Affiliation:
1Samik Diagnostic Pvt. Ltd., Bhubaneswar, Odisha 751024, India
ORCID: https://orcid.org/0009-0005-7254-5549
Affiliation:
2Kalinga Institute of Medical Sciences, Bhubaneswar, Odisha 751024, India
ORCID: https://orcid.org/0000-0002-4949-3585
Affiliation:
2Kalinga Institute of Medical Sciences, Bhubaneswar, Odisha 751024, India
ORCID: https://orcid.org/0000-0001-8039-5142
Affiliation:
3Department of Community Medicine, Subbaiah Medical College, Shimoga, Karnataka 577201, India
ORCID: https://orcid.org/0000-0002-5155-9010
Affiliation:
4Department of Internal Medicine, Kings College Hospital London, Dubai 340901, UAE
ORCID: https://orcid.org/0009-0005-2842-8468
Affiliation:
1Samik Diagnostic Pvt. Ltd., Bhubaneswar, Odisha 751024, India
Email: himadritanayabehera@gmail.com
ORCID: https://orcid.org/0000-0002-8372-9109
Explor Digit Health Technol. 2026;4:101188 DOI: https://doi.org/10.37349/edht.2026.101188
Received: November 11, 2025 Accepted: January 16, 2026 Published: March 05, 2026
Academic Editor: Atanas G. Atanasov, Medical University of Vienna, Austria
Background: Urine screening is a critical diagnostic tool in healthcare that supports the detection of a wide range of health conditions, including kidney diseases, metabolic disorders, and infections. Traditionally, urine tests are performed in clinical settings with results that often take time to be delivered. Such delays can hinder timely diagnosis, treatment initiation, and effective disease management. Recent advancements in digital health technologies, particularly the Internet of Things (IoT), machine learning (ML), and artificial intelligence (AI) algorithms, create opportunities for real-time data acquisition, integration, and analysis within routine urine screening. This systematic review synthesizes the current landscape of IoT-enabled urine screening technologies and evaluates their clinical, engineering, and computational foundations. The review also examines their integration with digital health architectures, edge computing systems, and tech driven personalized care.
Methods: A structured literature search was conducted across PubMed, IEEE Xplore, Scopus, and Google Scholar for studies published between 2000 and 2025. Predefined search terms related to urinalysis, IoT, digital health, and microfluidics were applied. Sixty-five studies met the inclusion criteria. Data extraction focused on sensor technologies, digital health platforms, and reported case studies that demonstrated successful system deployment across diverse healthcare settings.
Results: IoT-based urine screening technologies support real-time monitoring of biomarkers such as glucose, protein, and pH, which are essential for diagnosing conditions including diabetes, kidney disease, and urinary tract infections (UTIs). Emerging devices utilize optical, and acoustofluidic modalities, while BLE, Wi-Fi, and LPWAN serve as the primary connectivity standards.
Discussion: IoT-driven digital transformation demonstrates strong potential to enhance the accessibility, efficiency, and diagnostic accuracy of urine screening. The convergence of biosensing, microfluidics and HDTs enables scalable, continuous, and personalized urine monitoring solutions. Despite these advancements, challenges related to data privacy, infrastructure readiness, and regulatory compliance remain significant barriers.
Routine urine screening is a fundamental diagnostic tool in clinical practice. It provides critical insights into an individual’s health status and enables the early detection of a wide range of diseases. Urinalysis plays a key role in identifying early signs of kidney disease, diabetes mellitus, urinary tract infections (UTIs), and various metabolic disorders [1–5]. It can also yield ancillary information related to hydration status, dietary intake, medication use, and lifestyle habits. Alterations in urine characteristics such as color, turbidity, or odor may indicate underlying pathology, although these features may also reflect benign influences such as diet, medications, physical activity, and hydration levels [6–10].
Urinalysis typically consists of three components: i) Physical examination, which evaluates color, clarity, volume, and specific gravity; ii) Chemical analysis, which tests for substances such as protein, glucose, ketones, blood, bilirubin, and pH; and iii) Microscopic examination, which identifies cells, casts, crystals, and other particulates [11]. Together, these analyses provide a noninvasive yet comprehensive assessment of renal, metabolic, and infectious conditions.
Despite its diagnostic value, routine urine screening faces notable barriers, particularly in resource-limited settings. Manual processing, reliance on centralized laboratories, the requirement for trained personnel, and delays in result reporting can reduce accessibility and hinder timely clinical decision-making. These limitations have increased interest in the integration of digital health technologies, particularly the Internet of Things (IoT), into diagnostic workflows.
IoT-enabled systems support real-time data collection, automated analysis, and remote transmission of results. Through the integration of smart sensors and cloud-based platforms, these systems can detect key urinary biomarkers and support rapid interpretation through AI and ML algorithms. Such innovations have the potential to decentralize urine diagnostics, reduce diagnostic delays, and improve diagnostic accuracy, especially in settings with limited infrastructure [12–15].
This systematic review aims to synthesize current evidence on the role and impact of routine urine screening in modern healthcare, with particular emphasis on digital transformation and IoT integration. The review explores the clinical utility of urine screening, evaluates technological advancements in diagnostic methods, examines implementation challenges, and highlights the potential of IoT-based systems to improve large-scale public health outcomes.
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure transparent and reproducible reporting.
The review aimed to address the following research question:
What is the current evidence on the use of digital health technologies, particularly IoT-enabled urine screening devices, and how do their engineering architectures, sensing modalities, and computational workflows support the diagnosis and management of urological and related health conditions?
The inclusion criteria encompassed studies involving adults and children who underwent urine screening or monitoring for urological or systemic health conditions. Eligible studies examined interventions that used digital health technologies such as IoT-enabled devices, mobile health applications, telemedicine platforms, and data analytics related specifically to urine screening. Outcomes of interest included diagnostic accuracy, clinical effectiveness, patient adherence, usability, health outcomes, and improvements in healthcare accessibility. The review considered a range of study designs, which included randomized controlled trials, cohort studies, case-control studies, cross-sectional studies, and relevant pilot or feasibility studies. To ensure relevance to contemporary technological developments, only studies published from the year 2000 onward and written in English were included. Exclusion criteria comprised studies that did not involve urine screening or urological diagnostics, non-primary research articles such as editorials or conference abstracts without full datasets, and studies that did not clearly evaluate the impact of digital health technologies.
A structured literature search was conducted across multiple academic databases to identify relevant studies related to routine urine screening and the integration of IoT technologies into healthcare diagnostics. The databases searched included PubMed, Scopus, Google Scholar, and IEEE Xplore.
The search strategy combined keywords and MeSH terms related to Urine Screening, Urine Analysis, Routine Urine Testing, Point-of-Care Urinalysis, IoT in Healthcare, IoT-enabled Devices, Healthcare Digitalization, Digital Transformation, and Public Health IoT. Additional studies were identified through manual examination of reference lists from relevant reviews and eligible articles.
Three independent reviewers screened titles and abstracts to identify potentially eligible studies. Full-text articles were retrieved and evaluated for inclusion according to the predefined eligibility criteria. Any discrepancies were resolved through discussion or consultation with an additional reviewer.
Three independent reviewers performed quality assessments and data extraction. Titles, abstracts, and full-text articles were screened according to the predefined inclusion criteria. Extracted data included study type and design, type of IoT devices and sensors used, integration with AI, ML, or cloud computing, clinical impact such as accuracy, speed, and accessibility, and public health implications such as scalability, cost-effectiveness, and patient engagement. Each included study corresponded to a single report; no multiple publications from the same study population were included. Where potentially overlapping reports were identified, only the most complete and relevant publication was retained for data extraction.
Discrepancies in study selection or data interpretation were resolved through consensus. This systematic approach ensured transparency and minimized bias in the analysis.
A total of 265 articles were initially identified. Of these, 241 were obtained from database searches, and 24 were identified from other sources, including citation tracking of relevant studies. After duplicate removal and eligibility screening, 65 studies met the inclusion criteria and were selected for analysis, as illustrated in the PRISMA flow diagram (Figure 1). Notably, 14 of the 21 articles identified through citation searching were included in the final dataset. Of the 99 reports initially sought for retrieval from the database search, 28 full-text articles were not accessible through databases at the time of the study. Additional attempts were made through institutional library access and publisher platforms to access these 28 full-text articles, but they could not be retrieved due to access restrictions at the time of the review. While this may introduce potential retrieval bias, the review synthesis was conducted using the 51 available full-text studies, supplemented by 14 additional studies obtained through alternative methods, encompassing a range of device types, methodologies, and populations.
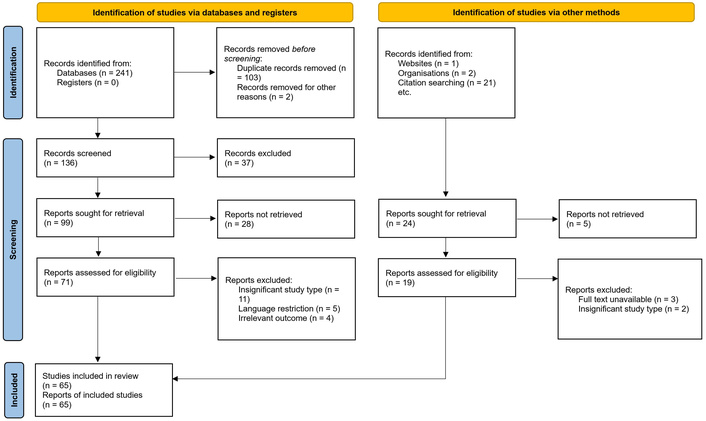
PRISMA 2020 flow diagram for study selection. Adapted from [16], under CC BY 4.0 license.
Urinalysis is a critical diagnostic tool that can identify a wide range of medical conditions, including kidney disease, diabetes, UTIs, dehydration, and certain cancers. Through the assessment of the physical, chemical, and microscopic properties of urine, clinicians can detect disease markers before clinical symptoms appear. This capability supports early diagnosis and preventive care.
Key markers such as proteinuria, hematuria, and abnormal creatinine levels often represent the earliest signs of renal dysfunction, particularly in individuals with diabetes or hypertension. Urine testing also plays an important role in chronic disease management, evaluation of treatment effectiveness, and detection of complications.
Recent studies have emphasized the value of urinary biomarkers in the detection of bladder cancer, including markers such as DJ-1, as well as conditions such as coronary artery disease and diabetic complications [17–21]. In addition, urine has emerged as a promising noninvasive medium for the detection of viral infections such as HIV, Zika virus, hepatitis C, and HPV [22–25].
Routine urine screening enables healthcare providers to initiate timely interventions, adjust treatment strategies, and encourage appropriate lifestyle modifications, ultimately improving patient outcomes and overall health [26].
Urine screening across the lifespan remains clinically essential; however, from an engineering and digital health perspective, the primary challenges lie in accommodating age-specific variability in sample acquisition, biomarker interpretation, and usability requirements. Neonatal and pediatric screening is constrained by noninvasive collection and minimal sample volumes [27–32], whereas adult and geriatric populations require systems capable of longitudinal monitoring, low user burden, and integration with chronic disease management workflows [33–46]. Pregnancy-specific screening further necessitates high analytical sensitivity and reliable data transmission for timely clinical intervention [47–51]. These age-dependent constraints underscore the need for adaptable IoT architectures, robust signal normalization, and privacy-aware data handling frameworks rather than purely clinical stratification.
Our analysis indicates that current IoT-driven urinalysis technologies exist along a continuum that ranges from digitally enhanced dipstick-based assays to fully automated microfluidic lab-on-chip platforms and sensor-integrated smart toilets. Early-generation devices primarily incorporate colorimetric test strips that are interpreted through smartphone-based imaging applications [52–55]. These systems generally rely on optical reflectance analysis of reagent pad color changes that occur in response to urinary analytes such as protein, glucose, leukocyte esterase, nitrites, or ketones. Captured images are converted into standardized color spaces to normalize lighting variations, after which algorithmic or ML models quantify analyte concentrations. Many of these devices communicate through Bluetooth Low Energy (BLE) or Wi-Fi, which enables automated data upload to cloud platforms or local mobile applications. One such IoT-enabled device is EzeFind by EzeRx (Odisha, India). This system uses colorimetry and computer vision to interpret urine test strips and connects to a mobile application and web portal for remote tracking and analysis. The device is portable and user friendly, which makes it particularly valuable for community health programs, home care, and resource-limited settings where access to traditional laboratories is restricted. These systems offer portability and affordability, but few of them remain vulnerable to environmental and device-related variability, including illumination fluctuations, camera sensor noise, and inconsistencies in reagent pad manufacturing [56–58]. In addition, conventional laboratory-based methods continue to face challenges related to accessibility, cost, and efficiency, particularly in rural or underserved areas [59–63].
More advanced systems move beyond smartphone-based imaging through the integration of electrochemical, microfluidic, or acoustofluidic sensing modalities that are specifically designed to improve analytical precision. Electrochemical urine biosensors use ion-selective electrodes, enzyme-linked detection mechanisms, or impedance spectroscopy to quantify analytes such as creatinine, urea, sodium, and potassium with greater sensitivity than purely optical approaches. These sensors often incorporate low-power microcontrollers that pre-process raw signals on the device before transmitting compressed features to mobile or cloud-based platforms. Wearable biosensors represent a growing subdomain within this category [64]. Although many wearable systems do not measure urine directly, they can detect urine-derived biomarkers through sweat analysis or moisture-detection patches integrated into undergarments. Such technologies have been applied to hydration monitoring, urinary incontinence detection, and continuous renal function assessment in vulnerable populations, including older adults and bedridden patients. Given their role in long-term monitoring, wearable systems commonly employ BLE, Low-Power Wide-Area Network (LPWAN), or hybrid connectivity architectures that prioritize low energy consumption and intermittent data transmission.
Microfluidic point-of-care diagnostic devices represent the most technically sophisticated category identified in this review. Several recent studies describe gravity-driven or capillary-driven microfluidic metering systems that provide precise volumetric control, multiplexed biochemical assays, and automated sample handling without the need for external pumps [65–69]. Acoustofluidic platforms have demonstrated automated microscopic urinalysis through controlled manipulation, focusing, and imaging of urinary particles such as cells, casts, and crystals [70–72]. These technologies increasingly integrate smartphone optical modules or embedded cameras, which transforms the traditionally operator-dependent process of microscopic urinalysis into an automated and high-throughput workflow. The underlying microfluidic architecture also allows integration of filtration, reagent mixing, incubation, and optical or electrochemical readouts within a single disposable cartridge. As a result, these systems are emerging as high-fidelity alternatives to centralized laboratory testing [73].
At the extreme end of system integration, smart toilets represent a paradigm shift toward passive and continuous urine monitoring. These devices embed spectroscopic sensors, chemical cartridges, computer vision modules, and IoT connectivity directly within household toilet infrastructure [74]. By analyzing urine during routine toileting, smart toilets eliminate the need for manual sample collection and enable the generation of longitudinal datasets that are not achievable through episodic clinical testing [65]. Several prototypes demonstrate advanced functionalities such as automatic user identification, volumetric flow analysis, uroflowmetry pattern assessment, and continuous biochemical surveillance [75, 76]. However, system complexity and high costs currently limit large-scale deployment [77].
Across all device categories, the review identified substantial heterogeneity in connectivity standards and computational workflows. BLE remains the predominant protocol for wearable and portable systems, whereas Wi-Fi is more common in imaging-based or high data rate applications. NFC is used primarily in battery-free passive systems with minimal data requirements. LPWAN technologies support rural or remote deployments that require extended transmission range and multi-month battery life. A small but growing subset of studies investigates 5G connectivity for high-bandwidth, real-time biosensing, particularly in conjunction with digital twin frameworks.
Computational models varied considerably across studies. Several investigations employed cloud-centric architectures in which raw or minimally processed data were transmitted to cloud servers for model inference, visualization, and long-term storage. Notably, a subset of studies positioned urine-derived data within broader digital health and HDT frameworks. Within these systems, continuous or high-frequency urinalysis data contribute to dynamic computational representations of individual physiology. This integration supports personalized risk prediction, adaptive therapeutic guidance, and simulation-based optimization of treatment strategies.
Security and privacy measures showed marked variability. While some studies implemented robust end-to-end encryption, token-based authentication, blockchain-based integrity verification, or data governance frameworks aligned with HIPAA or GDPR requirements, most provided only limited descriptions of their cybersecurity architecture. Few studies specified encryption standards or authentication protocols at the implementation level, which highlights a significant research gap in protecting sensitive biosensor-derived health data.
From a clinical perspective, the reviewed systems targeted a wide range of urological and systemic conditions. Commonly assessed biomarkers included protein, glucose, pH, nitrites, leukocyte esterase, and ketones, whereas more advanced platforms also quantified creatinine, specific gravity, and microscopic particulates. Diagnostic performance was generally favorable, with reported sensitivities ranging from 78%–100% and specificities from 72%–98%. However, variability in study design and methodological rigor limited direct comparability across studies. Smart toilets and microfluidic devices demonstrated the highest analytical consistency, which likely reflects improved control over sample handling and measurement conditions. To enhance cross-platform comparability, Table 1 summarizes diagnostic performance metrics, connectivity standards, and computational architectures across major device categories.
Comparative summary of IoT-enabled urine screening device categories.
| Device category | Key biomarkers | Reported sensitivity (%) | Reported specificity (%) | Primary connectivity | Computational architecture | Ideal for |
|---|---|---|---|---|---|---|
| Smartphone-based strip readers | Protein, glucose, pH, ketones, etc. | 78–95 | 72–90 | BLE, Wi-Fi | Cloud-centric ML inference | Home, community screening |
| Wearable urine-adjacent sensors | Hydration markers, electrolytes | 80–92 | 75–88 | BLE, LPWAN | Edge preprocessing + cloud analytics | Long-term monitoring |
| Microfluidic lab-on-chip platforms | Creatinine, cells, proteins | 90–100 | 85–98 | Wi-Fi, USB | Edge-assisted inference | POC and clinics |
| Acoustofluidic microscopic systems | Cells, casts, crystals | 92–100 | 88–98 | Wi-Fi | Hybrid edge–cloud | Advanced POC |
| Smart toilets | Multi-analyte longitudinal profiles | 85–98 | 80–95 | Wi-Fi, 5G | Edge + cloud + HDT integration | Home-based continuous monitoring |
BLE: Bluetooth Low Energy; POC: point of care; LPWAN: Low-Power Wide-Area Network.
Overall, IoT-enabled urine diagnostics demonstrate substantial potential for automation, remote monitoring, personalized healthcare integration, and the digital transformation of urological care [78–87]. Nonetheless, critical gaps remain in large-scale clinical validation, standardization of engineering protocols, calibration stability, cybersecurity robustness, and the advancement of HDT-based integration pathways.
Through automated urine analysis and integration with cloud-based dashboards, healthcare systems can monitor trends, predict disease outbreaks, and manage resources more efficiently. Figure 2 illustrates the multifaceted role of IoT technologies in enhancing routine urinalysis.
This systematic review addresses a rapidly evolving domain at the intersection of medical informatics, health service delivery, and technological innovation. Its primary aim is to improve healthcare outcomes through the use of digital technologies, including the IoT, mobile applications, and cloud computing, to support efficient care delivery at local, regional, and global levels [88]. Although digital health tools have been under development since the early 2000s, their widespread adoption, particularly within primary healthcare (PHC), accelerated markedly during the COVID-19 pandemic. This period highlighted their critical role in improving healthcare access, continuity, and system resilience [89, 90].
Evidence drawn from 65 studies demonstrates clear momentum toward automation, decentralization, and digital transformation in urinalysis. At the same time, the findings reveal substantial variability in engineering rigor, cybersecurity implementation, and clinical validation. Synthesis of the available evidence indicates that IoT-based and digitally transformed urine testing systems extend beyond improvements in convenience. These technologies fundamentally reshape the diagnostic paradigm through multimodal sensing, real-time analytics, and seamless integration with broader digital health ecosystems, including telemedicine platforms, electronic health records (EHRs), and HDT frameworks ultimately increasing the patient engagement [91].
The prominent trend is the transition from episodic, laboratory-based urinalysis to decentralized systems capable of generating high-frequency or continuous health data. Smart urine strips and smartphone-based analyzers, once viewed primarily as consumer-grade tools, have evolved into technically robust systems that leverage algorithmic color correction, ML-based interpretation, and encrypted wireless data transmission to deliver clinically relevant insights. Despite these advances, early-generation systems continue to face limitations related to environmental variability, reagent instability, and dependence on user technique. In contrast, microfluidic and acoustofluidic point-of-care platforms represent a more substantial departure from conventional dipstick testing. Their capacity for automated sample metering, multiplexed biochemical assays, and high-resolution microscopic analysis elevates their function from basic screening to tightly controlled analytical evaluation. These characteristics reduce operator error, enhance analytic reliability, and enable integration with mobile imaging and computational modules, which positions these platforms as credible alternatives to centralized laboratory urinalysis.
Across device categories, the engineering architectures that support IoT-enabled urine diagnostics are progressing toward more advanced computational models. Early systems relied primarily on cloud-based analytics, with transmission of raw or minimally processed data for remote interpretation. Although cloud platforms provide scalability and facilitate integration with clinical dashboards, they also introduce latency, bandwidth constraints, and dependence on network reliability [92, 93]. Several studies address these challenges through the incorporation of edge computing, which allows on-device preprocessing, feature extraction, and, in some cases, complete model inference. More advanced systems leverage dynamic computation offloading, accuracy-aware optimization, and multiuser scheduling strategies to maintain diagnostic timeliness under constrained network conditions [94]. Collectively, these findings emphasize the importance of aligning computational architecture with clinical context, particularly for time-sensitive conditions such as UTIs and acute kidney injury.
Recent advances in mobile edge computing provide valuable insights into optimizing computational workflows for IoT-enabled urine diagnostics. Studies on multi-user computation offloading and transmission scheduling for delay-sensitive applications demonstrate how latency constraints can be managed through dynamic task partitioning between edge nodes and cloud servers [95, 96]. Such frameworks are particularly relevant for time-critical urinalysis applications, including acute infection detection and renal injury screening, where diagnostic delays may have direct clinical consequences.
Accuracy-aware online optimization strategies, such as those proposed for dynamic HDT deployment at the edge, further highlight the importance of balancing inference precision with computational and energy constraints. These approaches enable adaptive decision-making regarding where analytic tasks should be executed, allowing systems to prioritize edge-based inference when network conditions degrade while preserving cloud-based model refinement during stable connectivity.
Game-theoretic and ML–based computation offloading mechanisms proposed for cloudlet-based mobile computing also offer transferable design principles for distributed urinalysis platforms [97]. In large-scale deployments such as community screening or smart toilet networks, such strategies can support fair resource allocation, scalability, and robust performance across heterogeneous user populations.
Recent surveys on HDT architectures in personalized healthcare emphasize the role of continuous physiological data streams in enabling individualized health modeling [98]. IoT-enabled urinalysis contributes uniquely to HDT frameworks by providing noninvasive, biochemical, and longitudinal indicators of renal, metabolic, and hydration status. Unlike episodic laboratory testing, continuous or high-frequency urine data enable dynamic updates to digital twin representations, supporting early anomaly detection and personalized intervention strategies.
Generative AI–driven HDT models further enhance this capability by simulating physiological trajectories and predicting disease progression under varying behavioral or therapeutic conditions [99]. Additionally, privacy-preserving approaches such as differentially private federated multi-task learning offer promising solutions for safeguarding sensitive urine-derived data while enabling collaborative model training across distributed healthcare environments [100].
Additionally, the growing use of generative AI models, reconfigurable intelligent surfaces (RIS) to optimize network quality of experience, and mobile AI-generated content (AIGC) for personalized guidance reflects a maturation of the field beyond isolated diagnostic tools [101, 102]. Collectively, these studies position IoT-enabled urinalysis as a foundational data modality within a broader computational healthcare system.
Despite these promising developments, several important gaps remain. Most notably, there is a persistent lack of large-scale, clinically validated trials that evaluate diagnostic accuracy, reproducibility, and device performance across diverse populations and real-world settings. Many studies report encouraging sensitivity and specificity; however, these metrics often originate from small, controlled laboratory evaluations rather than robust cohort-based testing. Issues such as calibration drift, reagent shelf life, optical variability, and sensor degradation received limited attention, which raises concerns regarding long-term reliability.
Taken together, the findings of this review indicate that IoT-enabled urinalysis is progressing toward becoming an integral component of digitally transformed healthcare. As sensing technologies mature and computational architectures advance, future systems will likely evolve into hybrid platforms that combine microfluidic precision, edge-based intelligence, multimodal sensor fusion, and seamless integration with digital twin ecosystems. Despite these advances, the clinical value of such devices remains dependent on user adoption, usability, and accessibility. Smart toilets, for example, provide exceptional diagnostic continuity but may be impractical in low-resource settings. In contrast, low-cost smartphone-based systems offer broad accessibility but may lack biomarker diversity or analytical precision. Bridging this divide requires translational research that addresses engineering performance alongside workflow integration, clinician acceptance, and cost-effectiveness. Furthermore, although several studies describe theoretical or proposed system architectures, few provide implementation-level detail related to cybersecurity, interoperability, or regulatory compliance. Only a limited number of studies specify encryption standards, blockchain-based data integrity mechanisms, or federated learning frameworks designed to protect sensitive biosensor-derived health data. As these technologies become increasingly embedded within clinical systems, the development of secure architectures that comply with HIPAA and GDPR requirements remains essential.
Addressing current gaps in security, validation, and standardization [103–105] will be critical to realizing this vision.
The digital divide also remains a significant barrier to widespread adoption. Limited digital literacy, unreliable internet connectivity, and restricted access to smartphones or other smart devices can impede effective use of digital health technologies, particularly within marginalized communities. This tension between technological utility and individual rights or knowledge varies by disease type, local legal frameworks, and cultural norms. Responsible navigation of these factors is necessary to ensure equitable and ethical deployment.
Overall, routine urine screening remains an essential, noninvasive diagnostic tool for the early detection of conditions such as kidney disease, diabetes, infections, and certain cancers. Innovations such as EzeFind, smart toilets, and related platforms demonstrate how affordable, user-friendly, IoT-based solutions can address long-standing limitations of traditional urinalysis through real-time diagnostics, remote monitoring, and seamless clinical data exchange. These advances expand diagnostic access, empower patients, strengthen preventive care, enhance health system responsiveness, and improve outcomes at both individual and population levels. IoT-enabled urine screening systems therefore, represent a promising frontier in decentralized, patient-centered healthcare. By bridging gaps in early diagnosis, strengthening surveillance, and supporting equitable access, particularly in underserved communities, such technologies have the potential to contribute meaningfully to more resilient, efficient, and inclusive health systems.
BLE: Bluetooth Low Energy
IoT: Internet of Things
LPWAN: Low-Power Wide-Area Network
POC: point of care
UTIs: urinary tract infections
SS: Conceptualization, Writing—review & editing, Supervision. DM: Conceptualization, Investigation, Writing—review & editing, Supervision. NKM: Methodology, Writing—review & editing, Supervision. HR: Methodology, Investigation, Visualization, Data curation. BPS: Methodology, Formal analysis. HTB: Writing—original draft, Formal analysis, Investigation, Visualization, Data curation. All authors have read and approved the submitted version of the manuscript.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
All data used in this systematic review were extracted exclusively from previously published and publicly available articles indexed in databases such as PubMed, Scopus, IEEE Xplore, and Google Scholar. No primary data were generated for this study. The complete list of included studies is available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1902
Download: 126
Times Cited: 0
