, Barthel et al. [61] reported that almost half of CD patients who regularly consume coffee do report a negative association between coffee and their symptoms (this number is twice that of UC patients); despite this association, CD patients did not alter their coffee intake. This shows that perception may not always influence action and that patients’ subjective symptoms do not always correlate with intestinal inflammation, as the same study by Neamți et al. [49] found that the fecal calprotectin values were significantly lower amongst CD patients who were coffee drinkers [60, 61].
The previously mentioned cross-sectional study by Yang et al. [60] describing the U-shaped phenomenon between bowel habits in IBD patients and coffee intake suggests that coffee has different effects at different doses, it can help with either constipation or diarrhea depending on the dosage, this is of therapeutic interest as due to the nature of the transmural inflammation of CD and it’s tendency to affect the small intestine and thus cause malabsorptive diarrhea, this dose-response relationship can be of particular benefit to patient’s with CD that may present with alternating constipation and diarrhea [19, 60].
While caffeine has many positive effects on the gastrointestinal tract (Figure 2), analyses of dietary patterns also place coffee and caffeine-containing beverages within broader eating habits. Western or carnivorous style diets rich in processed foods, red meat, and low fiber appear to worsen IBD more than coffee itself, while some recent studies show that overall diet quality, including fiber intake and less processed food consumption than any single drink, is more strongly linked to reduced disease activity [36, 63, 72, 73]. This further reinforces the fact that coffee’s effects may depend on the overall dietary context as well as its dose-dependent effects. Moderate (100–300 mg/day) intake within a balanced diet that is low in processed foods may be even beneficial [36, 72, 74] (Figure 3).
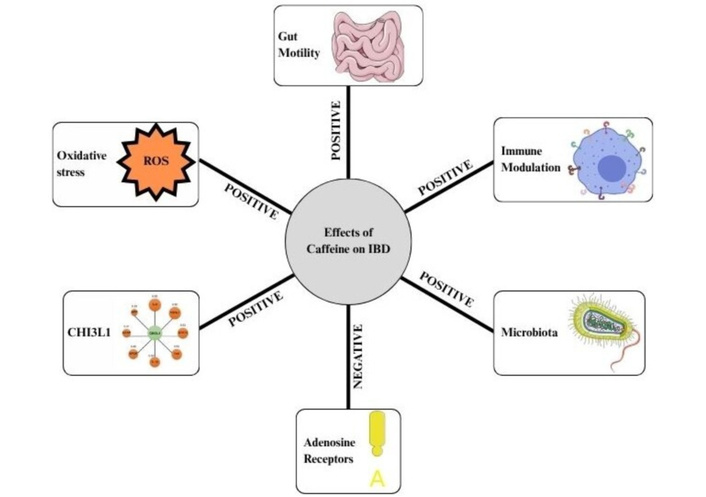
This schematic figure illustrates 6 major pathways that caffeine affects, relevant to the pathophysiology of inflammatory bowel disease (IBD). (1) Gut motility: Caffeine has a positive effect by increasing intestinal motor activity. This property can help those patients with constipation. (2) Immune modulation: Caffeine has a positive effect by reducing pro-inflammatory cytokines and downregulating chitinase-3-like protein 1 (CHI3L1). (3) Microbiota: Caffeine alters the gut microbiota by increasing beneficial bacteria, thereby decreasing intestinal inflammation. (4) Oxidative stress: Caffeine displays antioxidant properties by reducing reactive oxygen species and damage to DNA. (5) CHI3L1: Caffeine downregulates CHI3L1, which reduces the severity of colitis. (6) Adenosine receptors: Caffeine has a negative impact by antagonizing adenosine receptors, which blocks its anti-inflammatory effects, which could potentially negate caffeine’s net anti-inflammatory effect in IBD.
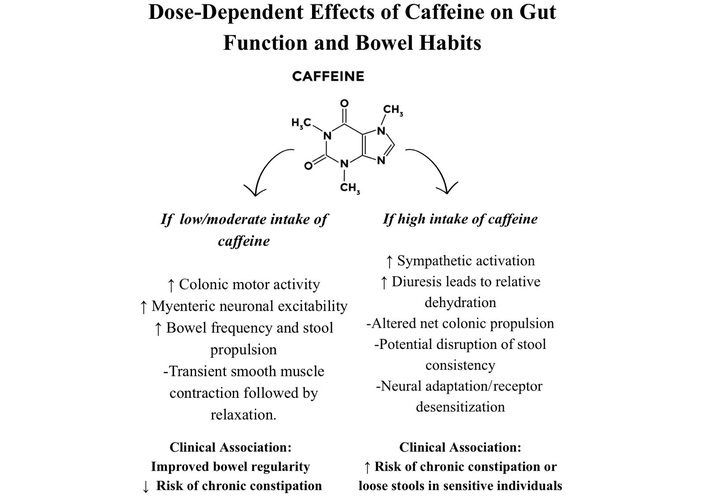
This illustration is a simplified mechanism model of the dose-dependent effects of caffeine on bowel habits. Low–Moderate (< 200 mg/day) intake of caffeine is associated with increased neuronal excitability and smooth muscle contraction that manifests as more frequent stools. Higher doses of caffeine (≥ 200 mg/day) are associated with desensitization and may paradoxically lead to decreased stool frequency and disrupted bowel habits.
A notable limitation to these findings is the fact that most of the evidence is based on preclinical models and observational cohorts, and thus should not be interpreted as causal inference but instead be hypothesis-generating and perhaps guide future investigational studies.
No two IBD patients are the same, and there is a considerable lack of literature delving into the effect of caffeine on different disease phenotypes. Another important limiting factor is the fact that most of the studies reporting the association between caffeine and IBD are from North America, Europe, and Asia, and since the consumption of caffeinated beverages varies between cultures, the lack of data from Africa and South America limits the generalizability of these findings. Finally, caffeine content differs between different beverages, and the concentration of other molecules such as polyphenols may play a role in the effects described so far. Ultimately, with the evidence provided thus far, caffeine can be consumed in IBD safely with moderation (with more caution paid to CD), provided that patients do not complain of significant symptoms and are not in active remission.
Caffeine exerts diverse physiological effects on the intestines and has been hypothesized to reduce intestinal inflammation by downregulating inflammatory markers, maintaining gut permeability, and causing positive shifts in the gut microbiome through its effects on adenosine receptors, CHI3L, and influencing the gut microbiome, which are specific to the intestines, could prove to be potential therapeutic targets. Overall, caffeine found in beverages does not significantly affect symptoms in IBD patients, and may be associated with a decreased incidence of IBD and an improvement in gut inflammation.
5-ASA: aminosalicylates
CD: Crohn’s disease
CHI3L1: chitinase-3-like protein 1
CRC: colorectal cancer
CRP: C-reactive protein
DSS: dextran sodium sulfate
ESR: erythrocyte sedimentation rate
GERD: gastroesophageal reflux disease
IBD: inflammatory bowel disease
IL: interleukin
JAK: Janus kinase
SCFA: short-chain fatty acid
TNF-α: tumor necrosis factor α
UC: ulcerative colitis
The authors acknowledge the research training support from INSCRIBE by INCREDOC, Tbilisi, Georgia.
MAM: Conceptualization, Project administration, Supervision, Writing—review & editing. DC: Visualization, Supervision, Writing—original draft, Writing—review & editing. HS: Visualization, Project administration, Writing—original draft, Writing—review & editing. SH: Visualization, Writing—original draft, Writing—review & editing. ZA: Writing—original draft. SS: Writing—original draft, Writing—review & editing. FB: Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 5221
Download: 33
Times Cited: 0
Neelakanta Sarvashiva Kiran ... Ankita Chatterjee
