Affiliation:
Sports Physical Therapy Residency, Carolinas Rehabilitation, Atrium Health, Charlotte, NC 28211, US
Email: Tommywyattdpt@gmail.com
ORCID: https://orcid.org/0009-0006-9900-4440
Explor Musculoskeletal Dis. 2025;3:1007102 DOI: https://doi.org/10.37349/emd.2025.1007102
Received: June 08, 2025 Accepted: August 11, 2025 Published: August 26, 2025
Academic Editor: Fernando Pérez-Ruiz, Cruces University Hospital, Spain
Arterial thoracic outlet syndrome (aTOS) is a rare condition, but it has an elevated incidence among athletes due to high mechanical demands placed on the upper extremities. Post-surgical rehabilitation guidelines for aTOS are not well defined, especially in high-performance populations. Mechanically loaded neurodynamics (MLND) is a novel technique that introduces controlled external load during neurodynamic movements to optimize neurovascular adaptation and musculoskeletal function. A 47-year-old professional long-distance cyclist presented with left upper extremity pain, paresthesia, and vascular symptoms three weeks after undergoing left first rib and cervical rib resection with scalenectomy for aTOS. The patient had significant scapular dyskinesis, thoracic spine hypomobility, glenohumeral joint stiffness, and posture-related thoracic outlet compression, alongside hyperalgesia and allodynia in the ulnar nerve distribution. A structured 11-week physical therapy protocol was implemented, including traditional and MLND techniques, progressive scapular and thoracic mobility training, and sport-specific strengthening. MLND was introduced to progressively load neural tissues in median, ulnar, and radial nerve distributions while addressing musculoskeletal impairments that contribute to thoracic outlet compression. The patient demonstrated a significant reduction in pain and neurological symptoms, resolution of allodynia, restoration of full active range of motion, ≥ 93% limb symmetry index in strength testing, and an improvement in DASH score from 86.7 to 2.5. The athlete returned to unrestricted cycling at her previous level within 3.5 months post-operation. No adverse effects were reported with MLND use. This case highlights the potential role of MLND in accelerating recovery following thoracic outlet surgery in athletic populations. The approach emphasizes progressive mechanical loading to stimulate neurovascular adaptation and addresses the underlying musculoskeletal impairments contributing to neurovascular compression. Given the absence of standardized protocols for aTOS rehabilitation, MLND may serve as a safe and effective intervention. Future research should further investigate its mechanophysiological effects and clinical efficacy through controlled trials.
Thoracic outlet syndrome (TOS) is a rare group of conditions in which there is compression or entrapment of the neurovascular structures at the thoracic outlet, defined by the space between the clavicle and the first rib. The affected structures include the brachial plexus, subclavian artery, and subclavian vein, each classified into distinct subtypes of TOS. The three primary subtypes include neurogenic (nTOS), accounting for approximately 90% of cases; venous (vTOS), comprising 5–10% of cases; and arterial (aTOS), comprising only 1–2% of cases [1]. TOS is a multifactorial condition, fundamentally rooted in musculoskeletal dysfunction leading to neurovascular compression in the upper extremity. TOS is accurately conceptualized as a neurovascular syndrome with a primary musculoskeletal origin; compression being of bony origin, and entrapment being of soft tissue origin. Musculoskeletal impairments such as scapular dyskinesis, thoracic kyphosis, forward head posture, and glenohumeral joint (GHJ) restrictions can narrow the thoracic outlet and increase mechanical stress on the brachial plexus and subclavian vessels. These postural and kinematic deficits or demands often exacerbate neural tension and vascular compromise during upper extremity loading, particularly in athletes. The most common treatment for nTOS is physical therapy management, while surgery is often indicated for vascular TOS [2]. Vascular TOS (aTOS, vTOS) is primarily caused by a bony abnormality, seen commonly in the first rib, clavicle, or cervical rib. These structures compress the neurovascular structures passing through the thoracic outlet, causing a cascade of symptoms which may include but are not limited to weakness, numbness, tingling, muscle wasting, loss of sensation, discoloration, coldness of the fingers and hand, hyperalgesia, allodynia, and claudication of the upper extremity [3].
Although rare in nature, TOS affects the athletic population at a higher incidence rate than the general population. This is likely due to repetitive upper extremity movements, muscle hypertrophy, traction injury in collision sports, and increased mechanical demands of the neurovascular structures in the upper extremity. Sports like baseball, swimming, weightlifting, and football have notably higher incidence rates than other sports, with studies suggesting 30% of professional baseball pitchers experience TOS related symptoms in their careers [4, 5]. Return to play timelines have not been extensively researched and reported, but the available data shows athletes typically resume their sport in 4–6 weeks for physical therapy management of nTOS (surgical management of nTOS, extended timeline to approximately 4 months), 3.5 months post-surgery for vTOS, and 4.7 months post-surgery for aTOS. Studies indicate long-term effects of TOS such as strength, power, and endurance deficits may continue to persist for up to 1 year [6]. Common surgical management includes the decompression via scalenectomy and resection of the bony abnormalities, such as the first rib, clavicle, or cervical rib resection [6]. Systematic reviews highlight the absence of rigorous diagnostic criteria and standardized physical therapy management guidelines for athletes with TOS [7]. Musculoskeletal impairments such as scapular dyskinesis, limited thoracic spine (T/S) mobility, postural asymmetries, and GHJ stiffness often accompany neurovascular compression. Such impairments may perpetuate neurovascular compression, contribute to symptom recurrence, and must be concurrently addressed through integrated orthopedic rehabilitation strategies. High mechanical demand sports like cycling require optimal neuromuscular and postural control, making musculoskeletal-focused rehabilitation essential for safe and effective return to sport.
Recent expert consensus emphasizes the need for standardized diagnostic and rehabilitation pathways in TOS [7]. Typical post-surgical management of TOS includes pain reduction, neural and vascular mobility, restoration of range of motion (ROM), strength, and scapulothoracic control [8]. Neurodynamic techniques are common in physical therapy management and are widely utilized in improving neural mobility. Traditional neurodynamic techniques involve passive mobilization of neural tissue to improve nerve excursion and reduce mechanosensitivity [9]. However, passive neurodynamics often fail to adequately prepare athletes for return to high-level mechanical load required of the neurovascular structures in their sport-specific movements. Mechanically loaded neurodynamics (MLND) builds upon this concept by introducing a progressive, controlled external load to the neurovascular structures, using graded tension and movement to optimize neural and vascular mobility, and promote adaptations in healing and tolerance [10]. From a physiological perspective, MLND influences neural and vascular remodeling. Increased mechanical load enhances neurovascular compliance, promotes increased endoneurial blood flow, decreases intraneural edema, and improves axoplasmic transport [11]. In vascular structures, MLND facilitates shear stress adaptation, improves dilation, capillary recruitment, and overall vascular resilience under load. These mechanophysiological adaptations differentiate MLND from traditional neurodynamics, making it a promising intervention for post-surgical TOS rehabilitation in the athletic population [2, 11].
This case study examines the application of MLND in the post-surgical rehabilitation of an athlete with a unique presentation of aTOS, evaluating its effectiveness in facilitating a timely return to sport. This report aims to contribute to the scarce but growing body of evidence supporting evolving neurovascular rehabilitation strategies and refine rehabilitation protocols for TOS in the athletic population. Recent literature supports the growing complexity of TOS in high-demand athletes and emphasizes the need for individualized and load-responsive rehabilitation approaches.
The patient was a 47-year-old female professional long-distance cyclist. The right hand is dominant, with no previous history of left shoulder, arm, or neck pain. Prior medical history of psoriatic arthritis, rheumatoid arthritis, iron deficiency disorder, gastroesophageal reflux disease (GERD), breast cancer (treated with cryoablation), and gastric bypass. She was re-hospitalized post-surgery due to pleural effusion, delaying the start of physical therapy management by 2 weeks.
The patient showed a functional and medical diagnosis of acute left shoulder pain with strength, mobility, and endurance deficits, secondary to aTOS, 3 weeks post left first rib and cervical rib resection, with scalenectomy. Symptoms included 8/10 pain on a numerical pain rating scale (NPRS). Sharp shooting pain was radiating from the left-sided cervical spine (C/S) down the left upper extremity (LUE) to the fingers. Numbness and tingling (N/T) were in the median and ulnar nerve patterns, with some radial nerve distribution. Pain was exacerbated by shoulder abduction beyond 90 degrees, especially during exertion. Pain eased with rest and arm support. The referred trigger point (TrP) was at the medial border of the left scapula. Hyperalgesia and allodynia were in the ulnar nerve distribution. Upper limb tension test (ULTT) was positive in the median and ulnar nerve patterns. A significant limitation was in T/S mobility in extension. Physical examination also revealed notable scapular winging and poor scapular upward rotation during active shoulder flexion, suggestive of scapular dyskinesis. The patient demonstrated excessive thoracic kyphosis and forward head posture, which further increased mechanical stress on the thoracic outlet. GHJ stiffness was observed during overhead motion, with compensatory motion noted at the cervicothoracic junction.
Physical therapy management consisted of 11 visits over the course of 11 weeks. The care began 20 days post-surgery. A summary of the patient’s subjective symptom resolution and objective improvements is provided in Table 1. Interventions over the course of care are detailed in Table 2. MLND was implemented using green medium-resistance bands (approximately 4.6–6.9 lbs. at 100% elongation). The following protocol followed along a progressive loading sequence: week 3–4: Low tension isometric holds (5–10 seconds) in partial range tensioner positions using green bands; week 5–7: Dynamic repetitions (10–15 reps, 3 sets/session) were used in full range nerve tension patterns against moderate resistance; week 8–11: Sport-specific load progression involved incorporating resisted cycling postures and closed chain upper extremity tasks with integrated and progressive MLND. Each repetition was performed in a slow and controlled manner, with verbal cueing to avoid guarding or Valsalva. Progression was based on symptom response, NPRS scores, and fatigue tolerance. MLND was performed 1–2 times per week in clinic, with daily home exercise reinforcement. Traditional neurodynamics and MLND were implemented using the procedure shown in Figure 1A–C for median nerve, Figure 2A–C for ulnar nerve, and Figure 3A–C for radial nerve.
Symptom progression and clinical milestones
| Week | NPRS pain score/DASH | Allodynia/hyperalgesia | ROM limitation | Strength (LSI) | Notes |
|---|---|---|---|---|---|
| Initial–2 | 8/10DASH: 86.7 | Present | 10% | N/A | Median/ulnar pain; trigger points |
| 3–4 | 6/10–3/10 post-MLND | Present | Full ROM restored | N/A | Centralization of pain |
| 5–7 | 2/10 | Absent | Normalized | 85–90% | Resumed biking |
| 8–11 (discharge) | 0/10DASH: 2.5 | Absent | Full | ≥ 93% | Returned to unrestricted sport |
The patient’s DASH score improved from 86.7 at initial evaluation to 2.5 at discharge, reflecting near complete resolution of functional disability. NPRS: numerical pain rating scale; DASH: Disabilities of the Arm, Shoulder and Hand questionnaire; ROM: range of motion; LSI: limb symmetry index; MLND: mechanically loaded neurodynamics
Rehabilitation phases, interventions, and outcomes
| Phases | Intervention | Symptoms/outcomes |
|---|---|---|
| Initial evaluation–week 2 | Patient education on rehabilitation process. TE focused on C/S, T/S, scapular mobility, along with activation of the biceps, triceps, forearm, and grip. Gentle introduction to weightbearing.MT: focused on C/S for pain modulation, and introduction to traditional nerve glides in the median, ulnar, and radial patterns. | NPRS pain score 4/10, sharp and achy pain from left-sided C/S down LUE in the ulnar nerve pattern, worse with exertion. Centralization of T/S pain to the left-sided C/S. Continued decreased LUE strength and endurance. Signs of allodynia in the ulnar nerve pattern. 8/10 pain on exertion. Left shoulder active ROM limited 10%. C/S active ROM limited 10%. DASH: 86.7. |
| Week 3–4 | TE: progressive scapular, C/S, T/S, and GHJ load against gravity, and light resistance bands. Scapular stability in open and closed chain.Upper body ergometer intervals, light resistance, reduced time, and reps based on symptoms.MT: focused on C/S for pain modulation, and introduction to manually loaded nerve glides in the median, ulnar, and radial patterns via tensioners, and isometric load throughout ROM as tolerated.Week 4 introduced band resisted nerve glides in the median, ulnar, and radial patterns. | NPRS pain score 6/10, pain left hand continues to be intense burning. No pain in the shoulder, more pain in the medial forearm. Allodynia symptoms continue without change. Full left shoulder active ROM. Full C/S active ROM. Pain reduces to 3–4/10 post nerve glides and 2/10 post tensioners. |
| Week 5–7 | TE: progress weightbearing, overhead presses, cable loaded neurodynamics in the median, ulnar, and radial nerve glide motions. Farmer’s carry. Progressive load of distal muscle groups, and periscapular muscles. Loaded T/S mobility. Home exercise program: MLND. | NPRS pain score 2/10, discomfort in the left scapular region. Discontinued allodynia symptoms, no hand symptoms. Normalized sensation in the left hand. No ROM limitations. Progressing strength in LUE well, compared to RUE. Initiate biking outside of therapy as tolerated. |
| Week 8–11 (discharge) | TE: return to sport testing, limb symmetry of deltoids, periscapulars, biceps, triceps, ability to bear weight.Postural control on bike, with max effort. Compared to the prior level of function, monitor symptoms if applicable. Testing overhead and in closed chain.The patient was discharged after this visit, back to sports. | Shoulder front raise: 93% LSI. Shoulder lateral raise: 94% LSI. Bicep curls: 100% LSI. Triceps extension 100%.Able to bike at the prior level of function with no onset of symptoms.NPRS pain score 0/10. DASH: 2.5. |
TE: therapeutic exercise; C/S: cervical spine; T/S: thoracic spine; MT: manual therapy; NPRS: numerical pain rating scale; ROM: range of motion; MLND: mechanically loaded neurodynamics; LUE: left upper extremity; DASH: Disabilities of the Arm, Shoulder and Hand questionnaire; GHJ: glenohumeral joint; RUE: right upper extremity
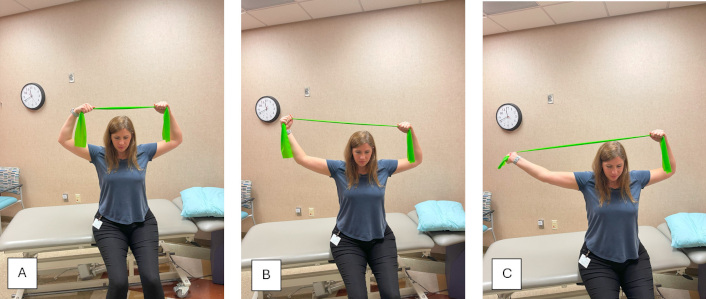
MLND in median nerve pattern using a green resistance band. The patient applies tension through shoulder abduction and external rotation, while pulling into elbow and wrist extension, to stress the median nerve pathway. (A) Starting position; (B) middle position; (C) ending position. MLND: mechanically loaded neurodynamics

MLND in ulnar nerve pattern using a green resistance band. The patient starts in shoulder abduction and pulls tension through elbow flexion and wrist extension. (A) Starting position; (B) middle position; (C) ending position. MLND: mechanically loaded neurodynamics
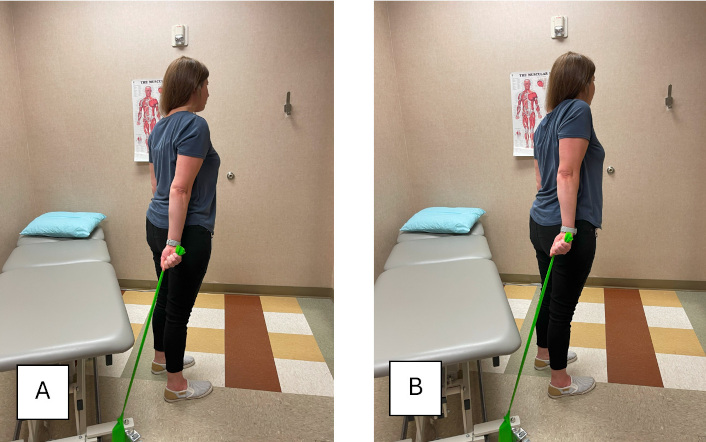
MLND in radial nerve pattern using a green resistance band. Starting in shoulder and scapular depression, shoulder internal rotation with wrist flexion. Pulling tension through a scapular and shoulder elevation (shrug). (A) Starting position; (B) ending position. MLND: mechanically loaded neurodynamics
This case study describes the rehabilitation of a 47-year-old professional cyclist following first rib and cervical rib resection with scalenectomy for aTOS. The patient presented significant neurovascular symptoms and complications, including sharp radiating pain, allodynia, paresthesia in median and ulnar distributions, and activity-limiting dysfunction. MLND was introduced as a central component of post-surgical rehabilitation, targeting graded tension and excursion of neurovascular structures to promote functional adaptation. In addition to improvements in neurovascular symptoms, the patient demonstrated significant gains in musculoskeletal function. Scapular mechanics normalized with symmetrical upward rotation and posterior tilt on dynamic testing. Thoracic extension improved by over 20 degrees, and postural assessment revealed improved head and shoulder positioning during bike fitting and sport-specific assessments.
The incorporation of MLND corresponded with progressive symptom resolution and return to sport within 3 months, quicker than reported post-surgical timelines for vascular TOS rehabilitation [1]. Compared to standard post-operative protocols for aTOS that report return to sport between 4–5 months with limb symmetry index (LSI) deficits often persisting beyond discharge [5], this case demonstrated accelerated return and ≥ 93% LSI symmetry within 11 weeks. These functional gains were supported by a dramatic reduction in the DASH, patient reported outcome measure score, which improved from 86.7 (severe disability) to 2.5 (minimal functional impairment) by discharge, indicating significant improvements in upper extremity function and participation. While strength symmetry was tracked as a functional outcome, validated tools such as DASH [12] were only taken at initial and final sessions. Importantly, these functional outcomes were supported by concurrent improvements in musculoskeletal performance. Restoration of scapular control and thoracic mobility played a key role in reducing mechanical stress on the thoracic outlet and facilitating vascular decompression during overhead and endurance tasks. The integration of scapulothoracic strengthening and thoracic extension training complemented MLND by correcting biomechanical contributors to symptom recurrence and improving overall kinetic chain function. Unlike many reports in the literature that rely heavily on symptom-guided progression and passive mobility interventions [2, 3], this case emphasized deliberate mechanical loading of neurovascular tissue to mimic the functional demands of high-level cycling. These findings align with recent reports showing that individual rehabilitation, including progressive loading strategies, improves functional outcomes in overhead and endurance athletes with TOS [4]. Compared to similar cases, the timeline for return to sport in this case (11 weeks) is shorter than the reported 4–5 months average or aTOS rehabilitation [4]. However, these comparisons must be interpreted cautiously given differences in surgical extent, symptom presentation, comorbidities, and population/sport. Mechanical loading of peripheral nerves is known to stimulate Schwann cell alignment, facilitate axoplasmic transport, and enhance neurotrophic signaling, all contributing to improved neural conductivity and resilience [11]. Similarly, graded vascular loading may promote shear stress-mediated nitric oxide release, facilitating arteriogenesis and endothelial health [6]. The intervention was well tolerated and led to clear improvements in pain, mobility, and function without adverse effects, aligning with previous findings on the benefits of progressive loading in neural and vascular tissue recovery [4]. MLND is a unique intervention that targets the interplay between musculoskeletal mechanics and neurovascular physiology. Unlike passive neurodynamics, MLND applies graded external load to the limb through the full ROM, challenging scapulothoracic musculature, GHJ, and the T/S concurrently with neurovascular tissue to improve tolerance and decrease symptoms.
This case contributes to a significantly limited but emerging body of literature addressing evidence-based rehabilitation following vascular TOS surgery. Systematic reviews consistently cite a lack of standardized physical therapy protocols and note the limited guidance available for return-to-sport progression [2, 5]. The novelty of this report lies in both its structured integration of MLND and its application in a high-level athlete. MLND differs from traditional passive neurodynamics by incorporating resistance and position-specific loads that reflect the real-world stress on neurovascular structures, particularly relevant in endurance athletes with high circulation demands and repetitive limb use. By targeting mechanical resilience and vascular remodeling, MLND may offer a more sport-specific and mechanophysiologically grounded approach to post-surgical recovery [6, 7]. Clinicians considering MLND should introduce low-tension resistance banded load after the patient can achieve and tolerate full ULLT positions with prolonged holds. Progression should be guided by symptom response, with 2–3 sessions/week and a home program to reinforce tolerance adaptations and healing.
The patient’s presentation included both vascular and neural symptoms, complicating the clinical picture and highlighting the need for a rehabilitation strategy that balances neural sensitivity with vascular healing. The observed improvements, specifically rapid reductions in allodynia and restoration of functional capacity, suggest a favorable neuromodulatory and vascular response to controlled mechanical input. From a physiological standpoint, MLND is theorized to enhance axoplasmic transport, reduce intraneural edema, and facilitate vascular shear adaptation, although these mechanisms require further validation in human trials [6, 8]. Objective diagnostic tools such as magnetic resonance imaging, Doppler ultrasound, or nerve conduction studies were not available during the course of care and were not used to confirm vascular decompression. While physical examination findings and symptom response guided clinical reasoning, future cases would benefit from incorporation of these tools to validate physiological recovery and document post-surgical change.
Although this case demonstrated favorable outcomes, there are theoretical risks associated with early application of MLND after vascular surgery, including thrombus formation, endothelial injury, or vascular irritation. In this case, MLND was not introduced until three weeks post-operation, and only after surgical clearance. Safety measures included pulse symmetry checks, capillary refill assessment, and monitoring for cold intolerance or progressive neurological symptoms. MLND progression was paused if any of the following occurred: Onset of vascular signs (e.g., cyanosis, diminished pulse), increase in pain > 2 points on the NPRS lasting > 24 hours, development of new or progressing neurological deficits. No adverse events occurred, and the intervention was well tolerated. Nonetheless, further research should define clear dosing parameters and physiological monitoring strategies for MLND in post operative musculoskeletal, neurogenic, and vascular populations.
There may be concerns surrounding the safety and dosing of loaded neural mobilization post-surgically, particularly in populations with vascular compromise. Existing literature on neural mobilization post-TOS surgery has raised caution regarding load progression and re-irritation risk [9, 10]. However, this case demonstrates that, when introduced judiciously and progressively, MLND may be a safe and effective tool for accelerating recovery. Still, generalizing these results is limited by the single-subject design, lack of objective imaging or electrophysiological data, and the absence of long-term follow-up beyond return to full training. While the patient in this case achieved an expedited recovery, the generalizability of MLND in older, sedentary or comorbid populations remains unclear and warrants further research. Although the neurogenic and vascular effects of MLND were emphasized in this case, it is crucial to recognize the central role of musculoskeletal impairments in the pathophysiology and recurrence of TOS. Clinical rehabilitation must holistically target these impairments to reduce reinjury risk and promote a sustained return to sport/performance. Musculoskeletal biomarkers such as scapular upward rotation angle, T/S excursion, and glenohumeral strength symmetry may serve as useful metrics for rehabilitation progress and discharge readiness in this population. While many aTOS case reports utilize traditional diagnostics, recent work using thermal imaging and Doppler-assisted assessment provides objective confirmation of vascular compromise and tracking of recovery trajectories [12, 13]. By contrast, this care relied on clinical findings and presentation, future reports may benefit from inclusion of vascular imaging to enhance monitoring and validation.
Future research should prioritize prospective studies comparing traditional neurodynamic protocols with MLND in both neurogenic and vascular TOS populations. Establishing dose-response relationships, monitoring physiological changes, and validating clinical outcomes through larger sample sizes are the next essential steps. Furthermore, collaboration between vascular surgeons, sports physical therapists, and neurophysiologists is needed to create standardized, interdisciplinary post-operative guidelines tailored to athletic populations. In conclusion, this case illustrates the potential utility of MLND in the accelerated rehabilitation of aTOS, particularly in the athlete population. The intervention corresponded with rapid symptom reduction, restoration of neuromuscular control, and return to full sport within 3 months. Strengths of the case include a structured intervention progression, use of validated patient reported outcome measures (DASH), clear symptom response monitoring, and integration of sport specific demands. However, generalizability is limited by the single-subject design, absence of objective imaging data, and lack of long-term follow-up. Future studies should explore MLND in broader populations with validated outcome measures and physiological monitoring. The high-functioning nature of the patient, combined with the rare presentation of aTOS, further limits generalizability to sedentary, older, or multi-morbid populations.
aTOS: arterial thoracic outlet syndrome
DASH: Disabilities of the Arm, Shoulder and Hand questionnaire
GHJ: glenohumeral joint
LSI: limb symmetry index
MLND: mechanically loaded neurodynamics
NPRS: numerical pain rating scale
nTOS: neurogenic thoracic outlet syndrome
TOS: thoracic outlet syndrome
T/S: thoracic spine
vTOS: venous thoracic outlet syndrome
TW: Conceptualization, Investigation, Writing—original draft, Writing—review & editing.
The author declares that there are no conflicts of interest.
All investigating authors/participants are certified by the Collaborative Institutional Training Initiative (CITI Program) for human subject research, ethics compliance, reasonable conduct of research, and biomedical-basic. Atrium Health IRB Associate Director formally exempted this project from IRB oversight. Exemption number IRB #11-23-46EX. This study complies with the Declaration of Helsinki (2024).
Informed consent to participate in the study was obtained from the participant.
Informed consent to publication was obtained from the participant.
All data are included in the manuscript. For further discussion regarding this report, please contact the corresponding author.
Not applicable.
© The Author(s) 2025.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2025. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 4365
Download: 48
Times Cited: 0
